Abstract
Objectives
To assess the pattern of antimicrobial use among pediatric inpatients in a secondary care hospital in the United Arab Emirates.
Materials and Methods
The prospective study was conducted based on the electronic records of 600 patients, collected over a period of 11 months, from September 2020 to July 2021. The primary patient population was the pediatric patients receiving antimicrobials admitted to Saqr Hospital, Ras Al Khaimah (RAK), United Arab Emirates (UAE). The data gathered includes general patient information, lab investigations, diagnoses, chronic medical conditions, and antimicrobials used. The number of defined daily doses (DDDs) administered per patient was calculated for each antimicrobial prescribed as per the WHO anatomical therapeutic chemical classification.
Results
Out of 1400 patients admitted to the pediatric unit during the 11-month study period, 600 (42.8%) received antimicrobials. The mean duration of hospital stay was 3.44 days, and each patient received a mean of 1.41 antimicrobials per prescription. The mean days of antimicrobial therapy were 6.9 days. The majority of the patients were aged 0–5 years (61.1%), and 58% of the total sample was male. Amongst a total of 41 different antimicrobials prescribed, the beta-lactam co-amoxiclav (J01CR02) was the most frequently (19.3%) used one, followed by cefuroxime (J01DC02) (16.3%), amoxicillin (J01CA04) (15.0%), and azithromycin (J01FA10) (5.99%). These were administered mainly via the parenteral route, and the most common indication was respiratory disease.
Conclusion
Our study concludes that most of the prescribed antimicrobials for pediatric patients are within the WHO access and watch group. Co-amoxiclav, cefuroxime, and amoxicillin are the most frequently used antimicrobials. The main indication for use was respiratory illness.
Keywords
Introduction
The discovery of antimicrobials has revolutionized medical practice by curing a vast majority of patients from fatal bacterial infections. These medications have truly made a great positive impact in the medical field. However, unnecessary over-prescription of these medications has become a common issue worldwide, prompting the World Health Organization (WHO) to declare it a public health emergency. 1 Moreover, the incidence of pediatric hospitalization is increasing exponentially for various reasons, resulting in an increase in the use of antimicrobials in children worldwide. 2 This has resulted in selection pressure associated with genetic or mutational alterations, leading to augmented antimicrobial resistance among micro-organisms.3, 4
Among all infectious causes requiring hospitalization in children, upper respiratory tract infections are the most common. They are the primary cause of antimicrobial misuse, specifically in primary care, where viruses are the main culprit. 5 In order to maintain antimicrobial effectiveness for future generations, rational antimicrobial usage is crucial. 1 Rational drug prescription refers to the use of the least number of drugs to obtain the best possible effect in the shortest period and at a reasonable cost. The aim of studying drug utilization is to identify the root problems and issues associated with them, as well as provide useful feedback to the prescriber. This is done in order to increase awareness of the irrational use of antimicrobials. 6
Over-prescription has inevitably led to increased adverse effects, ineffective treatment, and increased costs.7, 8 This, along with antimicrobial resistance, is becoming increasingly prevalent in areas with low micro-organisms status. Not only is this population at increased risk of infection, but they also face a lack of access to diagnostic resources and medical aid. 1 Children are particularly vulnerable to these issues because they fall sick frequently and thus, are exposed to more antimicrobials compared to adults. They have unique physiological features, thus altering pharmacokinetics and pharmacodynamics, putting them at a higher risk of the consequences of antimicrobial resistance. The most common type of prescription error in this age group is dosing errors, which may easily be avoided by using individualized adjustments. 9 There are also variations in the prevalence of antimicrobial use among inpatient pediatricians.10–12 The easy accessibility of antimicrobials has further contributed to the global crisis of antimicrobial resistance. 13 Several studies have been conducted to evaluate antimicrobial use; nevertheless, there exist wide variations in antimicrobial use among children.14, 15 Hence, our aim was to focus on its usage in pediatric patients of a secondary care hospital in the Emirate of Ras Al Khaimah in the UAE. By breaking down the different antimicrobials administered to pediatric patients, this study aims to evaluate the drug utilization pattern of antimicrobials prescribed in hospitals.
