Abstract
Monoclonal antibodies (mAbs), which are commonly used to treat rheumatoid arthritis (RA), have been linked to a variety of adverse events (AEs). The objective of the study was to compare the safety profiles of six FDA approved mAbs (sarilumab, tocilizumab, adalimumab, golimumab, infliximab, and rituximab) marketed for the treatment of RA. A systematic review of the literature was conducted using the databases PubMed, Cochrane Library, and Science Direct. The manuscript comprised a total of 23 clinical studies. The percentage of patients who had AEs was calculated and presented using box-whisker and forest plots. Infections and infestations were found to be the most common AEs in RA patients treated with mAbs. Raised alanine aminotransferase (ALT), aspartate aminotransferase (AST), upper respiratory tract infection (URTI), and nasopharyngitis were frequently reported. The most common AEs were reported with adalimumab. The highest percentage of patients reporting AEs was associated with golimumab (52%), while rituximab had the fewest AEs (4.9%). In conclusion, rituximab appears to be a safer treatment option for RA as it is found to be associated with a lower risk of AEs, particularly respiratory infections.
Keywords
Introduction
Rheumatoid arthritis (RA) is a long-term autoimmune and inflammatory disorder that is characterized by painful, swollen joints, persistent synovitis, and autoantibodies, accounting for a large percentage of disability worldwide. Globally, RA was found to affect 460 people per 100,000 people during the period of 1980–2019. 1
The conventional management of RA includes pharmacological therapy with disease-modifying antirheumatic drugs (DMARDs) [which includes chemically synthesized (csDMARDs), biological origin (boDMARDs), biosimilar (bsDMARDs), and targeted synthetic (tsDMARDs)], nonsteroidal anti-inflammatory drugs (NSAIDs), and steroids, along with non-pharmacological measures such as physical therapy, emotional support, and rest. The updated guidelines from the European Alliance of Associations for Rheumatology (EULAR) and the American College of Rheumatology (ACR) recommend that DMARDs should be started as soon as possible after diagnosis and that ts/bDMARDs should be added if treatment goals are not achieved. The use of corticosteroids has been advised with regular monitoring of disease progression and adverse events (AEs), as they can increase the risk of infections, especially viral ones. As the management of RA usually spans a lifetime, the high risk of AEs with existing therapy poses a practical challenge. The rheumatologists are progressively looking at biological drugs, particularly monoclonal antibodies (mAbs), as a viable option. 2 In the current COVID-19 pandemic, the management of patients with RA has been fraught with numerous challenges. First, these patients have a higher risk of COVID-19 infection than the general population; second, the outcomes are worsened by COVID-19 infection; and third, during lockdowns, these patients experienced increased pain and functional impairment that severely affected their quality of life (QoL) and mental health. 3 Although most of the guidelines advised patients to continue the ongoing treatment, including NSAIDs, glucocorticoids (GCs), and cs/ts/bDMARDs, in clinical settings, the choice of therapy was often governed by the increased risk of AEs, particularly during COVID-19 infection and the subsequent recovery period.
Biological drugs/mAbs are most often used for the treatment of RA, particularly during the COVID-19 pandemic. The mAbs that have been approved for the treatment of RA (sarilumab, tocilizumab, adalimumab, golimumab, infliximab, and rituximab) have also been repurposed for use in the COVID-19 prevention and/or management. 4 The overview of approved mAbs is presented in Table 1. 5
Overview of Approved mAbs Used for the Treatment of RA.5
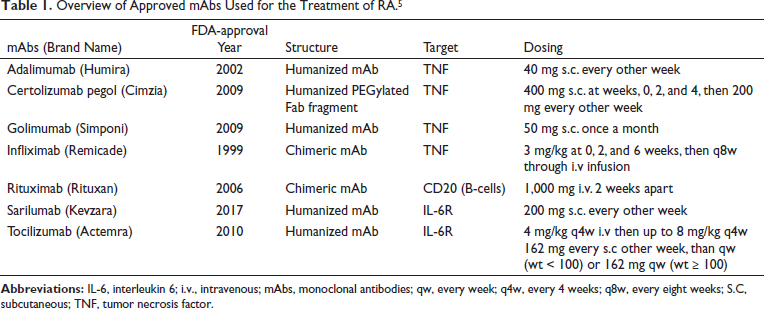
To the best of authors’ knowledge, scattered, singular reports of AEs and serious AEs associated with mAbs in RA treatment are available, but a comprehensive comparison of the safety profile is not available in the literature. This is essential as it can assist the clinicians to make rational drug choices that can improve QoL and safety of the patients, especially in the COVID-19 scenario, and the present systematic review addresses this lacuna.
