Abstract
Cognitive decline is a late adverse event in brain tumor survivors. The patients receiving radiation treatment exhibit a wide range of damage and impairment in attention, memory, and executive function compared to the untreated group. After radiation treatment, various changes are observed in astrocytes, oligodendrocytes, white matter, and vasculature. The major affected areas are the hippocampus and prefrontal cortex. Neurogenesis impairment is one of the primary mechanisms responsible for cognitive dysfunction. Various cytokines and growth factors are responsible for inducing apoptosis of neural cells, which results in impaired neurogenesis in response to radiotherapy. Transforming growth factor (TGF-β) is one of the key cytokines released in response to radiation. TGF-β plays a major role in neuronal apoptosis through various pathways such as the MAP kinase pathway, JAK/STAT pathway, and protein kinase pathway. In contrast, activation of the ALK5 pathway via TGF-β improves neurogenesis. So, the current review article focuses on the detailed effects of TGF-β on neuronal cells concerning radiation exposure. This in-depth knowledge will help researchers focus more on the TGF-β pathway and come up with new treatment schedules which will help reduce cognitive dysfunctions in brain tumor patients produced as a result of radiation therapy.
Introduction
Brain tumors are said to be the most deleterious form of cancer. In the United States, nearly 210,000 people are diagnosed with principal and metastatic brain tumors yearly. These tumors are the most common form of solid tumor in children under the age of 20, and they are the second, increasing source of cancer-related fatalities in this age group. Some of the risk factors associated with these tumors are environmental factors, genetic disorders, and electromagnetic fields. Many brain tumor patients experience cognitive impairment at some point during treatment, and cognitive deficits are present in more than 90% of patients with primary brain tumors and brain metastases before treatment. 1 For these patients, retaining cognitive function is important, and, in most cases, a deterioration in cognitive function is escorted by a decline in mental independence and success grade. This intellectual loss may be caused by tumor growth, anxiety or depression, exhaustion or sleep problems, or the medications they have taken, in general, prefrontal functions, including expression, attention, memory, and executive function, are the most impaired in brain tumor patients because of cognitive impairment, which can be caused by the tumor itself, treatment options, or a combination of both. Currently, radiotherapy is often used in the treatment of tumors. Every year in the United States, nearly 200,000 patients receive radiation treatment. Around 50% to 90% of survivors exhibit cognitive impairment following treatment, and these effects follow a biphasic process. Initially, three months after the treatment, there is a transient decline in memory. This is followed by a progressive, irreversible decline after one year. Various changes are observed in astrocytes, oligodendrocytes, white matter, vasculature, etc., following radiation which can lead to various neurological complications. 2 Radiotherapy is a profound treatment in low-grade glioma brain tumors like astrocytoma, oligoastrocytoma, and oligodendroglioma.
