Abstract
Monkeypox is a self-limiting zoonotic disease caused by the monkeypox virus belonging to the genus of orthopox viruses. Initially considered an ‘African disease’, this infection has crossed the boundaries to affect other continents and it has raised tremendous concerns among the general public as well as the medical fraternity all over the world, particularly because of the lack of specific vaccinations and drugs for the management of the illness. Epidemiological evaluation of the current infection has reported that it is mainly transmitted through sexual contact in bisexual men, mostly whites, and in those with pre-existing human immunodeficiency virus infection. The most common presentations were skin rash, anogenital lesions, or mucosal lesions along with systemic symptoms. It has been established that the vaccines and drugs approved for the management of smallpox could be used for the management of the current monkeypox outbreak. Vaccinia Immune Globulin (VIG) and vaccines like JYNNEOS and ACAM2000 and antiviral drugs like tecovirimat, cidofovir (CDV), and brincidofovir are being considered for those patients with serious diseases. It is imperative for physicians to understand the pharmacological aspects of these drugs for delivering better care to patients with monkeypox, which is eventually essential for the containment of this infection. This review covers updates on vaccines as well as drugs for the prevention and management of monkeypox.
Keywords
Introduction
Monkeypox is a zoonotic disease caused by monkeypox virus (MPXV), which belongs to the genus orthopox virus similar to the variola virus that causes smallpox. Monkeypox was a disease that was endemic to western and central Africa until recently. But since the last few months, isolated cases have been reported from the United States and European countries which has raised concern in global politics. As of 9 August 2022, more than 27,500 cases of monkeypox have been reported from 80 non-endemic countries. 1 Hence, the World Health Organization (WHO) declared monkeypox as a Public Health Emergency of International Concern on 23 July 2022, in order to strengthen global efforts to contain this illness. 2
Monkeypox transmits through large respiratory droplets, direct contact with skin lesions of infected patients as well as through contaminated fomites. Transmission can also occur via the placenta from mother to foetus, leading to congenital monkeypox. It is unclear whether transmission will occur through seminal or vaginal fluids. 3
Monkeypox is generally a self-limited infection with clade dependent fatality rates of 1% to 10%. 4 The infection begins with fever, followed by the appearance of popular, vesicopustular, and ulcerative lesions on the face and body with lymphadenopathy. 5 Complications like secondary bacterial skin infections, pneumonitis, encephalitis, and keratitis can occur in immunocompromised patients. 3
Even though monkeypox was endemic to African countries since the 1970s, 6 the disease was not adequately reported. Research about this zoonotic disease was often neglected and underfunded. Hence, the spread of this disease outside the African continent during recent years has caused concern as there are no specific drugs or vaccines for the disease. However, as MPXV is genetically similar to the smallpox virus, it has been considered that antiviral drugs and vaccines developed for the management of smallpox may be used for the prevention and management of monkeypox infection. 7 This review is a deliberate attempt to consolidate all the available evidences for the clinical management of monkeypox and the relevant pharmacological considerations recorded in the literature.
Management of Monkeypox
The primary aim of the management of a case of monkeypox should be to alleviate the symptoms, manage the complications, and prevention of long-term sequelae. The patient should be isolated and lesions in the skin and mucous membranes should be protected. The nutritional status of the patient should be well maintained with adequate fluids as well as oral supplementation. The patient should be on vigilant monitoring for any signs of deterioration of general condition like blurring of vision, breathing difficulty, altered consciousness, decrease in urine output, poor oral intake, or lethargy. 8
Specific Antiviral Therapy for Monkeypox Virus Infection
Monkeypox is usually a self-limiting disease and does not usually require any specific therapy except in certain cases of immunocompromised patients such as those with coexisting HIV infection, and young children who are at increased risk of developing complications. The prognosis of the disease depends on factors such as previous immunization status, general health of the individual, presence of concurrent illness, or other comorbidities. The Centre for Diseases Control (CDC) has outlined the following categories of patients who might require specific antiviral therapy for monkeypox:
Patients with severe illness like those presenting with the hemorrhagic disease, confluent lesions, septicemia, encephalitis, or those who require hospitalization. Immunocompromised patients like those with HIV-AIDS, malignancies, post-transplant patients, or those with autoimmune diseases with immunodeficiency. Paediatric patients. Patients with history or those presenting with dermatological conditions like atopic dermatitis, eczema, burns, impetigo, varicella zoster virus infection, and so on. Pregnant and lactating mothers. Patients developing complications like the secondary bacterial infection of the skin lesions, gastroenteritis, dehydration, bronchopneumonia, or other comorbidities.
