Abstract
Objective
To evaluate the effect of p-coumaric acid (p-CA) on haloperidol-induced catalepsy in Swiss albino male mice.
Methods
To induce catalepsy, haloperidol (1 mg/kg i.p.) was administered for 21 consecutive days. p-CA (50, 75, and 100 mg/kg, PO) was administered 30 min before haloperidol injection for 21 consecutive days. For catalepsy, locomotor activity and motor coordination scores were recorded on the 17, 14, and 21 days of drug treatment, while the gait analysis score was recorded on day 21. After behavioral testing, animals were sacrificed, and various biochemical and histopathology tests of the brain were conducted. Dopamine, malondialdehyde, reduced glutathione (GSH), superoxide dismutase (SOD), and catalase activity were examined in the brain.
Results
Chronic administration of haloperidol significantly increased catalepsy in mice. It also produced hypolocomotion, motor coordination, and gait disturbance in mice. p-CA significantly inhibited haloperidol-induced catalepsy. Haloperidol significantly increased malondialdehyde levels in the brain. While dopamine levels in the brain dropped along with GSH, SOD, and catalase activity levels, which also had an impact on the histology of the brain. p-CA significantly reduced haloperidol-induced increases in brain oxidative stress, dopamine levels in the brain, and brain histology in mice.
Discussion
p-CA significantly reduced haloperidol-induced catalepsy, possibly through reducing oxidative stress and increasing brain dopamine levels. It can be a good candidate drug for extrapyramidal symptoms in Parkinson’s disease and adjuvant therapy with antipsychotic drugs.
Introduction
Numerous healthy components can be found in our diet, primarily plant-based foods. Polyphenols are one of the essential natural molecules among these substances. Phenolic chemicals are likely the largest type of secondary plant metabolite, and their primary role is to protect plants from ultraviolet radiation and diseases. p-Coumaric acid (p-CA), a hydroxycinnamic acid-family phenolic acid, is synthesized biologically via the shikimate pathway using phenylalanine and tyrosine as precursors. p-CA is present in free and bound forms in various plants and mushrooms. p-CA is found in a large variety of fruits and vegetables (e.g., apples, pears, grapes, oranges, tomatoes, and berries), as well as cereals (e.g., maize, oats, and wheat). Extensive research has demonstrated that they exhibit a variety of bioactivities, including antioxidant, anti-inflammatory, antimutagenic, antiulcer, antiplatelet, and anticancer properties, in addition to reducing neuronal injury, anxiety, gout, and diabetes. 1
According to previous studies, p-CA is responsible for the inhibition of oxidative stress by scavenging reactive oxygen species, 2 a neuroprotective action, 1 a potent inhibitor of 5-s-cysteinyldopamine, 2 a catechol-O-methyltransferase (COMT) inhibitor, the promotion of the brain-derived neurotrophic factor (BDNF). 3 Since p-coumaric acid has been reported to possess neuroprotective and antioxidant activities, this bioactive compound may have the potential to meet haloperidol-induced catalepsy. Catalepsy is a central nervous system disorder classified as extrapyramidal movement disorders. It is characterized by extremity muscular rigidity, postural fixation, and diminished sensitivity to pain. 4 Catalepsy is a symptom of several different types of nervous system disorders or conditions, including Parkinson’s disease (PD) and epilepsy. Haloperidol, an antipsychotic drug often used to treat schizophrenia, may be a factor in the development of this condition. 5
Haloperidol is the first of the butyrophenone series of major tranquilizers, and it is also the most widely used. Haloperidol is indicated for the treatment of psychotic disorders and their manifestations, such as hallucinations. 6 It has been connected to the inhibition of monoamine transport in the brain, specifically the cessation of impulse transmission in dopaminergic neurons. 7 Haloperidol, a commonly used neuroleptic for psychosis, is limited by its proclivity for causing extrapyramidal movement disorders, including tardive dyskinesia, akathisia, akinesia, dystonia, and Parkinsonism.
