Abstract
Objectives
To investigate the role of miR-142-3p in the bleomycin-induced idiopathic pulmonary fibrosis (IPF) mouse model and elucidate its targets.
Methods
In vitro model: Alveolar epithelial cells (AECs) were isolated and treated with bleomycin (50 µg/mL) or PBS for 12 h. In vivo model: Bleomycin (5 mg/kg) was injected into the trachea under anesthesia and aseptic conditions, and controls were treated with equal saline. After the completion of modeling, proteins and RNA were extracted. p53/NF-κB signaling factors were evaluated by western blot or immunohistochemistry. IL-1β and MMP-9 levels were measured by ELISA. The lentiviral transfection technique was used to overexpress miR-142-3p.
Results
In IPF, miR-142-3p was identified to play a negative regulatory role in lung epithelial cell senescence. Bleomycin treatment significantly reduced miR-142-3p expression in a concentration-dependent manner in vitro. miR-142-3p overexpression inhibited bleomycin-induced cellular senescence in vivo. In particular, miR-142-3p negatively regulated collagen deposition in pulmonary fibrosis by regulating p53/NF-κB expression.
Conclusion
MiR-142-3p plays an important role in the development of IPF by negatively regulating the p53/NF-κB network.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, fibrotic lung disease of unclear etiology. It is increasingly recognized as a serious global public health problem due to limited treatment choices and a poor prognosis. 1 According to the epidemiological survey, IPF mainly occurs in the older age group, and the incidence of IPF is positively correlated with age. Damage to alveolar epithelial cells (AECs) is known to play a critical role in the development and progression of IPF. 2 AEC injury leads to impaired epithelial barrier integrity, which in turn triggers a feedback mechanism that leads to aberrant production of profibrotic factors such as transforming growth factor-beta 1 (TGF-β1). 3 Furthermore, persistent or irreparable damage to AECs may further induce cell apoptosis or trigger the progression of stress-induced senescence. During senescence, abnormally activated AECs promote the excessive secretion of extracellular matrix by fibroblasts and myofibroblasts, leading to collagen deposition and progressive structural changes in the lung. Subsequently, the expression of senescence-associated beta-galactosidase (SA-β-gal), a marker of cellular senescence, is significantly upregulated in the IPF fibroblasts. 3
Cellular senescence can be triggered by a variety of factors, and therefore, senescence is mainly divided into two forms, namely, replicative senescence and stress-induced senescence. Senescent cells display distinct gene expression patterns relative to non-senescent cells, which are termed the senescence-associated secretory phenotype (SASP). According to senescence-driven transcriptional factors, SASP can be further divided into NF-κB-dependent SASP (N-SASP) and p53-dependent SASP (P-SASP), 4 which are both involved in the occurrence and development of IPF. The SASP factors secreted by senescent cells are rich in a variety of bioactive molecules, such as growth factors and inflammatory cytokines, which can affect the senescent cells themselves and promote the hyperplasia and degeneration of nearby cells through autocrine and paracrine mechanisms. 5 However, despite accumulating evidence that the extent of cellular senescence is associated with the severity of IPF, the molecular mechanism remains unclear.
Although many microRNAs have been reported to be involved in the occurrence and development of IPF, 6 the microRNAs that regulate the p53 and NF-κB signaling pathways in IPF remain unclear. Therefore, the microRNA target prediction tool Starbase was used to predict the potential microRNAs that target p53 and NF-κB.7, 8 We found that miR-142-3p may be able to interact with NF-κB/p53. In this study, we will try to explore the relationship between miR-142-3p and NF-κB/p53 in IPF.
