Abstract
Antivitamins are compounds that negate the biological effects of vitamins. They have been successfully exploited for the development of various classes of drugs. In the early 19th century, the antifolate prontosil was developed for the treatment of puerperal fever. Since then, numerous other antifolates have been used to treat a wide range of infections. Antifolates, such as methotrexate, are potent anticancer agents and antivitamin K, such as warfarin, are used as anticoagulants. Despite several years of research, most antivitamin-based drugs are limited to vitamin K and B9, and the development of antagonists for other vitamins is still in the nascent stage. In the era of antimicrobial resistance, antivitamins can be considered as a promising alternative to develop newer antimicrobials and are worth exploring further. This review discusses key antivitamins at different stages of development which have potential utility as antibiotic drug candidates. The summary of studies of antivitamins in clinical development is also narrated.
Introduction
Antivitamins are the compounds that hamper the essential effects of vitamins. Based on their mode of action, antivitamins are broadly divided into two classes: Class A and B. Class A antivitamins are inhibitors whereas the class B are structural modifiers. 1
Antivitamins hold countless medicinal applications. Over the years, a wide array of drugs has been developed from antivitamins ranging from anticoagulants to anticancer drugs. Prontosil, the first commercially available antimicrobial agent for humans, is a B9 antivitamin.1, 2 Another important example of antivitamins is the anticoagulant warfarin, an antivitamin K. 1
Antivitamins are attractive antimicrobials for several reasons. First, many microorganisms have vitamin transporters, making the delivery of the vitamin analogs to the target molecules very efficient. Second, vitamin analogs act on multiple cellular targets, thus reducing the odds of resistance development. 3
Excessive and unnecessary usage of antibiotics has led to the ever-growing menace of antibiotic resistance. To overcome the rising army of antibiotic-resistant organisms, novel antibiotics are required; and the antivitamins are promising candidates. Despite the tremendous potential of antivitamins as antibiotics, this area of research is considerably unexplored. In this review, we discuss the potential antivitamins B1, B2, B9, and B12 and their antimicrobial actions.
Thiamine (B1) Antivitamins
Vitamin B1, thiamine, is a crucial vitamin for the maintenance of metabolic functions of most living organisms. Thiamine diphosphate (ThDP) acts as a coenzyme for several enzymes, such as oxoglutarate ferredoxin oxidoreductase, 2-oxoglutarate dehydrogenase, oxalate oxidoreductase, pyruvate ferredoxin oxidoreductase, pyruvate dehydrogenase, pyruvate oxidase, dihydroxyacetone synthase, transketolase, 1-deoxy-D-xylulose 5-phosphate synthase, phosphoketolase, pyruvate decarboxylase, and indolepyruvate decarboxylase, in prokaryotes involving carbohydrate, lipid, and amino acid metabolism (Figure 1). Furthermore, bacteria, fungi, and plants possess the de novo pathways for biosynthesis of ThDP or thiamine pyrophosphate (TPP) unlike mammals. 4
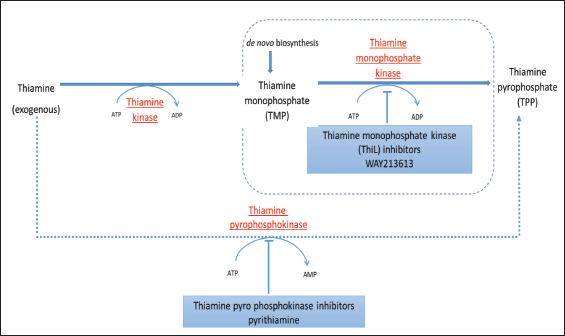
Thiamine antivitamins were first developed to induce thiamine deficiency in experimental models.5, 6 Nowadays, they show great prospect in the field of medicine. Some examples of synthetic thiamine antivitamins include oxythiamine, amprolium, pyrithiamine, and 3-deazathiamine. 6 Recently, a natural antivitamin of thiamine was discovered known as 2′-methoxy-thiamine 7 (Figure 1). The biosynthesis of TPP has been an appealing target for developing antibiotics against bacteria. The Pseudomonas aeruginosa thiamine monophosphate kinase (PaThiL) is a pivotal enzyme at last step of biosynthesis of TPP. Furthermore, it is demonstrated that noncompetitive inhibition of PaThiL by WAY213613 exhibits potent action against P. aeruginosa. 8 This biosynthesis pathway involving ThiL is also found in other bacteria such as Salmonella typhimurium and Escherichia coli. However, in fungi, such as Saccharomyces cerevisiae, synthesis of TPP occurs from thiamine through thiamine pyrophosphokinase 9 (Figure 2).
