Abstract
Objectives:
To analyse the adverse drug reactions (ADRs) reported from clinical departments of a tertiary care hospital.
Materials and Methods:
A prospective, observational study to analyse the reported ADRs to the pharmacovigilance unit, Department of Pharmacology, East Point College of Medical Sciences and Research Centre, between 2019 and 2021. Institutional Ethics Committee approval was taken before doing the study. The data pertaining to various parameters were recorded in the Central Drugs Standard Control Organization (CDSCO) approved ADR reporting form and were analysed with respect to each reported data using descriptive statistics and expressed as numbers and percentages using Microsoft Excel.
Results:
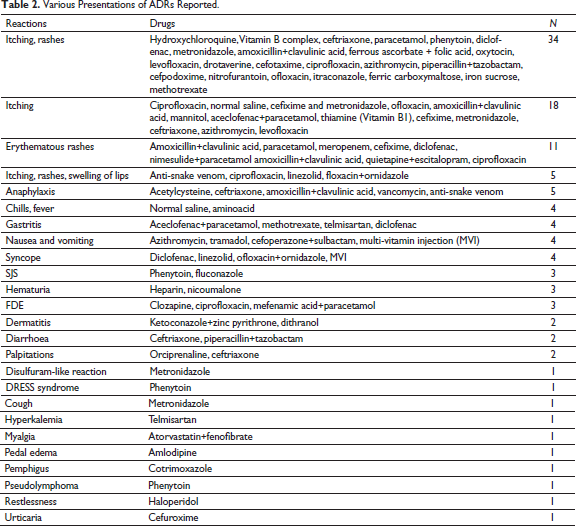
Overall, 114 ADRs were reported during the study duration, and ADRs were most commonly reported amongst females (69) and 31–45 years (27.2%) age group. Causality assessment was done using the World Health Organization-Uppsala Monitoring Centre (WHO-UMC) scale, which showed 75 (66%) probable and 39 (34%) possible ADRs. The highest number of ADRs were reported by Medicine department (48.2%), emergency and intensive care unit (ICU) (16.6%) followed by Dermatology department (9.6%). The majority of them were due to antimicrobial agents (53.5%). The most commonly affected organ system was found to be dermatological (68.4%) followed by the body as a whole (15.7%) and gastrointestinal system (8.7%). The presentations of ADRs were diverse; itching and rashes (34 cases) were most commonly reported.
Conclusion:
This study gives an overall understanding of the current situation and trends in ADRs and their reporting status by health professionals in a tertiary care hospital, which would help to strengthen the pharmacovigilance activities at all levels of health care.
Introduction
The development of newer drugs has brought remarkable benefits for the patients, along with an increased incidence of adverse drug reactions (ADRs). 1 ADR is defined by WHO as ‘a response to a medicinal product which is noxious, unintended and which occurs at doses normally used in man for prophylaxis, diagnosis or therapy of disease or for the restoration, correction or modification of physiological function’. 2 As said, ‘No drug is absolutely free from side-effects’. It is observed that 5% of all hospitalizations were associated with drug-induced problems, and 10%–20% of in-patients are developing ADRs. 3 Several factors like complexity of diseases, drug interactions, polypharmacy and possible negligence are associated with ADRs in hospitalized patients. 4
The ADRs increase drug-related morbidity and mortality, enhancing the economic burden on the patients and the health care establishment, mainly in a developing country like India. 2 Health care professionals have the responsibility to report ADRs; however, due to the need for motivation, time, knowledge and attitude, several reactions are certainly not reported. Under-reporting is a major drawback in India. Hence, there is a necessity to boost health care professionals’ responsiveness to the prevention, identification and reporting of ADRs. 5
In India, epidemiological data of ADRs regarding burden and spectrum are limited, 6 also, there are differences among various regions within India in the occurrence of ADRs. This study aims to strengthen the ADR database through analysis of spontaneous reporting patterns of ADRs from various clinical departments of a tertiary care teaching hospital, thereby increasing awareness and improves the reporting culture among health care practitioners.
Materials and Methods
Study Design
A prospective, observational study to analyse voluntarily reported ADRs to the pharmacovigilance unit, Department of Pharmacology, East Point College of Medical Sciences and Research Centre, Bengaluru, between 2019 and 2021. Prior Institutional Ethics Committee approval was obtained (EPCMSRC/ADM/IEC/2019-20/013) before initiating the study.
Inclusion Criteria
Patients (wards, casualty and ICU), of either sex, of any age, who developed an ADR.
Exclusion Criteria
Patients who developed an ADR due to drug poisoning (accidental or intentional), blood or blood products and vaccines, and ADRs caused by alternate systems of medicines such as homeopathy, ayurvedic and unani were excluded from the study.