Material and Methods
Reagents
Bleomycin was purchased from the Shanghai YuanMu Biological Technology Company. The hematoxylin and eosin (H&E) staining kit was purchased from Sigma-Aldrich, and Masson’s trichrome stain kit was obtained from Solarbio Science & Technology. Primary antibodies against p53 and NF-κB were acquired from Beijing Biosynthesis Biotechnology, and primary antibodies against glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were obtained from Multiscinces Biotech. Protein signals were detected using the rabbit polymer HRP detection system (ZSBG-BIO). Total RNA was extracted using the RNA Pure Total RNA Kit (Generay) and further reverse transcribed into cDNA using the PrimeScript™ RT Reagent Kit (Riobobio). The expression level of mRNAs was detected using the HiScript-II Q RT SuperMix for qPCR (Vazyme) and the ChamQ SYBR Color qPCR Master Mix (Vazyme). The levels of interleukin 1-beta (IL-1β) and matrix metalloproteinase-9 (MMP-9) were purchased from Jianglai Bio and Multi Science.
Animal Experiments
Specific pathogen-free (SPF) C57BL/6J mice (20 ± 2 g) were obtained from Shanghai Jihui Laboratory Animal Care Company. Thirty mice were randomly divided into three groups (10 mice per group): the control group, the bleomycin (BLM) group, and the BLM+miR-142-3p group. Adenovirus-miR-142-3p was purchased from WZ Biosciences Inc. Mice in the BLM+miR-142-3p group was injected intratracheally with adenovirus-miR-142-3p (5 × 10 8 pfu). The interventions were performed one week after transfection. All mice were acclimatized in an experimental room with ad libitum access to chow and water of 24 ± 2°C and a 12-h light–dark cycle. Mice were anesthetized with ketamine. Under sterile conditions, the trachea was intubated with a 26-gauge needle, and bleomycin (5 mg/kg) was then administered into the trachea through the catheter.9, 10 An equal volume of normal saline was used as a blank control. All mice in each group were sacrificed for further analysis after 28 days of bleomycin administration (Figure 1a). All animal experiments in this study were approved by the Institutional Animal Care and Use Committee of Hangzhou Hibio Technology Co. Ltd. (HBFM3.68-2015).
Flowchart of the Experiment.
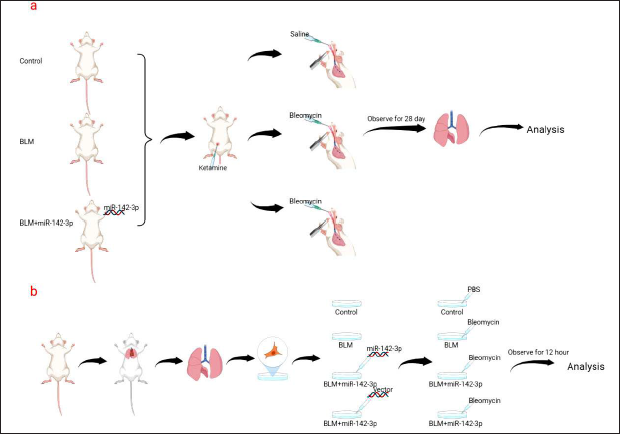
Primary AECs Isolation, Culture, Transfection, and Bleomycin Treatment
Mouse lungs were harvested and homogenized under sterile conditions. Primary AECs were isolated by trypsin digestion, followed by differential centrifugation and differential adhesion. Cells were transferred to 6-well culture plates and incubated to reach 90% confluency. The miR-142-3p overexpression plasmid and vector plasmid were purchased from WZ Biosciences, Inc. Primary AECs were transfected with miRNA-expressing plasmids following the manufacturer’s instructions. 24 h after transfection, primary AECs were treated with bleomycin (50 µg/mL) or PBS for 12 h, and then cells were harvested and analyzed for RNA and protein (Figure 1b). 11
Quantitative Real-Time PCR (qRT-PCR)
Total RNA was extracted from cells using the TRIzol reagent. The purity and integrity of the extracted total RNA were examined by agarose gel electrophoresis, and the concentration was determined by measuring the absorbance using a UV spectrophotometer. The extracted RNA was then reverse-transcribed into cDNA and used as a template for PCR amplification. The expression of miR-142-3p was normalized with U6 RNA as an endogenous control, which was amplified using U6-F and U6-R primers. Table 1 shows the primer sequences for miR-142-3p and U6.