Material and Methods
Results
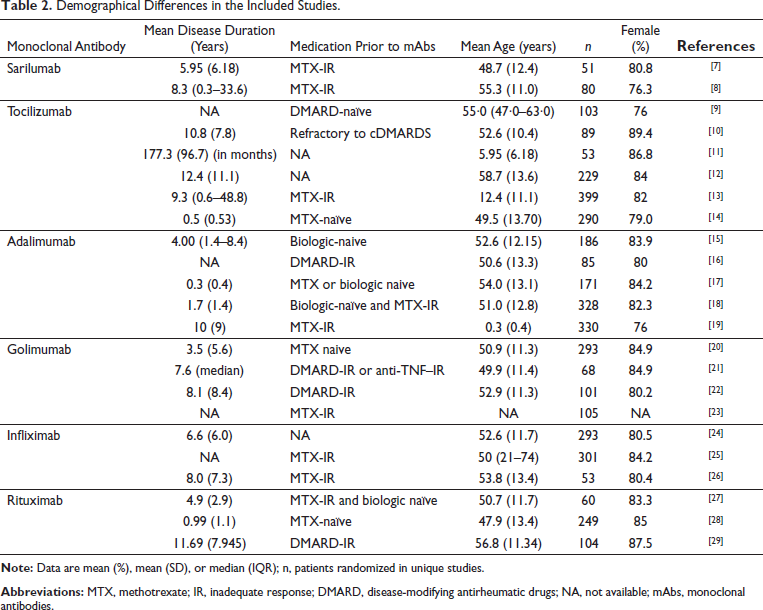
Demographical Differences in the Included Studies.
Adverse Events (AEs)
AEs are any unfavorable and unintended signs, including abnormal laboratory results, symptoms, or a disease associated with treatment.
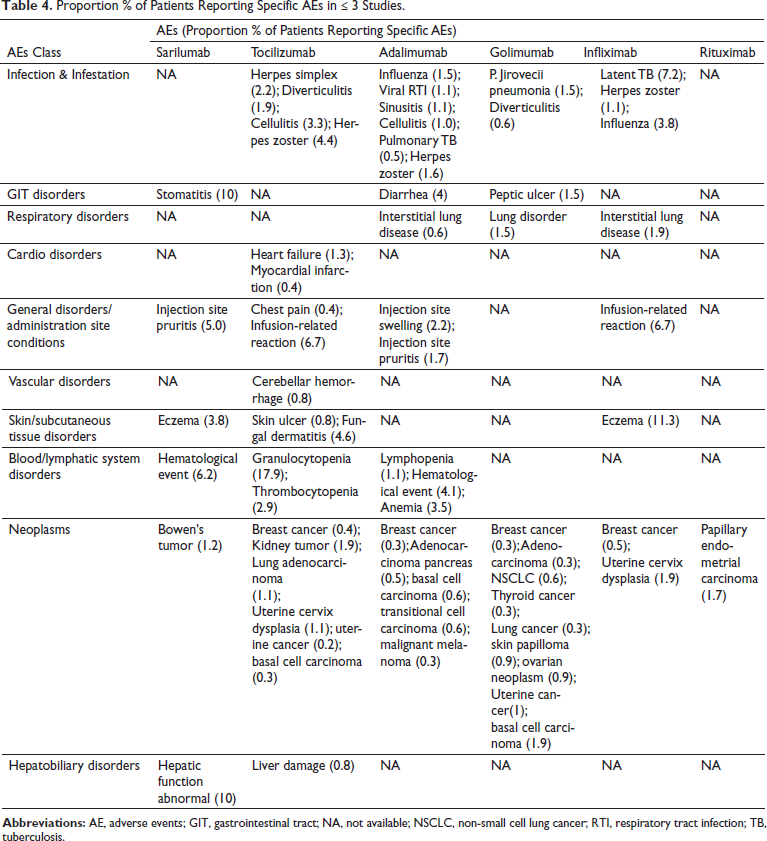
The reported AEs from each unique study were categorized according to System Organ Class (SOC), which are groupings by etiology (e.g., infections and infestations), manifestation site (e.g., gastrointestinal disorders), or purpose (e.g., surgical and medical procedures). In addition, there is a SOC to contain issues pertaining to products and one to contain social circumstances. The reported AEs were subcategorized according to the preferred term (PT). The proportion percentage of patients reporting specific AEs with different mAbs are presented in Table 3.
Comparison of Reported Adverse Events Among Six Approved mAbs in 23 Included Studies.