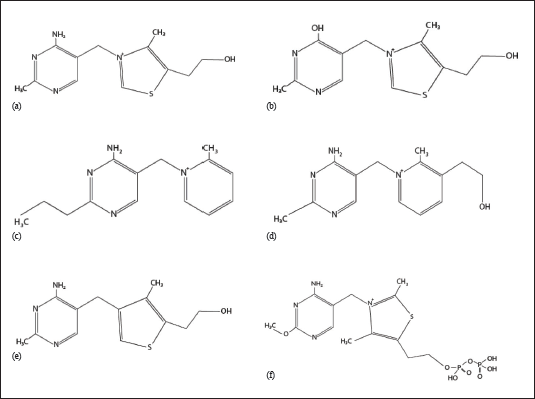
Oxythiamine, pyrithiamine, and 3-deazathiamine inactivate ThDP-dependent enzymes. In addition, pyrithiamine may also impede conversion of thiamine to ThDP by inhibiting the enzyme thiamine pyrophosphokinase. Moreover, action of oxythiamine and pyrithiamine on microbes and fungi is further mediated by action on riboswitches.6, 10 Amprolium exhibits antivitamin action by preventing thiamine uptake. 11 The potential applications of each of these thiamine antivitamins are discussed.
Oxythiamine
Oxythiamine is shown to inhibit the growth of malarial parasite Plasmodium falciparum by acting on a thiamine-related pathway. 12 It also reduces proliferation of yeast, such as S. cerevisiae.13, 14 It could act synergistically with ketoconazole to reduce fungal growth especially those of the genus Malassezia. Hence, this antithiamine can be considered for being a supportive agent in superficial mycoses treatment. 13 Similarly, oxythiamine also confers resistance to Lansing strain of poliomyelitis virus in mice, shedding light on its potential as an antiviral agent which needs to be explored further. 15 In addition, oxythiamine shows interesting antitumor properties. It inhibits cell proliferation in MIA pancreatic carcinoma lines and Ehrlich’s tumor and demonstrates antimetastatic effect against Lewis lung carcinoma. Moreover, when combined with other anticancer agents, it is effective against drug-resistant colon adrenocarcinoma and chronic myeloid leukemia.6, 16 Thus, it is a promising drug candidate with potential in targeted therapies.
Amprolium
Amprolium is widely used in the treatment of coccidiosis in cattle and poultry.6, 17 It has also been used in humans for treatment of severe coccidiosis in acquired immunodeficiency syndrome (AIDS) patients unresponsive to standard treatment. 18
Pyrithiamine
Pyrithiamine, when given in small doses, inhibits growth of fungi and bacteria. It is used to induce thiamine deficiency in experimental conditions 6 (Figure 2).
2′-Methoxy-Thiamine
2′-methoxy-thiamine (MTh) is a naturally occurring antivitamin B1 derived from bacimethrin. 19 It inhibits ThDP-dependent enzymes of bacteria. It shows differential effects on human and bacterial enzymes, with no inhibitory effects on the former. 20
Thiamine antivitamins can be considered as useful agents in the treatment of fungal and bacterial infections as well as cancer. However, exposure to trace amounts of B1 antivitamins in food can adversely affect metabolism. Thus, there is a need to develop new methods to detect these trace amounts in our diet and environment and study the effects of such contamination on our health. 20
Riboflavin (B2) Antivitamins
Riboflavin, vitamin B2, is a direct precursor for the cofactors flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD), which carry out a wide range of biochemical functions. 3 Riboflavin antivitamins can be divided into natural and synthetic agents.