Before 1970, the human brain was resistant to radiation i.e., Central Nervous System (CNS) syndrome and white matter necrosis occurred only at high doses. Between1980 and 1990, the brain damage caused by radiation was known by changes in white matter, demyelination, and vascular irregularities, which occurred six months after radiation. Some of the preclinical studies show that there are significant reductions in hippocampal neurogenesis in animal models. Besides impaired neurogenesis, neuroinflammation has also been shown to play a major role in radiation-induced cognitive damage. Severe cognitive dysfunction can occur in patients receiving fractionated partial or whole-brain irradiation (fWBI), generally six months after irradiation. Therefore, the functional effects of brain irradiation are the subject of existing attempts to examine radiation-induced brain injury. 3 Despite these advancements, radiation exposure to normal brain tissues has some negative consequences. A study revealed that 11% of patients who received WBRT suffered from postirradiation dementia; other 89% cases complained of visual defects, gait disturbances, and dysarthria. 4
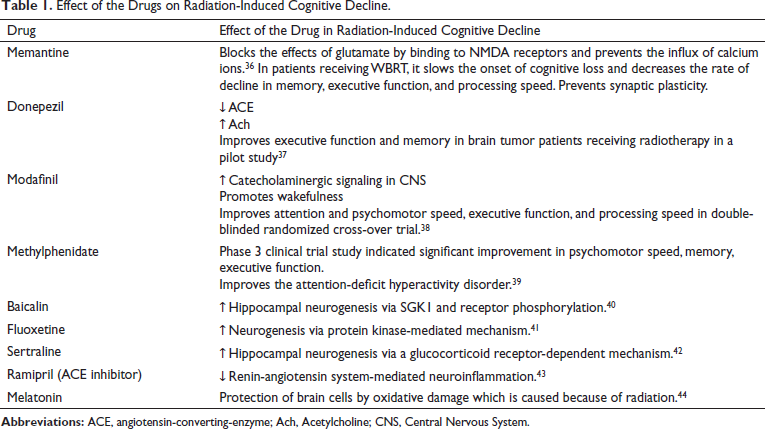
Radiation therapy has the benefit of being organ-specific and local in the case of brain tumors. Three-dimensional conformal radiotherapy (3DCRT), strength modulated radiation therapy (IMRT), and gamma knife radiosurgery (GKS) are examples of recent technological advancements that include safer and more sophisticated treatments. In the case of secondary tumors with multiple lacerations, whole-brain irradiation therapy is used. The large dose of the radiation, excess volume of the radiation, radiation vasculopathy, and age contribute to radiotherapy-associated cognitive impairment. 5 The brain effects of radiation are classified as acute, early, delayed, and late. The early-delayed shifts often consist of transitory demyelination and occur one to six months after treatment. Early delayed and acute improvements are reversible and can resolve spontaneously, whereas late effects are usually permanent and progressive. The current existing and ongoing treatments available for radiation therapy-induced cognitive decline are mentioned below in Table 1. So, the current review articles cover the role of TGF-β signaling in radiotherapy-associated cognitive decline in brain tumors.
Effect of the Drugs on Radiation-Induced Cognitive Decline.
Materials and Methods
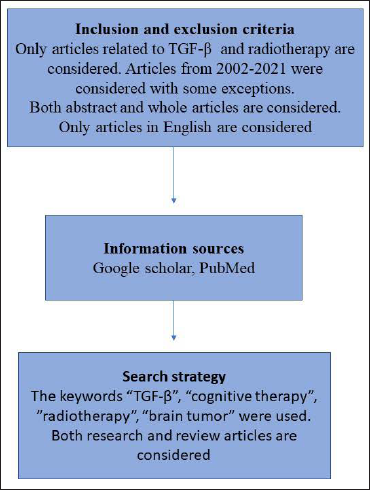
To collect the data about cognitive decline in brain tumors, a search was performed with PubMed and Google Scholar from 2002 to 2021. The keywords used include “cognitive decline,” “brain tumor,” “radiotherapy,” “molecular mechanism,” “TGF signaling,” “neurodegeneration,” “cognitive impairment,” and “tumor.” Both research and review articles were included, but only the publications in English were considered.
Pathophysiological Mechanisms Involved
The affected brain areas because of radiation therapy are the hippocampus and prefrontal cortex which are involved in cognitive function. The hippocampus is associated with learning and memory, whereas the prefrontal cortex is associated with executive functioning. The radiation injury can affect these areas, and some of its neurological complications include vascular abnormalities, inflammation, gliosis, demyelination, and white matter necrosis. 6
Effect of Radiation on Brain
Radiation affects both cancerous and healthy cells. Radiation causes necrosis, which is a serious adverse effect. These side effects, together with neurological problems, cause cognitive deterioration. Because several radiation therapies are required for malignancies, the rate of cognitive impairment accelerates. Radioresitance is another factor to consider. Because certain tumor cells develop resistance to radiation, the radiation dose must be increased to achieve the treatment dose. Necrosis is caused if a high dose of radiation falls into a small region of the brain. The effect may be nonrecoverable. These effects are usually late toxicities. The main mechanism of this toxicity is the necrosis of oligodendrocytes and endothelial. This caused demyelination because of vascular modeling and chronic hypoxia. Vascular insufficiency in the radiate area leads to nutrient deficiency, chronic hypoxia, ischemia, and oxidative stress. These effects in the brain cause necrosis and progression of cognitive decline.
