Abstract
Objective:
To study the safety and efficacy of low-dose etoricoxib and low-dose paracetamol versus ibuprofen and low-dose paracetamol treatments in patients who experienced acute pain after tooth extraction.
Methods:
A total of 80 patients were recruited and randomized to two study groups, i.e., EP and IP. Group EP received etoricoxib 30 mg once a day and add-on paracetamol 325 mg eight-hourly, and Group IP received ibuprofen 400 mg and paracetamol 325 mg eight-hourly for three days. The analgesic efficacy was assessed by a visual analog scale, pain relief score, and global evaluation score. Patients were assessed at 0, 6, 24, 48, and 72 h. Safety was assessed by the patient’s estimation of the severity of adverse drug reactions using a 3-point scale and the type of adverse drug reactions reported by the patients after 72 h.
Results:
Mean pain intensity reduction, mean pain relief score, and global evaluation score all showed better analgesic efficacy results in Group EP as compared to Group IP but were not significant (
Conclusion:
Low-dose etoricoxib with low-dose paracetamol has comparable analgesic efficacy with better safety than therapeutic dose ibuprofen and low-dose paracetamol.
Introduction
Physical injury, infection and antibody formation result in overproduction of prostaglandins thereby initiating the inflammatory process with pain being one of the most important features of inflammation. 1 Tooth extraction, except for the third molar, is a common minor dental surgical procedure associated with mild to moderate pain in patients. 2 Ibuprofen is a traditional nonsteroidal anti-inflammatory drug commonly prescribed for the control of pain and inflammation after tooth extraction. Physical injury, infection and antibody formation result in overproduction of prostaglandins thereby initiating the inflammatory process with pain being one of the most important features of inflammation; however, studies reported an increased risk of gastric mucosal damage and bleeding because of the inhibition of the COX-1 enzyme.3,4 Later, selective COX-2 inhibitors, such as celecoxib, parecoxib, and etoricoxib, were introduced and frequently used, 5 but because of an increased risk of cardiovascular thrombotic episodes, drugs, such as rofecoxib and valdecoxib, are banned. 6
Several studies reported that etoricoxib is a better anti-inflammatory and analgesic drug as compared to ibuprofen, but it is under vigilance for an increased risk of myocardial infarction because of COX-2 inhibition. 7 Two drugs acting by the same mechanism if given together in small doses have better efficacy with lesser incidence of adverse effects. 8 Given the above-mentioned facts, there is a need to conduct studies using a low dose of etoricoxib as an analgesic to minimize adverse effects, such as thrombosis, headache, and fatigue. 9 Paracetamol is an analgesic antipyretic drug with poor anti-inflammatory activity and is devoid of gastric mucosal damage like ibuprofen and thrombotic episodes like rofecoxib. 10 There is no convincing scientific evidence to support the finding that low-dose etoricoxib with low-dose paracetamol has superior analgesic efficacy than the frequently used combination of ibuprofen with low-dose paracetamol. Therefore, the present study is planned to compare the safety and efficacy of the addition of low-dose paracetamol to low-dose etoricoxib with the commonly used drug ibuprofen in patients suffering from acute pain after tooth extraction.
Materials and Methods
Study Design
This was a prospective, randomized, interventional, double-blind comparative study conducted in the Department of Pharmacology and the Department of Dentistry at J.A. Group of Hospital, Gajra Raja Medical College, Gwalior (M.P.). The study was conducted from February 2020 to August 2021 after obtaining approval from the Institutional Ethics Committee (No.431/IEC-GRMC/2019). The study was also registered prospectively in the Clinical Trials Registry of India (CTRI/2020/09/027587).
Sample Size Calculation
The sample size was calculated by using the Epi Info software tool. By considering the power of 80%, a significance level of 0.05, expected population size, and expected tooth extraction frequency from the previous study, the required sample size was calculated to be 40 patients in each group.
Interventions
A total of 80 patients who underwent tooth extraction were enrolled for the study and were randomly divided into two groups, i.e., EP and IP, consisting of 40 patients each. Group EP received etoricoxib 30 mg once a day and paracetamol 325 mg eight-hourly, and Group IP received ibuprofen 400 mg and paracetamol 325 mg eight-hourly for three days. Informed consent was taken from all the study patients. Randomization was done using a random number table.
