Abstract
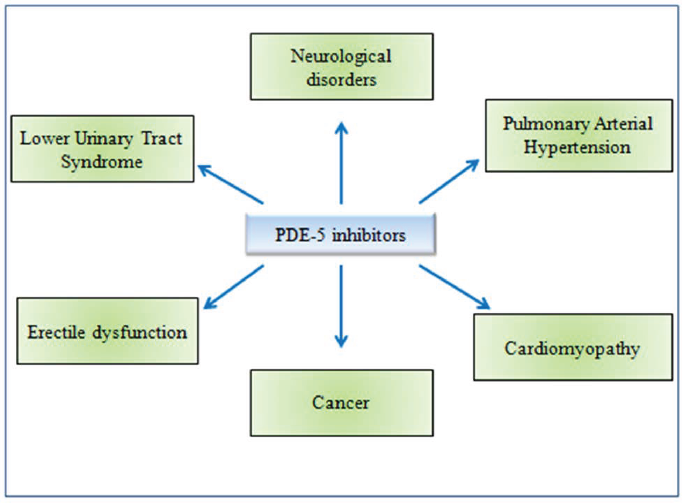
Phosphodiesterase type 5 (PDE5) is a cyclic GMP (cGMP) specific protein. It hydrolyzes the phosphodiesterase linkage and catalyzes the conversion of cGMP to 5’ GMP, which controls different physiological activities of the body. PDE5 is associated with biomedical conditions like neurological disorders, pulmonary arterial hypertension, cardiomyopathy, cancer, erectile dysfunction, and lower urinary tract syndrome. Inhibition of PDE5 has now been proven pharmaceutically effective in a variety of therapeutic conditions. Avanafil, tadalafil, sildenafil, and vardenafil are the most commonly used PDE5 inhibitors (PDE5i) today which are often used for the management of erectile dysfunction, lower urinary tract syndromes, malignancy, and pulmonary arterial hypertension. However, these synthetic PDE5i come with a slew of negative effects. Some of the most common side effects include mild headaches, flushing, dyspepsia, altered color vision, back discomfort, priapism, melanoma, hypotension and dizziness, non-arteritic anterior ischemic optic neuropathy (NAION), and hearing loss. In light of the potential negative effects of this class of medications, there is a lot of room for new, selective PDE5 inhibitors to be discovered. We have found 25 plant botanical compounds effectively inhibiting PDE5 which might be useful in treating a variety of disorders with minimal or no adverse effects.
Introduction
Phosphodiesterase 5 (PDE5) is a cyclic guanosine monophosphate (cGMP) specific protein and belongs to the metalo-hydrolase superfamily. 1 The enzymatic activity of PDE5 is found in the vascular and visceral smooth muscle, skeletal muscle, platelets, kidney, lungs, spinal cord, cerebellum, pancreas, spleen, prostate, urethra, bladder, and smooth muscle cells of the corpus cavernosum.2-6 It regulates various physiological functions of the body, such as growth, viability, smooth muscle relaxation, secretion, ion transport, endothelial permeability, and gene transcription. 7
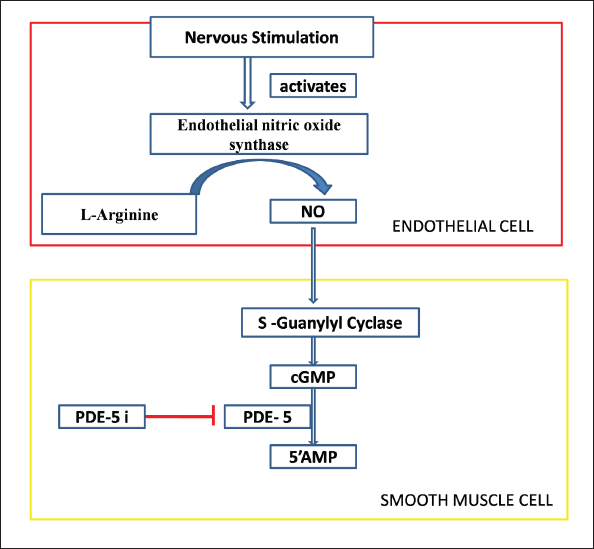
The structural unit of PDE5 consists of two units, one acting as a regulatory unit and the other acting as a catalytic unit. The GAF-A and GAF-B domains of the regulatory unit work in concert to control the catalytic activity and dimerization of PDE5. 8 The primary target of PDE5 is the secondary messenger cGMP, which catalyzes the conversion of cGMP into inactive 5’ GMP by hydrolyzing the phosphodiesterase (PDEs) linkage. 1 PDE5’s affinity for cGMP is explained by the existence of cGMP binding sites on the N-terminal regulatory GAF-A domain. 9 The activation of protein kinases, ion channels, or PDEs by cGMP results in a wide range of physiological reactions. Thus, understanding the mechanisms of cGMP generation and downstream signaling cascades is critical for understanding molecular physiology and pathophysiology, as well as the development of drugs targeting these pathways, which have already been shown to have significant therapeutic implications in recent years. 10 The second messenger molecule, guanosine 3’, 5’-cGMP, regulates a variety of downstream effects, including vasodilation, neurotransmission, calcium homeostasis, and retinal photo transduction. Through effects such as activation and modification of specific ion channels, interaction with PDEs, and association with GMP-dependent protein kinases, cGMP plays a role in these processes. The synthesis of cGMP on target cell is aided by a neurotransmitter nitric oxide (NO) upon stimulation. NO and cGMP are components of an autocrine, paracrine, and perhaps endocrine signal transduction pathway. The soluble guanylyl cyclase (sGC) within the target cells is activated by NO generated from its own cell and surrounding cells within a tissue. 