8
As the above categories of patients are likely to develop complications of monkeypox, due to their underlying diseases and immune deficient status, it is recommended to administer specific drug therapy for these patients.
Pharmacological Classification of Drugs for the Management of Monkeypox
Table 1 summarizes the immunoglobulin, vaccines, and antiviral drugs used in monkeypox infection. The medications available for the management of monkeypox can be classified as follows:
Immune globulin and Vaccination
Vaccinia Immune Globulin (VIG) JYNNEOS ACAM 2000 Other vaccines—Aventis Pasteur Smallpox Vaccine (APSV), IMVANEX, Imvamune, LC16m8 Antiviral drugs Tecovirimat Cidofovir (CDV) Brincidofovir
List of Immunoglobulin, Vaccines, and Antiviral Drugs Used in Monkeypox.

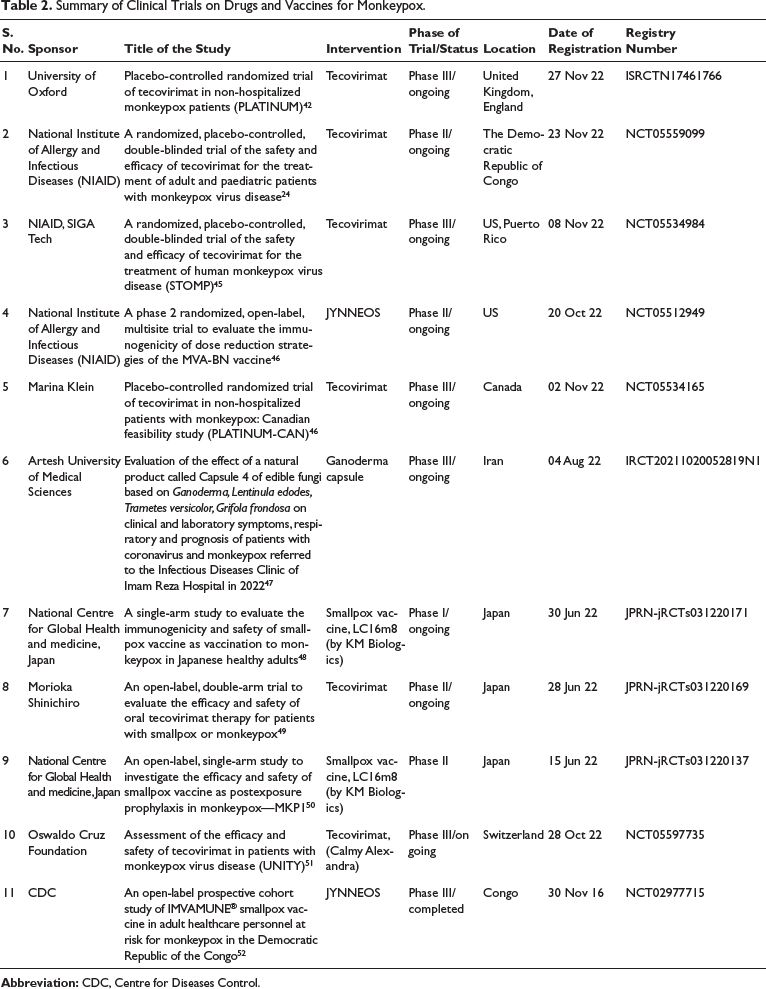
Summary of Clinical Trials on Drugs and Vaccines for Monkeypox.
Among the above, VIG and antivirals has been used in the management of monkeypox and the rest of the vaccines have been used in the prevention of the disease.
