Abstract
Background
Non-alcoholic steatohepatitis (NASH) is a clinical condition with a global prevalence of 25.24%. Peroxisome proliferator-activated receptors (PPAR) have been significantly associated with the pathogenesis of NASH.
Aim
To evaluate the efficacy of saroglitazar in an animal model of NASH by evaluating the magnitude of changes in liver function tests (LFT) and histopathology.
Materials and Methods
The baseline parameters of 14 male Sprague–Dawley rats were recorded and then grouped into four groups: treatment groups (high high-dose saroglitazar [HDSG] and low low-dose saroglitazar [LDSG] doses of saroglitazar), normal control, and disease control. Initially, except for the normal control, the other three groups were fed a fructose diet for 5 weeks and then all four groups were fed a standard chow diet for the next 2 weeks during which the two treatment groups were orally gavaged with saroglitazar. Changes in LFT, body weight (BW), lipid profile, oxidative stress, and histopathology were evaluated at different time points.
Results
A statistically significant reduction was found in the mean serum glutamic-oxaloacetic transaminase (SGOT) (p = 0.0267) and serum glutamate-pyruvate transaminase (SGPT) (p = 0.0059) between the groups at the end of treatment. As with BW changes (p < 0.001), a significant difference was observed between the time points in HDSG and LDSG with respect to all parameters of the lipid profile assessed (p < 0.05). Amelioration of hepatocellular ballooning and lobular inflammation in histopathology was evident in both treatment groups. Immunohistochemistry revealed loss of cytokeratin CK8/18 in disease control while it was preserved in LDSG and HDSG.
Conclusion
The study has explicitly illustrated the improvement in the biochemical and pathological changes in the rat model of NASH induced by a high fructose diet.
Introduction
Non-alcoholic steatohepatitis (NASH) is a severe form of non-alcoholic fatty liver disease (NAFLD) affecting 35 million people globally, associated with the presence of liver inflammation and hepatocyte injury (ballooning) due to fat accumulation. 1 NAFLD, being one of the multiple comorbidities in diabetes and India, with a high nationwide prevalence of diabetes, substantially contributes to the global burden of disease. 2
The global prevalence of NAFLD is 25.24%, with the highest prevalence in the Middle East and South America. Metabolic comorbidities associated with NAFLD include obesity (51.34%), type 2 diabetes (T2D) (22.51%), hyperlipidemia (69.16%), hypertension (39.34%), and metabolic syndrome (42.54%). 3 By 2030, 33.5% of the global adult population is projected to suffer from NAFLD, with a further 27% suffering from NASH. 4
Hepatic steatosis in NAFLD is characterized by the hepatocytes accumulating fat droplets. The resulting hepatocellular injury and inflammation, with or without hepatic fibrosis, can lead to NASH. These conditions are predisposed to factors such as insulin resistance, oxidative stress, inflammation, altered production of adipokines and cytokines, altered lipid metabolism, and endoplasmic reticulum stress. 5
These factors make NASH a clinically aggressive variant of NAFLD and patients with NASH are at higher risk of progression to cirrhosis. Hence, the disease has been targeted for drug research and development efforts over the past few years. 6
Due to the multifaceted nature of NAFLD and NASH, the ideal drug for managing these conditions would be expected to show beneficial effects on mitochondrial dysfunction, oxidative stress, insulin resistance, steatosis, inflammation, and fibrosis. 7
Histopathological analysis shows that NASH has a specific injury pattern characterized by ballooning and hepatocellular injury in combination with macrovesicular steatosis and inflammation. Fibrosis and Mallory-Denk bodies (MDB) are often seen but not essential for diagnosis. 8
Peroxisome proliferator-activated receptors (PPAR) are nuclear receptors that regulate a host of processes associated with inflammation and energy homeostasis, making them attractive treatment options for metabolic syndrome-related disorders. 9
In India, saroglitazar magnesium (Lipaglyn), approved for the treatment of T2D and dyslipidemia, has shown promising results in clinical trials evaluating its therapeutic role in NASH. 10
Saroglitazar is a non-thiazolidinedione and non-fibric acid derivative that acts as a dual regulator of lipid and glucose metabolism by activating PPARα/γ. It has insulin-sensitizing effects by activating PPARγ, which results in a reduction in blood glucose. PPARα activation by saroglitazar increases the hepatic oxidation of fatty acids and reduces the synthesis and secretion of triglycerides (TGL). It causes increased lipolysis and the elimination of TGL-rich particles from plasma by activating lipoprotein lipase (LPL) and reducing the production of Apo C-III, an inhibitor of LPL activity. Besides, there is a reduction in low-density lipoprotein (LDL), an increase in the synthesis of apolipoproteins A-I, and A-II and also high-density lipoprotein (HDL). 11 It is currently approved for the treatment of diabetes dyslipidemia in patients not well controlled on statin therapy.
The present study aimed to evaluate the effect of saroglitazar on NASH as a potential therapeutic target in an animal model. The justification stems from the fact that evidence exists for the potential involvement of PPAR nuclear receptors in the pathogenesis of NAFLD. The objectives of the study were to evaluate the magnitude of changes in liver function tests (LFT) and histopathology in saroglitazar-treated rat groups compared to control animals, to assess the changes in body weight (BW) and serum lipid profile, and to estimate the amelioration of oxidative stress by studying the changes in superoxide dismutase (SOD). Studies have shown that rats fed fructose-enriched diets are a better model for NASH than rats fed fat-enriched diets, as the characteristic changes in NASH are well pronounced in this animal model, and second, this model typically induces all features of metabolic syndrome, which is a common concordance in NASH patients. 12 There are no studies to date evaluating the effectiveness of saroglitazar in the fructose diet model of NASH.
Materials and Methods
After approval by the Institutional Animal Ethics Committee (IAEC) with proposal number 370/2017/IAEC, 14 male Sprague–Dawley rats of weight 150–200 g were selected for the study. The animals were housed as 2–4 animals per cage in polypropylene cages at normal ambient room temperature with a 12 h dark–light cycle. They were allowed food and drinking water ad libitum. After obtaining the baseline parameters (LFT, lipid profile, SOD, and BW), the animals were grouped into four groups.
Group 1 (low-dose saroglitazar [LDSG] group): five animals; fed a 70% fructose diet for 5 weeks, followed by saroglitazar (1 mg/kg orally) for 2 weeks (sixth and seventh weeks).
Group 2 (high-dose saroglitazar [HDSG] group): five animals; fed a 70% fructose diet for 5 weeks, followed by saroglitazar (3 mg/kg orally) for 2 weeks (sixth and seventh weeks).
Group 3 (disease control [DC]): two animals; fed a 70% fructose diet for 5 weeks. 13
Group 4 (normal control [NC]): two animals; fed a normal rat chow diet for 5 weeks.
All animals were provided with the standard rat chow diet for 2 weeks prior to the commencement of the study and the following fifth week. BW was recorded at baseline, after NASH induction, and at the end of treatment. Blood was collected at baseline and after induction at the end of 5 weeks using the tail snip method and the parameters studied included the lipid profile (TGL, HDL, total cholesterol [TC], LDL), LFT (serum glutamic-oxaloacetic transaminase [SGOT], serum glutamate-pyruvate transaminase [SGPT], gamma-glutamyl transferase [GGT]) and SOD.
At the end of the study, all animals were sacrificed to collect blood and liver samples for biochemistry and histopathological examination, respectively. The rats were anesthetized using an intraperitoneal injection of an overdose (45 mg/kg BW) of sodium pentobarbital. Terminal blood collection was done by cardiac puncture using a disposable syringe with needle no. 21 and collected into two microcentrifuge tubes. The abdominal walls were opened to harvest the liver, which was fixed in 10% formalin for histopathological examination.
Data collected was entered in Microsoft Excel and analyzed using statistical software R version 4.2.1. Categorical variables were represented by frequency and percentage. Continuous variables are given in mean ± SD/median (Min, Max) form. The normality of the variable was checked by the Shapiro–Wilk’s test and QQ plot. A one-way analysis of variance (ANOVA) was used to compare the means of variables across the groups. Tukey’s HSD was used as a post hoc analysis. A one-way repeated measures ANOVA was used to compare the means of variables over the time points. A pairwise t-test was used as a post hoc analysis. A p-value less than or equal to 0.05 was considered statistically significant.
