Abstract
Objective
To evaluate whether a fixed dose combination (FDC) with remogliflozin and vildagliptin as an add-on therapy can improve the glycemic control in the management of type 2 diabetes mellitus and also the non-glycemic effects on physical profile, blood pressure, lipids, and insulin resistance.
Materials and Methods
An observational study that included 50 poorly controlled diabetics from April 2021 to September 2021. Patients were divided into two groups – those who were prescribed this FDC by their treating physician as an add-on drug were formed as group 1 (n = 28). Comparison group was age-matched patients who received other standard anti-diabetic medications, categorized as group 2 (n = 22). Fasting and postprandial sugars were done at baseline, the third and sixth month; glycated hemoglobin, body mass index, blood pressure, and lipids were done at baseline and the sixth month. Changes in blood sugar levels and glycated hemoglobin (HbA1C) at the third and sixth month from the baseline were compared using the Mann-Whitney U test. P-value less than 5% was considered statistically significant.
Results
A statistically significant reduction in mean HbA1c was noted in group 1 [–1.80 (−3.20, −0.15)] when compared to group 2 [0.50 (0.05, 0.80)] at the end of the third month. At the end of the sixth month, a significant reduction in the HbA1c level was noted in group 1 [(7.83 ± 0.87 %) when compared to baseline (10.3 ± 1.75%)]. Change in PPBS value at the third month from baseline was also statistically significant between groups 1 and 2 (−62.0 mg%, 19.0, P = 0.003). With respect to the body mass index and blood pressure, we did not find any significant difference.
Conclusion
The fixed drug combination improves glycemic control by significantly reducing mean HbA1c at the third and sixth month from baseline and there was no significant effect on body mass index, blood pressure, and lipids.
Introduction
Diabetes mellitus is a chronic, progressive disease characterized by hyperglycemia, affecting the carbohydrate, protein, and fat metabolism and associated with microvascular and macrovascular complications. The pathogenesis of type 2 diabetes is complex, involving multiple mechanisms, and include reduced insulin secretion, decreased insulin sensitivity, and increased hepatic glucose production.1, 2 Hence, multiple strategies targeting this pathogenesis are often required for effective control of hyperglycemia to prevent complications. Also, an effective combination of drugs with complementary mechanisms of action may enhance the glucose-lowering effect without compromising drug safety. Over the last decade, a few newer classes of orally administered antihyperglycemic agents (OHAs) have been developed and they can effectively reduce glycated hemoglobin (HbA1c) and favorably affect body weight. This includes sodium-glucose cotransporter-2 (SGLT2) inhibitors and dipeptidyl-peptidase 4 (DPP4) inhibitors. Remogliflozin, which acts by inhibiting SGLT2, was licensed in India following the results of a clinical trial demonstrating its efficacy and safety when compared to dapagliflozin. 3 Despite the drawback of twice daily administration, this drug is considerably less expensive than other SGLT2 inhibitors. It may result in a treatment cost of less than half that of the other molecules in the same class. 4 Remogliflozin could be a helpful alternative for providing SGLT2 inhibitor therapy in developing countries where out-of-pocket expenses for drug purchases matter significantly for the general population and its favorable tolerability profile based on the safety data that are presently available.
SGLT2 inhibitors (SGLT2i) and DPP4 inhibitors (DPP4i) have a complementary mode of action that address multiple pathophysiological aspects; hence, the combination of these two drugs could be effective and safe in type 2 DM. Various studies have demonstrated that SGLT2i/DPP4i achieved better glycemic control and more significant weight reduction without increasing the risk of hypoglycemia in patients with inadequately controlled T2DM.5, 6 Although this combination therapy of SGLT2i–DPP4i provides multiple benefits in managing T2DM, only limited FDCs are available in India. The combinations available in the market include empagliflozin with linagliptin, dapagliflozin with saxagliptin, and dapagliflozin with sitagliptin. Another combination added to this list is the recent FDC of remogliflozin 100 mg + vildagliptin 50 mg.
