Abstract
Sarcopenia, which is defined as an age-related decline in muscle mass and strength, has been found to have a strong bidirectional relationship with Type 2 diabetes. Both problems are associated with advancing age, and have now become of critical importance in view of increasing lifespan and a large increase in the elderly population. Sarcopenia significantly worsens the quality of life and increases morbidity and mortality in patients with Type 2 diabetes. Uncontrolled diabetes, on the other hand, predisposes to worsening sarcopenia. This review discusses the common risk factors and pathophysiology related to these two conditions. We also discuss pharmacological as well as non-pharmacological ways of combating this dual threat.
Introduction
Type 2 diabetes mellitus (T2DM) is one of the most widespread metabolic diseases, and affects more than 500 million people worldwide.[1] More than 90% of diabetes is Type 2 diabetes with an underlying mechanism characterised by insulin resistance. Sarcopenia, which is defined as an age-related decline in muscle strength and muscle function, is found to have a strong association with Type 2 diabetes.[2] Risk factors for sarcopenia include age, dietary factors, systemic inflammation and physical inactivity. Sarcopenia is associated with various negative health outcomes such as increased risk of falls and fractures, cognitive impairment, and reduced overall survival. Sarcopenia is the result of a complex interaction of intrinsic and extrinsic factors. Sarcopenia has a bidirectional relationship with T2DM with each worsening the other. The course of T2DM is associated with declining muscle mass, which reduces physical activity and in turn accelerates the progress of disease. The bidirectional relationship between T2DM and sarcopenia may be related to the common underlying risk factors such as inactivity, inflammation, malnutrition and mitochondrial dysfunction.[3]
The connection between T2DM and sarcopenia has been observed in a number of studies.[4,5] Associations have also been found between complications of T2DM and sarcopenia. Uncontrolled diabetes (high haemoglobin A1c [HbA1c]) and the presence of vascular complications have been found to be associated with an increased prevalence of sarcopenia.[5,6] Another study demonstrating a reciprocal relationship, reported a decrease in the risk of T2DM in older adults with increasing grip strength.[7] Lower limb muscle mass was found to be lower in older adults with T2DM, compared to those without T2DM.[5] Sarcopenia leads to a poor quality of life and increases mortality in T2DM. Patients with T2DM and sarcopenia have been shown to have higher mortality than those without sarcopenia.[8] This review explores recent evidence on the connection between T2DM and sarcopenia, including established and emerging treatment strategies for managing these disorders.
Epidemiology
Population prevalence of sarcopenia in older adults depends on the patient population studied and the definition used. The prevalence among community dwelling older adults ranges between 9.9% and 40%[9] and prevalence among those with T2DM ranges between 15% and 44%[10] using various diagnostic criteria. The Longitudinal Aging Study in India (LASI) focused on adults aged more than 60 years, and using Asian Working Group on Sarcopenia (AWGS) definitions, reported a 27% prevalence of sarcopenia.[11] Participants with diabetes were found to have a significantly higher odds ratio of 1.14 for sarcopenia. Lack of physical activity, physical disability, chronic health conditions and age more than 80 were the other significant risk factors in this study. A study by Sambashivaiah et al. found that muscle strength was significantly lower in patients with T2DM even after adjusting for age and other covariates.[12]
Definition and Diagnosis of Sarcopenia
The diagnostic criteria for sarcopenia have been evolving over the past few years. Two of the most widely used definitions are the ones proposed by the AWGS, and the European Working Group on Sarcopenia in Older People (EWGSOP) (Table 1). They include measures of low muscle strength, low muscle mass and poor physical performance to define sarcopenia. The AWGS defines the condition as an ‘age related decline of skeletal muscle plus low muscle strength and /or physical performance’. It requires low muscle mass with either low muscle strength or poor physical performance to define sarcopenia. The EWGSOP requires both low muscle mass and strength and uses measures of physical performance to describe severity. The European working group recommends a stepwise process for diagnosis and stratification. Initial assessment of risk can be done by the SARC-F (Strength Assistance with walking Rise from a chair Climb stairs and Falls) questionnaire. Muscle strength is measured next using tests such as grip strength (for upper limbs) and chair stand strength (for lower limbs). Total skeletal muscle mass is quantified using methods such as computed tomography (CT), magnetic resonance imaging (MRI), dual energy X-ray absorptiometry (DXA) or bioelectrical impedance analysis (BIA). Measures of muscle function are necessary to quantify the severity of sarcopenia. Some of the commonly used tests include chair rising test (CRT), daily pace assessment and stand up and go test.