There are two possible mechanisms for haloperidol-induced catalepsy. (1) Auto-oxidation of haloperidol or degradation by an oxidase, which produces significant amounts of oxyradicals and a hazardous pyridinium-like metabolite that attacks the complex 1 reaction. 8 (2) Chronic blockage of dopamine D2 receptors in nigrostriatal neurons of the brain by neuroleptics results in an increase in dopamine turnover in the basal ganglia, which may result in an excess of free radicals like dopamine quinone and hydrogen peroxide via monoamine oxidase activity. 9 The purpose of the present study was to evaluate the neuroprotective effect of p-coumaric acid (p-CA) in haloperidol-induced catalepsy in mice.
Materials and Methods
Experimental Animals
Healthy male Swiss albino mice ranging between 25 and 30 g were used in the study. Animals were obtained from the R. C. Patel Institute of Pharmaceutical Education and Research, Shirpur. Animals were housed in well-ventilated polypropylene cages (Size: 465 × 300 × 150 mm). A total of six cages and six animals per cage were maintained under standard conditions such as 25 ± 2°C and 12–12 h light/dark cycle in the departmental animal house. The animals were given regular pelletized feed (Nutrivet Life Science, Pune) and water. The study protocol was approved by the Institutional Animal Ethics Committee (Approval No. IAEC/RCPIPER/2020-21-15) of the R. C. Patel Institute of Pharmaceutical Education and Research, Shirpur, India.
Drugs and Chemicals
Haloperidol (RPG Life Sciences Ltd., Mumbai, India), (p-Coumaric acid, Sigma-Aldrich, St Louis, USA), dopamine hydrochloride (SRL Diagnostics), and bromocriptine (Serum Institute of India). All the other reagents used in the study were of analytical grade only. p-CA was suspended in a 0.5% w/v carboxymethyl cellulose solution (CMC). The haloperidol injection was diluted in normal saline (0.9% w/v sodium chloride). The pharmacological doses were chosen as per the previous studies, that is, 1 mg/kg for haloperidol; 50, 75, and 100 mg/kg for p-coumaric acid; 10 and 2.5 mg/kg for bromocriptine. 11
Experimental Design
After a 1-week acclimation period, mice were randomly divided into six groups, each containing six animals (n = 6), and subjected to various drug treatments for 21 days. The first group was taken as the normal control group, and animals were administered orally with vehicle (0.5% CMC) alone. The second group was considered the positive control group. Animals were administered haloperidol 1 (0.9% w/v NaCl) mg/kg i.p. once a day for 21 days. 12 The third group was considered a standard group and received bromocriptine 2.5 mg/kg orally with haloperidol 1 (0.9% w/v NaCl) mg/kg i.p., while groups 4th, 5th, and 6th received p-CA p.o. at doses of 50 mg/kg, 75 mg/kg, and 100 mg/kg, respectively, for 21 days with haloperidol 1 mg/kg i.p. Catalepsy, locomotor activity, gait analysis, and motor impairment were measured in animals every week up to the end of the study (on days 7, 14, and 21). The animals were decapitated after treatment, and brain samples were dissected and rinsed in ice-cold saline. Brain samples were immediately fixed in a 10% formalin solution for histopathological analysis. The remaining midbrain was utilized to estimate oxidative parameters, whereas the forebrain was used to estimate dopamine. 4
Behavioral Assessments
Catalepsy Test
According to a modified protocol, catalepsy was assessed with a bar catalepsy instrument on days 7, 14, and 21 after haloperidol and p-CA administration, respectively. After 60 min of haloperidol administration, the animal’s forepaws were placed on a horizontal bar mounted 3 cm above the surface. The step-down duration was recorded on three consecutive trials using a 300-s stop criteria. 13
Rotarod Test
The apparatus consists of a horizontal metal rod (6 cm in diameter) connected to a variable-speed motor. A partition disc divides the rod into five parts (10.5 cm in diameter). Prior to the start of the study, the mice were trained on the rotarod apparatus, and rotarod tests were conducted on days 7, 14, and 21. The average length of the mice from each group stayed on the rotarod apparatus on average. 14
Locomotor Activity
Locomotor activity was used to determine the effect of p-CA on locomotion. An actophotometer (orchid) was used to determine the locomotor activity for 5 min; mice were placed in the actophotometer for 5 min, and the total (X and Y axes = T) number of line crossings was recorded. 15
Footprint Test
The mice were walked on the manually made catwalk apparatus (which consists of a glass slide), and images were captured from the downside. The images were then sent for fingerprint analysis with the help of ImageJ software. The stride length was measured in centimeters. 16
Oxidative Stress Parameters
Preparation of Brain Homogenate for Oxidative Parameter
Homogenate was prepared using a homogenizer with 10% (w/v) phosphate-buffered saline (0.1 mol/L, pH 7.4) and KCl. The supernatant was obtained by centrifuging at 10,000 rpm for 10 min at 40°C. The supernatant prepared in PBS was used to measure SOD, catalase, and GSH. The lipid peroxidation was measured using the supernatant prepared in KCl.