Since the drug is recently introduced, information regarding its efficacy on glycemic control, body weight, blood pressure, lipid profile, and other markers of atherosclerosis is sparse. Various studies have shown that SGLT2i, such as empagliflozin, dapagliflozin, and canagliflozin, provide cardiovascular benefits in addition to glycemic control. 7 Regarding DPP4 inhibitors, none of the available drugs confer cardiovascular or renal benefits, although they are relatively safe and well tolerated. 8 The cardiovascular and renal effect of this combination therapy (remogliflozin + vildagliptin) is unknown; its effectiveness as an add-on therapy to existing anti-diabetic therapy is unknown. Hence, this study was planned in individuals with poor glycemic control to evaluate and compare the reduction in HbA1c between those who were prescribed this drug and those who were not prescribed it in addition to their existing anti-diabetic medications.
The primary objective of the study is to evaluate whether the fixed-dose combination of remogliflozin 100 mg + vildagliptin 50 mg can improve glycemic control in patients with poorly controlled type 2 diabetes mellitus. The secondary objective is to evaluate the non-glycemic effects of this fixed-dose combination on blood pressure, body mass index, lipid levels, and insulin resistance.
Materials and Methods
Study Design, Population, and Eligibility Criteria
A prospective, observational study was conducted in a tertiary care center from April 2021 to September 2021 after getting the Institutional Human Ethics Committee (PSG project number 21/103) approval. Adults diagnosed with type 2 diabetes attending general medicine OPD with inadequate glycemic control (glycated hemoglobin HbA1c ≥ 7% with existing anti-diabetic medications ± insulin) were included in the study. Patients who were prescribed remogliflozin 100 mg + vildagliptin 50 mg twice daily along with existing medications for better control by their treating physicians were considered as group 1; patients who were not prescribed remogliflozin 100 mg + vildagliptin 50 mg were taken as group 2. However, they continued to receive their existing anti-diabetic medications. Patients who are unwilling to participate, type 1 diabetes mellitus, history of prior diabetic ketoacidosis, estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m 2 , and pregnant or lactating women were excluded.
The sample size was estimated based on the HbA1c as an outcome at the end of 6 months. In order to observe a minimum difference in HbA1c as 0.7% between the various subgroups based on the study by Dharmalingam et al., 3 the number of subjects required would be 150 with a 5% level of significance and 80% power, after accounting for multiple comparisons and 20% attrition rate.
Methodology and Data Collection
Through this observational study, primary and secondary outcomes were assessed in the third and sixth month and were compared between groups 1 and 2. A detailed history regarding the duration of diabetes-associated co-morbidities and current medication was taken. A complete physical examination – measurement of height, weight, systolic blood pressure (SBP), diastolic blood pressure (DBP), and thorough systemic examination was done. Baseline laboratory evaluation includes the estimation of fasting blood sugar (FBS), post-prandial blood sugar (PPBS), glycated hemoglobin (HbA1C), renal and liver function tests, and fasting lipid profile. Insulin resistance was assessed for all the study participants and controls by calculating homeostatic model assessment for insulin resistance (HOMA-IR) using the formula HOMA-IR = (fasting insulin in mU/L × fasting glucose in mg/dL)/405; value > 2 indicates insulin resistance. 9 Blood sugars were estimated by hexokinase method COBAS C 501, Integra 400 plus; glycated hemoglobin was measured by high-performance liquid chromatography-Tosoh G8 variant; fasting insulin levels were measured by electrochemiluminescence E411 and fasting lipids were measured by COBES C501. Enzymatic endpoint for total cholesterol and triglycerides, direct method for high- and low-density lipoprotein, and calculation method for very low-density lipoprotein. Blood pressure and body weight were recorded by standard physical examination methods.
The primary outcome measure was the change in HbA1c at the third and sixth month from baseline. Secondary outcome measures included changes in mean blood pressure, body mass index, and lipid ratios in the sixth month from baseline.