All the patients who came for tooth extraction, except the third molar, in the dental OPD of both genders between 25 and 60 years of age, weighing between 40 and 70 kg, and having an active mobile number were included in the study. Pregnant and breastfeeding women; patients intolerant to paracetamol, etoricoxib, or ibuprofen; patients with diabetes, coronary artery disease, cerebrovascular disease, hepatic insufficiency, renal insufficiency, gastric disease; patients on any ongoing medication except antibiotics; and patients who had taken analgesic in the previous 48 h were excluded from the study.
Blinding Procedure
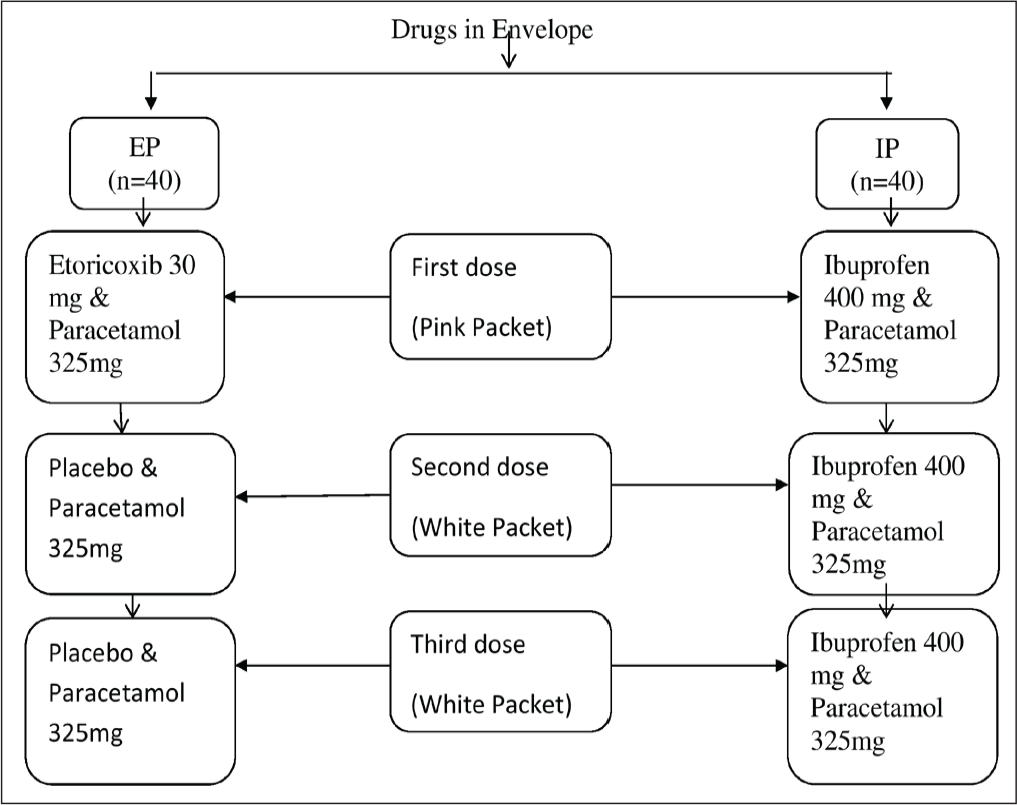
After tooth extraction, each patient was given a sealed coded envelope containing three pink and six white packets of tablets, each packet containing two tablets. Eighty such envelopes containing drugs for each of the patients enrolled in two study groups were prepared, and then the third person who had not been part of the study coded them. The code mark on the envelope assigned to the patient was noted by the investigator. Patients were instructed to take the first dose from the pink packet 2 h after surgery and then the second and third doses from white packets after 8 h and 16 h, respectively, of the first dose per day for three days. All study personnel and participating patients were blinded to treatment assignment for the whole duration of the study. The third person who was not part of the study broke the codes for the final calculation (Figure 1).
Flow Diagram Showing the Preparation of Envelope Containing Packets of Study Drugs.