11 The sGC is a heterodimeric hemeprotein composed of the sGC alpha and sGC beta subunits. 12 The activation of sGC by NO triggers the activation of downstream cGMP-dependent protein kinases G (PKG) family and cGMP-regulated ion channels, which initiates a signaling cascade that governs a wide range of physiological effects. 13 PDE5 targets those physiological pathways that utilize cGMP by converting it into inactive 5’ GMP via hydrolysis of the PDEs linkage of cGMP (Figure 1).
PDE5 isoforms have been discovered in wide range of animal and human tissues. In humans, three isoforms have been identified: PDE5A1, PDE5A2, and PDE5A3 with similar cGMP-catalytic activity and slight differences in the 5′ end of the mRNA and the N-terminus of the protein. 7 PDE5A1 and PDE5A2 are found in almost all tissues, but PDE5A3 is only found in smooth muscle. 3 These three isoforms of PDE5 are suppressed by its inhibitors in different ways, with PDE5A1 being more resistant than PDE5A2 or PDE5A3. The inhibition of PDE5 has been found to be pharmacologically effective in many clinical aspects (Figure 4). Currently the most widely used PDE5 inhibitors include avanafil, tadalafil, sildenafil, and vardenafil. The pharmacological interventions of PDE5 inhibitors are extensively recommended for patients with erectile dysfunction (ED), lower urinary tract syndromes, and pulmonary arterial hypertension (PAH). 14 Looking at the possible application of this class of drugs, there is enough scope for the discovery of novel, selective PDE5 inhibitors over the existing drugs.
Representation of the role of PDE5. PDE5 ends the role of cyclic GMP by converting it into inactive 5’ GMP via hydrolysis of the phosphodiesterase linkage of cyclic GMP.
PDE5 and Biomedical Conditions
PDE5 and Neurological Disorders
The NO–cGMP mediated signaling cascade plays an important role in the central nervous system. sGC serves as the acceptor of NO. When this enzyme is activated, it converts GTP into cGMP. cGMP acts as a second messenger that initiates a cascade of reaction eventually activating PKG. The active cGMP/PKG pathway plays a significant role in memory development and synaptic plasticity. A decreased concentration of cGMP in the cerebrospinal fluid may be associated with neurodegenerative diseases such as Alzheimer’s disease. 15 PDE5 is a major component of the cGMP–PKG (Protein Kinase G) axis of cellular communication in neurons, and most PDE isoforms are strongly expressed in the brain (cortex, cerebellum, and hippocampus) as well as in the blood vessels of the brain (for example, cerebral, basilar, and mesenteric arteries). 16 Many studies have found that cyclic adenosine monophosphate (cAMP) and cGMP, both play a crucial role in learning and memory, and inhibition of PDE5 has thus been shown to be therapeutic in a range of neurologic illnesses in animal models, including Alzheimer disease.17, 18 In rat cerebellar slices, local zaprinast, a PDE5 inhibitor treatment increases the onset of long-term depression. 19 These suggest that the cGMP/PDE5 pathway regulates vascular and central nervous system functioning and is involved in a variety of cerebral activities as well as brain metabolism.
PDE5 and Pulmonary Arterial Hypertension (PAH)
A continuous obstruction of blood arteries, hypertrophy of the endothelium inner wall, thrombosis, and edema cause PAH, which raises blood pressure in the lungs. Immune system abnormalities and endothelial dysfunction may be the causes of this pathogenesis. Strategies to increase NO bioavailability or cGMP levels in the cells are available treatments for endothelial dysfunction. Hence, the inhibition of PDE5 is often linked with the treatment of PAH. 20 Despite being the primary therapy for ED, PDE5 inhibitors are now approved for the treatment of PAH in adults. 7 It is evident that PDE5 plays a significant role in controlling pulmonary responses since sildenafil citrate, a medication that is used to treat ED, has also been used to treat PAH. 21 Inhaled NO is also used to treat persistent pulmonary hypertension in neonates, but it’s a costly alternative, and about a third of babies don’t respond, prompting the development of other treatments.22-24 There are reports that a phytocompound, Quercetin, could increase the expression of PDE5 in the lungs resulting in smooth muscle relaxation and diminished trans vascular leakage. 24
PDE5 and Cardiomyopathy
Since the discovery of sildenafil in 2002, there has been extensive research into its potential use in cardiovascular disease. PDE5 inhibitors’ vasodilatory effects have been shown to be beneficial in vascular coagulopathy and improving endothelial functionality throughout the body. These inhibitors are useful against heart failure and myocardial ischemia. Research into the impact of PDE5 inhibitors on ischemia–reperfusion injury, pressure overload–induced hypertrophy, and chemotoxicity revealed that each of these drugs could have clinical relevance. 25
PDE5 expression in the heart is a contentious topic. 26 While anti-PDE5 antibodies have been found to be positive in coronary vascular smooth muscle cells, 27 healthy myocardium does not appear to express large quantities of the enzyme. 26 However, in congestive heart failure and right ventricular hypertrophy, Angiotensin II stimulation of vascular smooth muscle cells raises PDE5,27-29 which lowers cGMP/PKG reactivity. 30 The role of PDE5 is best understood in the heart, where PDE5A inhibitors reduced cardiomyocyte apoptosis and infarct size. 31 Korkmaz-Icoz et al. 32 discovered similar cardioprotective evidence in their meticulous repurposing of these inhibitors for the treatment of heart failure and myocardial ischemia. 32 Although PDE5 inhibitors have a well-established role in ED treatment, their implications on the vascular system are not very popular. Preclinical research into the impact of PDE5 inhibitors on ischemia–reperfusion injury, pressure overload–induced hypertrophy, and chemotoxicity revealed that each of these drugs could have a clinical relevance. 25
PDE5 and Cancer
The NO/cGMP signaling system has been discovered to play a contradictory and diverse role in cancer research.33, 34 Chronic inflammation raises NO levels and increases inducible nitric oxide synthase. Both of these can provide a carcinogenic environment for DNA alteration by enhancing the prevalence of p53 mutations, which is a key tumor suppressor oncogene. 35 Increased NOS expression in stromal and tumor cells has also been shown to promote tumor growth through increased cell motility, invasiveness, and angiogenesis. 36 Contradictory to this, there is a report which shows that increased NOS activity and NO generation leads to higher NO-mediated tumor cell death. 37