Statistical Analysis
The assumption of normality was checked using Q-Q plot. Descriptive statistics were reported as mean and standard deviation for the normally distributed continuous variables; median with interquartile range for the non-normal data; numbers and percentages for the categorical variables. Chi-square test was used to test the association between categorical variables. An independent t-test was used to compare the glucose parameters between the study cases and the control groups. Within each group, change in glucose parameters from baseline to the third and sixth month of therapy was analyzed using paired t-tests. Change in the HbA1c at the third month and sixth month from baseline was compared using the Mann-Whitney U test. P value less than 5% was considered statistically significant. Analysis was performed using SPSS version 25.0.
Results
The present study was conducted in 50 diabetic patients with sub-optimal glycemic control. Since the study was conducted during the COVID-19 pandemic, we could not achieve the estimated sample size. Group 1 comprised of 28 people with diabetes with a mean HbA1c level of 10.3 ± 1.75%; group 2 comprised of 22 diabetic patients with a mean HbA1c level of 8.85 ± 1.91% at baseline. In group 1, one was on insulin only and not on any oral drugs due to intolerance (his HbA1c was 13.5%), four were on insulin in addition to OHAs, 27 were on metformin, 16 had received second generation sulfonylureas, four were on vildagliptin + sulfonylurea combination, five were on voglibose + sulfonylurea combination, and two had received pioglitazone + sulfonylurea combination. In group 2, almost all were on metformin; 20 were on two or more classes of OHAs and six were on insulin and oral anti-diabetic medications. Eleven in group 1 and one in group 2 were lost to follow-up in the third month. There was not much change in the physical activity or dietary pattern in both groups and there were no hypoglycemic events in either group. Genito-urinary infection (mild) was noted in one patient, which was treated appropriately; oral anti-diabetic drug dosage was reduced in three patients in group 1.
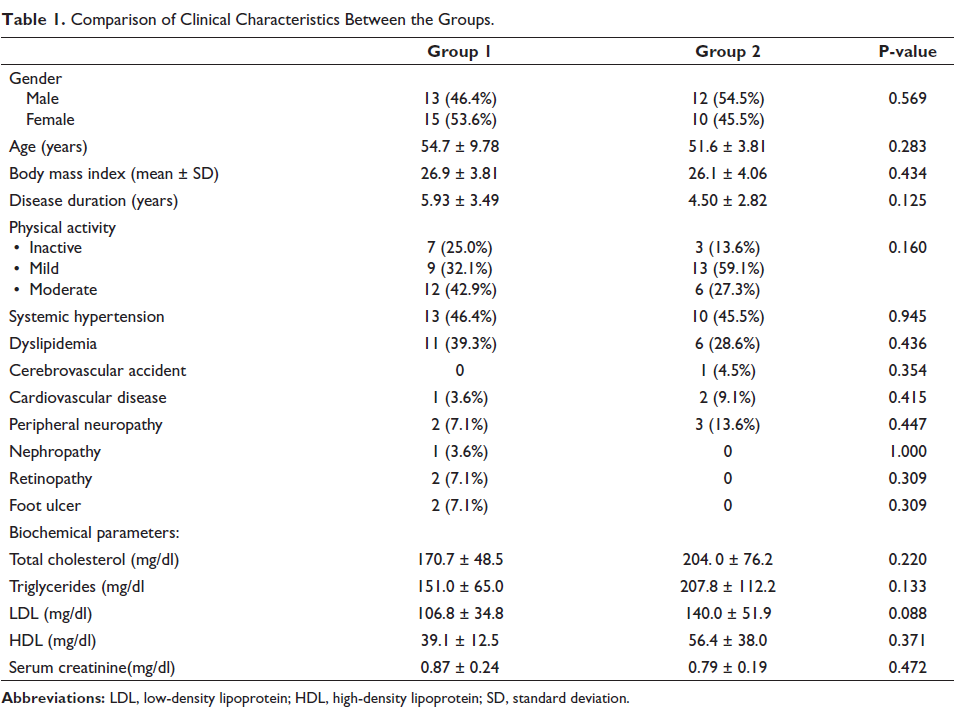
The mean age of the patients studied was 53.4 ± 9.97 years. Around 50% of the subjects in each group were males. The clinical characteristics such as age, body mass index, duration of diabetes, level of physical activity, presence of comorbid conditions such as systemic hypertension, dyslipidemia, cardiovascular disease, presence of target organ involvement, and lipid profile were comparable between the cases and the control groups, as given in Table 1.