Materials and Methods
This prospective study was conducted over a period of 11 months, from September 2020 to July 2021, and included a total of 600 patient records. The medical records of pediatric inpatients were evaluated. The information has been obtained from medical records by visiting the hospital every week. The data gathered includes general patient information, lab investigations, diagnoses, chronic medical conditions, and antimicrobials used. The defined daily doses of antimicrobials prescribed have also been calculated as per WHO guidelines. The data has been organized in tables and charts and compared with WHO standards as well as findings from similar studies.
Our study included patients under the age of 18, hospitalized in a secondary care hospital, Saqr Hospital in Ras Al Khaimah, and those that were given antimicrobials, whereas patients over the age of 18, pediatric patients who did not receive antimicrobials, pediatric outpatients, neonatal intensive care unit patients, COVID-positive patients, and prescriptions with incomplete patient information were excluded from the study. The MOHAP Research Ethics Committee approved the study (MOHAP/REC/2020-21/30-2020-UG-M).
Results
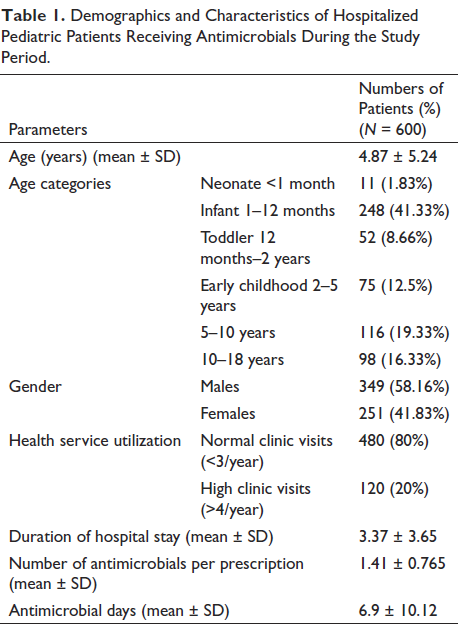