Pharmacological Aspects of VIG and Vaccinations for Monkeypox
Vaccinia Immune Globulin (VIG)
VIG was approved by the FDA in 2005 for the management of complications due to smallpox vaccination like eczema vaccinatum, progressive vaccinia, generalized vaccinia, vaccinia infections in patients with dermatological disorders, and those with accidental implantation of vaccinia virus in eyes, mouth, or other areas in whom vaccinia infection would be hazardous. 9 It is administered intravenously in a dose of 6000 U/kg and a repeat dose at 9000 U/kg if the patient is not responsive to initial dose, if necessary.
VIG is a solvent treated, sterile solution of purified gamma globulin (IgG) fraction of human plasma containing antibodies to vaccinia virus. This is collected from those donors who were administered booster shots of vaccinia virus vaccination. Hence, VIG provides passive immunity for patients with complications of vaccinia vaccination.
However, there are no data on the efficacy and safety of VIG in monkeypox. CDC has recommended to consider VIG for prophylaxis of monkeypox infection in those exposed individuals with immunodeficiency in T-cell function, in whom monkeypox vaccination is contraindicated. It is available as Expanded Access Investigational New Drug Application (EA-IND). 8
VIG is contraindicated in isolated vaccinia keratitis, history of anaphylaxis to parenteral administration of any human immune globulin preparation, and in IgA deficient patients. VIG is found ineffective in post-vaccinal encephalitis.
VIG should be used with caution in patients with pre-existing renal insufficiency and in those patients with a risk of developing renal insufficiencies like geriatric patients, history of diabetes mellitus, volume depletion, paraproteinemia, sepsis, and those receiving nephrotoxic drugs. VIG also exerts a potentially increased risk of thrombosis and is hence administered in patients at risk of hypercoagulability after assessing the risk-benefit ratio. Other complications that can occur following parenteral administration of VIG are aseptic meningitis syndrome, hemolysis, transfusion-related acute lung injury (TRALI), and transmission of other infectious agents through human plasma. 9
VIG administration may impair the efficacy of live attenuated vaccines like measles, rubella, mumps, and varicella. Hence, vaccination with live virus vaccines should be deferred till three months after the administration of VIG. 10 It belongs to pregnancy category C and there is no data regarding its safety in pregnancy and lactation.
JYNNEOS Vaccine
The JYNNEOS vaccine was approved for Emergency Use Authorization (EUA) by FDA on 9 August 2022, for individuals at high risk of monkeypox infection via two doses which are 4 weeks apart. It is a live, attenuated, non-replicating vaccine. It elicits humoral and cellular immune responses to orthopoxvirus infection and is originally approved in 2019 for the prevention of smallpox and monkeypox in adults over 18 years, who are at high risk of infection. 11 It is intended for individuals who are 18 years or older and is given via a subcutaneous route. In individuals less than 18 years, it is administered via the intradermal route. 12 It is a modified Vaccinia Ankara (MVA) vaccine. MVA is a live virus which does not replicate well in humans. 13
There are no data available at present regarding the efficacy of a single dose of this vaccine, in monkeypox which is crucial to control the current outbreak. It has been found to have a very good tolerability and safety profile and be safe in immunocompromised individuals as well during the clinical studies. 11 However, it was found to produce clinically significant ECG changes and caused elevation of Troponin-I levels two times above the upper limit of normal due to unknown reasons and were not considered to be serious. 11 It was not found to have any deleterious effect on pregnancy and lactation during preclinical studies and there are no evidence of its safety and effectiveness in paediatric population.