PDE5 plays a vital role in the control of intracellular cGMP pool; it has been reported that cGMP and protein kinase-G diminished with increased expression of PDE5 in a variety of human cancers, including colon adenocarcinoma, bladder squamous carcinoma, metastatic breast, prostate, pancreatic, and lung cancers.38-43 The increased expression of PDE5 has also been confirmed in various cell lines originating from breast cancer (MCF-7, HTB-26, MDA-MB-468), prostate cancer (LNCAP, PC3), bladder cancer (HTB-76, HT1376), and colorectal cancer (HT29, HCT116, SW480, T84). 39 These findings imply that PDE5 may play a role in carcinogenesis, and that targeting PDE5 could be a viable anticancer strategy for reducing the risk of developing cancer. Numerous studies have been conducted to support the role of PDE5 inhibitors in cancer prevention. Several studies have looked into the anticancer effects of sildenafil and other PDE5 inhibitors. Under in vitro conditions, sildenafil and vardenafil have been shown to inhibit tumor cell proliferation and trigger caspase-dependent death in B-cell derived chronic lymphocytic leukemia cells. 44 Sildenafil dramatically inhibited cell growth, cell cycle arrest, and death in human colorectal cancer cells, which was accompanied by evident alterations in associated proteins such as CDKs, cyclins, and PARP, among others. 45 Exisulind and its analogs, a nonspecific PDE5 inhibitor, preferentially caused apoptosis in diverse human prostate, colon, and breast cancer cells, owing to their inhibitory effects on PDE5 expression, boosting the cGMP-driven activation of PKG.40-49
PDE5 and Erectile Dysfunction
One of the most common and under-treated sexual disorders in men is ED, which affects over half of all men between the ages of 40 and 70, or roughly 150 million men globally. 50 It is defined as a lack of ability to keep erections strong enough to engage in sexual activity. 51
The NO–cGMP route is the primary mechanism of penile erection in primates, including humans. 2 The major biomolecule responsible is NO, which is triggered via the NO–cGMP dilator pathway; thus, a decrease in its amount or poor production could result in ED. 52 Sexual stimulation activates neurological pathways that cause NO to be released directly into the penis from neurons and endothelial cells. NO penetrates smooth muscle cells’ cytoplasm and binds to guanylyl cyclase. When NO binds with guanylyl cyclase, it undergoes a conformational change, allowing it to catalytically produce 3’-5’-cyclic guanosine monophosphate from guanosine 5’-triphsophate (Figure 2).7, 53 The intracellular trigger for penile erection is cGMP, which activates PKG. PKG reduces intracellular calcium levels, causing arterial and trabecular smooth muscle to relax, resulting in arterial dilation, venous constriction, and penile erection stiffness. 2 In the corpus cavernosum, PDE5 is the most common PDEs. PDE5 usually destroys cGMP by hydrolyzing it. 1 The reduced production of cGMP causes malfunctioning of the penile tissue leading to ED. 2
Representation of the production pathway of NO and its role in smooth relaxation of penile tissue.
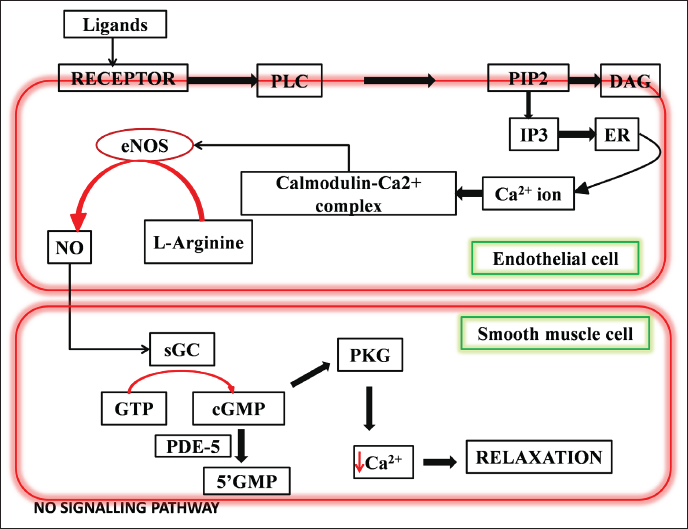
The receptor in the endothelium responds to any penile erection-inducing stimulation, activating PLC, which then acts on PIP2. IP3, which functions as an intracellular messenger, is produced from PIP2. The endoplasmic reticulum releases calcium ions as a result of induction of IP3. Calmodulin and calcium interact to generate the calcium–calmodulin complex. This compound stimulates the conversion of L-arginine to NO by activating eNOS. This NO diffuses to the corpus cavernosum where it triggers the cGMP synthesis, which relaxes the smooth muscles and leads to erection.