The Primer for miR-142-3p Overexpression.

ELISA Assays
Tissues were washed with pre-cooled phosphate-buffered saline (PBS) to remove residual blood, weighed, and then cut into pieces. The ground tissues were mixed with 10 volumes of PBS and further minced on dry ice. The supernatant was obtained by centrifugation at 5,000g for 5–10 min. ELISA assays of MMP-9 (Multi Science, EK2MO9-24) and IL-1β (Jianglai Bio, JL18442) were conducted using a commercial ELISA kit (J&L Biological and R&D Systems) according to the manufacturing instructions.
Western Blotting Analysis
Mouse pulmonary tissues (100 mg) were minced and homogenized with 500 µL of RIPA lysis buffer. The protein concentration of each group was measured using the bicinchoninic acid assay (BCA) method. Proteins were resolved by SDS-PAGE and electrotransferred onto polyvinylidene difluoride (PVDF) membranes. The membranes were blocked with 5% non-fat milk tris Buffered saline (TBS) for 1 h at room temperature and then incubated with primary antibodies overnight at 4°C. After incubation with the secondary antibodies for 1 h, the protein on the membrane was visualized using the ECL detection system. The signals were scanned and quantified using Image-Pro Plus 6.0. The relative expression level of each protein was calculated by normalizing it with GAPDH expression levels.
Masson Trichrome Staining and H&E Staining
Lungs were harvested and manufactured into paraffin sections for further analysis. Paraffin sections were dewaxed and hydrated using xylene, ethanol, and water. H&E staining was performed using H&E staining kits (hematoxylin: SIGMA, H9627; eosin: SIGMA, E6003). Masson staining was performed using Masson Trichrome staining kits (Solarbio, G1345), according to the protocols, and finally observed under a light microscope. Photographs were taken and retained. Percentage of the area of collagen deposition (%) = [collagen-positive area (blue)/(collagen-positive area + collagen-negative area)] × 100%.
Immunohistochemistry
Paraffin sections were dewaxed and hydrated with xylene, ethanol, and water. The sections were incubated with 3% H2O2 for 15 min to block and inactivate endogenous peroxidase, placed in 0.01 M citrate buffer (pH 6.0), boiled for 10 min, and cooled naturally to room temperature. Subsequently, primary antibody was added and incubated overnight at 4°C; secondary antibody was added and incubated for 30 min at 37°C. 100 µL of DAB was added for color development. After washing, the film was restained with hematoxylin, sealed with neutral gel, and photographed by light microscopy.
Statistical Analysis
The experimental data were statistically analyzed using SPSS software version 17.0. Data are shown as the mean ± standard deviation (SD). Data were analyzed using the t-test; p < 0.05 is considered statistically significant. GraphPad Prism 8.0 was used to generate plots.