Study Methodology
The data regarding demographic parameters, ADR parameters, medication parameters and causality and outcome assessments were recorded in the Central Drug Standard Control Organization certified ADR reporting form 7 and were analysed with respect to each reported data. The data were kept confidential.
Statistical Analysis
Data obtained were analysed using descriptive statistics and expressed as numbers and percentages wherever appropriate with the help of Microsoft Excel version 2010.
Results
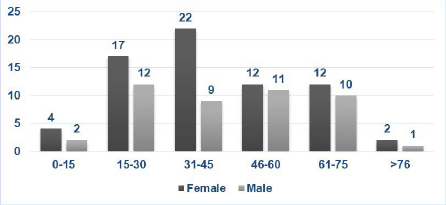
A total of 114 ADRs were reported during the study duration from 2019 to 2021, of which 45 were males and 69 were females.
The age group of patients in whom ADRs were reported is shown in Figure 1.
Age-wise Distribution of ADRs.
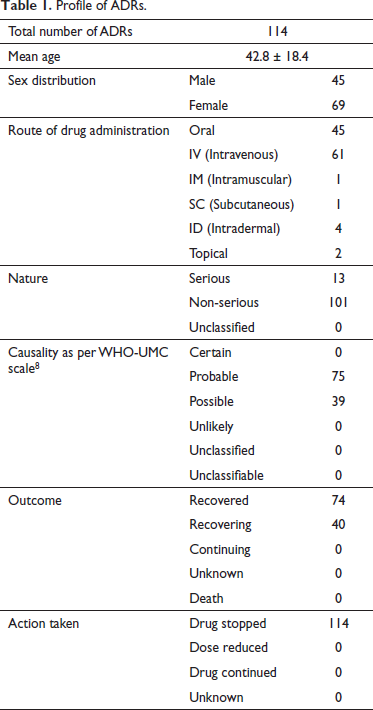
The profile of various reported ADRs during the study period is shown in Table 1. Among the 114 reported cases, serious ADRs were 13 (11%). Causality assessment through WHO-UMC scale 8 indicated around 75 (66%) probable and 39 (34%) possible ADRs. There were no ADRs which were certain (Table 1).
Profile of ADRs.
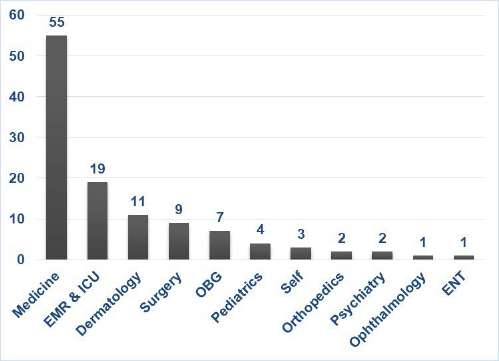
Most of the ADRs were reported by the Medicine department (48.2%), Emergency and ICU (16.6%) followed by the Dermatology department (9.6%) (Figure 2).
Department-wise Distribution of ADRs.
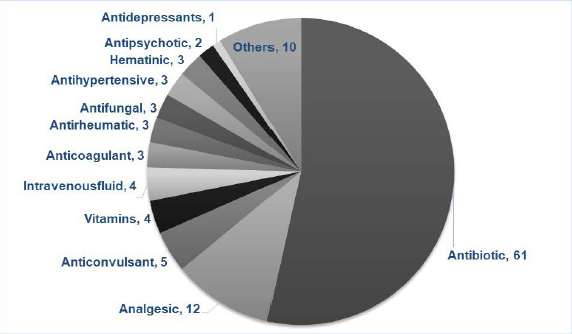
Most ADRs were due to antibiotics (53.5%) (Figure 3).
Drug-wise Distribution of ADRs.
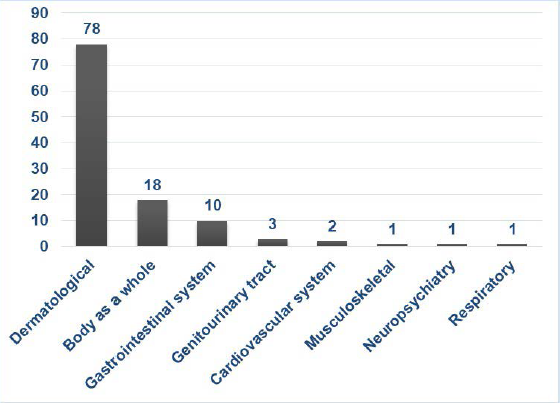
The most frequently involved organ system was noticed to be the dermatological (68.4%) followed by the body as a whole (15.7%) and gastrointestinal system (8.7%) (Figure 4).
System-wise Distribution of ADRs.
The presentations of ADRs were diverse; itching and rashes (34 cases) were the most common presentation reported (Table 2).
Various Presentations of ADRs Reported.
