Abstract
We report a case of terbinafine-induced myositis in a 37-year-old Asian male patient treated for Tinea cruris. The patient complained of severe generalized body aches, weakness, fever, fatigue, and passing of concentrated urine after consuming one dose of terbinafine 250 mg tablet, which worsened after the second dose. At the presentation, the patient was afebrile, fatigued, and had difficulty moving. All the laboratory parameters were normal, except N-acetyl-cysteine-(NAC)-activated creatine kinase, which was elevated to 276 U/L. There was a slight elevation in alanine transaminase (ALT) levels (44 U/L) and albumin-to-globulin ratio (1.8). The oral terbinafine was stopped. Patient was prescribed with tablet acetaminophen 650 mg thrice daily for two days and as needed thereafter, to manage myalgia. The patient reported being symptomatically better after six days of terbinafine withdrawal. The Naranjo’s causality assessment scale score was eight, indicating a probable relation between drug exposure (terbinafine use) and adverse drug reaction (myositis). The severity of terbinafine-induced myositis in this patient was moderate (level 3) as categorized by the Modified Hartwig and Siegel scale.
Introduction
Terbinafine is a synthetic allylamine anti-fungal agent used to treat several superficial fungal infections. It is a potent fungicide against many dermatophytes that affect skin and nails.1, 2 Terbinafine-induced myalgia and rhabdomyolysis have been observed in post-marketing surveillance studies; however, there are no reports of myositis.3, 4 We report a case of terbinafine-induced myositis in a 37-year-old Asian male patient treated for Tinea cruris.
Case
The patient presented to the treating dermatologist with complaints of severe generalized body aches, weakness, fever, fatigue, and passing of concentrated urine after consuming one dose of terbinafine 250 mg tablet, which worsened after the intake of the second dose. The patient had experienced similar symptoms, slight fever, muscle weakness, and generalized body aches when he took oral terbinafine 1-month ago (250 mg, once daily for 14 days); however, he explained that the symptoms were mild as compared to the present episode. At the presentation, the patient was afebrile, fatigued, and had difficulty moving. The dermatologist advised complete blood count, urea, serum creatinine, N-acetyl-cysteine-(NAC)-activated creatine kinase (CK-NAC), liver function tests, and urine routine measurements (Table 1). All the laboratory parameters were normal, except CK-NAC, which was elevated to 276 U/L (normal reference range: 20−200 U/L), and urine specific gravity was 1.014 units (normal reference range: 1−1.03 units). There were also slight clinically insignificant elevations of alanine transaminase (ALT) levels to 44 U/L against expected levels of 41 U/L and albumin-to-globulin ratio (1.8 against 1.5). The possibility of exercise-induced myalgia was ruled out, as the patient reported normal physical activity, prior to, during, and after terbinafine use. Additionally, there was no evidence of dehydration in this patient, and his daily fluid consumption was normal. The oral terbinafine was stopped and the topical antifungal agent luliconazole was continued to treat the Tinea cruris infection. Patient was prescribed with tablet acetaminophen 650 mg thrice daily for two days and as needed thereafter, to manage myalgia.
Laboratory Investigations Performed at the Time of Presentation.