Questionnaire
All the patients were provided with the questionnaire having a visual analog scale (VAS) and a 5-point pain relief scale before the start of the treatment. Patients were asked to mark on these scales simultaneously at 0, 6, 24, 48, and 72 h after taking drugs. The questionnaire was collected after three days and analyzed.
Efficacy Assessments
Pain intensity was evaluated by the patients’ marking on a horizontal visual analog scale of 10 cm. Patients were asked to mark “no pain” at 0 and “worst possible pain” at 10. 11
Pain relief was measured by the markings done by the patients on a 5-point categorical scale (1 = “poor,” 2 = “average,” 3 = “good,” 4 = “very good,” and 5 = “excellent”). 12
Overall assessment of medication was judged by the global evaluation score (GES) measured after 72 h by asking the patients using a 4-point scale (0 = “poor,” 1 = “fair,” 2 = “good,” and 3 = “excellent”). 13
Safety Assessments
Adverse drug reactions, if any, experienced by the patients were recorded after 72 h of treatment. Patients were also asked about the intensity of estimated adverse drug reactions and were marked on a 3-point scale (1 = “mild,” 2 = “moderate,” and 3 = “severe”). 14
Statistical Analysis
All the data analyses were performed using SPSS version 20 software. Quantitative variables were expressed as the mean and standard deviation. Intragroup (within-group) statistical analysis was carried out using a paired
Results
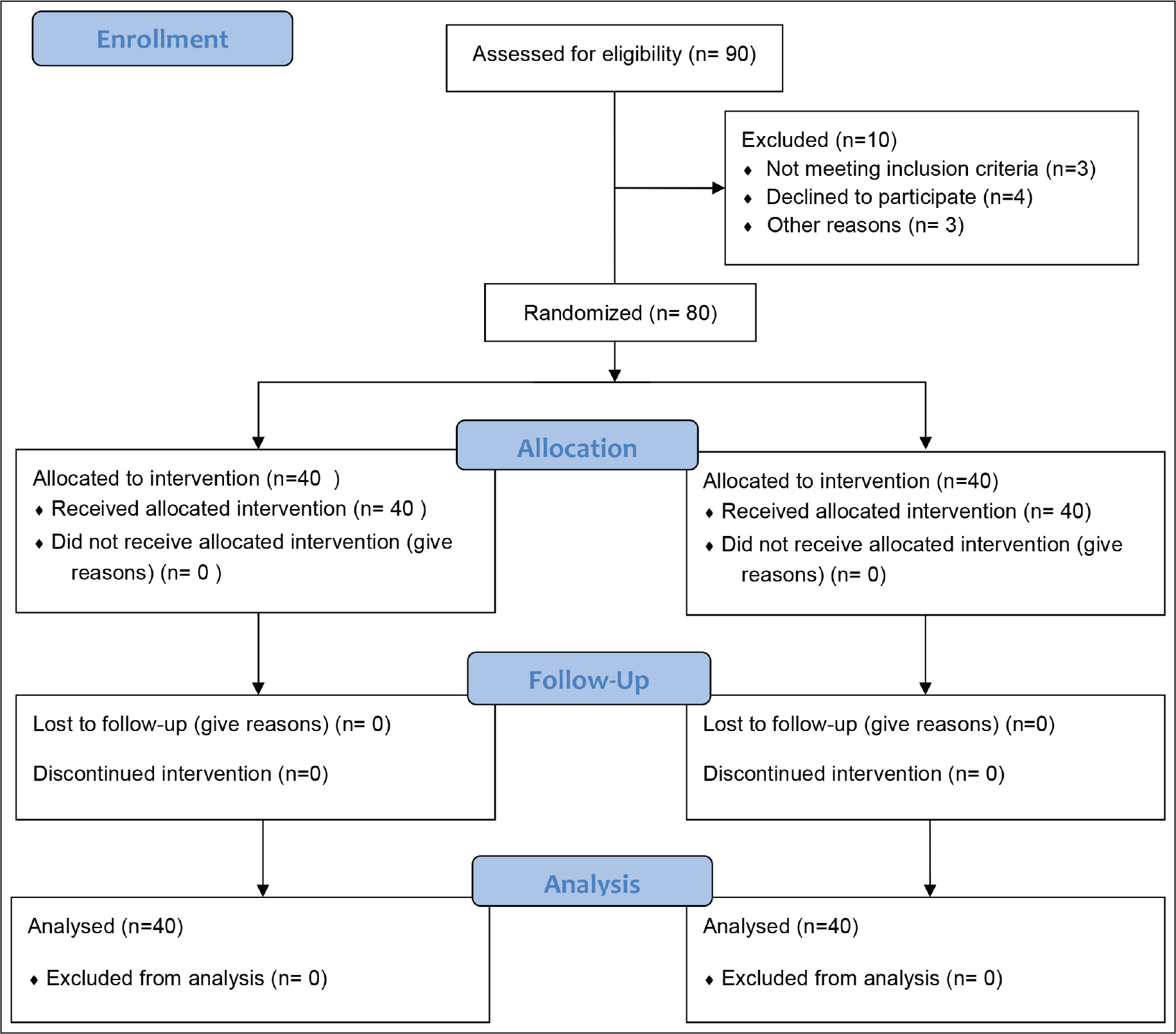