Natural Riboflavin Antivitamins
Cofactor F420, molybdopterins, roseoflavin (RoF), and 8-demethyl-8-amino-riboflavin (AF) are natural riboflavin analogs.21, 22 RoF and AF are the only natural antivitamins B2 with antibiotic action. 22 Both of them are synthesized by Streptomyces davawensis. They demonstrate antimicrobial action not only against gram-positive bacteria, such as Bacillus subtilis, but also against gram-negative bacteria if uptake systems for flavins/flavin analogs are present.3, 23, 24 Both RoF and AF inhibit flavoenzymes and block the FMN riboswitch, ultimately resulting in riboflavin deficiency. In addition, RoF also shows antioncogenic potential. One study has demonstrated an economical method for the large-scale production of RoF. 25 Natural B2 antivitamins have a few disadvantages. A single mutation in ribG FMN riboswitch or in the flavokinase/FAD synthetase gene ribC can result in resistance to RoF. 26 Also, the adverse effects of natural riboflavin antivitamins on human metabolism cannot be dismissed. 22 AF is less toxic for humans than RoF making it a better alternative for the development of novel antibiotics. 3
Synthetic Riboflavin Antivitamins
The synthetic RF analogs isoriboflavin (5, 6-dimethyl-9-Dribitylisoalloxazine), dichloroflavin and galactoflavin effectively inhibit the growth of various organisms by inhibition of flavoenzymes. However, the in vivo toxicity of these synthetic antivitamins overrides their use as antibiotics. 3
Folate (B9) Antivitamins
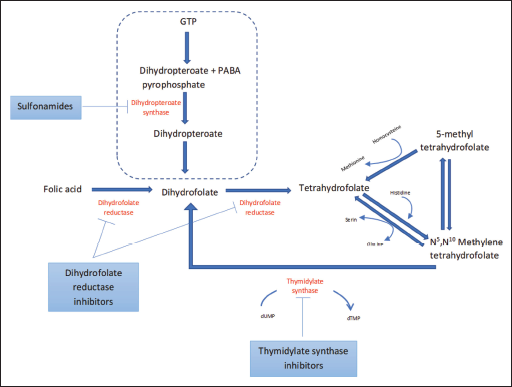
Folate plays an essential role in the synthesis of DNA, RNA, and amino acids. Folate antivitamins, also known as antifolates, are classified into two groups: classical antifolates and nonclassical antifolates. Classical antifolates are structural analogs of folic acid whereas nonclassical antifolates are structurally dissimilar compounds. 27 Antifolates have three mechanisms of action: (a) inhibition of folic acid biosynthesis, (b) hindering cellular uptake, and (c) prevention of folate-dependent enzymatic processes (Figure 3).
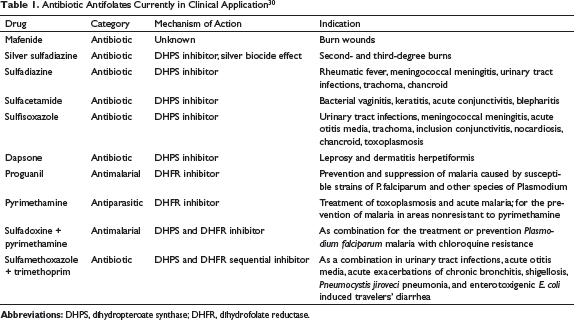
Antibiotic Antifolates Currently in Clinical Application 30
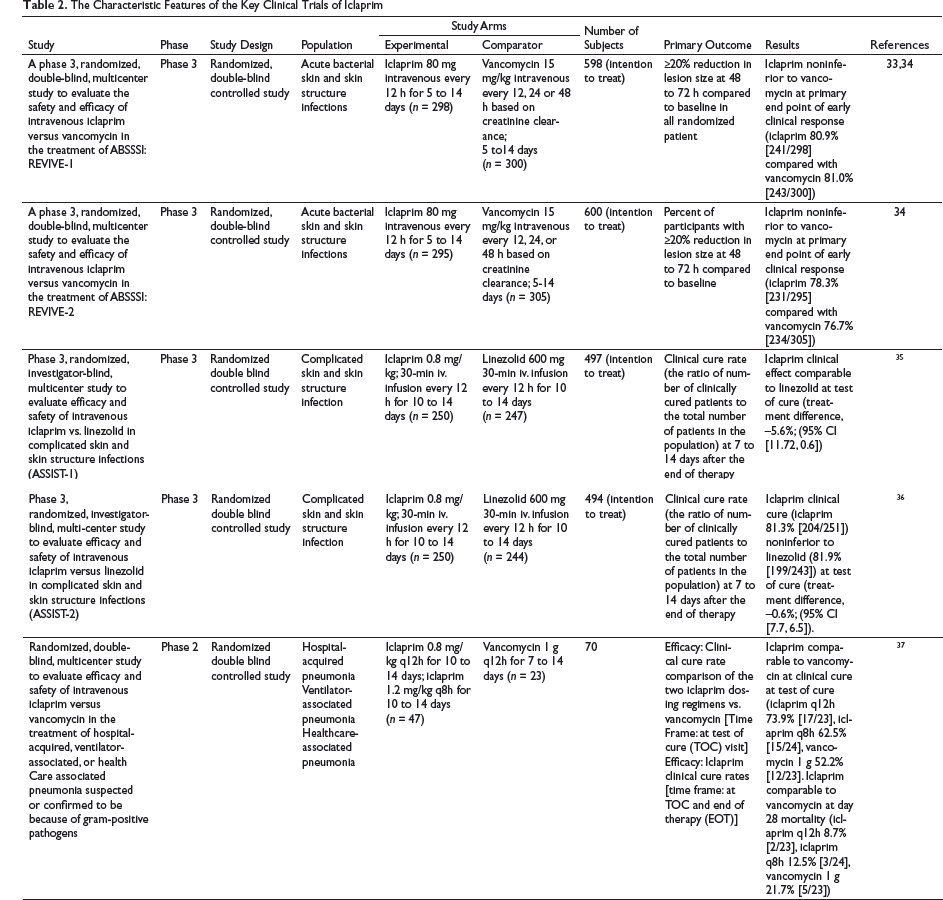
Iclaprim
The Characteristic Features of the Key Clinical Trials of Iclaprim
Propargyl-Linked Antifolates
The propargyl-linked antifolates (PLA) are an upcoming class of drugs designed to target both TMP-sensitive and TMP-resistant organisms. These compounds are found to be effective against both gram-positive and gram-negative bacteria, including multidrug-resistant Staphylococcus aureus.38, 39 In addition, PLAs also inhibit Mycobacterium tuberculosis, with two compounds being very potent inhibitors of multi-drug resistant (MDR) and extensively drug resistant (XDR) strains as well. 38 The PLAs have low frequency of inducing resistant mutations. 39