Results
Bleomycin Upregulated p53 and NF-κB Expression in AECs and Promotes Cellular Senescence
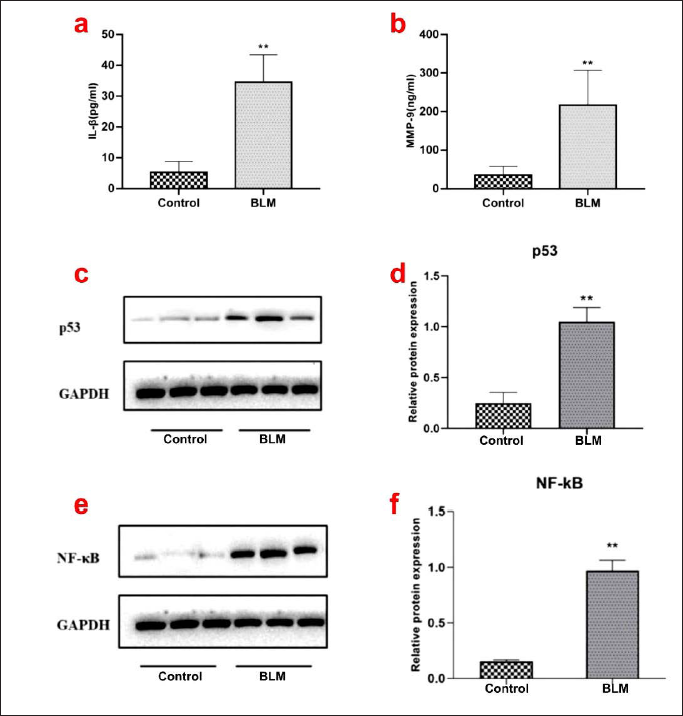
To investigate the role of cellular senescence in IPF, bleomycin, a potent inducer of lung fibrosis, was used to stimulate AECs, and the expression of senescence biomarkers was examined. As shown in Figure 2, bleomycin treatment significantly upregulated the expression levels of IL-1β and MMP-9, which are marker molecules of SASP. On the contrary, the expression levels of p53 and NF-κB in AECs were also examined. The results showed that both p53 and NF-κB were significantly upregulated in AECs after bleomycin treatment. In summary, bleomycin promoted cellular senescence and upregulated the expression of p53 and NF-κB, suggesting that p53 and NF-κB may be involved in the regulation of IPF.
miR-142-3p Targeted p53 and NF-κB and Inhibited Cellular Senescence in AECs
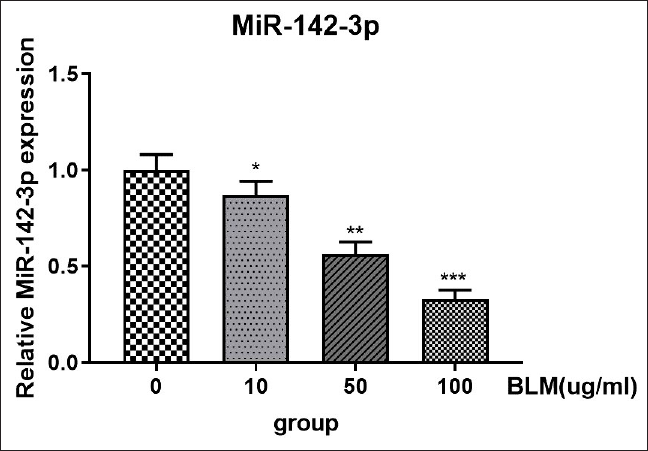
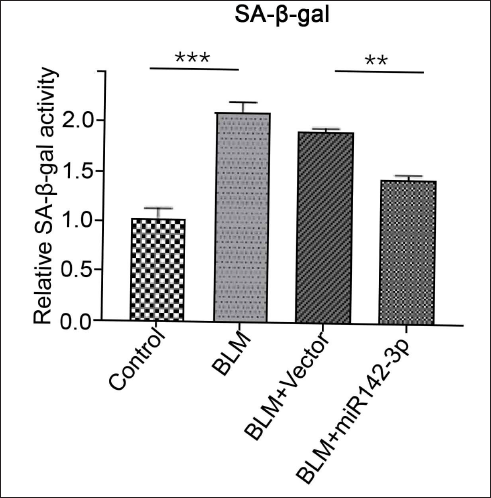
To further elucidate the molecular mechanisms between stress-induced aging and IPF in AECs, we found that the expression of miR-142-3p was significantly decreased in bleomycin-treated AECs in a concentration-dependent manner (Figure 3). This suggests that miR-142-3p may play an important role in bleomycin-induced senescence in AECs. Several papers have demonstrated that p53 and NF-κB are involved in the regulation of cellular senescence. In our study, we found that miR-142-3p overexpression inhibited the expression levels of p53 and NF-κB (Figure 4). We further examined senescence-associated β-galactosidase (SA-β-gal), a cell senescence marker, and found that SA-β-gal was significantly elevated by BLM induction, while the elevation of SA-β-gal is eliminated by overexpression of miR-142-3p, suggesting that miR-142-3p overexpression can alleviate bleomycin-induced senescence (Figure 5). In summary, we guess that miR-142-3p may be involved in regulating p53 and NF-κB to regulate pulmonary fibrosis in AECs.
