Abstract
Objective
To evaluate the rationality of drug promotional literature (DPL) using World Health Organization (WHO) ethical criteria and to evaluate the perception of clinicians regarding DPLs using prevalidated criteria.
Methods
The promotional material was collected at a tertiary care teaching hospital over a period of 3 months. The perception of different prescribers has been changed. The data were analyzed using descriptive analysis.
Results
191 DPLs were collected from various outpatient departments at a tertiary care hospital. The information regarding brand name, generic name, and active ingredients was mentioned in 100% of the collected material. Therapeutic uses of the promotional agents were mentioned in only 60.20%; references were cited in only 15.18%. The adverse effects were listed in only 17.80% of the DPLs. The perception regarding DPLs was gathered from 27 physicians at the same institute. The percentage of clinicians who were aware of the WHO criteria used for analyzing DPL was only 37.03%. All healthcare workers have agreed on the necessity of including critical appraisal of the DPLs in the undergraduate curriculum.
Introduction
The aggressive marketing of products by pharmaceutical companies plays a major role in today’s era. The possibility of promotion being unethical is high as companies compete for better market share. 1 The baseline standards coded by the International Federation of Pharmaceutical Manufacturers and Associations are applicable universally for the purpose of marketing. 2 The ethical criteria for medicinal drug promotion were laid down by World Health Organization (WHO) in 1988 for the purpose of assisting and encouraging the rational use of drug products. 3 WHO recommends that DPLs provide genuine, satisfactory, and reliable information consistent with the scientific literature.4,5 In India, the Organization of Pharmaceutical Producers of India, the Self-Regulatory Code of Pharmaceutical Marketing Practices, January 2007, and the national legislation governs such promotional activities. 6
The WHO has defined drug promotion as “All the information and persuasive activities of manufacturers and distributors, the effect of which is to induce prescription, supply, and purchase and/or use of medicinal drugs.” 7 The different modes of drug promotions, such as medical representatives activities, holding conferences, offering incentives/gifts, and providing free drug samples to prescribers, are used by pharmaceutical companies. The materials that company representatives use for drug promotion are important information for physicians. 8 However, these commercial sources of drug information are not considered good sources of information, but even then, they are the first choice for providing drug information. 9 The different modes of drug promotion used by pharmaceutical companies include directed-to-prescriber advertisements, directed-to-consumer advertisements (DTCA), continuous medical education (CME), electronic detailing, internet-based drug promotion, and visual aids. 3
The purpose of such drug promotional materials is to raise awareness, but the information they provide might be misleading, wrong, and incomplete to draw any conclusions. 10 DPL can be highly instructive when providing authenticated information. 11 However, the evidence supports that DPLs used as the primary source of information by prescribers tend to cause irrational use of medicines. 12 The inclusion of different promotional strategies influences the prescribing pattern and is a topic of ethical concern. 13 Thus, it is becoming essential to gather information regarding the perception of physicians toward DPLs.
In the other studies, the prescribing patterns of the clinicians were stated to be influenced by the provision of samples and gifts from the medical representatives. 14 The need for clinicians to critically analyze the promotional literature before prescribing will be helpful in reducing the economic burden on patients. The rational use of medicines requires that patients receive medicine as per their needs and in doses that can fulfill their own requirements; this medicine too should be used for an adequate period of time at the lowest cost to the patients and to the community (as per WHO). 15 Pharmaceutical companies spend billions on the marketing of their products. Thus, such marketing might affect the prescribing patterns of the clinicians. 14
This study aims to evaluate the drug promotional literature (DPL) using the ethical criteria laid out by the WHO and assess clinicians’ perceptions of the validity of such materials so that relevant interventions can be made.
Materials and Methods
An observational study was conducted by the Department of Pharmacology at a tertiary care teaching hospital in North India. The purpose of the study was to collect DPLs from various outpatient departments for a period of 3 months (November 2021 to January 2022). The collected DPLs were evaluated for rationality as per the WHO ethical criteria. 7 The critical evaluation of DPL has been included in the curriculum of MBBS students by the National Medical Council of India, so the perception of students regarding DPLs has been collected.
The perception of clinicians regarding DPLs was assessed on the basis of a prevalidated questionnaire. The questionnaire was prepared to assess knowledge regarding WHO criteria for DPL evaluation, the influence of promotions on prescribing patterns, and the updation of knowledge about the new and in-the-market drugs. The questionnaire included 12 questions. Nine of the questions were closed-ended, and three questions were open-ended: (1) the time a medical practitioner can spend with a medical representative, (2) the other mode of drug promotion on which clinicians rely other than DPLs, and (3) which factor is considered as a priority in DPLs (Table 1). The responses were kept anonymous.
Results
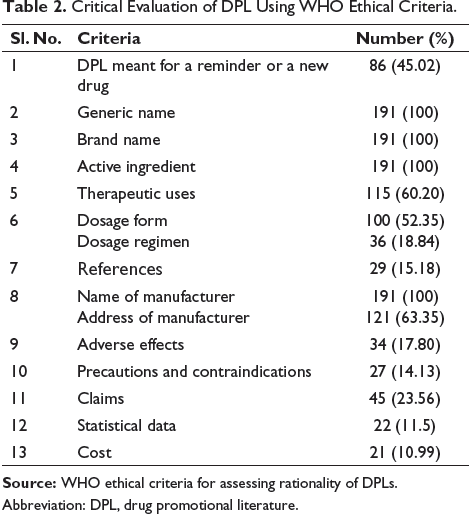
A total of 191 DPLs were collected and evaluated using WHO criteria. DPLs that promote devices and types of equipment and appliances in the orthopedic department were excluded. From the collected DPLs, the criteria given by WHO include generic name, brand name, and active ingredients, as well as the ethical promotion of the drugs. The brand names, generic names, active ingredients, and manufacturer names were mentioned in all of the collected DPLs. Therapeutic uses of the drugs were displayed in 60.20% of the DPLs.
The information regarding the dosage form was included in 52.35%. However, the dosage regimen was mentioned in only 18.84%. All of the promotional literature has the manufacturer’s name, though the complete address was only written by 63.35%. DPLs were also analyzed for mentioning the references, and the references for the promotional drug were mentioned in 15.18% of the DPLs. The information on the adverse effects, precautions, and contraindications was included in 17.80% and 14.13%, respectively. All the collected DPLs have used small fonts to write the information on the safety of the drugs. The claims were made in 23.56% of the collected data. The price of products was mentioned in 10.99% of the DPLs. The statistical data was visualized in 11.5% of the collected materials.
The strategy adopted by pharmaceutical companies is to market new drugs to physicians. Thus, it was necessary to know the perception of clinicians. The questionnaire to collect perceptions regarding DPLs was distributed to 58 clinicians, and it was filled out and collected from 27 clinicians (Table 1). Only 44.44% of prescribers read the complete information provided in promotional literature. Thus, the chances of ethical educational aspects getting compromised are very high. Only 37.03% are aware of the WHO guidelines.
The Perception of Clinicians Regarding DPLs.

In total, 7.40% of healthcare providers have agreed that DPL influences the prescribing pattern. Though none of the prescribers in this study have agreed that their prescribing integrity gets compromised by incentives/gratitudes from medical representatives. The DPL has been accepted as the only source for updating knowledge by 40.74% of clinicians.
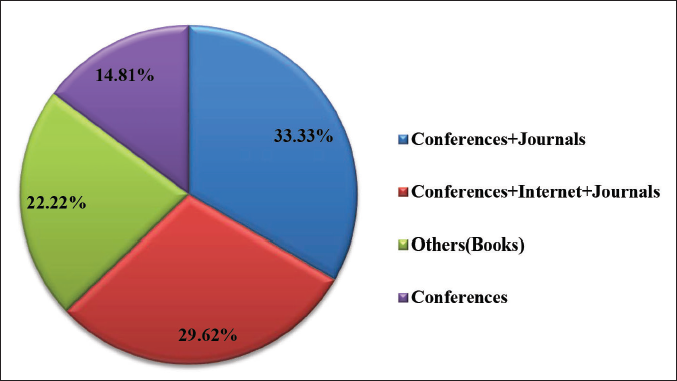
In the present study, 100% of clinicians agreed to spend 10–15 min with the medical representatives. The clinicians (33.33%) rely on conferences and going through journals to update their knowledge. The help of conferences, journals, and the internet is taken by 29.62% of prescribers. Another mode, such as books for updating knowledge, is preferred by 22.22% of clinicians.
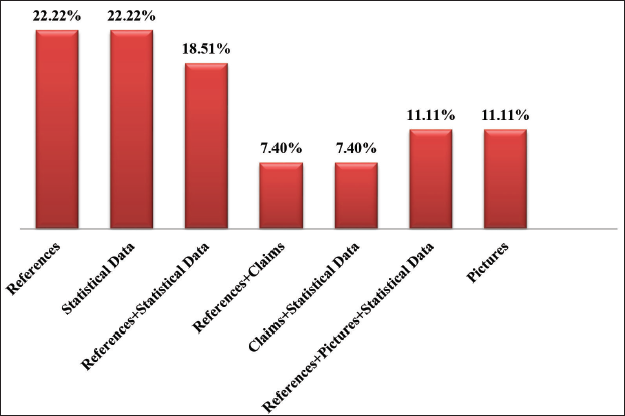
Statistical data and references are both important aspects of drug promotional advertisements and are considered a priority by 22.22% of healthcare providers. In total, 18.51% of clinicians agreed to use references and statistical data as a priority factor. References and claims are used by 7.40% of professionals, and claims, along with statistical data in DPLs, are used by 7.40% of professionals. 11.11% of clinicians have agreed to rely on pictures provided in advertisements, and the other 11.11% have agreed to use pictures, references, and statistical data. The MBBS students who have attended the class on DPL have found the topic to be very interesting, and 100% of them have agreed that it will be helpful in their future practice.
Discussion
The study was done for the purpose of evaluating the validity of the DPL as per the WHO criteria for the ethical promotion of drugs. In addition to that, the DPLs are a source of information about newer drugs or act as reminders for existing drugs for the healthcare workers. 16 Therefore, the perception of clinicians regarding the promotional materials has been evaluated.