Neurological Complications
Vascular Abnormalities
The microvascular network is considered to be the most sensitive part in response to radiation. This is because the radiation due to gamma rays induces cerebral microvascular injury. This radiation-induced vascular disruption is somewhat similar to the vessel defects in vascular dementia. Radiation destabilizes the plasma membrane and also other cells including vascular endothelium. The endothelial cells show an appreciable sensitivity to the radiation. 7 These endothelial cells die and adhere the platelets to the exposed matrix. The radiations also cause broadening of basement coverings and lumen replacement by collagen, which ultimately results in vascular damage. This leads to the formation of thrombus and occlusion of blood vessels. Following radiation, vascular insufficiency and infarction can occur because of enhanced atherosclerosis and mineralizing capillary disease in small vessels. These radiations also damage the DNA, leading to senescence of the brain, causing the impairment in the functioning of the cortex, white matter, and hippocampus. Ischemia and white matter necrosis (lesions) are considered to be the primary indications of vascular damage. 8 Ischemia occurs first, accompanied by white matter necrosis in the late stages, with clinical manifestations ranging from moderate cognitive neuropathy to dementia and death. Some evidence also suggests that the radiation has deteriorated the structural integrity of cerebral microcirculation, leading to cognitive impairment.
Inflammation
Radiation modifies the differentiation of stem cell precursors via changes in the microenvironment. It has been suggested that neuroinflammation has been responsible for this altered fate of stem cell precursors. Brain irradiation causes uncontrolled proliferation and activation of microglial cells and release of macrophage-secreted cytokines, chemokines, etc. 9 There is a decline in working memory and a decrease in processing speed, which can be because of the high dose of radiation received in orbitofrontal regions and temporal lobes. 10
In the studies conducted on rats, it has been shown that there is an activation of microglial cells which has led to the release of a higher amount of oxygen reactive species. This oxidative stress has led to the expression of various pro-inflammatory intermediaries such as cytokines, adhesion molecules, and chemokines, thus leading to neuroinflammation. These chronic inflammatory processes were later associated with impairment in cognitive function. Microglial cells or astrocytes are responsible for maintaining hemostasis and modulation of neurotransmitters. Radiation causes the proliferation of these astrocytes, which causes gliosis. 6 A study reported that a single dose of radiation to a rat brain had increased the levels of inflammatory markers in astrocytes within 30 min.