Results
From the ANOVA test, a significant difference was found in the mean of SGOT (p = 0.0267) and SGPT (p = 0.0059) between the groups at the end of treatment. From one-way repeated measures of ANOVA, both LDSG and HDSG groups demonstrated a fall in mean SGOT and SGPT post-induction, though the difference was statistically significant in the LDSG group (Table 1). There was no significant difference in the mean of GGT between or within the groups at any time point (Table 1).
Comparison of LFT Within and Between Groups.
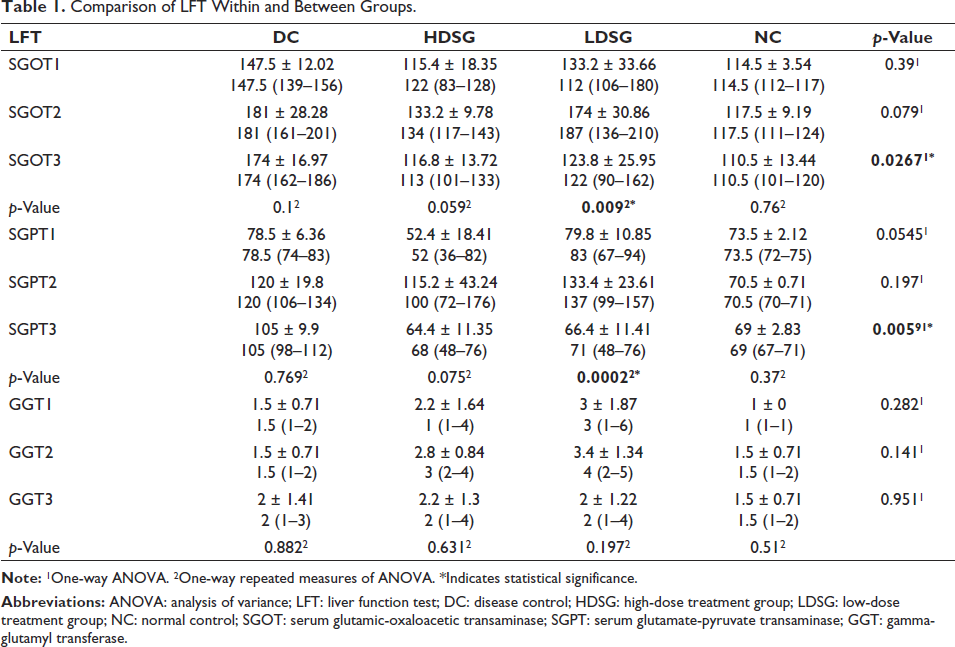
A one-way ANOVA demonstrated a significant difference in the mean of TGL (p = 0.0144) between the groups at the end of treatment. From one-way repeated measures of ANOVA, a significant difference was observed in the mean of TGL between the time points in HDSG (p = 0.002) and LDSG (p = 0.0004) (Table 2). Likewise, from the ANOVA test, a significant difference was also observed in the mean of HDL (p = 0.0423), TC (p = 0.0069) and LDL (p = 0.0059) between the groups at the end of 7 weeks. In addition, from the one-way repeated measures of ANOVA, a significant difference was observed in the mean HDL between the time points in HDSG (p = 0.0003) and LDSG (p = 0.0002), the mean TC in HDSG (p = 0.0002) and LDSG (p = 0.0002), and the mean LDL in HDSG (p < 0.001) and LDSG (p < 0.001) (Table 2).
Comparison of Lipid Profile Within and Between Groups.