Estimation of Superoxide Dismutase in Brain Tissue
The method described by Kono determined superoxide dismutase (SOD) activity.18, 19 A mixture of 100 µl of 500 mM Na2CO3, 100 µl of 1 mM EDTA, 100 µl of 240 M/NBT, 640 µl of distilled water, 10 µl of 0.3% Triton × 100, and 25 µl of 10 mM hydroxylamine were added to 25 µl of tissue supernatant. At 560 nm, the values were taken spectrophotometrically in kinetic mode at 1- to 3-min intervals. The enzyme activity was measured in units per milligram of protein.
Estimation of Reduced Glutathione in Brain Tissue
The reduced glutathione (GSH) was calculated using the Ellman method; 17 in brief, 20 µl of tissue homogenate was mixed with 180 µl of DTNB. Read the developed yellow color at 412 nm at 37°C. GSH concentrations were measured in µg/mg of protein.
Estimation of Lipid Peroxidation (MDA) in Brain Tissue
Lipid peroxidation was estimated as per Ohkawa et al. MDA concentration was used to evaluate lipid peroxidation in brain tissue. MDA concentration was used to assess lipid peroxidation in brain tissue; 18 100 µL of the tissue supernatant was mixed with 600 µL of 1% w/v phosphoric acid and 200 µL of 0.6% thiobarbituric acid w/v. The reaction mixture was heated to 85°C for 45 min before cooling in an ice bath. After cooling, 4 mL of n-butanol was added to it, vortexed, and centrifuged at 5,000 rpm for 10 min, the organic layer separated. The absorbance of the generated pink color in the organic layer was measured at 532 nm. The concentrations were represented as nmol/mg of protein.
Estimation of Catalase Level in Brain Tissue
Catalase activity was determined using the method published by Aebi (Aebi, 1984), 19 which consisted of mixing 50 L of tissue supernatant with 1.0 mL of 50 mM phosphate buffer (pH 7) and 0.1 mL of 30 mM hydrogen peroxide. The optical density was determined by the drop in absorbance, which was determined spectrophotometrically every 5 s for 30 s at 240 nm.
Estimation of Dopamine Levels in Brain Tissue
The dopamine level in the forebrain was determined using the UV spectrophotometer method. 20 The forebrain was weighed and homogenized in 3 mL HCl–Butanol (0.85 mL 37% HCl in 1 L n-butanol) for 1 min in a 0°C. The sample was then centrifuged at 0°C for 10 min at a speed of 2,000 rpm using a chilled centrifuge. A volume of 0.8 mL of the supernatant phase was transferred to a tube containing 2 mL of heptane and 0.25 mL of 0.1 M HCl. After 10 min of shaking the samples vigorously in the same way, the two phases were separated by spinning them in a centrifuge. For estimation, the top organic phase was discarded in favor of the aqueous solution.24, 25
The dopamine level in mouse brains was estimated by admixing the above prepared homogenized supernatant liquid (1 mL) with 1 mL of ferric chloride (1.5 × 10−2 M) and 1 mL of potassium ferricyanide (1.5 × 10−2 M) in 25 mL of distilled water. It was kept aside for 30 min, and the developed color was estimated using the UV visible double beam spectrophotometer at 735 nm.23, 26
Histopathological Examination
The brain tissue was removed from the mice immediately after they were sacrificed for histological analysis.