Comparison of Clinical Characteristics Between the Groups.
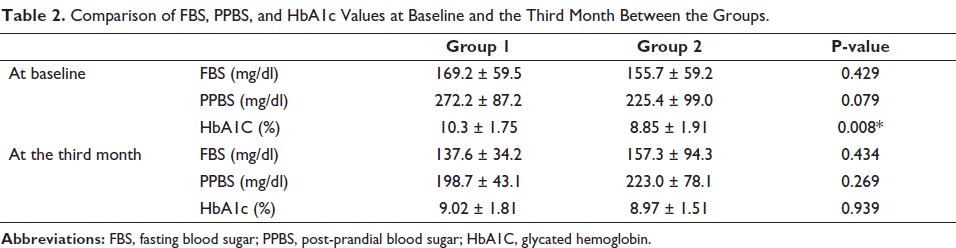
The baseline and change of blood glucose parameters between the two groups are presented in Table 2. The mean FBS and PPBS values in both groups were comparable at baseline. Group 1 had a significantly higher mean HbA1c than group 2 (10.3 ± 1.75% vs. 8.85 ± 1.91%, P = 0.008), as given in Table 2.
Comparison of FBS, PPBS, and HbA1c Values at Baseline and the Third Month Between the Groups.
Change in the blood glucose parameters at the third month of therapy from the baseline was compared between the two groups (Table 3). There was a reduction in FBS values in group 1 than group 2 at the third month, but this difference was not statistically significant. In group 1, FBS was reduced by 32.5 mg%, while in group 2, it increased by 1 mg% (P = 0.211). However, we noted that there was a more significant reduction in the PPBS (−62.0 mg%) and HbA1c (−1.80%) values at the third month in group 1, while in group 2, there was an increase in PPBS by 19.0 mg% and HbA1c by 0.5%. This change in the PPBS and HbA1c values at the third month from the baseline was statistically significant between the two groups (−62.0 mg% vs. 19.0 mg%, P = 0.003 and −1.80% vs. 0.5%, P = 0.007, respectively). Figure 1 depicts the comparison of the change in the HbA1c values between the two groups.
Change in FBS, PPBS, and HbA1c at the Third Month from the Baseline Between the Two Groups.