ACAM2000
ACAM2000 which was approved for immunization against smallpox in 2007 for high-risk individuals was approved for the current outbreak of monkeypox under the EA-IND. It is given as a single dose in individuals above one year of age to prevent monkeypox disease via percutaneous route via a bifurcated needle. 13 It is a live vaccinia vaccine. Vaccinia virus causes a localized infection of the skin at the site of injection, affecting the dermis and surrounding subcutaneous tissue and draining lymph nodes. The virus that may be present in the blood for a transient time will infect the reticuloendothelial system and other organs. The specific targets for virus replication in the early stage are the Langerhans cells in the epidermis. The development of protective immunity is marked by the formation of a pustule (pock or take) at the site of injection. The virus undergoes replication with the cells and viral antigens will be presented to the immune system. The long-term memory will be provided by neutralizing antibodies and T and B cells. 14 The efficacy and safety of ACAM2000 have not been studied in pregnant women, lactating mothers, and paediatric population. However, live vaccinia virus vaccination can cause serious harm to the foetus if administered during pregnancy like congenital malformations, preterm delivery, still birth or perinatal deaths. Live vaccinia virus may be transmitted through breast milk to the baby and can cause serious complications. 14 Additionally, it is marketed with special warning of risks of myocarditis and pericarditis. Rarely serious adverse effects like encephalitis, Steven Jonson Syndrome can occur.
Other Vaccines
Aventis Pasteur Smallpox Vaccine (APSV) was approved for smallpox under IND/EUA in the USA and is currently in the pipeline for a regulatory nod for emergency use in monkeypox. It is a live replicating virus vaccine that is derived from New York City Board of Health strain of vaccinia. IMVANEX is a live non-replicating vaccine produced from MVA attenuated, approved by the European Medicines Agency (EMA) for the protection of adults from monkeypox infection in July 2022.
This is marketed as ‘Imvamune’ in Canada and was approved in 2020 for post-exposure prophylaxis (PEP) in individuals with a recent high-risk monkeypox exposure. LC16m8 is a live replicating attenuated smallpox vaccine in the pipeline for regulatory approval for monkeypox in Japan.15–17
Post-Exposure Prophylaxis (PEP) for Monkeypox
It has been already established that vaccination is effective in preventing monkeypox infection in those health care workers (HCWs) exposed to the cases in Africa. 18 Multiple agencies like US FDA, National Institute of Health (NIH), and CDC are implementing vaccination strategies to contain the current outbreak. Individual can be vaccinated if there is suspected exposure to a case of monkeypox, within 4 days, which is considered as standard PEP. It is recommended to adopt preventive measures along with vaccination like self-isolation.
Expanded post-exposure prophylaxis (PEP++) is recommended in those individuals who are at high risk of developing monkeypox like recent exposure to monkeypox. This strategy is also employed in those areas with large numbers of monkeypox cases that suggest a high rate of MPXV transmission. 19
Pre-Exposure Prophylaxis (PrEP)
PrEP or vaccination before exposure to monkeypox is recommended in those individuals who have high risks of exposure to the virus. This includes individuals at risk of occupational exposure like laboratory personnel performing diagnostic testing for orthopox viruses.
At present two vaccines are being used to prevent monkeypox infection, the JYNNEOS which has FDA approval for the prevention of monkeypox and the ACAM 2000, which has been made available for use against monkeypox under EA-IND. The vaccination schedule has been discussed above.
Pharmacology of Antiviral Drugs Considered for Monkeypox
Tecovirimat
Tecovirimat (also known as TPOXX or ST-246) is an antiviral drug approved for the treatment of smallpox in 2018. It was proposed to use tecovirimat for monkeypox, and vaccination-associated adverse events as well as to counter the adverse effects of vaccinia oncolytic virus therapy. The antiviral action of tecovirimat is exerted through the inhibition of a major envelope protein involved in the production of new extracellular viruses. By stopping the virus from exiting an infected cell, it also reduces the likelihood of a virus spreading within the body. In vitro studies demonstrated that tecovirimat inhibited plaque formation and cytopathic effects induced by the virus.
The drug specifically binds to the orthopox virus protein F13 (also known as VP37 and p37), which is responsible for the production of new enveloped virions. The viral F13 target is highly conserved in all the members of the orthopox virus genus, hence, it can be used in a wide spectrum of orthopox infections. 20
Tecovirimat appeared to be readily absorbed on oral administration in both humans and animals. Absorption is enhanced by the presence of food in the stomach. In vitro metabolic stability studies has proven that tecovirimat is not a substrate of cytochrome P (CYP) enzymes and hence minimal drug interactions. It undergoes metabolism in the liver through glucuronide conjugation and is excreted mainly through the kidneys.