A total of six cages, with six animals in each cage, were obtained, and representative samples of each group were fixed in 10% phosphate-buffered formalin, dehydrated in graded alcohol, and embedded in paraffin sections of 3–4 micron thickness. This section was mounted on glass slides and counterstained with hematoxylin and eosin. The slides were examined under a light microscope at 40× magnification for signs of neuronal death in the substantia nigra.
Statistical Analysis
The resultant data were expressed as means and subjected to a one-way analysis of variance (ANOVA) using GraphPad Prism version 8.00 software for Windows. Bonferroni’s multiple comparison test was used to determine the statistically significant difference between the groups. A P-value less than 0.05 was considered a significant difference between the groups.
Results
Effect of p-CA on Catalepsy Time in Haloperidol-Induced Catalepsy in Mice
Haloperidol (1 mg/kg i.p.) significantly increased the catalepsy (Figure 1) on days 7 (P < 0.001), 14 (P < 0.001), and 21 (P < 0.001) of treatment as compared to the vehicle-treated control.
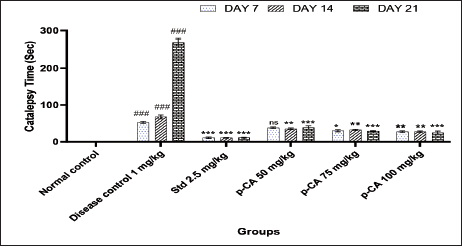
Treatment of p-CA at doses 75 (P < 0.05) and 100 (P < 0.01) mg/kg showed a significant reduction in catalepsy time on day 7 when compared to the disease control group. On days 7 and 14, catalepsy time was significantly reduced at all dose levels. Also, the p-CA showed a significant reduction in catalepsy time when compared to the disease control group (Figure 1).
Data were analyzed using a two-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, *P < 0.05, **P < 0.01, and ***P < 0.001 when compared to the disease control group, ns, non-significant, DF = 5.
Effect of p-CA on Motor Coordination in Haloperidol-Induced Catalepsy in Mice
As depicted in Figure 2, the retention time in the disease control group was significantly reduced at all intervals compared to the normal control group. The high dose of p-CA (75 mg/kg, significant but less than 100 mg/kg) showed a significant increment in retention in cataleptic mice on days 14 (P < 0.05) and 21 (P < 0.01). In addition, intermediate dose of 75 mg/kg showed a significant increment I retention on days 14 (P < 0.05) and 21 (P < 0.01). The standard treatment of bromocriptine showed a significant increment in retention time at all week intervals.
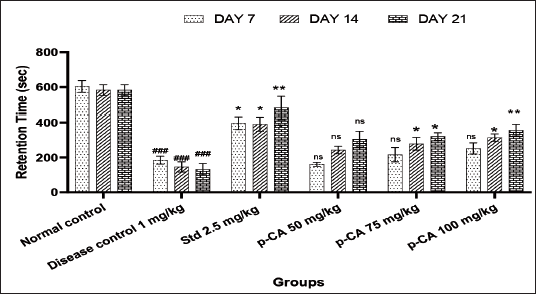
Data were analyzed using a two-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, *P < 0.05 and **P < 0.01 when compared to the disease control group, ns, non-significant, DF = 5.