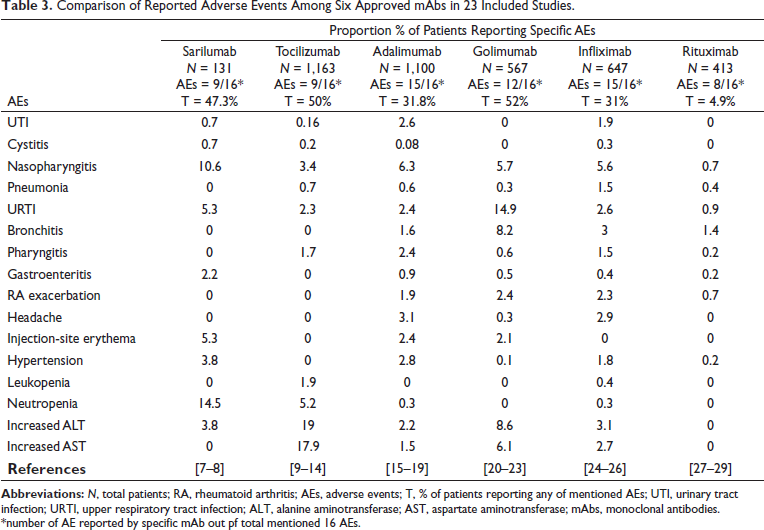
URTI was reported in 12 studies. The proportion of patients reporting AE was 14.9% and 5.3% with golimumab and sarilumab, respectively. Nasopharyngitis was reported in 13 studies. The proportion of patients reporting nasopharyngitis was 10.6% and 6.3% for sarilumab and adalimumab, respectively. Urinary tract infection (UTI) was reported in 7 studies. The proportion of patients reporting UTI was 1.9% and 2.6% with infliximab and adalimumab, respectively. Pneumonia was reported in 11 studies. For infliximab and tocilizumab, the proportion of patients reporting pneumonia was 1.5% and 0.7%, respectively.
Pharyngitis was reported in eight studies. The proportion of patients reporting pharyngitis was 1.7% and 2.4% with tocilizumab and adalimumab, respectively. Cystitis was reported in four studies. The proportion of patients reporting AE was 0.7% and 0.3% with sarilumab and infliximab, respectively. Bronchitis was reported in nine studies. For golimumab and infliximab, the proportion of patients reporting bronchitis was 8.2% and 3.0%, respectively. Gastroenteritis was reported in seven studies. The proportion of patients reporting gastroenteritis was 2.2% and 0.9% with sarilumab and adalimumab, respectively.
Neutropenia was recorded in seven studies. The proportion of patients reporting AE was 14.5% and 5.2% with sarilumab and tocilizumab, respectively. Leukopenia was reported in four studies. The proportion of patients reporting leukopenia was 1.9% for tocilizumab. Raised ALT was reported in 12 studies. The proportion of patients reporting raised ALT was 8.6% and 19% for golimumab and tocilizumab, respectively. Raised AST was reported in nine studies. The proportion of patients reporting raised AST was 6.1% and 17.9% for golimumab and tocilizumab, respectively.
Headache was reported in six studies. The proportion of patients reporting headache was 2.9% and 3.1% for infliximab and adalimumab, respectively. Hypertension was reported in eight studies. The proportion of patients reporting hypertension was 3.8% and 2.8% for sarilumab and adalimumab, respectively.
Exacerbation of the disease was recorded in seven studies. The proportion of patients reporting AEs was 2.4% and 2.3% for golimumab and infliximab, respectively. Erythema at the injection site was recorded in four studies. For sarilumab and adalimumab, the proportion of patients reporting erythema was 5.3% and 2.4%, respectively.
Proportion % of Patients Reporting Specific AEs in ≤ 3 Studies.