In a series of Phase 1 clinical trials of Tecovirimat in human healthy volunteers single daily dose of oral Tecovirimat (250 mg, 400 mg, 800 mg or placebo) given for a single day or 14 days and 28 days did not produce remarkable changes in lab assessment parameters, vital signs or ECG and was found to be well tolerated with similar adherence level to placebo.21–23 Currently, Tecovirimat is undergoing phase 3 trials in various locations around the world. 24
Tecovirimat is given in monkeypox at a dose of 600 mg per oral twice daily for 14 days (for 40–120 kg). For 25–40 kg, it is administered as 400 mg every 12 h and for 13–25 kg, it is given as 200 mg twice daily. If >120 kg, it is given as 600 mg thrice daily for 14 days. If the patient is unable to take it orally, tecovirimat is given as an intravenous infusion at the rate of 200 mg IV over 6–12 h for 14 days.
Nervous system disorders like migraine, disturbance in attention, headache, dizziness, gastrointestinal disorders like nausea, diarrhoea, vomiting, upper abdominal pain, constipation, dry mouth, dyspepsia, infections and infestations, nasopharyngitis, general and administration site adverse effects like fatigue, pyrexia, pain, and chills were the commonly reported adverse reactions with the use of Tecovirimat.
The major drug interaction reported with co-administration of Tecovirimat and repaglinide/midazolam is hypoglycemia/reduced effect of midazolam. Tecovirimat is contraindicated in patients with severe renal impairment. 24
There are evidences on the efficacy and safety of tecovirimat in case studies of monkeypox infection in the US in 2019 and the 2022 outbreak in several countries25, 26; however, we require more data from real-world evidences to substantiate the same. As of September 2022, enrolment of participants into Phase 3 clinical trial to evaluate tecovirimat has been started in the US and other countries to evaluate its efficacy on painful monkeypox symptoms and prevention of serious outcomes. 27
At present Tecovirimat is available in the US through the Strategic National Stockpile. Clinicians and care facility pharmacists needing to obtain oral tecovirimat supply should contact their state or territorial health department. In Europe, it is available for those patients with monkeypox-related hospital admissions.
Cidofovir (CDV)
CDV is a nucleoside analog approved in 1996 for the management of cytomegalovirus (CMV) retinitis in AIDS patients. It belongs to the class of acyclic nucleoside phosphonate (ANP). It exerts a broad spectrum of antiviral activity against DNA viruses like herpes, adeno, polyoma, papilloma and pox viruses. CDV has shown in vitro activity against orthopox and parapox viruses. It has been used in the management of recalcitrant molluscum contagiosum and orf virus in immunocompromised patients. 28
The ANP carries a negative charge due to the phosphonate moiety. This delays the cellular uptake of the moiety. CDV undergoes active membrane transport via an endocytosis like process characterized by slow kinetics and shows temperature dependence. The uptake of CDV is slower and less efficient than other nucleoside analogues which cross the cell membrane by nucleoside transport carrier system or via passive diffusion. 29 Inside the cytoplasm, CDV is converted to its active form, a diphosphoryl derivative, by cellular enzymes, the first step being catalyzed by pyrimidine nucleoside monophosphate (PNMP) kinase and the monophosphorylated derivative is converted to diphosphorylate form by nucleoside diphosphate (NDP) kinase, pyruvate kinase or creatinine kinase.30, 31 CDV has a significantly long intracellular half-life because of the diphosphoryl metabolites, which allow infrequent dosing and long duration of antiviral action.