Oligodendrocyte Loss
These are the specialized cells that form the myelin sheath around axons. The oligodendrocyte loss is because of radiation-induced white matter necrosis. Radiation exposure results in oligodendrocyte progenitor apoptosis, thereby contributing to oligodendrocyte cycle depletion. This leads to the loss of white matter credibility through demyelination. 11
Impaired Angiogenesis
Tumor angiogenesis (tumor-induced blood vessel growth) is the most common angiosarcoma in CD-31-positive patients. Irradiation and also other angiogenesis inhibitors such as bevacizumab reduces capillary density in CD-31 positive angiosarcoma patients. 12 Bevacizumab which is also an anti-vascular endothelial growth factor sold under the name of Avastin which is used in the treatment of this angiosarcoma and also other carcinomas and gliomas has been said to deteriorate the cognitive condition by constraining hippocampal plasticity. 11
Radiation Effect on Hippocampus
Radiation Reduces Hippocampal Neurogenesis
In one of the studies, it has been assumed that the primary mechanism responsible for cognitive dysfunction is reduced neurogenesis activity after radiation therapy. Ionizing radiation causes DNA damage which thus leads to a reduction in neurogenesis. 13 In a recent review, it has been shown that the first fractionated dose of the radiation itself is said to cause damage to the tissue structure, cellular water ionization, generation of free radicals, etc. This means that ionizing radiation’s primary target is DNA. Reactive oxygen species or secondary electrons may damage this DNA, resulting in irreversible double-stranded breaks. These breaks can be repaired using error-prone nonhomologous end joining, which is orchestrated by DNA-dependent protein kinase, or homologous recombination, which only occurs in the S or G2 phases. Genomic instability and cell death can occur because of unrepaired DSBs. In a recent journal, it has been shown that radiations have direct interactions with the molecules in the cell, which leads to ionization. Direct ionization leads to the breakage of single or double strands of DNA, whereas indirect ionization leads to the formation of hydroxyl free radicals after water ionization, and this most commonly occurs with LET irradiations. This has been linked to neurogenesis. 14 The subgranular zone (SGZ) of the hippocampus and the subventricular zone (SVZ) of the lateral ventricles are the two areas of the brain where neurogenesis occurs. From there, the neural progenitors are derived from multipotent stem cells capable of differentiating into neurons and glial cells. In the hippocampus, it has been shown that radiation exposure reduces neurogenesis significantly. 15 Superoxide dismutase, an extracellular enzyme, has been involved in neurogenesis and the preservation of cognitive function after radiotherapy. The deficiency of this enzyme has been linked with reduced progenitor cells in the subgranular zone of the dentate gyrus. 16 Some studies suggest that changes that occurred in apoptosis and differentiation of neuronal stem cell precursors in the dentate gyrus part of the hippocampus have been associated with cognitive impairment in rodent models. Also, many animal studies showed an alteration in the neurogenesis process after LET (low linear energy) irradiation, such as X-rays, electrons, and medium-high energy protons with doses of 1Gy. 17 These neuronal stem cells have the capability of self-renewing and generating new neurons, astrocytes, and oligodendrocytes.
A preclinical rat study has shown a dose-dependent reduction and decreased differentiation of these stem cells postradiation. Neurogenesis is a neurogenic microenvironment-dependent process where the endothelial cells and astrocytes are involved in the regulation of neurogenesis. Apart from the neural cells, the radiation also affects the multiplication and discrepancy of stem cell precursors. 18
Pieces of evidence suggest that some growth factors are synthesized by the endothelial cells in the brain, which are involved in the regulation of neurogenesis. In mice studies, the endothelial cells of the irradiated brain have induced apoptosis of neural stem cells through the TGF-β/Smad3 signaling pathway. TGF-β is a cytokine that is released in response to the injury. Its levels are upregulated in the brain following injury. The increase in the TGF-β causes accretion of proteins and leads to microvascular degeneration. The findings show a decrease in neurogenesis in the SVZ region, which is linked to the activation of the TGF pathway, which causes multi-progenitor neuronal stem cells to die. Therefore, the study results also revealed that inhibition of this pathway is the hypothesis, which could considerably improve neurogenesis. 19
On exposure to radiation and other injuries, it was observed that there is a drastic increase in the TGF-β1 synthesis by the endothelial cells of the brain stem cells. From the animal studies conducted, it was found that the high radiation exposure in adult mice caused a decrease in the formation of new neurons. They have proposed that it could be because of the apoptosis of the progenitor cells through the TGF-β1/Smad3 pathway. This hypothesis was further proved through the reversal of the condition when the same TGF-β were blocked, resulting in increased production of new neurons. The cognitive functions of the brain are related to the efficiency of neurogenesis in the brain. This brings us to the conclusion that the fall in neurogenesis in the brain will lead to the reduced cognitive functions associated with the radiation and other injuries in the brain. Progenitor cells in the hippocampus cause the production of more neuronal bodies in the hippocampus, but it was again proved through further animal studies which focused on the TGF-β, and the results were the same as the increased expression of TGF-β and decreased the number of immature neurons in the hippocampal tissues. It further showed that the mice with TGF-β1 elevation had an almost 60% reduction of neurogenesis in the hippocampus. 20 This further supports the view that there is a decrease in neurogenesis in the hippocampal region through the TGF-β/Smad3 pathway in response to injury because of radiation which elevates the levels of TGF-β in the brain.