Discussion
RA is an autoimmune disease that is frequently associated with progressive disability, premature death, high morbidity, and a high socioeconomic burden. While there is no cure, physiotherapy and medication can help slow the disease’s progression, and DMARDs can be used to manage the majority of cases. 5
Several pathogenic mechanisms that may underpin RA have been identified over the last decade, which has aided in the development of targeted drugs. The therapeutic perspectives of RA patients have changed dramatically due to the development of biologics. Till now, the US Food and Drug Administration has approved more than ten biologics, including seven mAbs, fusion proteins, or soluble receptors. Biological agents are very large, complex molecules, and patents for many of these agents have expired, providing third parties the opportunity to develop their own biologicals or biosimilars. Biosimilars are not generic; they are similar but not identical to the original product. 30
The seven mAbs include four TNF-blocking agents (infliximab, 1999; adalimumab, 2002; golimumab, 2009; and certolizumab pegol, 2009), one ß-cell depleting mAbs (rituximab, 2006) and mAbs directed against the IL-6 receptor (tocilizumab, 2010 and sarilumab, 2017). To the best of authors’ knowledge, no originator mAbs are approved for the treatment of RA in India, but their biosimilars have been approved and marketed. Acellbia, Maball (rituximab); Adfrar, Exemptia (adalimumab); and Infimab (infliximab) are some of the approved biosimilars for the Indian market. 5 Current therapy for patients with RA includes a treat-to-target strategy rather than a non-targeted approach. 2
The mAbs (subcategory of bDMARDS) are a group of complex and effective biologic agents that have become an important series of options in the treatment of RA, but they also pose the problem of serious AEs, such as an increased risk of infections [like bacterial infections (particularly pneumonia), herpes zoster, tuberculosis, and opportunistic infections]. So, active pharmacovigilance (PV) in the form of maintaining a “patient registry”, electronic medical health records, and so on. would be necessary to pool safety data and further statistical analysis to provide a better understanding of the toxicity profile of each drug, which will further help to improve patient care and reduce the risk of negative side effects.
The post-marketing changes of biotherapeutics are one of PV’s primary global concerns. These changes can be low risk, moderate risk, and high risk. These changes suggest that biotherapeutics have an impact on the medicine’s safety and efficacy, and they can cause AEs in patients. 31
mAbs have a highly selective mechanism of action, a rapid onset, and a high rate of response. However, they are associated with high production costs and an increased risk of AEs, particularly infections, but not as much as cDMARDs. 5 Patients treated with bDMARDs, on the other hand, may be at a lower risk of COVID-19 infection, and their use is not associated with severe COVID-19 manifestations. 32 According to some studies, patients receiving anti-TNF agents do not have a higher risk of heart failure. When compared to cDMARDs, they did not have an increased risk of malignancies in general, but the risk of melanoma may be slightly increased. They can be used during pregnancy, as recommended by EULAR, and for this purpose, certolizumab pegol was recently approved by the EMA due to its low rate of transplacental passage. 30
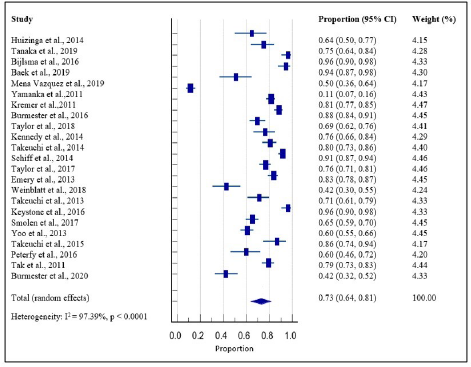
Here, the AEs of the mAbs were systematically assessed on the basis of the evidence from clinical studies of RA patients. A total of 23 clinical studies, investigating the safety of mAbs in RA patients, were retrieved. The pooled proportion estimate was found to be 0.73 with a 95% CI = 0.64, 0.81. In this study, it was identified that some of the AEs were rare but serious and may occur with the long-term use of mAbs. The pooled results show that infections and infestations [URTI and nasopharyngitis-PT] were the most common AEs associated with the use of mAbs. Elevations in liver enzymes (ALT and AST) were the second most common. AEs related to the hepatobiliary system and metabolism/nutritional disorders were the least common. Out of the published reports on clinical studies selected for this analysis, 8.8% of patients with an average treatment duration of 49 weeks discontinued the treatment due to AEs.
Of the six mAbs, most of the AEs were reported with adalimumab, while the highest percentage of patients reporting AEs was associated with golimumab (52%). URTI and nasopharyngitis were the most common AEs reported with all mAbs. Adalimumab was associated with the widest range of patients reporting UTI, headache, hypertension, and gastroenteritis. Tocilizumab was associated with the widest range of patients reporting nasopharyngitis, pharyngitis, neutropenia, leukopenia, raised ALT, and AST; sarilumab was associated with erythema at the injection site; infliximab with cystitis and pneumonia, golimumab with URTI, RA exacerbation and bronchitis; and rituximab was not widely associated with any unique AEs. Taken together, it may be concluded that rituximab was found to be the safest of all mAbs.
Tocilizumab was frequently associated with less common AEs like neoplasms and infections. Breast cancer was the most commonly reported malignancy with all mAbs except sarilumab and rituximab. The majority of the neoplasms were associated with golimumab, whereas sarilumab, infliximab, and rituximab appear to be safe.
On a positive side, with a better understanding of the pathogenesis of COVID-19, many anti-rheumatic drugs have been studied for its treatment. Tocilizumab is helpful in lowering biomarkers of COVID-19 infection, particularly CRP and ferritin levels. Physicians may consider using tocilizumab as a treatment for COVID-19 patients, particularly those with severe disease. 33
Exposure to mAbs was seen to be associated with many instances of AEs. Anti-TNF agents have been associated with an increased risk of infections as they can suppress the immune system 34 . Similarly, raised liver enzymes (AST and ALT) may be associated with the induction of autoimmunity or immune modulation. 35
Repeated electronic searches did not reveal any previous reports that assessed and compared the safety of all the classes of mAbs used for the treatment of RA. Previous report by Minozzi and co-workers, included anti-TNF agents and concluded that there was an increase in the occurrence of any infections, including serious infections. 36 In contrast to our study, Aaltonen et al. found that risk ratios reached statistical significance only in the number of patients experiencing injection or infusion reactions with the use of anti-TNF agents; however, results were expressed in comparison with the control group. 37 In our analysis, the injection or infusion reactions were reported in seven studies. Navarro and co-workers recorded that infections, liver function test abnormalities, and gastrointestinal side effects were among the clinically significant AEs associated with tocilizumab, and here we add blood disorders as another clinically significant AE. 38 An important report on rituximab revealed that the risk of developing AEs was comparable to that of other anti-TNF agents and that infections were more frequent when compared to controls. 39
There are very few safety and efficacy studies of mAbs in Indian patients with RA. Relevant Indian studies show that most of the AEs were mild to moderate in severity. At least one AE occurred in 36% of the patients. The most commonly reported AEs were infections and infestations. There were two serious AEs reported: sinusitis and viral infection. No deaths have been reported during the study. 40
Cases of tocilizumab-induced pancreatitis and rituximab-induced psoriasis and progressive multifocal leukoencephalopathy have been previously reported in the literature.41–43
In addition, our systematic analysis has numerous limitations, such that available data are scarce in terms of sample size and follow-up time. The safety profile of certolizumab pegol was not presented because no study was found using our search strategy. A high degree of heterogeneity [I2 = 97.3%; p < 0.0001] was observed in the effect estimates. Estimated effects differ across studies, partly because of random sampling error and partly because of heterogeneity. The statistic I2 is used in meta-analysis to estimate the fraction of variance due to heterogeneity. We calculate the bias of I2 in situations where the number of studies in the meta-analysis is small. 44
As the sample size for these studies varies and experience with the more recently introduced drugs is limited, the data pertaining to AEs for individual drugs may have been modified in comparison to their true incidence, introducing bias in this study.
As the treatment of RA requires long-term therapy and revised treatment guidelines are coming up with the introduction of newer mAb therapies, it is critical to monitor their safety profile to explore and ensure their long-term safety. In the future, PV studies must be able to swiftly identify new safety issues, such as which individuals are at risk of experiencing an AE and how the AE would manifest.
Evaluation of the mentioned factors during admission may aid in risk assessment so that efforts can be made for disease management as soon as possible.
In clinical decision-making, AEs are definitely important considerations in drug selection; however, this has to be balanced against effectiveness, affordability, and availability of information while making the actual choice. The decision regarding the clinical use of a drug should be balanced with all considerations.
This review may be useful in selecting appropriate treatment options. The need for active PV regarding the use of mAbs in RA is also highlighted.
Conclusion
Managing patients with RA, who appear to be particularly vulnerable to COVID-19 infection due to an already weakened immune system, was one of the challenges in the field of rheumatology. According to our study, rituximab may be recommended as a safer treatment option, particularly during COVID-19 pandemic, because it was found to be associated with a lower risk of AEs, especially respiratory infections, over the other approved mAbs. More direct studies are needed to further validate these findings.
Footnotes
Acknowledgment
The authors thank Mrs. Arshvir Kaur, Department of Pharmacology, DIPSAR, for helping in manuscript formatting and editing.
Authors’ Contributions
All authors have sufficiently participated in the intellectual content, conception, and design of this systematic review and meta-analysis. The collection, interpretation, and writing of the manuscript are done by Sweety Sharma and Dr. Rajani Mathur. Data analysis and manuscript editing are done by Dr. Ramesh K. Goyal, Sh. Somnath Basu, and Parbhat K. Sahoo. All authors have contributed equally to the manuscript revision and take public responsibility for the validity of the content therein.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