PDE5 and Lower Urinary Tract Syndrome (LUTS)
NO mediated signals play a key role in the relaxation of the urogenital tract including urinary bladder and urethra. 54 NO relaxes smooth muscle by activating soluble guanylate cyclase (sGC) and boosting tissue levels of cGMP in those tissues. 55 Sometimes a problem may persist, which may affect the bladder retention and discharge. This condition is referred to as LUTS, which may be associated with bladder outlet blockage caused due to benign prosthetic enlargement. 56 Several investigations have shown the presence of PDE5 in the urinary system, which lowers cGMP levels in those tissues. PDE5 inhibitors have been discovered to impede the process of cGMP hydrolysis and are consequently utilized to treat LUTS, particularly LUTS caused by benign prostatic hyperplasia.57-60 PDE5 inhibitors are hypothesized to improve the lower urinary tract by relaxing smooth muscle, reducing prostate stromal cell growth, modulating afferent nerve activity, and lowering the inflammatory response in the prostate.7, 61 However, regardless of erectile function, tadalafil is the only currently approved drug for treating LUTS. 61
Synthetic PDE5 Inhibitors and Risk Factors Associated with Their Uses
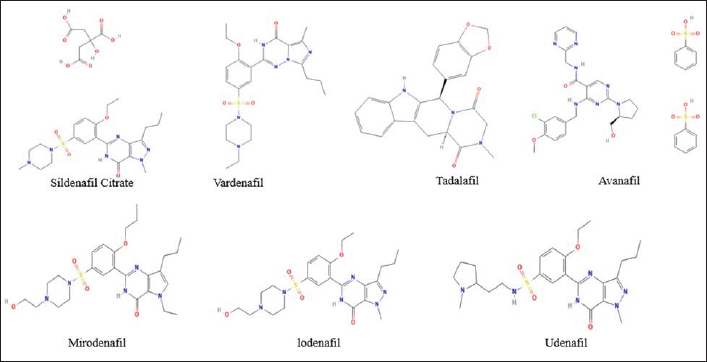
PDE5 inhibitors are the primary line of treatment for ED patients. 62 However, as PDE5 receptors are ubiquitous in the pulmonary vasculature, brain, lower urinary tract, and heart, PDE5 inhibitors are also an essential treatment option for pulmonary hypertension, neurological diseases, cardiomyopathy, and cancer.40, 41, 46, 47–49 ED has become a big concern, with recent predictions estimating that 320 million men will be affected by the disease by 2025. 63 FDA-approved sildenafil, vardenafil, tadalafil, and avanafil, as well as non-FDA approved lodenafil, udenafil, and mirodenafil, are among the PDE5 inhibitors available (Figure 3).8, 64 However, none of the PDE5 inhibitors are truly selective for the PDE5 receptor, and the majority of the negative effects are caused by cross-reactivity with other PDE isoenzymes. 64 The majority of these effects are dose-dependent.
Structures of Synthetic PDE5 Inhibitors.
Mild headaches, flushing, dyspepsia, altered color vision, back discomfort, myalgias, priapism, melanoma, hypotension and dizziness, rhinitis, nonarteritic anterior ischemic optic neuropathy (NAION), and hearing loss are some of the most prevalent side effects associated with the use of PDE5 inhibitors.64, 65 The presence of a high concentration of PDE6 enzyme in the rods and cones of the retina makes these cells a vulnerable target for PDE5 inhibitors causing altered color vision, particularly by sildenafil, which has a high affinity for blocking both PDE6 and PDE5. This effect has not been linked to any retinal structural or functional alterations.66, 67 Back pain and myalgias are caused by a high concentration of the PDE11 enzyme in skeletal muscle, which has a strong cross-reactivity with tadalafil. The FDA issued a warning on the possibility of hearing loss linked with PDE5I usage due to the pathophysiology that causes sensorineural hearing loss, which has been described in a few case studies. 64 Additionally, using nitrates or nitroglycerin alongside PDE5 inhibitors increases the risk of hypotension. Vardenafil, tadalafil, and sildenafil have all been linked to longer QT intervals in ECGs, with vardenafil having the longest QT intervals, tadalafil having the shortest QT intervals, and sildenafil having intermediate QT intervals. Patients taking PDE5Is should wait at least 1–2 days following their last PDE5I dose before taking nitrates, according to the American College of Cardiology.63, 68 Erythromycin and HIV protease inhibitors are two examples of medications that interact with PDE5 inhibitors since they share the same metabolic pathway. 68
Schematic Representation of the Role of PDE5 Inhibitors.
Natural PDE5 Inhibitors
Phyto-Compounds Inhibiting PDE5.