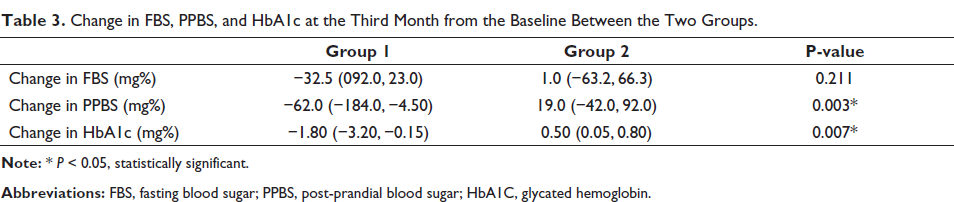
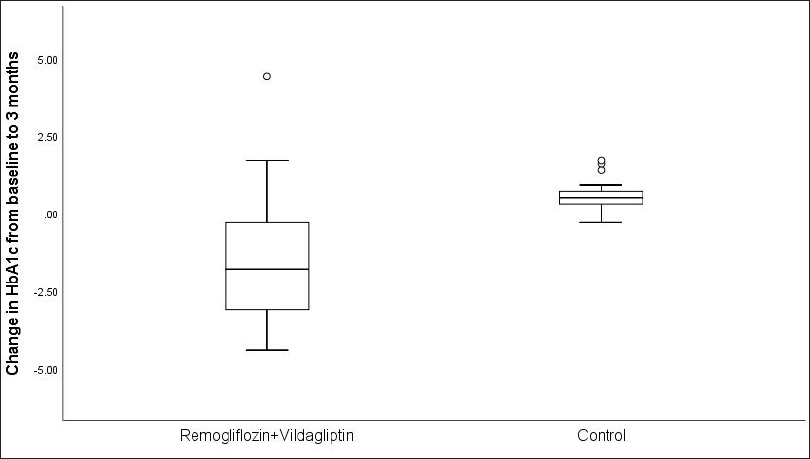
Eighteen patients in group 1 and almost all the group 2 patients did not complete the study period. The mean HbA1c at the baseline, third month, and sixth month were 10.3 ± 1.75%, 9.02 ± 1.81, and 7.83 ± 0.87 %, respectively, in group 1. The reduction in HbA1c at the third and sixth month from the baseline is statistically significant in the remogliflozin + vildagliptin-treated group (p < 0.05).
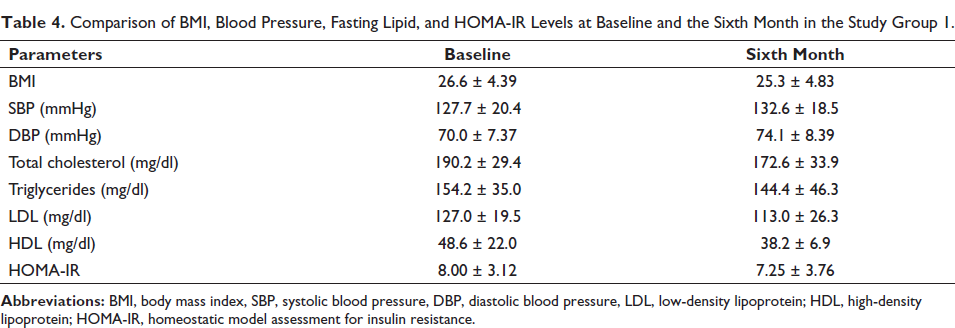
With respect to the body mass index and blood pressure, we did not find any significant difference in group 1 when compared to their baseline values (Table 4). We could get the fasting lipid profile and insulin levels at the sixth month in only five patients in group 1; the results shown in Table 4 showed no significant difference from baseline, although we did not perform statistical analysis because of lesser sample size.
Comparison of BMI, Blood Pressure, Fasting Lipid, and HOMA-IR Levels at Baseline and the Sixth Month in the Study Group 1.
Discussion
The effective glycemic control plays a crucial role in the management of type 2 diabetes mellitus to prevent complications. Therapeutic strategies targeting the various pathogenic mechanisms help to achieve better glycemic control. Newer classes of drugs such as gliptins (DPP4i) and SGLT2 inhibitors have been proven to be beneficial not only in glycemic control but also in providing additional benefits in terms of cardiovascular and renal protection, body weight, and blood pressure management. Although few FDCs combining these two classes of drugs are available in the market, the FDC of remogliflozin 100 mg + vildagliptin 50 mg has been available in the Indian market since 2020, which can be administered twice daily and is relatively less expensive.
The present study conducted in 50 diabetic patients with sub-optimal glycemic control has shown that this FDC of remogliflozin 100 mg + vildagliptin 50 mg effectively improves glycemic control in these patients with a significant reduction in HbA1c by 1.80% at the third month of the study period. This improvement in glycemic control was achieved with a greater significant reduction in PPBS values than FBS values. Interestingly, the reduction in the HbA1c at the third month was noted not only between the two groups but also within the group who received the FDC from the baseline to the third month and even extended to the sixth month. Since we did not have adequate controls at the sixth month, we could not analyze the change in HbA1c at the sixth month between the two groups.