Effect of p-CA on Locomotor Activity in Haloperidol-Induced Catalepsy in Mice
Haloperidol (1 mg/kg i.p.) significantly decreased locomotor activity (Figure 3) on days 7 (P < 0.001), 14 (P < 0.001), and 21 (P < 0.001) of treatment as compared to the vehicle-treated control. p-Coumaric acid at 50, 75, and 100 mg/kg orally on day 7 (ns, P < 0.01 and P < 0.001, respectively), day 14 (P < 0.01, P < 0.01, and P < 0.001, respectively), and day 21 (P < 0.001, P < 0.001, and P < 0.001, respectively) increased locomotor activity significantly in dose-dependent manner when compared to the disease control group.
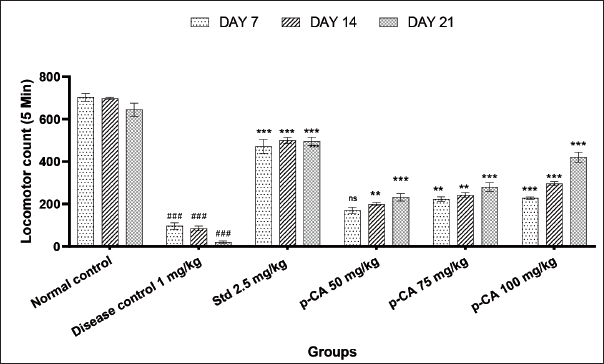
Data were analyzed using a two-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, *P < 0.05, **P < 0.01, and ***P < 0.001 when compared to the disease control group, DF = 5.
Effect of p-CA on Gait Impairment in Haloperidol-Induced Catalepsy in Mice
As depicted in Figure 4, haloperidol (1 mg/kg i.p.) significantly decreased stride length (Figure 4) on day 21 (P < 0.001) of treatment as compared to the vehicle-treated control. Treatment with p-CA of 75 mg/kg (P < 0.05) and 100 (P < 0.01) mg/kg increased stride length significantly in dose-dependent manner when compared to the disease control group on day 21.
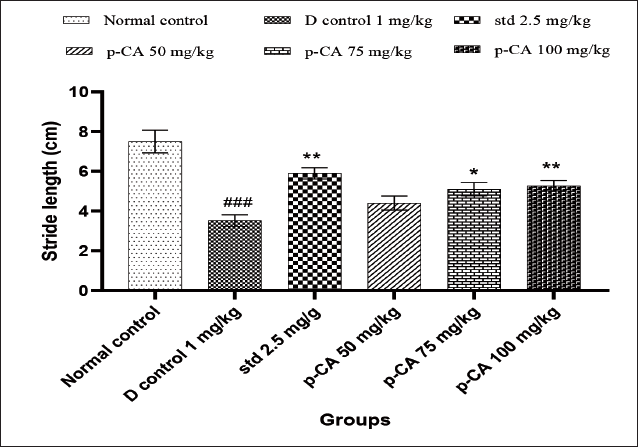
Data were analyzed using a one-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, *P < 0.05 and **P < 0.01 when compared to the disease control group.
Effect of p-CA on Haloperidol-Induced Changes in Brain Levels of GSH, Malondialdehyde (MDA), Catalase (CAT), and SOD Activities
As shown in Table 1, haloperidol (1 mg/kg) significantly decreased GSH, SOD, and CAT activities when compared to the normal control and significantly increased MDA activity when compared to the normal control, while treatment with daily doses of p-CA (50, 75, and 100 mg/kg) for 21 days increased GSH, SOD, and CAT activities and also decreased MDA activity when compared to the disease control group. A daily dose of p-CA (50 mg/kg) for 21 days significantly increased SOD (P < 0.05) and CAT (P < 0.05) activities. In addition, intermediate (75 mg/kg) and high (100 mg/kg) doses of p-CA showed significantly increased GSH, SOD, and CAT activities and decreased MDA activity.
Effect of p-CA on Haloperidol-Induced Changes in Brain Levels of Reduced Glutathione (GSH), Malondialdehyde (MDA), Catalase (CAT), and Superoxide Dismutase (SOD) Activities.