The patient disposal has been depicted in the consolidated standards for reporting trials (CONSORT) style flow diagram in Figure 2. The groups were evenly matched at baseline concerning age and other baseline clinical characteristics.
CONSORT Flow Diagram.
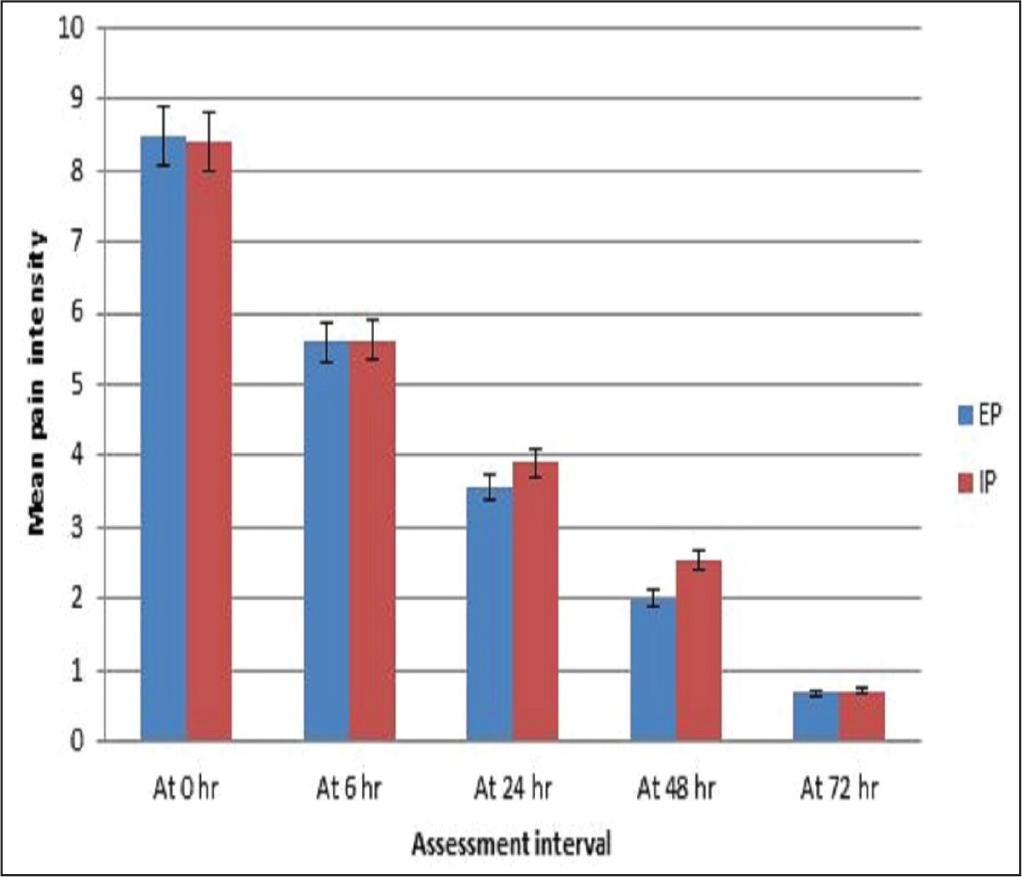
Efficacy Assessment
In Group EP, the mean pain intensity showed a percent reduction of 34%, 58%, 76%, and 92% from baseline at 6, 24, 48, and 72 h, respectively. A significant reduction was seen within the group as compared to baseline at 6, 24, 48, and 72 h (
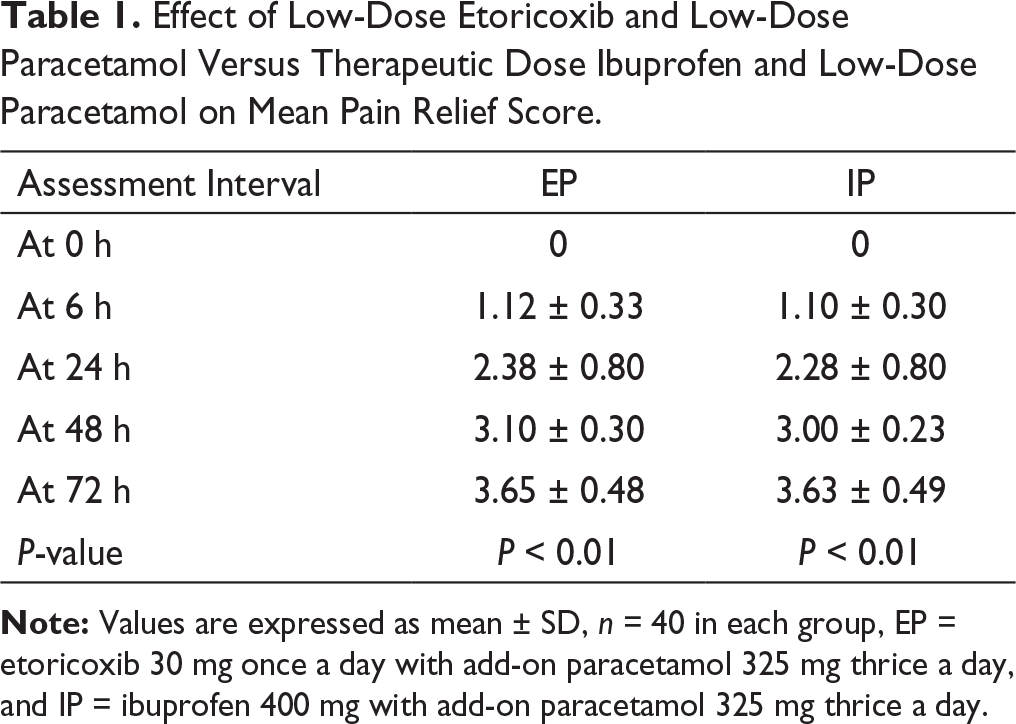
Effect of Low-Dose Etoricoxib and Low-Dose Paracetamol Versus Therapeutic Dose Ibuprofen and Low-Dose Paracetamol on Mean Pain Relief Score.