The mean age of the study population noted in our study was similar to the one observed by Bhattacharyya et al., which was 54.8 ± 11.5 years. 10 A phase III study conducted by Khaladkar et al. compared the efficacy and safety of this fixed drug combination remogliflozin 100 mg + vildagliptin 50 mg (RV) added to metformin, with empagliflozin 25 mg + linagliptin 5 mg (EL). 11 It showed a significant reduction in the FBS, PPBS, and HbA1c in both the treatment arms and concluded that RV is non-inferior to EL. The mean change in the HbA1c value from baseline at week 16 in that study was −1.46% in the RV arm, similar to our observation of a decrease in HbA1c by 1.80% at the third month in group 1.
A study similar to our design was conducted by Bhattacharyya et al., which included 50 patients. 10 There was a statistically significant reduction in FBS, PPBS, and HbA1c after three months of treatment; the mean change of HbA1c at the third month from baseline was −1.9% (P < 0.0001) similar to the present study. They noted a significant reduction in the mean FBS value from baseline by 38 mg/dl (P < 0.0001); however, a non-significant reduction in FBS by 32.5 mg/dl (P = 0.211) was noted in the present study. The reduction in PPBS was comparable in both studies; they noted a change of −78 mg/dl (P < 0.0001), which was comparable with the present study (P = 0.003).
Body mass index, blood pressure, HOMA-IR, and lipid levels remain unaltered at the sixth month in the present study, although a small non-significant increase in DBP was noted in the present study. However, it should be remembered that there was a significant number of dropouts by the sixth month. A significant reduction in SBP (−3.5 mmHg, P = 0.03) was also noted in the study conducted by Bhattacharyya et al., but not for DBP and total body weight. 10
The present study is significant as it is the first of its kind that assessed the efficacy of the FDC of remogliflozin 100 mg + vildagliptin 50 mg. To the best of our knowledge, we could not find any other research articles on this fixed drug combination although we found a very few paper presentations in conferences.
Small sample size, non-randomized, and single center-based study design, and a short follow-up period with huge dropouts at the sixth month are some of the major limitations of the current study. Since the study was conducted during the COVID-19 pandemic, we experienced difficulties in recruitment and follow-up of the patients. The present study can be considered a pilot study. Given the abovementioned limitations, the study results need to be backed up by multicentric, randomized controlled trials with adequate sample size and follow-up. Future studies can also focus on other parameters such as carotid intima thickness and urine albumin creatinine ratio to study the cardiovascular and renal effect of this drug combination.
Conclusion
The fixed-dose combination of remogliflozin 100 mg + vildagliptin 50 mg is effective as an add-on therapy in improving the glycemic control in patients with poorly controlled type 2 diabetes mellitus with a 1.8% reduction in HbA1c at the third month when compared to baseline. No significant change was noted in the physical profile, blood pressure, lipid levels, and insulin resistance (HOMA-IR) at the sixth month. No major adverse effects were noted.
Footnotes
Abbreviations
FDC: Fixed drug combination
HbA1C: Glycated haemoglobin
OHA’s: Orally administered anti-hyperglycemic agents
SGLT2 i: Sodium-glucose Cotransporter-2 Inhibitors
DPP4 i: Dipeptidyl-peptidase 4 Inhibitors
T2DM: Type 2 diabetes mellitus
FBS: Fasting blood sugar
PPBS: Post-prandial blood sugar
HOMA-IR: Homeostatic Model Assessment for Insulin Resistance
Authorship Agreement
The manuscript has been read and approved by all the authors, and the requirements for authorship as stated have been met, and each author believes that the manuscript represents honest work.
Disclaimer
The views expressed in the submitted article are our own and not an official position of the institution.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethics approval was obtained from Institutional Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
Sources of Support
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