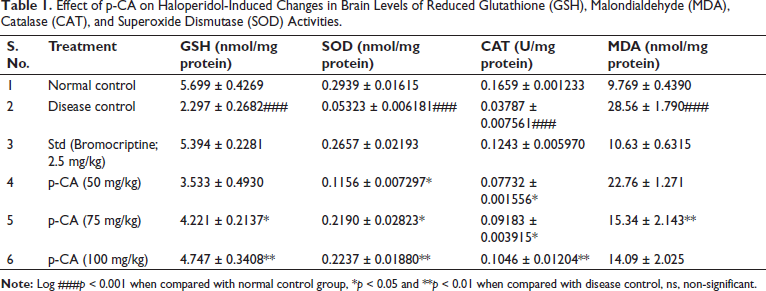
Data were analyzed using a one-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, *P < 0.05 and ** P < 0.01 when compared to the disease control group.
Effect of p-CA on Dopamine Level in Haloperidol-Induced Catalepsy in Mice
As depicted in Figure 5, brain dopamine level in the disease control group was significantly lower than normal. Intermediate (75 mg/kg) and high (100 mg/kg) doses of p-CA showed significant increments in brain dopamine levels (P < 0.01 and P < 0.001, respectively).
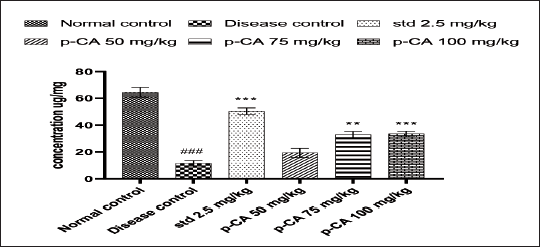
Data were analyzed using a one-way ANOVA followed by Dunnett’s post-hoc multiple comparison test. Values are mean ± SEM; n = 6 in each group. Log ###P < 0.001 when compared to the normal control group, * P < 0.05 and ** P < 0.01 when compared to the disease control group.
Effect of p-CA on Histopathology of Brain in Haloperidol-Induced Catalepsy in Mice
The effect of p-CA on histopathological changes in the mouse brain is shown in Figure 6. The histopathological examination reveals that the micrograph for the brain shows degenerative changes in neurons and angiectasis.
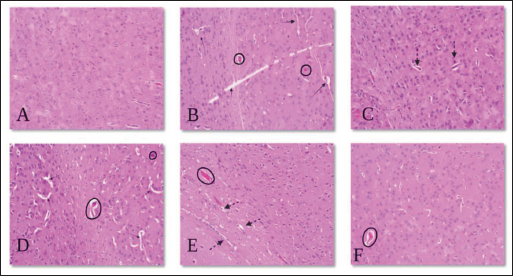
 This type arrow represents neurodegeneration.
This type arrow represents neurodegeneration.  This type circle represents angiectasis.
This type circle represents angiectasis.  This type arrow represents improve neurogenesis.
This type arrow represents improve neurogenesis.
As a result, from Figure 6, brain histopathology revealed pathological degenerative changes in neurons and angiectasis due to the treatment of haloperidol (1 mg//kg) for 21 days, which were significantly attenuated by the treatment of p-CA.
Discussion
Catalepsy is characterized by continuous insensitivity to external stimuli or to fixed body posture; limbs remain in whatever position they were placed in. Several different conditions can cause catalepsy. 5 Catalepsy is a major symptom of PD. PD is a neurodegenerative disease caused by the loss of dopaminergic neurons in the substantia nigra pars compacta region of the basal ganglia and the accumulation of Lewy bodies inside neurons caused by the clumping together of synuclein proteins. PD is characterized by motor symptoms like tremors, stiffness, and slow movement, as well as problems with standing up straight. 21 In the current work, we evaluate the neuroprotective and anticataleptic activity of p-CA in haloperidol-induced catalepsy in mice. Our research shows that p-CA is helpful and protects mice from haloperidol-induced catalepsy.