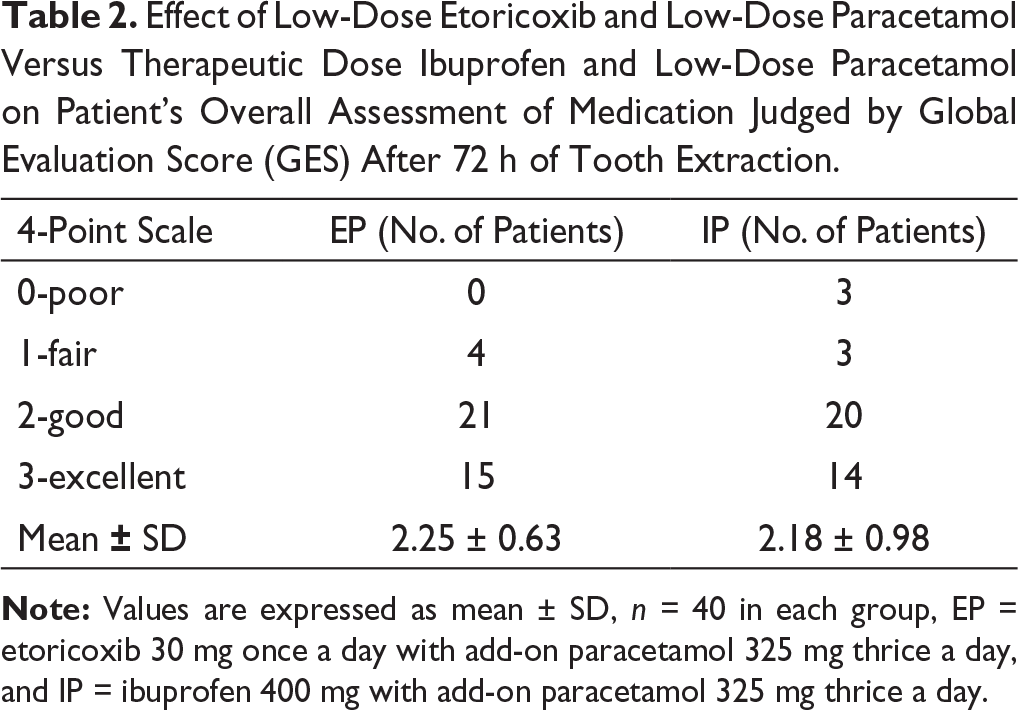
In Group EP, no patient rated their medication as poor, 10% (
Effect of Low-Dose Etoricoxib and Low-Dose Paracetamol Versus Therapeutic Dose Ibuprofen and Low-Dose Paracetamol on Patient’s Overall Assessment of Medication Judged by Global Evaluation Score (GES) After 72 h of Tooth Extraction.
Safety Assessment
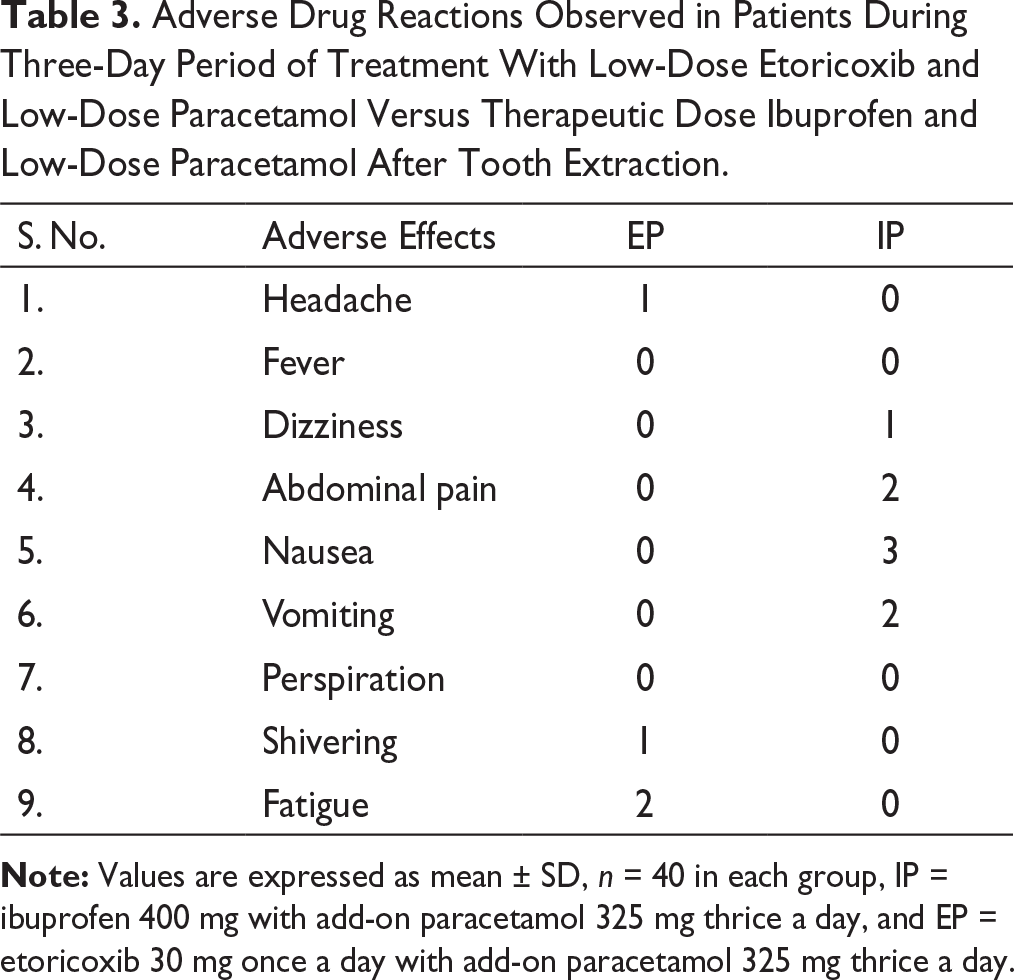
Adverse Drug Reactions Observed in Patients During Three-Day Period of Treatment With Low-Dose Etoricoxib and Low-Dose Paracetamol Versus Therapeutic Dose Ibuprofen and Low-Dose Paracetamol After Tooth Extraction.