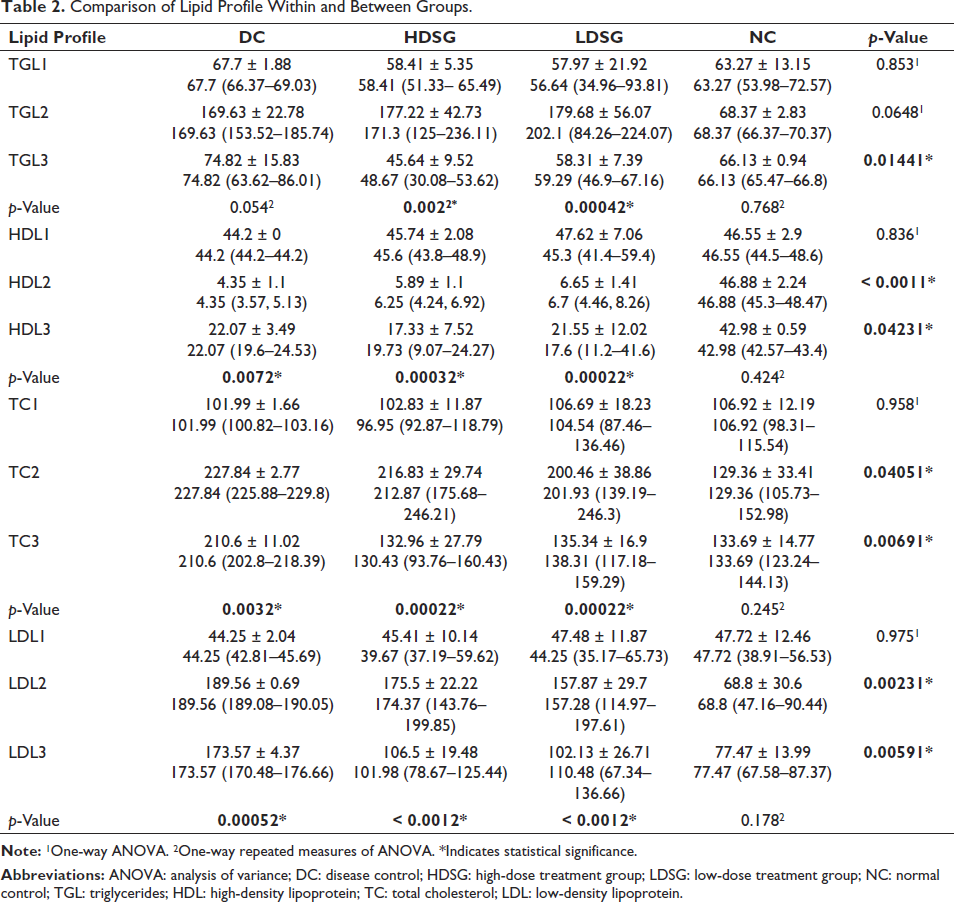
Comparing the SOD levels within and between all groups, no significant difference was observed in the distribution of SOD between the groups at any time point. There was no significant difference observed in the distribution of SOD between the timepoints in any group (Table 3).
Comparison of SOD Within and Between Groups.
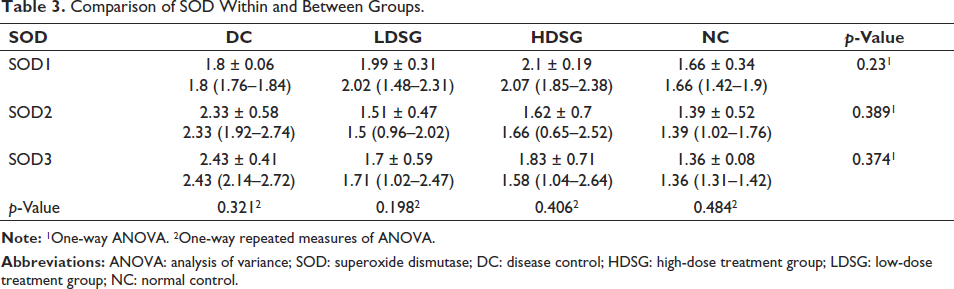
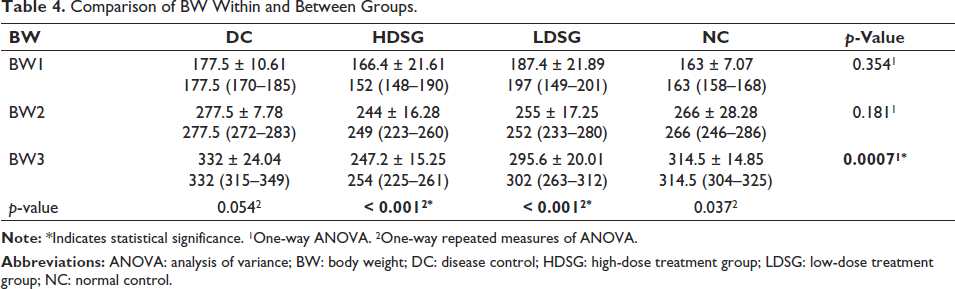
Table 4 compares the BW within and between groups. From the ANOVA test, a significant difference was observed in the mean of BW over the groups at the third time point (p = 0.0007). A significant difference was observed in the mean of BW between the timepoints in both HDSG (p < 0.001) and LDSG (p < 0.001) (Table 4).