It was also evident through animal studies that the increased production of TGF-β1 from the astrocytes can lead to cognitive deficit neurodegenerative diseases like Alzheimer’s disease. From the animal study, the overexpression of the active form of TGF-β in the astrocytes will lead to the increased deposition of astrocytes and amyloids, which accelerates the formation of Aβ, leading to the cognitive deficit symptoms synonymous with Alzheimer’s disease. 21 It is evident that the radiation causes an overproduction of TGF-β, and from this data we come to the conclusion that the induction of radiation will lead to the loss of cognition because of the lack of neurogenesis in the hippocampal region of the brain.
An Increase in TGF-β/Smad Signaling Pathway Leads to a Reduction in Neural Stem Cells
TGF-β is a cytokine that controls the primary cellular response to injurious agents and is a central mediator of tissue repair, inflammation, immune response, and fibrosis. It works through a variety of molecular pathways, the most common of which is the Smad pathway. Smad proteins are the key inflammatory mediators. 22 The various activities of TGF-β are schematically represented in Figure 1.
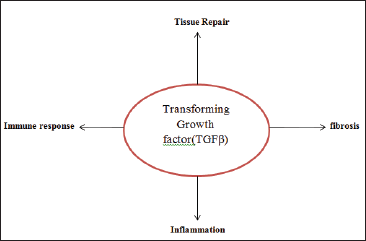
In embryonic growth and adult tissue homeostasis, TGF signaling molecules control various biological processes, including cell proliferation, differentiation, and the induction of apoptosis. In a zebrafish model study, it was identified that Drosophila Mad protein is a cytoplasmic transducer of TGF-β. In the same way, several Mad-related vertebrate proteins, called Smads, were isolated and characterized as the key cytoplasmic TGF mediators. These Smad family proteins are involved in mediating the downstream signaling process. 23
TGF-β induces a long-lasting inhibition of neural stem and progenitor cell proliferation and reduced neurogenesis in adult neural stem and progenitor cell cultures after intracerebroventricular infusion. TGF-specifically arrested neural stem and progenitor cells in the G0/1 process in vitro, even though their self-renewal ability and differentiation fate were unaffected, whereas it did not affect the differentiation fate of newly developed cells in vivo, as shown by bromodeoxyuridine incorporation experiments. As a result, TGF-β is a key signaling molecule in regulating and controlling neuronal stem and progenitor cells in the brain. 23
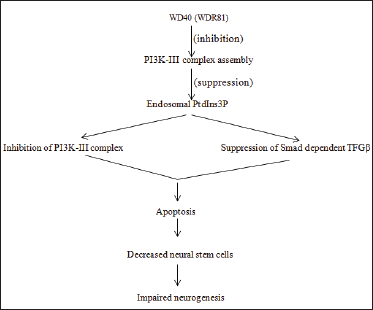
TGF-β signaling involves ligand binding to the constitutively active Type 2 (R2) serine/threonine kinase receptor, followed by recruitment of Type 1 (R1) receptor into the signaling complex. In recent preclinical studies, hippocampal neurogenesis, which is thought to be an important function of the hippocampus, was discovered to be regulated at multiple molecular levels. It revealed that mutations in the WD40 gene, which encodes the protein WDR81, are linked to neurological disorders like cerebellar ataxia, mental retardation, and microcephaly. According to the findings, destroying this protein in adult neural progenitor cells reduced hippocampal neurogenesis and hippocampus-dependent learning. WDR81 inhibits the formation of the PI3K-III complex, which inhibits the synthesis of endosomal PtdIns3P. Inhibition of PI3K-III activity/suppression of Smad-dependent TGF-β has greatly improved the WDR81-deficient mice’s defective adult neurogenesis. According to the findings, defective hippocampal neurogenesis has been linked to the etiology of gene-related neurological disorders. 24 The process is schematically represented in Figure 2.