In the penile tissues, quercetin and rutin significantly modulate the activities of ED-related enzymes (phosphodiesterase-5′, arginase, acetylcholinesterase, adenosine deaminase, and angiotensin-I converting enzyme) and biomolecules (malondialdehyde and non-protein thiols). Because of their multi-specificity, quercetin and rutin were shown to be more effective than sildenafil. 69 Gallic acid, chlorogenic acid, caffeine, ellagic acid, rutin, quercetin, and luteolin, all extracted from Cylicodiscus gabunensis and Anogeissus leiocarpus have comparable inhibitory effects on ED enzymes. 76 Shin et al. reported that the inhibitory activities of five flavonoids isolated from S. flavescens, Kushenol H, Kushenol K, Kurarinol, Sophoflavescenol, and Kuraridine were measured against cGMP-specific PDE5 to identify potent inhibitors of PDE5. 82 It has been found that sophoflavescenol, a C-8 prenylatedflavonol, has the most potent inhibitory activity against PDE5. Chlorogenic acid isolated from H. umbellata and A. leiocarpus showed similar results on PDE5 and arginase activity, as well as pro-oxidant induced lipid peroxidation in rat penile tissue. 52 Another study found that isoliquiritigenin, a flavonoid derived from the roots of Glycyrrhiza glabra, raised intracellular cGMP levels in cultured tracheal smooth muscle cells, while inhibiting PDE5 activity in human platelets. 93
Icariin, a major constituents of epimedium herbs, potentially inhibits human PDE5 and represents a unique pharmacophore for treating ED, pulmonary hypertension, and other diseases. Icariin is now recognized as a natural PDE5 inhibitor with improved potency and specificity. 71
(A and B). Structures of Natural PDE5 Inhibitors.
Icariside II (ICS II), derived from the traditional Chinese herb Epimedium brevicornum, was effective treatment for cognitive defects and inflammation induced by intracerebroventricular-streptozotocin (ICV-STZ) in rats by inhibiting PDE5. 88 Resveratrol was seen to alter the reperfusion injury by inhibiting the activity of PDE5, leading to an increase in intracellular cGMP, which accounts for the cardioprotection via the cGMP/PKG/GSK-3 signal pathway.77, 78
Catechin, garcinal, garcinoic acid, and d-tocotrienol compounds isolated from Garcinia kola were targeted against receptor linked to ED. These processes include the molecular docking of catechin, garcinal, garcinoic acid, d-tocotrienol, and sildenafil to receptor PDE5 via Auto Dock Vina. 53 Similar results were observed by Temkitthawon et al., 80 when they investigated the chemical constituents of the tubers of Eulophia macrobulbon and found phenanthrenes as a new class of PDE5 inhibitors. 80 Clerodendrumcole brookianum Walph has ethno-medicinal importance for the treatment of hypertension and reported to show activity against anti-hypertensive drug targets rho-associated coiled-coil protein kinase (ROCK), angiotensin-converting enzyme, and PDE5. 81 Another group of researchers observed that three chemical constituents (acteoside, martinoside, and osmanthuside β6) derived from plants interact effectively with the anti-hypertensive drug targets by docking and simulation studies. The molecular binding study of the plant constituents could help to design new drugs to treat hypertension. 81
Osajin, pomiferin, scandenone, and auriculasin extracted from M. pomifera were subjected to docking study. The docking study highlighted how small structural differences may heavily influence the binding mode and the interaction pattern with PDE5. The binding motif and predicted binding energy suggest that Scandenone has a good potential to interact with PDE5 and could be investigated as a novel PDE5 inhibitor. 84 In another study, the mode of action of luteolin on PDEs (PDE1–5) was studied and it was found that according to the Lineweaver–Burk analysis, luteolin (3–30 µM) competitively inhibited PDE1–5 activities.90, 91
Conclusion
This review summarizes the usefulness of PDE5 inhibition. PDE5 is a dimeric enzyme present in the endothelium. This enzyme plays an important role in maintaining the tissue level of cGMP. It hydrolyzes the phosphodiester bond of cGMP, the second messenger molecule, responsible for many biological processes. Though PDE5 inhibitors are used to treat ED, now a days, these inhibitors have also gained special attention as an efficient management strategy to high blood pressure, CNS disorders, cardiomyopathy, and some forms of cancer. In this respect, sildenafil, verdenefil, tadanafil, and avanafil are widely used PDE5 inhibitors. Considering the negative effects associated with the use of these medications, we have presented some of the plants and their active compounds that may prove beneficial as PDE5 inhibitors that are expected to have least or no side effects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