Important antipsychotics in the butyrophenone series begin with haloperidol. Haloperidol, sold as Haldol and under other brand names, is a common antipsychotic used to treat schizophrenia. It works by blocking the dopamine D2 receptor. Haloperidol is prescribed for the treatment of schizophrenia, Tourette’s syndrome, bipolar disorder mania, delirium, agitation, acute psychosis, and alcohol withdrawal hallucinations. In hospice, it is used to treat terminal delirium and severe agitation in dementia patients in their final stages. Haloperidol treatment cannot be replaced in end-of-life care for psychosis. Therefore, to overcome the side effects of haloperidol, the best co-therapy or pretreatment is required. Therefore, it is vital to investigate haloperidol-induced catalepsy in animal models. In the present study, catalepsy is induced by administering haloperidol for 21 consecutive days. Haloperidol is a typical antipsychotic for the treatment of schizophrenia that primarily blocks dopamine D2 receptors. In humans, neuroleptics cause two distinct forms of motor disturbances, termed extrapyramidal side effects: tardive dyskinesia and catalepsy. 6 Dopamine D2 receptors can be blocked directly or indirectly, resulting in these symptoms. These unwanted effects are the principal detriment associated with the application of traditional neuroleptics in therapeutic settings. The administration of haloperidol for 21 consecutive days resulted in a considerable rise in the number of animals exhibiting catalepsy.
There are two distinct hypotheses for the mechanism by which haloperidol induces catalepsy and neurotoxicity. The first is that haloperidol is broken down by an enzyme called oxidase, which makes a lot of oxyradicals and a dangerous compound that looks like pyridinium and leads to oxidative stress. 8 The second is that neuroleptics’ chronic blockade of dopamine D2 receptors in the brain’s nigrostriatal neurons increases basal ganglia dopamine turnover, which might result in an excess of free radicals such as dopamine quinone and hydrogen peroxide due to monoamine oxidase activity. Because of this, dopamine levels in the parts of the brain that control motor symptoms are disturbed, resulting in motor dysfunction. 12
In previous studies, it was proved that continuous administration of haloperidol for 21 days leads to extrapyramidal symptoms and increased catalepsy in rodents. 12 In the current investigation, haloperidol produced a significant amount of catalepsy in mice. This catalepsy was significantly reduced by the administration of p-CA for 21 days in a row. This was accomplished through a variety of possible mechanisms, including the inhibition of the COMT enzyme, the promotion of BDNF, and the inhibition of the 5-S-cysteinyl-dopamine metabolite. Chronic haloperidol therapy reduced locomotor activity. Researchers have discovered that dopamine D2 receptors regulate locomotion. It has been demonstrated that agonists of the D2 receptor induce hyperlocomotion, 15 whereas D2 receptor antagonists cause a decrease in movement (hypolocomotion). Due to the fact that antipsychotic medicines have an antagonistic action on dopamine D2 receptors, these drugs make animals’ hypolocomotion worse while simultaneously having a therapeutic effect. The current study revealed that p-CA significantly increased locomotor activity compared to haloperidol. 22
Also, it is well-established that chronic haloperidol treatment decreases motor coordination activity. It has been exposed that dopamine D2 receptors are also concerned with the regulation of various motor coordinations. 23 It has been proven that agonists of the D2 receptor improve motor coordination, whereas antagonists of the D2 receptor impair motor coordinations. 24 Antipsychotic drugs also aggravate motor impairment in animals simultaneously with their therapeutic effect due to their antagonistic impact on dopamine D2 receptors. 25 In the present study, p-CA significantly improved motor coordination and reduced motor impairment compared to a disease control group. In the current study, dopamine levels were increased and oxidative stress was decreased, which improved motor coordination. As previously reported, low dopamine levels 26 and increased oxidative stress 27 are major contributors to the development of motor deficiency in mice.