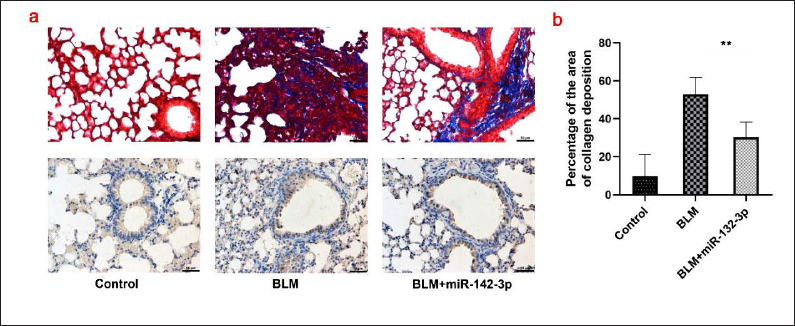
miR-142-3p Overexpression Attenuated Bleomycin-induced Pulmonary Fibrosis in Mice
To further investigate the role of miR-142-3p in vivo, mice were administered bleomycin to induce pulmonary fibrosis. Compared with the saline control, bleomycin-treated mice showed pulmonary fibrosis, evidenced by the thickening of the alveolar septa and increased mononuclear infiltration throughout the lung parenchyma (Figure 6a). However, the degree of lung fibrosis was significantly decreased after transfection of miR-142-3p. A marked increase in collagen deposition was observed in the lungs of bleomycin-treated mice compared with saline-treated mice, while transfection with miR-142-3p attenuated bleomycin-treated collagen deposition. The degree of lung fibrosis was further quantified by measuring the collagen deposition area, and the results showed that collagen deposition was significantly increased after bleomycin treatment (Figure 6b). Moreover, miR-142-3p transfection significantly decreased collagen deposition area compared with saline control. Taken together, these results indicate that miR-142-3p plays a protective role in preventing the development of bleomycin-induced pulmonary fibrosis in vivo.
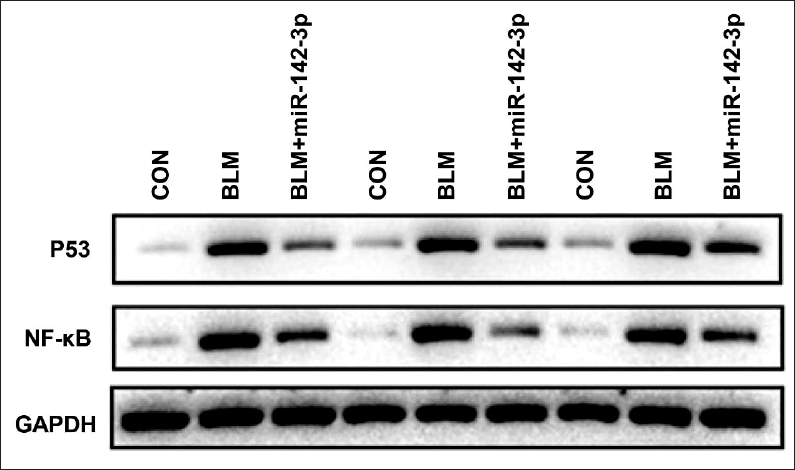
Transfection of miR-142-3p Suppressed p53/NF-κB-mediated Fibrosis in Mice
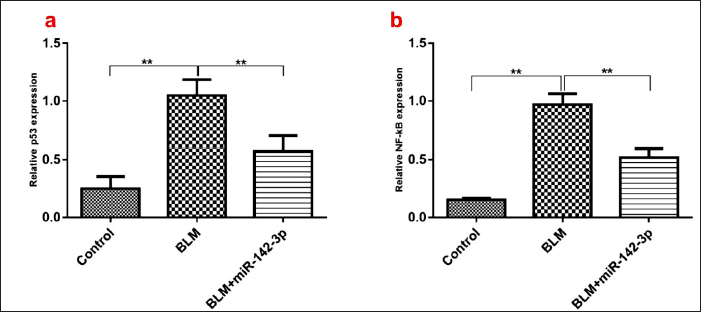
Previous studies have shown that NF-κB and p53 are involved in the progression of lung fibrosis.12, 13 Therefore, we examined whether miR-142-3p regulates the expression of NF-κB and p53. We found that both NF-Κb and p53 were activated in bleomycin-induced pulmonary fibrosis. As shown in Figure 7, bleomycin treatment