A total of 1,400 patients were admitted to the pediatric unit between September 2020 and July 2021, and 600 (42.8%) received antimicrobials. The mean duration of hospital stay was 3.44 days, and each patient received a mean of 1.41 antimicrobials per prescription. The majority of the patients were aged 0–5 years (61.1%) and males (58%). 80% of the total sample had less than three admissions per year (Table 1). The mean of antimicrobial days (mean ± SD) was 6.9 ± 10.12. The antimicrobial days for 0–5 years, 5–10 years, and 10–18 years were 6.39 ± 6.91, 4.62 ± 1.91, and 3.53 ± 2.70, respectively.
Demographics and Characteristics of Hospitalized Pediatric Patients Receiving Antimicrobials During the Study Period.
Amongst a total of 41 different antimicrobials prescribed, beta-lactam co-amoxiclav (J01CR02) was the most frequently (19.3%) used antimicrobial, followed by cefuroxime (J01DC02) (16.3%), amoxicillin (J01CA04) (15.0%), and azithromycin (J01FA10) (5.99%), as shown in Table 2.
Pattern of Antimicrobial Prescription.
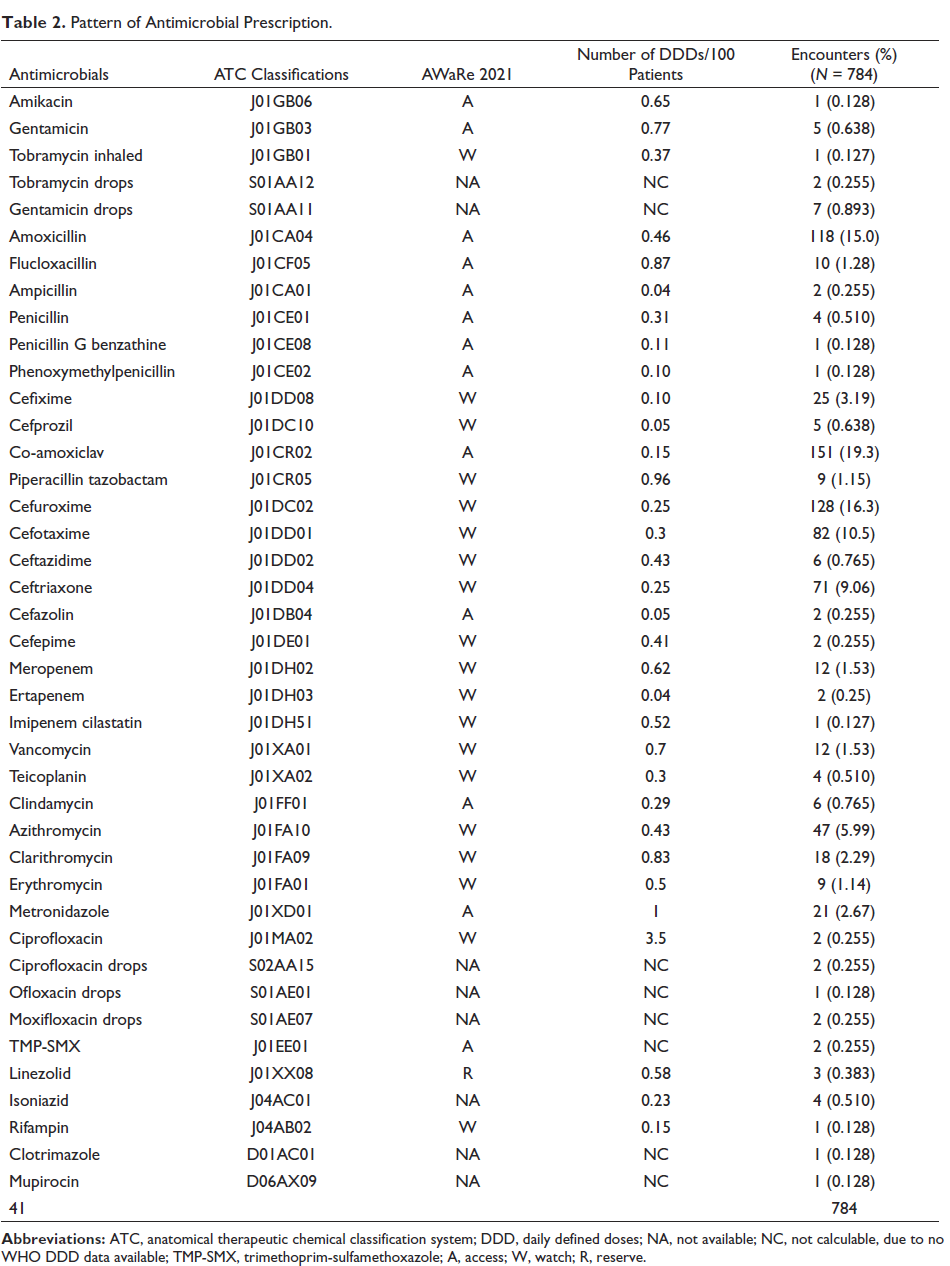
Among the total of 41 different antimicrobials prescribed, 13 (31.7%) were under “access,” 19 (46.3%) were under “watch,” and 1 (2.4%) was under the “reserve” category as per the WHO Access, Watch, Reserve (AWaRe) classification of antibiotics for evaluation and monitoring of use.16, 17 Antimicrobials were prescribed for a total of 784 times during our study period; among these, 506 (64.5%) were administered parenterally (IV), 264 (33.7%) orally (PO), and 14 (1.8%) were applied topically.