Movement disorders involving the basal ganglia, such as Parkinson’s disease, are characterized by slow movement (hypokinesia) and a shorter stride length. 16 In this investigation, we discovered that consecutive haloperidol administration impacts movement, muscle rigidity, and gait abnormalities, leading to decreased walking speed and stride length. The stride length was assessed by having an animal walk on a glass slide and taking a picture from the bottom of the slide; ImageJ analysis software was used to capture and measure the imprints. In the present work, we discovered that p-CA ameliorates gait disturbance in a dose-dependent way. In the present study, gait disruption is associated with a decrease in dopamine levels and an increase in oxidative stress in the brain, both of which are mitigated by p-CA.
The present study demonstrates that administration of haloperidol for 21 consecutive days significantly increased oxidative stress in the brains of mice, as evidenced by decreased antioxidant enzyme activity such as SOD, GSH, and catalase and an increase in malondialdehyde levels. It is also well known that consecutive use of haloperidol causes increased oxidative stress in the brain and other parts of the body. 28
Haloperidol blocks the dopamine D2 receptors, resulting in an increase in dopamine turnover in the basal ganglia. This region of the brain is extremely susceptible to extreme free radical production as a result of increased dopamine turnover. 29 Increased oxidative metabolism in the basal ganglia following continuous haloperidol treatment may be accompanied by a reduction in brain antioxidant defenses, as demonstrated by increased lipid peroxidation and significantly decreased catalase, GSH, and SOD activity.
According to the literature, increasing oxidative damage parameters in many brain regions following chronic haloperidol treatment may contribute to the onset of catalepsy. The present study shows that oxidative stress is ameliorated by p-CA in a dose-dependent manner through its antioxidant effect, continues p-CA treatment for 21 successive days significantly improved haloperidol-induced oxidative stress, as assessed by increased SOD, GSH, and catalase activity in the brain, and decreases malondialdehyde levels in the brain. The literature also confirms this, which indicates that p-CA exerts neuroprotective effects by inhibiting oxidative damage and activating antioxidant enzymes. 30
According to previously published research, continuous haloperidol administration may result in increased oxidative damage and blockage of the dopamine D2 receptor in brain regions, resulting in a drop in dopamine levels in the striatum and brain regions. 31 The results of this study demonstrate that continuous p-CA administration for 21 successive days dramatically increased dopamine levels in the striatum by alleviating oxidative stress-induced neurotoxicity.
According to previously published research, continuous haloperidol administration may result in increased oxidative damage and blockage of the dopamine D2 receptor in brain regions, leading to pathological degenerative changes in neurons and angiectasis. In the present study, we discovered that animals pretreated with p-CA showed restored neuronal death and reduced angiectasis in neurons. Restoring neuronal death is predicated on the reduction of oxidative stress in the brain, according to the fundamental hypothesis underlying this process. Future research should focus on specific mechanistic studies to gain further insight into the role of the p-CA in haloperidol-induced catalepsy in mice. The current study has some limitations, such as the fact that we only examined behavioral and dopamine levels.
Conclusion
The present study proves that p-CA exhibits excellent activity against haloperidol, which induces catalepsy in mice. The results revealed that catalepsy, locomotor activity, motor coordination, antioxidant activity, and dopamine level significantly improved after repeated administration of p-CA for 21 days, along with improved gait disturbance and histopathological changes. As p-CA was used in 3 different doses, and the p-CA dose of 100 mg/kg showed the most significant results when compared to the doses of 50 mg/kg and 75 mg/kg.
Footnotes
Acknowledgment
The authors are thankful to SES’s R. C. Patel Institute of Pharmaceutical Education and Research, Shirpur, India, 425405, for providing the facility.
Author Contributions
Dr. Pankaj Jain contributed to the conception and supervision of the study, checking, editing, and all revisions of the manuscript. Afsar Pathan and Dr Vivek Kumawat contributed to carrying out all the experiments, statistical analysis of the data, and preparing the first draft of the manuscript, including table and figures.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The experimental protocol was approved by the Institutional Animal Ethics Committee at R. C. Patel Institute of Pharmaceutical Education and Research, Shirpur, District Dhule (MS), India, 425405.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.