In the present study, DPLs have fulfilled the WHO criteria of mentioning the generic name, brand name, active ingredients, and the name of the manufacturer, though the other criteria provided by WHO are not fulfilled by DPLs. This finding indicates that drug promotional companies are more involved in building a commercial relationship with physicians, whereas the ethical education aspect is getting compromised. This finding is similar to that of other studies.17,18 Most of the data mentioned generic names (100%), similar to another study done in India. 19 However, the therapeutic indications were mentioned in only 60.20%, which is in contrast to another study. 11 Instead of adverse effects and precautions being described as the major drug-related problems. 20 The adverse effects and precautions were mentioned in 17.80% and 14.13%, respectively (Table 2). These findings are similar to those of other studies.21,22 Moreover, the small font size used for this information makes it unnoticeable and not crucial. This ultimately clarifies the purpose of the manufacturer is to sell their product and not to convey all the information. It is also a requirement for pharmaceutical companies to provide references to make the information more reliable and valid. However, only 15.18% have given references. This finding in promotional literature is similar to other studies.23,24 In our collected DPLs, the dosage form was mentioned in 52.35%, but the regimen was given in only 18.84%. A similar observation has been found in the study by Jadav et al., where the dosage form was mentioned in 98% of DPLs while the regimen was given to only 3.5% of them. 24 The claims for misguiding the practitioners were found in 23.56% of the collected materials.
Critical Evaluation of DPL Using WHO Ethical Criteria.
The International Federation of Pharmaceutical Manufacturers and Associations mentions that the purpose of all conferences or any event for healthcare professionals that is organized or sponsored by a pharmaceutical company is to provide scientific or educational information to professionals. 2 Knowing the attitude and perception of clinicians regarding promotional literature can help to understand the areas in which the relevant interventions are needed. 13 Sales representatives and commercial sources are the most frequently used first sources of drug information, in spite of the fact that clinicians doubt their credibility.13,25 The prescribing behavior gets influenced by the drug promotion, but the accuracy of the prescription gets compromised. The decisions on drug prescribing get influenced by pharmaceutical drug information, as stated in the study by Ziegler et al. and Vencelik et al., respectively.26,27 Only 44.44% of prescribers read the complete information provided in promotional literature. The level of awareness regarding WHO criteria among clinicians is very low. Thus, the chances of ethical educational aspects getting compromised are very high. 27 Only 37.03% are aware of the WHO guidelines, though it has been stated that the prescriber’s attitude gets affected by drug promotion. 28 Hence, the utmost importance should be given to create awareness at the beginning of their careers. All of the clinicians have felt the need to introduce the learning of appraising given promotional literature at the undergraduate level, which is also found in the other study done by Sharma et al. 17 In another study, it has been stated that the integrity gets compromised by incentives given to doctors, 15 though in our study, none of the clinicians agreed with this point. In our study, 96.29% of healthcare providers accepted that they use journals or books to validate the information given in DPLs; this finding is similar to another study. 16
This study’s results will help clinicians critically examine the information given in DPLs before prescribing. In the present study, 33.33% of clinicians mentioned attending conferences and reading journals to update their knowledge of newer drugs (Figure 1). The most influencing parameter in the DPL by clinicians has been mentioned as being the statistical data (22.22%) and references (22.22%), a combination of both statistical data and references (18.51%) in the present study (Figure 2).
The critical evaluation of the DPL has been included in the new curriculum to train them at the beginning of their medical career. 29 Moreover, the “Bad Ad Program” has been introduced by the US Food and Drug Administration to provide education to professionals so that inaccurate promotions can be identified and reported to the agency. 30 Several initiatives have been taken to train doctors in scrutinizing drug information.29,30 To provide practical training for medical and pharmacy students, WHO and Health Association International have collaborated to produce a manual. 29 The references form an integral and crucial part of the promotional material, and the claims that companies make should have been made on the basis of the references. 31
This study has also highlighted the importance of implementing educational training for undergraduate medical students, as they also get exposed to drug promotion during their internship. Thus, the critical appraisal of promotional literature has been implemented in second professional MBBS at our college. The students are being taught the WHO criteria for analyzing the promotional material, and different DPLs are distributed among them. Then they are given a specific time period to evaluate the promotional pamphlets. After conducting this study, we will be able to implement the critical appraisal of DPLs at the undergraduate level more enthusiastically, which will help them in the future to prescribe rationally as per the needs of the patient.
Conclusion
An easily accessible, available, and also crucial source of drug information is printed promotional literature. The WHO guidelines are not met by pharmaceutical companies while promoting their products. The concern regarding critical appraisal skills has led the National Medical Council to incorporate critical evaluation of DPL into the new curriculum, 31 which will help them in the future to do rational prescribing. All the codes regulating drug promotion effectiveness, laws, and self-regulation to control drug promotion will become effective if the prescribers are educated on how to do a CRITICAL APPRAISAL of the promotional material.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from institutional ethics committee.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Informed consent was obtained from participants.