Comparison of BW Within and Between Groups.
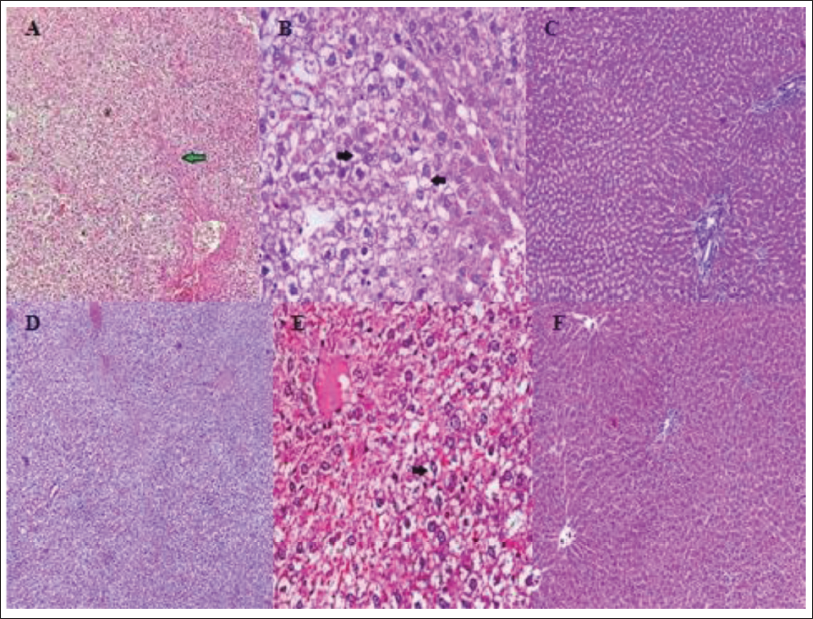
With regard to histopathology, using hematoxylin and eosin staining, DC showed diffuse areas of hepatocellular ballooning with occasional areas of lobular inflammation associated with NASH (Figure 1). In the LDSG group, one or two sporadic areas of hepatocellular ballooning in a few sections were observed, which were of lesser grade when compared to disease control, with no significant alterations in hepatocellular morphology. The HDSG group showed hepatocellular morphology resembling that of normal control with the absence of ballooning and other features of lobular inflammation (Figure 2)
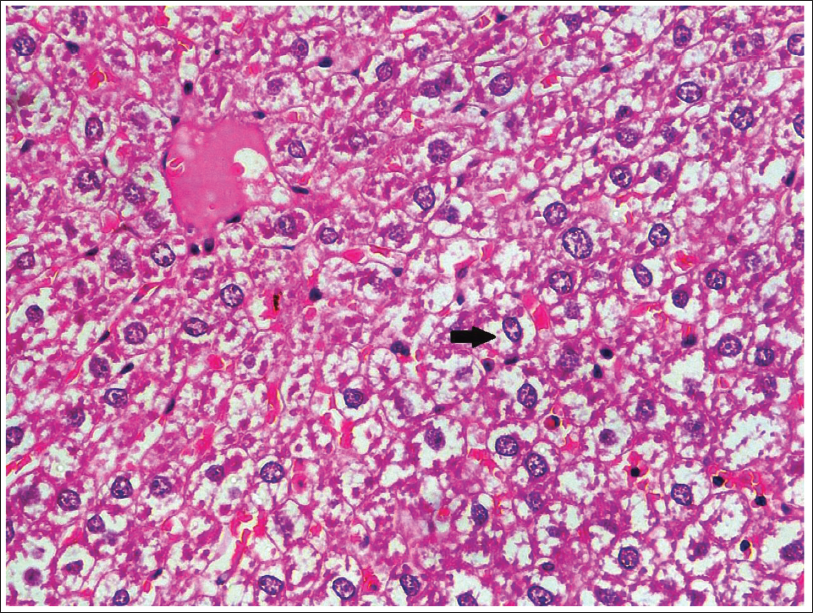
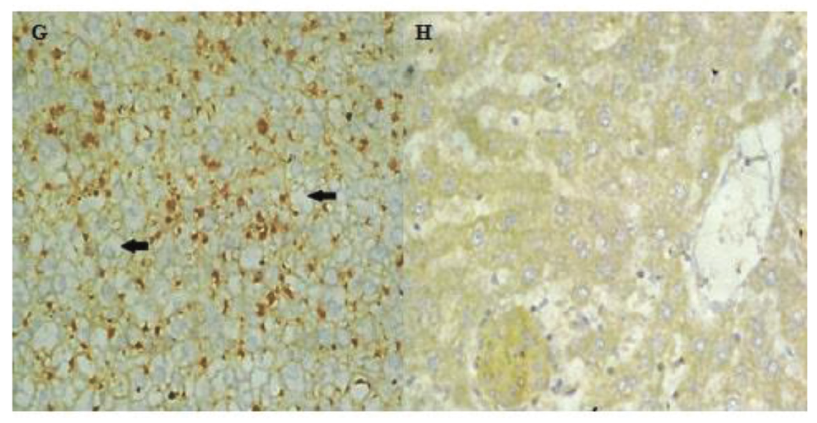
An immunohistochemical study was undertaken to evaluate K8/18 expression in ballooned hepatocytes. The cytokeratin stain revealed that the DC group depicted a depletion of cytoplasmic cytokeratin using CK8/18 immunostaining, while all other groups showed the presence of cytoplasmic cytokeratin, as highlighted by the CK8/18 stain (Figure 3).
Discussion
A key consideration in the therapeutic development of NASH is the identification of a rational therapeutic target, taking into account that NASH often develops in the context of excess adiposity and systemic insulin resistance. Saroglitazar is a PPARα/γ agonist and the rationale for its use in NASH includes the expected synergy of improved insulin resistance and increased lipid oxidation from its PPARγ and PPARα effects, respectively, which would reduce the lipotoxic load on the liver. 14 But its effects on NASH have not yet been fully evaluated in a relevant model. Hence, the present study was conducted to assess the efficacy of saroglitazar treatment in a rat model induced with NASH using a high fructose diet. The parameters were assessed at baseline, at the end of the fifth and seventh weeks.
The results of the study showed a rise in SGOT and SGPT during induction with a fructose diet and a definite fall in the mean values of these liver enzymes was illustrated following saroglitazar treatment. However, the effect in the LDSG group was statistically significant compared to the HDSG, which can be possibly attributed to the greater rise in levels with induction in this group of animals and a consequentially greater fall with treatment. When assessing the lipid profile, saroglitazar treatment effectively reduced all the parameters, which was evident from a significant difference in the mean TGL, HDL, TC, and LDL between the time points following induction and treatment (Table 2). Similarly, a demonstrable reduction in the mean BW of LDSG and HDSG was observed and found to be statistically significant. But no statistically significant change was seen in the SOD levels, both with induction and treatment.
The results of the present study were in agreement with those conducted by Hassan et al. which assessed deactivation of the hepatic LPS/TLR4 signaling pathway and amelioration of adipocyte dysfunction in rats with high-fat emulsion/LPS model-induced NASH. The study concluded that saroglitazar treatment counteracted the induced BW gain, activity of liver function parameters, and lipid profile levels, which were normalized alongside histopathological changes. 15 The study was very similar to the present study, except for the sample size used.