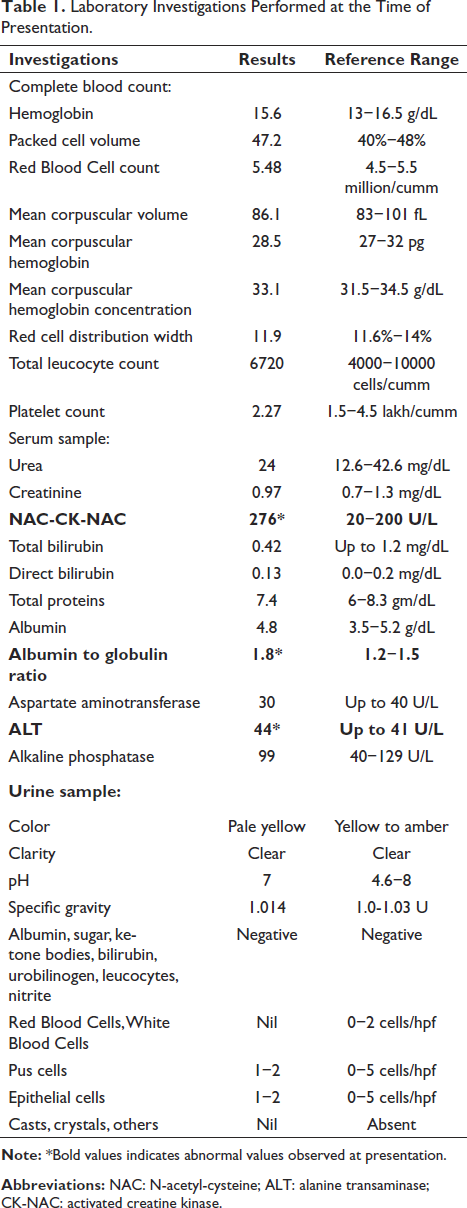
The patient reported being symptomatically better after six days of terbinafine withdrawal. As the patient was not willing to repeat all the laboratory investigations during follow-up, only CK-NAC was repeated. The serum CK-NAC measured after two weeks was within normal limits (195 U/L); however, it was still towards the upper end of the range (20−200 U/L). The Naranjo’s causality assessment scale score was eight, indicating a probable relation between drug exposure (terbinafine use) and adverse drug reaction (myositis). 5 The severity of terbinafine-induced myositis in this patient was moderate (level-3) as categorized by the Modified Hartwig and Siegel scale. 6
Discussion
Myositis is skeletal muscle inflammation, characterized by muscle weakness and is accompanied by elevated creatine kinase (CK) levels. 7 Necrotizing myopathies are usually characterized by muscle pain, tenderness, weakness, and a marked increase in serum CK (>10 times the normal upper limit). In contrast, the mitochondrial myopathies are usually painful, with wasting, tenderness, and muscle weakness affecting proximal musculature, with normal or moderately elevated serum CK and muscle biopsy demonstrating ragged red fibers. 8
The clinical presentation of our patient is most suggestive of myositis, as this patient had most of the clinical features including a slight elevation in serum CK levels. It could be because the serum CK was measured soon after the second dose, and terbinafine was stopped immediately by not allowing serum CK to rise further. Further, the serum CK-NAC measured after two weeks was within normal limits, though it was still towards the upper end of the range (195 U/L). This observation could be because of ongoing muscle damage due to terbinafine, as its terminal half-life is approximately three weeks representing its slow elimination from tissues, resulting in continued muscle weakness. 9 Though muscle biopsy would have given more insights into the type of myositis, in this case, it was not performed as the patient was not willing for the same. The patient was issued an adverse drug reaction alert card during the follow-up visit. He was instructed to keep the alert card and to show it to prescribers and pharmacists to alert them and avoid future use of the same or similar medicines.
Though the exact mechanism of terbinafine causing myositis is not well understood, the probable mechanism could be by directly affecting the mitochondria, lysosomes, and myofibrillar proteins.10, 11 Several modifiable and non-modifiable factors such as age, gender, ethnicity, and physical activity can alter CK levels by affecting enzyme tissue activities. 12 Decreased tolerance to muscle exertion leading to worsening of exercise-induced rhabdomyolysis was suspected in a Caucasian woman who was on terbinafine. The case report documents highly elevated CK levels (4774 IU/L, against 25–155 IU/L), probably due to increased frequency of physical exercise when the patient was on terbinafine. 13 However, in the present case, the mild elevation of CK-NAC was most probably due to terbinafine use alone, as the patient reports no changes in the physical activity and fluid intake prior to, during, and after terbinafine use. The drug-induced myotoxic symptoms may appear anywhere from one week to months after initiating the offending drug and can resolve within weeks after its withdrawal. The management involves early drug withdrawal, and supportive care with symptomatic management which can prevent further harm. In addition, risk assessment in identifying the drug-related factors (such as drugs known to cause myotoxicity, concomitant medications, and the possibility of drug interactions), patient-related factors (such as age, renal and/or hepatic impairment), and previous history of myotoxicity can prevent an individual from drug-induced myotoxicity. 7
Conclusion
Terbinafine-induced myalgia and rhabdomyolysis are reported in the literature through post-marketing surveillance. This is the first case report of terbinafine-induced myositis, where the patient had mild elevations of CK-NAC with myalgia.
Footnotes
Data Availability
All the subjective data and objective data (such as reports of the laboratory investigations) obtained by the patient are included in this case report and are available with the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Statement of Ethics and Informed Consent
The patient data were collected and included in this case report after obtaining written informed consent from the patient.