Other TGF-β-related pathways involved in neurogenesis control include MAP kinase, JAK/STAT pathway, protein kinase c pathway, and ALK5 receptor-mediated pathway. Activation of MAP kinase, JAK/STAT, protein kinase c pathway via TGF-β down-regulates neurogenesis, whereas activation of ALK5 receptor-mediated pathway up-regulates neurogenesis.
Effect of Radiation on Map Kinase Pathway via TGF-β
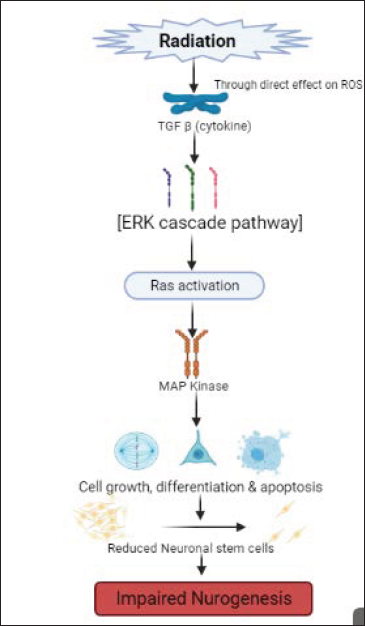
Many signaling pathways are activated in the presence of radiation and also get activated in mitogens like the classical MAPK. But the ability of MAPK signaling pathways to get activated in the presence of radiation also depends on the expression of various other factors like the release of the ligands of growth factors such as TGF-β and the TNF α. 25 Radiotherapy induces damage through the overproduction of reactive oxygen species (ROS) in the body, leading to DNA damage. Increased development of ROS in the body causes injury and activates the MAPK pathway and the NF-kB pathway, which is essential for cell proliferation. Activation of the MAPK signaling will eventually lead to apoptosis induced by the oxidative stress caused by radiation. 26 From the various studies conducted, it was evident that the mitogen-activated protein kinase (MAPK) will be activated by radiation via TGF-β, and on exposing the tissues to ionizing radiations, various signaling pathways which are mediated through the MAPK superfamily will either be down-regulated or activated. Majorly, ionizing radiations will activate all three MAPK via TGF-β in varying intensity. TGF-β causes MAP kinase activation through the ERK cascade pathway (ERK1, ERK2-major subgroups of MAP kinase also known as MEK’s and MKK’s). This causes activation of Ras, which in turn causes activation of Raf. Raf 1 then activates MAP kinase, which is involved in cell differentiation, proliferation, and apoptosis. This apoptosis leads to programmed cell death, leading to reduced neuronal stem cells. This causes impaired neurogenesis. 27
The various signaling pathways which are radiation stimulated and also mediated by MAPK include the c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase, and the p38 MAP kinase. The first wave of the JNK pathway is usually activated by radiation resulting mainly in apoptosis. The second wave of JNK activation because of radiation is mediated through the TGF α and EFGR rather than through the usual pathway because of the TNF α receptor activation. The blocking of the JNK cells during irradiation decreased the sensitivity of the radiation damage to the cancer cells. 28 The P38 pathway works in a similar mechanism as in the JNK pathway, 29 but the potential for the ionizing radiations to regulate the p38 signaling pathway from the various studies conducted was found to be either weak or absent, and, in some cases, it showed very strong activation as well 30 which is schematically represented in Figure 3.