The results of a study conducted by Kumar et al. to assess the improvement of NASH in the diet-induced animal model of NAFLD by the use of pioglitazone and saroglitazar along with controls were also in agreement with those of the current study, with the results revealing saroglitazar to be equal to or superior to pioglitazone by lowering BW, insulin resistance (HOMA-IR), TGL, TC, and SGPT while also improving steatosis, lobular inflammation, hepatocellular ballooning and fibrosis during histopathological analysis. 14
Hepatocellular ballooning is considered one of the histological hallmarks of steatohepatitis and represents a special form of liver cell degeneration associated with cell swelling and enlargement, found particularly in steatohepatitis. Yet, enlarged hepatocytes may be observed in other liver diseases as well. 16 Moreover, MDB, representing the accumulation of ubiquitinated cytokeratins, may not always be detectable in ballooned hepatocytes.
Hence, immunohistochemical staining for cytokeratins 8 and 18 (CK8/18) facilitates the diagnosis of NASH, in which ballooned hepatocytes are typically CK8/18 negative. These staining characteristics are consistent with the pathological process that is said to disrupt the turnover of intracellular proteins, namely ubiquitination and proteosomal degradation, leading to endoplasmic reticulum stress that sequentially contributes to hepatocyte injury. 17 Our study demonstrated that the loss of cytokeratin CK8/18 in immunostaining, an objective marker for the particular type of hepatocellular injury that results in morphologic ballooning in steatohepatitis, observed in the DC rat liver, was found to be resolved in both LDSG and HDSG.
Thus, the histopathological assessment conducted in this study showed improvement and normalization of NASH-induced changes on a cellular level and these results are in concordance with the results of previous studies, which concluded that saroglitazar treatment effectuated normalization of deteriorated histopathological changes in other animal models.14, 15
Saroglitazar treatment has been recently studied for its use in the control and treatment of NASH, which can be life-threatening if not adequately managed. The results of these studies have helped provide evidence for the potential therapeutic effectiveness of saroglitazar in clinical scenarios.18–21 A review has also shown that saroglitazar effectively improved lipid and glycemic parameters without significant adverse effects in patients with diabetic dyslipidemia in real-world clinical studies of up to 58 weeks duration. 22 Saroglitazar has also been compared to other drugs such as Hepano, a herbal formulation, and was found to be superior in high-fructose and high-fat-induced insulin resistance, obesity, and dyslipidemia. 23 Another PPAR agonist, pioglitazone, was also found to be equal to or inferior to saroglitazar with respect to non-metabolic diseases, including NASH, but its use was limited by its side effect profile.24, 25 A study conducted to assess the safety and efficacy of saroglitazar treatment in non-alcoholic fatty liver patients with diabetic dyslipidemia showed no major adverse reactions along with a significant improvement in liver parameters, a reduction in liver fibrosis and a reduction in serum TGL level. Thus, the evidence favors saroglitazar as a beneficial treatment approach for NASH. 26
The use of acetyl CoA carboxylase inhibitors as an augmented form of treatment alongside PPAR agonists has also been studied and further research could help produce better outcomes in patients suffering from NASH. 27
The current study has determined the therapeutic benefits of saroglitazar for the treatment of NASH using an animal model. Thus, the study has added to the scientific evidence for the potent therapeutic role of saroglitazar in NASH treatment. The limitation of the study was the small sample size of the groups. The results of the animal study, when supported by clinical trials, can help assert that saroglitazar is one of the favored therapeutic options for NASH.
Conclusion
NASH is a health condition that is on the rise due to the increased levels of unhealthy and sedentary lifestyle choices made by humans. It is projected to only increase in the near future. This study has ascertained saroglitazar to be a viable treatment option for NASH; further research could help in establishing it as a primary drug for this liver disease.
Footnotes
Acknowledgments
We acknowledge the animal house technicians for their support in the conduct of the study.
Authors’ Contribution
First author: Concept, design, definition of intellectual content, literature search, data acquisition, data analysis, manuscript preparation, manuscript editing and manuscript review.
Second author: Concept, design, definition of intellectual content, literature search, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Third author: Definition of intellectual content, literature search, data acquisition, data analysis, statistical analysis, manuscript preparation and manuscript review.
Fourth author: Definition of intellectual content, literature search, data acquisition, data analysis, statistical analysis, manuscript preparation and manuscript review.
Data Statement
The data that support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Study was conducted after obtaining ethical approval from the Institutional Review board (Ref no: 370/2017/IAEC).
Funding
This research was funded by Intramural grant for Faculty from the host institution.
Informed Consent
Not applicable