Discussion
The ADR adds to the burden of drug-related morbidity and mortality. 2 With the increased use of drugs, we see increased ADRs. Often ADRs are seen in hospitals, yet they can also occur with outpatients and with self-medication which majority of the time goes unnoticed due to lack of knowledge among the general public and even the health care professionals. 4 To overcome this gap, Pharmacovigilance Program of India (PvPI) has started a National Pharmacovigilance Program for spontaneous reporting of ADRs in the year 2010, which is accessible to everyone. 9 The PvPI collects the ADRs from all ADR monitoring centres (AMCs) and the public in India, and conveys the substantial data to drug regulatory authorities for essential action on the drugs; it also shares the health care professionals and the public regarding the risk of ADRs, by this, it improves the patient safety and welfare. It is the responsibility of all health care professionals to support the PvPI in promoting the safe use of medicine. 1 In this regard, we have reported a total of 114 ADRs to the PvPI according to the standard criteria given by National Coordinating Centre for monitoring ADRs.
Female preponderance (60.5%) was noted among the cases, similar to a study by James and Rani 10 (60%) and Godbharle and Padwal 11 showing that females may have more risk of developing ADRs. In our study, the adult age group (31–45 years) reported the majority of ADRs (27.2%), which was comparable to a study by Misra et al. 5 (42.8%).
Antibiotics (53.5%) were causing most ADRs which could be correlated to the increased frequency of use of antibiotics. Comparable results were obtained by Misra et al. 5 (36.1%) and Joseph and Badyal 12 (32%), where the maximum number of ADRs were related to antibiotics. Most of the adverse reactions were seen with beta-lactam antibiotics (26.3%) which were similar to the findings by James and Rani 10 (37.5%) as beta-lactam antibiotics are one of the commonly used antibiotics for a wide range of infections.10, 12
Medicine (48.2%) followed by Emergency and ICU (16.7%) departments reported the highest number of ADRs, which was also comparable to the study by James and Rani, 10 who reported 25% cases and Venkatasubbaiah et al. 1 reported 56.6% cases from the General Medicine department. This may be correlated to the usage of various drugs in these departments for the treatment and prophylaxis of various diseases. Also, the patients admitted in medicine and emergency departments have comorbidities requiring polypharmacy, accounting for the high number of ADRs. 10
In our study, the frequently affected organ system was found to be the dermatological (68.4%) with a predominance of cutaneous manifestations (itching and rashes) (29.8%), similar to studies by James and Rani (75%), 10 Godbharle and Padwal (34.4%) 11 and Misra et al. (51.1%), 5 but a study by Venkatasubbaiah et al. 1 and Joseph and Badyal, 12 showed the most commonly affected system was gastrointestinal system followed by skin.
To know if the reaction is because of drug alone or is there association of other factors in causing ADRs, doing a causality assessment is essential; which was done using WHO-UMC causality assessment scale 8 and found that the majority of ADRs were probable (66%), owing to recovery of ADRs on stoppage of suspected causative drugs, in contrast to a study by Venkatasubbaiah et al. 1 whose study showed the majority of cases were possible (48.8%). In our study, no cases were certain, as the re-challenge test was not performed for ethical reasons.
Through this study, we suggest there is a need for spontaneous ADR reporting from all the departments of this tertiary care hospital for the identification, monitoring and assessment of ADRs. This study also warrants the need for further research in this field for the development of possible intervention strategies to lower the burden of ADRs.
Conclusion
ADRs are an inevitable risk factor associated with the use of medicines. However, the risk of ADRs may be minimized by noting proper allergic history, rational use of medicines and prompt reporting of ADRs, which should be encouraged at all levels of health care. The present study gives an insight into the current situation and trends in ADRs in tertiary care teaching hospitals by analysing the reported ADRs, which would further strengthen pharmacovigilance activities. The main limitation of this study is the underreporting of ADRs for 3 years; which could be minimized by periodically educating the health care workers with respect to what to report, where to report, when to report and how to report the ADR and the importance of pharmacovigilance, also by giving incentives for regular spontaneous reporting of ADRs. There is also a need to train medical graduates regarding pharmacovigilance as they are future practitioners.
Abbreviations
ADR: Adverse drug reaction
CDSCO: Central drugs standard control organization
PvPI: Pharmacovigilance program of India
WHO-UMC: World health organization-Uppsala monitoring centre
NCC: National coordinating centre
AMC: ADR monitoring centre
ICU: Intensive care unit
Footnotes
Acknowledgments
We are thankful to the institution, principal, dean, director and medical superintendent for their support and permission to carry out this study and to all the staff for reporting the ADRs and helping us to conduct this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Prior Institutional Ethics Committee approval was obtained (EPCMSRC/ADM/IEC/2019-20/013) before initiating the study.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable