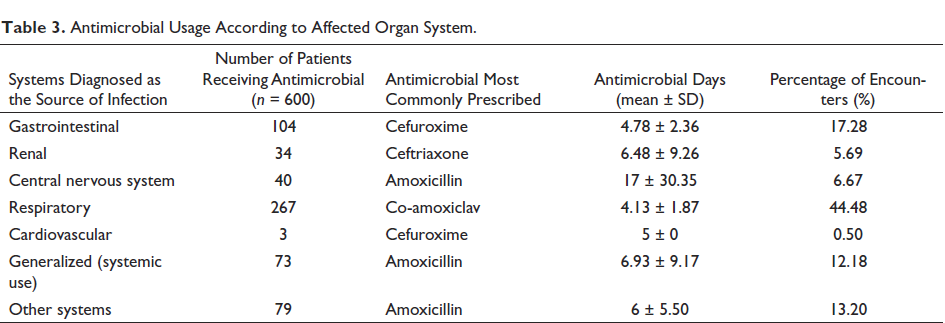
The highest incidence of antimicrobial prescriptions was 111 times, seen in November 2020. This was followed closely in June 2021, with a total of 104 prescriptions. The prevalence of the remaining prescriptions in descending order is as follows: December 2020 (80), October 2020 (75), January 2021 (65), July 2021 (61), April 2021 (60), May 2021 (59), September 2020 (58), March 2021 (56), and lastly, February 2021 (55). In our study, the most common reason for the use of antimicrobials was respiratory illness (Table 3). Among those, tonsillitis and bronchiolitis were the most prevalent. Other respiratory indications were pneumonia, sinusitis, and bronchitis. The second most common indication for antimicrobial use was gastrointestinal illnesses, of which gastroenteritis took precedence. The reserve antibiotics, in the WHO AWaRe classification, are antibiotics used in suspected or confirmed infections caused by multi-drug-resistant organisms. In our study, linezolid was the only reserve antibiotic used. It was prescribed three times for different bacterial infections: spinal abscess, mastoiditis, and cellulitis of the neck. The antimicrobial days (mean ± SD) for respiratory and gastrointestinal infections were 4.13 ± 1.87 and 4.78 ± 2.36, respectively (Table 3).
Antimicrobial Usage According to Affected Organ System.
Discussion
Of the 1,400 patients admitted to pediatrics during our study period, 42.8% received antimicrobials. This is in agreement with the point prevalence study, which reported 40.9% of antimicrobial use among inpatients in the United Kingdom. 11 Another web-based point prevalence study documented 49.7% of antimicrobial use among inpatients in South Africa, and ampicillin was the commonest one to be used for pneumonia. 18 Gibbons et al. reported 35% of antimicrobial use among hospitalized pediatric patients. 10 However, gentamicin was the most commonly used antimicrobial for clinical sepsis as opposed to amoxiclav for respiratory illness in our study. Thus, the findings may vary based on the pattern and burden of infections in that country. Gandra et al. reported 61.5% of in-patient children being on at least one antimicrobial, with 3rd generation cephalosporin being the commonest one prescribed in India. 19 Versporten et al. reported 36.7% of inpatients being on at least one antimicrobial in a large point prevalence study involving 226 centers and 41 countries. 20
There are studies that report higher use of antimicrobials. Labi et al. reported 70% of antimicrobial use among inpatients, and the third-generation cephalosporin is the one that is most commonly used. 21 Another study reported that 92% of pediatric inpatients are on antimicrobials, and ampicillin is commonly used for community-acquired infections. 12 Kebede et al. showed 86.4% of antimicrobial use among the admitted pediatric population. Penicillin G was the most common drug used, and the most common indication was pneumonia. 22
Among pediatric in-patients in Zimbabwe, a total of 93.1% of children were prescribed at least one antimicrobial, and benzylpenicillin was the most frequently prescribed medication. 23 Umar et al. reported 49.5% of drug encounters of antibiotics among the pediatric population, which also included a majority of 0–5 years old. 24 A similar study conducted in Sierra Leone has demonstrated that 74.8% of outpatient pediatric prescriptions consisted of antibiotics. 25
Skosana et al. reported percentages of antimicrobials in access (55.9%), watch (27.8%), and reserve (3.1%) as opposed to access (31.7%), watch (43.9%), and reserve (4.8%) in our study. 18 There is a lot of variation among countries regarding the share of access, watch, and reserve categories of antibiotic consumption. 26 The reporting of data regarding this is the first step to antimicrobial stewardship, and hence this study would facilitate such future measures.
There is a lack of consensus on the utility of DDD among the pediatric population. DDD is defined as “the amount of antibiotic prescribed for a 70 kg average adult weight for its main indication,” according to the WHO Collaborating Centre for Drug Statistics and Methodology. 27 However, it is not useful among children, especially for comparisons of drug exposure among different pediatric populations. We used DDD per 100 inpatients for the descriptive presentation of data, as suggested earlier. 11 Nevertheless, there is a need for another unit, such as pediatric DDD, for better comparability among populations.