significantly increased the expression levels of NF-κB and p53. However, transfection with miR-142-3p suppressed bleomycin-induced upregulation of NF-κB and p53. Immunohistochemistry was also conducted to confirm the expression of NF-κB and p53 in lung tissues. As shown in Figure 8, bleomycin treatment increased the expression of NF-κB and p53, while miR-142-3p transfection dramatically attenuated the bleomycin-induced elevation of NF-κB and p53. Taken together, NF-κB and p53 were upregulated in bleomycin-induced lung fibrosis in mice, and transfection with miR-142-3p suppressed the expression of p53/NF-κB and lung fibrosis in mice.
Expression Levels of p53 and NF-κB were Analyzed by Western Blotting Analysis among the Control Group, BLM Group, and BLM+miR-142-3p Group In Vivo. Western Blot Shows Samples from Three Separate Experiments.
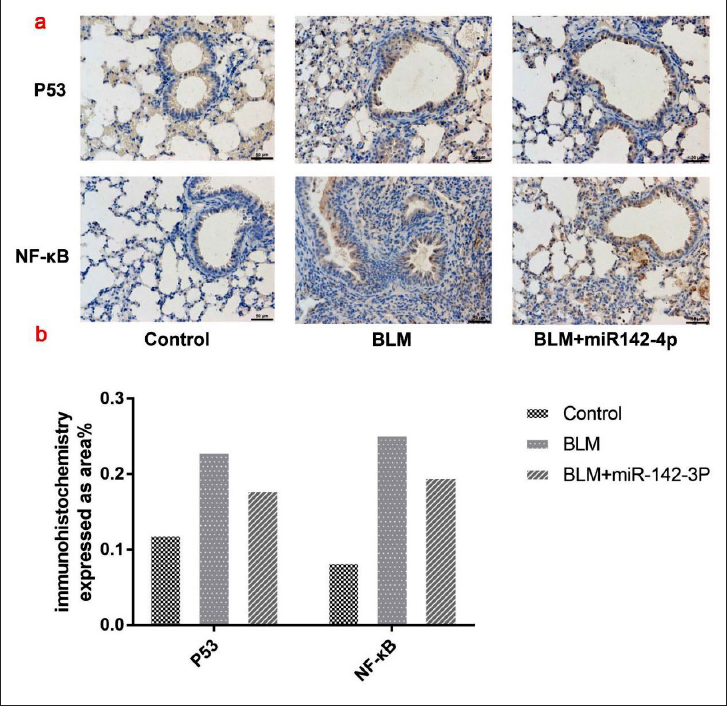
Discussion
IPF remains a difficult-to-treat disease with very limited therapeutic options and a poor prognosis, with a median survival of 2–3 years. 14 The important role of AEC senescence in the development of IPF has also been demonstrated in lung tissue samples from patients with IPF. 15 Senescent AECs secrete SASP to promote fibroblast proliferation and subsequent differentiation into myofibroblasts, which further contribute to basement membrane disruption and extracellular matrix production, ultimately leading to tissue remodeling and fibrosis formation. A recent study reported that the anti-aging drugs dasatinib and quercetin can significantly inhibit bleomycin-mediated cell senescence and fibrotic lesions. 16 Therefore, clarifying the mechanism of cellular senescence could provide a future therapeutic strategy direction for IPF.