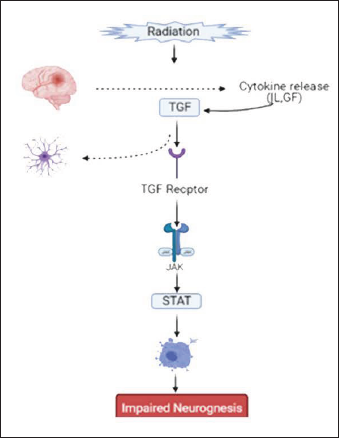
JAK/STAT Pathway
The JAK/STAT pathway is involved in various physiological processes, including cell formation, differentiation, and hematopoiesis. As previously mentioned, radiation is thought to induce the release of cytokines, including interleukins and growth factors. The central nervous system is said to be resistant to the radiation effects, but larger doses may lead to injury and brain damage. The factors responsible for this damage were dose per fraction, the volume of the tumor, and the total amount of dose administered. Radiation-induced brain injury can cause genetic, cellular, and functional changes in the neural, glial, and vascular compartments of the brain. The release of cytokine TGF-β related to brain injury leads to the formation of astrocyte scar. 31 In some studies, it is shown that TGF-β beta directly activates JAK/STAT3 in coordination through the Smad pathway. The TGF-β signaling is associated with binding to serine/threonine kinase receptors ((TGF_RI) and Type II (TGF_RII). In a study, it has been shown that JAK 1 is identified as the interacting protein of TGF-β R1, and it is also shown that JAK 1 activates STAT3 in both SMAD dependent and independent pathways in response to TGF-β. There were 15 identified TGF-β RI interacting proteins, out of which TAK1 and TAB1 are involved in TGF-β-induced JNK and MAPK activation. The JNK undergoes phosphorylation to produce STAT. The study has further shown that these STAT proteins were also phosphorylated in response to TGF-β. The JAK/STAT signaling pathway then gets activated. This completely involves the binding of cytokines (TGF-β) to cytokine receptors which in turn causes dimerization of molecules and causes the release of the Janus kinase enzyme. Phosphorylation of the Janus kinase enzyme at tyrosine sites activates the signal transducer and transcription. Thus, the JAK/STAT pathway is activated, triggering apoptosis, resulting in neuronal cell death and impaired neurogenesis. 32 The pathway is schematically represented in Figure 4.
Protein Kinase Pathway: (Radiation-Induced Apoptosis)
Apoptosis caused by the radiation is controlled by the balance between many proapoptotic and antiapoptotic signals. Many regulators have been identified and analyzed in radiation-induced apoptosis. One of the regulators is protein kinase c which belongs to the serine/threonine kinase family and is involved in regulating apoptosis caused by radiation. Protein kinase c appears to be involved in the control of many cellular processes, including differentiation, proliferation, apoptosis, and metabolism, according to preclinical studies. There are three main subtypes of protein kinase c: classical PKC (cPKC), novel PKC, and atypical PKC. The PKC γ is mainly found in the brain. As studied earlier, radiation causes damage to DNA and membrane, which is the primary step involved in apoptosis. Among many PKC subtypes, PKCδ is said to be involved in the signaling cascade of radiation-induced apoptosis. Firstly, radiation causes the degradation of PKC by caspase 3, and there is the activation of a catalytic factor produced after degradation, which results in apoptosis and interactions with some signaling molecules. This degradation and activation are complemented by translocation from cytosol to nuclear membranes but not in all cases. It may sometimes occur in cytosol also. It is further determined that radiation therapy inhibits protein kinase c by caspase c degradation, which leads to apoptosis, according to the findings. 33
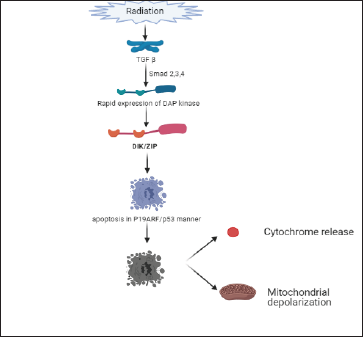
Role of Protein Kinase in Apoptosis via TGF-β
TGF-β induces apoptosis through the protein kinase pathway through the DAP-kinase enzyme (death-activated protein kinase). DAP-kinase is a calcium/calmodulin-dependent serine/threonine kinase that participates in various apoptotic processes. Certain cytokines and oncogenes cause apoptosis, and DAP-kinase is a positive mediator of this process. In a study, it has been revealed that TGF-β quickly induces the expression of DAP-kinase through action on Smad 2, 3, and 4 proteins. Rapid expression of DAP-kinase causes apoptosis in response to TGF. Apoptosis is induced by this DAP-kinase in a p19ARF/p53-dependent manner. One of the five members of the DAP-kinase family is Dlk/ZIP kinase. Overexpression of the Dlk/ZIP kinase has been shown to cause apoptosis in fibroblast cells. This cell death is characterized by various factors like cytochrome c release, apoptotic membrane blabbing, mitochondrial depolarization, and activation of caspase-3. 34 These findings reveal that DAP-kinase-mediated TGF-β-dependent apoptosis is because of the association of Smad proteins to mitochondrial-based proapoptotic events, schematically represented in Figure 5.