Hence, we also presented the data on antimicrobial days, which is defined as “the aggregate sum of days for which any amount of a specific antimicrobial agent was administered to individual patients as documented.”28–30 Further, we presented data on antimicrobial days per 100 admissions. The total antimicrobial days were 4,138 for 1,400 admissions during the study period. This accounted for 295.57 of antimicrobial days per 100 admissions. The mean ± SD of antimicrobial days was 6.9 ± 10.1. The antimicrobial days for 0–5 years, 5–10 years, and 10–18 years were 6.39 ± 6.91, 4.62 ± 1.91, and 3.53 ± 2.70, respectively. There is evidence that a shorter 3-day course is non-inferior to a longer 5-day course of antibiotics among 2–59-month-old children suffering from nonsevere community-acquired pneumonia. 31 However, there is a scarcity of data to conclude the same for intravenous antibiotics when used in the scenario of severe pneumonia. 32 Hence, randomized controlled studies are required to directly compare short-course and long-course intravenous antibiotics among hospitalized children. With regard to system-wise antimicrobial days, the longest was documented for central nervous infections (17 ± 30.35). This may be due to the severity of central nervous system infections. Nevertheless, there is sparse data to show that short-course and long-course antibiotics are equivalent in childhood bacterial meningitis. 33 Further studies are essential to draw conclusions on the same.
Another important factor with regard to the use of antimicrobials among pediatricians is their dose calculation. Moreover, there is a need for pediatric customized computerized provider order entry (CPOE) using artificial intelligence, which would allow dose calculation in real-time before starting the drugs. Similarly, dosing adjustment with respect to organ dysfunction needs to be incorporated into such systems.
Antimicrobial stewardship programs (ASP) with more active surveillance in pediatrics would make a difference in their use, as studied earlier. 34 ASP has been active in the studied hospital since 2014. The committee includes multidisciplinary team members, led by physicians and clinical pharmacists as coordinators and co-leaders. Other team members are pediatricians, internists, anesthetists, clinical microbiologists, infection control practitioners, and nurses. All members take part in the continual monitoring of antimicrobial usage, which might justify the low prevalence of antimicrobial use and limited order of “reserve” antimicrobials (0.5%) of drug encounters.
The prescribing practice of antimicrobials in pediatric patients in our study matches with the WHO recommendation in terms of giving preference to “access” and “watch” groups of antimicrobials whenever antimicrobials are required. However, ASP in pediatrics requires more precision, and the baseline data of this study would facilitate future studies with specific objectives.
A major limitation of this study was the occurrence of the COVID-19 pandemic, which restricted and delayed the data collection process. Adverse reactions to the antimicrobial drugs prescribed were not documented. There is no data proving the accuracy of dosage per unit body weight.
The web-based point prevalence study is proven to be a better study design for estimating antimicrobial consumption; however, we planned a prospective record-based review due to the logistics and magnitude of our single-center study. The results of this study cannot be generalized since it was a single-center study and reflected the practice at a single site. No control group was included in the study; therefore, there is a chance of confounding, bearing in mind the likelihood of seasonal tendencies in antimicrobial use. Moreover, due to the limited documentation of adverse drug reactions in electronic medical records, the pharmacovigilance data collection was not intended as an objective of this study.
Conclusion
The majority of the prescribed antimicrobials for pediatric patients are within the WHO access and watch groups. Co-amoxiclav, cefuroxime, and amoxicillin are the most frequently used antimicrobials. The main indication for use was respiratory illness, followed by gastrointestinal infections.
Footnotes
Acknowledgments
The authors are grateful to Dr. Muna Alayyan, Hospital Director, Saqr Hospital, and Dr. Haydar Ibrahim, Pediatrician, Saqr Hospital, for administrative support and encouragement during the study. The Saqr Hospital Antimicrobial Stewardship Committee is duly acknowledged for its intellectual insights during the conduct of the study. The authors are also thankful to Emirates Health Services (EHS) and the RAKMHSU administration for all the support during the study.
Authors’ Contributions
Conceptualization: SKS and SD; methodology: SD, YS, MM, and SKS; resources: DJ and LKB; data curation: SD, YS, and MM; Original draft preparation: SD; writing—review and editing: SKS, DJ, and LKB; supervision: DJ and SKS. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Statement of Ethical Approval and Informed Consent
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ministry of Health and Prevention (MOHAP) Research Ethics Committee (MO-HAP/REC/2020-21/30-2020-UG-M; date of approval: 3/22/2020). Patient consent was waived as the study involved no direct interaction with participants or their guardians.