SASP is characterized by the activation of multiple pro-fibrotic signals, including the p38 MAPK pathway and the Janus pathway, which are also associated with the activation of the p53 and NF-κB signaling pathways and subsequent activation of the inflammatory response.17–19 In other words, there is a complex cross-network between the NF-κB signaling pathway and the p53 signaling pathways in aspects of the immune response, inflammatory response, and cell apoptosis. In our in vivo and in vitro experiments, we found that bleomycin can upregulate the expression of p53 and NF-κB, which in turn regulate the expression of the senescence marker SA-β-gal and the production of SASP-related cytokines such as IL-1β and MMP-9. All these results suggest that AECs may undergo senescence changes during the development of IPF.
MicroRNAs are endogenous non-coding small RNAs that regulate protein expression by targeting their mRNAs. 20 Many studies have reported that various miRNAs, such as miR-205, miR-22, and miR-326, exert pro- and anti-fibrotic roles in pulmonary fibrosis through epithelial-mesenchymal transition, macrophage polarization, and collagen production.21–24 Furthermore, miR-142-3p is a recently discovered miRNA associated with the development of IPF. In the study by Njock et al., the levels of several miRNAs were dramatically changed in sputum samples of IPF patients, of which the miR-142-3p level was the most significant. 25 In addition, a higher level of miR-142-3p was reported to be negatively correlated with the carbon monoxide diffusing capacity of the lung and may alter the balance between mesenchymal cell proliferation and differentiation, indicating that miR-142-3p levels may be positively associated with the severity of IPF. 26 In this study, the expression level of miR-142-3p in AECs was decreased after bleomycin exposure. Moreover, bleomycin significantly inhibited the expression of miR-142-3p in a concentration-dependent manner. In the miR-142-3p rescue experiments, miR-142-3p overexpression reversed bleomycin-induced cell damage in AECs and reduced cellular senescence and collagen deposition. 19 In addition, miR-142-3p overexpression not only significantly reduced the levels of IL-1β and MMP-9 but also significantly reduced bleomycin-mediated p53 upregulation in the IPF model. Our findings suggest that miR-142-3p can negatively regulate the expression of p53 and NF-κB, thereby delaying cell senescence. Therefore, we speculate that miR-142-3p can functionally reduce the expansion of chronic inflammation by inhibiting the activation of the NF-κB pathway and suppressing the SASP, thereby achieving a certain regulatory effect on cell senescence. In conclusion, miR-142-3p plays an important role in regulating cell senescence and the subsequent development of IPF. Furthermore, our study highlights the therapeutic potential of miR-142-3p in the treatment of IPF.
Conclusion
In this study, miR-142-3p was identified to target p53 and NF-κB genes, and its expression level was significantly downregulated in bleomycin-treated AECs. Furthermore, in vivo, overexpression of miR-142-3p reduced pulmonary fibrosis in the bleomycin model of pulmonary fibrosis. More importantly, miR-142-3p overexpression reversed bleomycin-induced upregulation of P53 and NF-κB and collagen deposition. In conclusion, the results of this study revealed that miR-142-3p overexpression might ameliorate bleomycin-induced pulmonary fibrosis in mice by negatively regulating p53/NF-κB, and it may be a potential target for the treatment of IPF. But we did not find the signaling pathway of miR-142-3p regulates the p53/NF-κB network, which may be the direction of our next study.
Footnotes
Abbreviations
AECs, alveolar epithelial cells; IPF, idiopathic pulmonary fibrosis; SASP, senescence-associated secretory phenotype; IL-1β, interleukin 1-beta; MMP-9, matrix metalloproteinase 9; SA-β-gal, senescence-associated β-galactosidase.
Authors’ Contributions
SY, LHJ, ZK, and LSQ contributed equally to the conceptualization of this study, animal experiments, and preparation of the manuscript. XYG collected and statistically analyzed the data and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments in this study were approved by the Institutional Animal Care and Use Committee of the Hangzhou Hibio Technology Co. Ltd. (HBFM3.68-2015).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was supported by the Zhejiang Traditional Chinese Medicine Administration (2020ZB028).