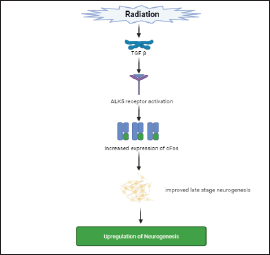
Neuroprotective Role of TGF-β in Hippocampal Neurogenesis: (ALK5 Receptor-Mediated Pathway)
Radiation exposure to the brain causes brain injury in many different ways. In some preclinical studies, it causes direct impairment of brain parenchymal cells and damage to the vasculature component. It is also revealed that radiation leads to brain ischemia. This brain injury causes the release of TGF-β cytokine (released in response to injury). 35
ALK5 is a TGF-β Type I receptor. TGF-β signaling regulates neural stem cell self-renewal in the developing midbrain, and the ALK5 receptor is abundant in migrating neurons in the developing cortex. In transgenic mice studies, it has been revealed that activation of the ALK5 receptor has enhanced the maturation of newborns and significantly improved hippocampal-dependent memory and working. In a preclinical study, it has been revealed that TGF-β Type 1 receptor has been activated by radiation. Radiation is said to activate the TGF-β Type 1 receptor ALK5. Radiation-induced inhibition of endothelial cell migration and sprouting in vitro is prevented by pharmacological ALK5 inhibition, as is angiogenesis impairment in vivo. Impairment of angiogenesis leads to neuronal degradation. Therefore, it is concluded that activation of the ALK5 pathway is required for maintaining angiogenesis. The pathway of upregulation of neurogenesis is schematically represented in Figure 6. The various drugs used in radiation-induced cognitive decline are described in Table 1.
Conclusion
Brain tumors are the most dangerous forms of cancer that are harmful to children’s health and are the second leading cause of cancer-related deaths in children. Many brain tumor patients and survivors exhibit cognitive dysfunction, especially affecting their attention, working, memory, and performance status. Various changes are observed in astrocytes, oligodendrocytes, white matter, vasculature, etc., following radiation treatment in brain tumor patients. Cognitive decline is said to be caused because of the significant effect of radiation on hippocampal neurogenesis. The associated neurological complications include vascular abnormalities, neuroinflammation, oligodendrocyte loss, and impaired angiogenesis.
It has been concluded that activation of TGF-β has significantly reduced neurogenesis through various pathways, leading to an apoptotic effect on neural cells. But some controversial studies are reported that TGF-β showed a neuroprotective effect through the ALK5 receptor-mediated pathway. So detailed studies are required to determine the role of the TGF-β and radiation-associated conative decline because this is one of the potential targets that can be used for the treatment of radiation-associated cognitive decline.
Footnotes
Acknowledgment
All images are adapted and modified by using bio-render version, Bio rad 2021.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