Discussion
Patients’ experience acute pain in several inflammatory diseases, such as rheumatoid arthritis, acute gouty arthritis, ankylosing spondylitis, osteoarthritis, bursitis, tooth infection, and posttrauma, so management of pain is a primary clinical necessity. 15
Etoricoxib is currently a widely used analgesic and anti-inflammatory drug found to be advantageous in gastroduodenal mucosal protection but is under supervision for cardiovascular adverse effects. In the present double-blind study, paracetamol in a low dose was added eight-hourly (short half-life of 6–8 h) to a single low-dose etoricoxib (long half-life of 24 h) for the treatment of pain in patients after tooth extraction. 16 The two drugs are not used as a fixed-dose combination, which is irrational because of their pharmacokinetic differences. 17 The results of the study showed that the addition of paracetamol with low-dose etoricoxib showed better efficacy in all the three parameters used to assess analgesic activity, such as reduction in pain intensity on the visual analog scale, pain relief score, and overall satisfaction by treatment using the global evaluation score, than the addition of low-dose paracetamol to ibuprofen in patients after tooth extraction. The study suggests that paracetamol could increase analgesic efficacy when added to low-dose etoricoxib better than ibuprofen in patients undergoing tooth extraction. An earlier study found that paracetamol has predominant COX-2 inhibiting activity 18 than COX-1 inhibiting activity and COX-2 enzyme expression is increased in acute inflammation. 19 Furthermore, COX-1 generated prostaglandins also play some role in producing inflammation, and the addition of paracetamol with etoricoxib broadens the efficacy of COX-2 inhibitor etoricoxib more than that of therapeutic dose ibuprofen. 20
In the present study on comparison of safety, it was observed that none of the patients reported any severe adverse reactions in both groups. Mild to moderate adverse drug reactions were reported in 10% of the etoricoxib-treated group and 20% of the ibuprofen-treated group, suggesting that etoricoxib is an overall safer drug than ibuprofen. GIT intolerance was seen in 17% of cases in the ibuprofen-treated group, whereas no incidence of GIT intolerance was noticed in the etoricoxib-treated group, which confirms their gastroprotective nature. 21 Further adverse effects reported in the ibuprofen 400 mg and paracetamol 325 mg received groups were mostly of gastrointestinal intolerance even in a short treatment period; this supports the concomitant use of proton pump inhibitors with these drugs, especially when treatment is to be continued for a longer period that will increase treatment cost also. 22 This is a small study of a short duration, which is the limitation of the study. There is a need to conduct several multicenter studies in different pain-producing conditions to validate the efficacy and safety of low-dose etoricoxib coadministered with low-dose paracetamol.
Conclusion
Thus, low-dose etoricoxib with add-on paracetamol is found not inferior in analgesic efficacy but superior in safety as compared to conventional drugs. It can be used safely as an alternative to traditional NSAIDs for the treatment of pain.
Footnotes
Acknowledgment
We are thankful to Dr Sameer Gupta Dean, Gajra Raja Medical College, Gwalior (M.P.), for his unstinted help in conducting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