The active form of CDV, CDV-diphosphoryl derivative (CDVpp), interacts with the DNA polymerase of the virus either by competitive inhibition or gets incorporated into DNA as alternate substrate. When two consecutive CDVpp chain are incorporated into the growing DNA chain, termination of the chain elongation happens leading to blockade of 3′ to 5′ exonuclease activity of the viral polymerase. Thus, it inhibits DNA synthesis and viral replication18–23. CDV resistance has been reported due to substitutions in the exonuclease domain and in the polymerase domain. 32
CDV is given intravenously at a dose of 5 mg/kg weekly for two weeks followed by 5 mg/kg once every alternate week. Renal function has to be strictly assessed before initiation of therapy. Oral probenecid as well as intravenous hydration are essential to minimize drug-induced nephrotoxicity. The nephrotoxicity is CDV is attributed to the accumulation of CDV and its metabolites in the renal tubular cells.
CDV has been found to have significant interactions probenecid, acyclovir and non-steroidal anti-inflammatory drugs. The major adverse effect noted with intravenous administration of the drug is nephrotoxicity. Local reactions at the application site have been reported with topical or intra-lesional CDV. 33
Data from animal studies proved that CDV may be used in the treatment as well as pre- and PEP of smallpox, monkeypox and vaccinia virus infections in humans. 34 In the current outbreak as well as in the previous outbreaks in 2018–2021 in the US, CDV has been found beneficial in the management of monkeypox in reducing the duration of skin lesions and duration of hospital stay.25, 26
Brincidofovir
Brincidofovir (BCV) is a prodrug containing CDV linked to a lipid through its phosphonate group. When administered orally, brincidofovir crosses the intestinal wall and penetrates the virus infected tissue and then it is cleaved to the antiviral agent CDV. CDV, in turn, is converted to cidofovir diphosphate, which has structural similarity to nucleotides, inhibits the viral DNA polymerase by competing with deoxycytosine 5-triphosphate (dCTP) and is incorporated into the growing viral DNA strands and blocks viral replication. 35
The conjugation of CDV with the lipid conjugate enhances the delivery of the drug to the target tissue as well as reduces the nephrotoxicity that is usually associated with CDV therapy.
Akin to CDV, brincidofovir has been investigated for the prevention and treatment of CMV, BK virus, adenoviruses, Epstein–Barr virus, and several ds DNA viruses in in vitro models.36–38 It was FDA approved for the treatment of smallpox in June 2021. Brincidofovir has been successfully used in the management of monkeypox in the 2018 outbreak in the US, where it was found to reduce the severity of skin lesions and their duration. 39
Brincidofovir (BCV) is administered as 200 mg per oral weekly × 2 doses (i.e., on Days 1 and 8) (for ≥48 kg, tablet or oral suspension) and for <48 kg as 4 mg/kg per oral weekly × 2 doses (i.e., on Days 1 and 8). 40
In preclinical studies BCV demonstrated a significantly higher penetration into lung, spleen, and liver tissues, albeit with lower concentrations in the kidneys. 41 Unlike CDV, BCV does not accumulate in the kidneys and has a lower risk for nephrotoxicity 42 and is reported to have no significant drug interactions. 43
Current Status of Clinical Trials on Monkeypox
The health agencies in the US and UK have launched several clinical trials to investigate the efficacy and safety of vaccines and antiviral drugs previously approved for monkeypox in various countries. As these drugs and vaccines were approved based only on animal studies and healthy participant data, these will be the first clinical trials investigating the efficacy of these drugs and vaccines. 44 Table 1 enlists few of the major studies registered in the International Clinical Trials Registry Platform.
The current monkeypox outbreak situation provides the opportunity for accrual of real-world evidence for correlation of protection against monkeypox and the duration of immunity. It is also possible to obtain additional safety data. Additionally, characterization of the immune response can identify potential biomarkers which are predictive of protection for monkeypox.
Conclusion
In this review, we have discussed in detail the pharmacology of vaccines and antiviral drugs approved for the management of monkeypox or being used under EA-IND, for sensitizing physicians. Meanwhile, the regulatory authorities should evaluate the feasibility and necessity of vaccination for the prevention and control of monkeypox.
Footnotes
Acknowledgment
The authors acknowledge the faculties of the department of pharmacology, SRM Medical College Hospital & Research Centre, who contributed towards various aspects of this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
There is no ethical approval associated with this paper.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed consent
Not applicable