Abyssomicins
Abyssomicins are antifolates that inhibit the para aminobenzoic acid (PABA) synthesis in the chorismate pathway. They are class I spirotetronate polyketides belonging to the class of tetronate antibiotics. Not only do the drugs inhibit mycobacteria and gram-positive bacteria but they also demonstrate action against latent HIV reactivation and antitumor activities. 40 Abyssomicins selectively inhibit the enzyme 4-amino-4-deoxychorismate synthase which catalyzes the transformation of chorismate to PABA, an intermediate in the folic acid pathway. 41
4′-Desmethyltrimethoprim (4′-DTMP)
Recently, a derivative of TMP called 4′-desmethyltrimethoprim (4′-DTMP) was identified to overcome antibiotic resistance to TMP. The 4′-DTMP is more effective than TMP against mutant strains of E. coli and exhibits antibiotic action similar to TMP against several gram-negative and gram-positive species. Moreover, it confers mutations at a markedly slower rate as compared to TMP and hampers the development of resistant mutations. 42
Novel DHPS Inhibitors
Novel inhibitors of the DHPS enzyme are currently being explored as alternate antifolates. Pterin–sulfonamides conjugates, pterin site inhibitors, and pterin/pABA site binders are some of the compounds being studied.28, 43
Cobalamin (B12) Antivitamins
Vitamin B12 is a cofactor in DNA synthesis and contributes to the normal functioning of the nervous system and the maturation of red blood cells. Antivitamins B12 are metabolically inert cobalamins (Cbls) with molecular structures resembling vitamin B12 (CNCbl or AdoCbl) that induce “functional” B12 deficiency.44, 45 Examples are the aryl-cobalamins such as 4-ethylphenyl-cobalamin (EtPhCbl). A limitation of EtPhCbl is its light sensitivity; exposure to light converts EtPhCbl into Cbls which are precursors for the B12-cofactors, thus branding them as “conditional antivitamins” whose effects can be reversed by irradiation. Recently, a group of light-stable antivitamins called as organometallic alkynyl-Cbls (e.g., 2-phenyl-ethynyl-cobalamin, PhEtyCbl) was created in order to overcome the limitation of EtPhCbl.44, 46
Antivitamins B12 inhibit vitamin B12-dependent enzymes by reducing synthesis and availability of B12 cofactors, thus impairing growth of bacteria.44, 45, 47, 48 They also impair the uptake of essential B12 derivatives by B12-dependent microorganisms. Antivitamins B12 enhance the antibiotic action of sulfonamides when added to the latter. 48 Thus, antivitamins B12 show great promise as novel antibiotics. Moreover, they also show potential as anticancer agents. 1
Despite tremendous efforts, not a single antivitamin B12 has reached clinical trials. Thus, there is a long road ahead in the development of antivitamins B12 for medical applications.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