Diagnosis of sarcopenia

The 2022 South Asian Working Group on SARCOpenia (SWAG SARCO) consensus focuses on the South Asian population, who have a smaller body size and adiposity as compared to western populations.[13] Using AWGS guidelines also showed a spuriously high prevalence of sarcopenia in elderly South Asian individuals, in view of ethnic differences between the South Asian and South East Asian populations.[14] The SWAG SARCO consensus defines sarcopenia as a syndrome where two of the following three parameters, of muscle mass, muscle strength and muscle function are suboptimal, without giving importance to any one parameter over the others. It also highlights secondary causes of sarcopenia in view of the increasing non-communicable disease burden. SWAG-SARCO proposes the 5-S pathway for screening and diagnosing sarcopenia:
Sarcopenic Obesity
Sarcopenic obesity (SO) is the coexistence of obesity and sarcopenia. Loss of muscle mass is common in individuals with obesity due to metabolic changes seen with a sedentary lifestyle, comorbidities, adipose tissue derangements and the aging process. Sarcopenia may potentiate weight gain through reduced total energy expenditure. Sarcopenia and obesity both worsen each other by mechanisms such as insulin resistance, systemic inflammation and oxidative stress. Coexisting sarcopenia and obesity have been associated with higher cardiovascular risk than either disease alone.[15] Obesity is associated with both sarcopenia and T2DM with more than 50% of patients being obese.
SO should be considered in individuals with an elevated body mass index (BMI)/waist circumference and indicators of impaired muscle mass or function. The European Society for Clinical Nutrition and Metabolism (ESPEN) and European Association for the Study of Obesity (EASO) consensus suggest diagnosis of SO following in high-risk individuals (e.g., elderly, presence of comorbids). They suggest diagnosis using a sequential assessment of muscle strength (e.g., hand grip strength, chair stand test), followed by body composition analysis using DXA, BIA or CT. Diagnosis of SO requires both altered skeletal muscle functional parameters and altered body composition. Additionally, diagnosis requires a relative reduction in skeletal mass adjusted to body weight.[16] Anthropometric measurements such as calf circumference, when adjusted for BMI, can play a role in the rapid estimation of muscle mass.[17] Weight-adjusted waist index (WWI), calculated as waist circumference divided by the square root of weight, showed significant correlation with standard measures of SO in male participants.[18]
Osteosarcopenia
The term osteosarcopenia (OS) refers to the concurrent presence of sarcopenia and osteopenia/osteoporosis. Aging is associated with a chronic low-grade inflammation with a progressive fatty infiltration of bone and muscle tissue.[19] Other factors commonly seen at an older age such as reduced physical activity, poor nutrition and a low vitamin D also predispose to OS. OS is more common in patients with T2DM and their co-occurrence may lead to an increase in falls and fractures. A study by Liu et al. showed that diabetic patients with OS have lower BMI and worse β-cell function, and surmised that better beta cell function could be protective against OS.[20] Another study showed a higher prevalence of OS in patients with chronic complications of T2DM.[21]
Pathophysiology
Aging is one of the most important factors contributing to both T2DM and sarcopenia. A decline in muscle mass starts in the fourth decade and is progressive.[22] Aging is accompanied by an increase in the rate of muscle catabolism. An increase in fat deposition in the visceral as well as intramuscular depots is observed with aging. Impaired anabolic pathways mediated by growth hormone (GH) and insulin-like growth factor 1 (IGF-1), chronic inflammation and a reduced protein intake contribute to an age-related decline in muscle quality (Figure 1). A decrease in the regenerative capacity of skeletal muscle stem cells, which occurs with aging, contributes to muscle loss.
Pathophysiology-intrinsic versus extrinsic risk factors for sarcopenia
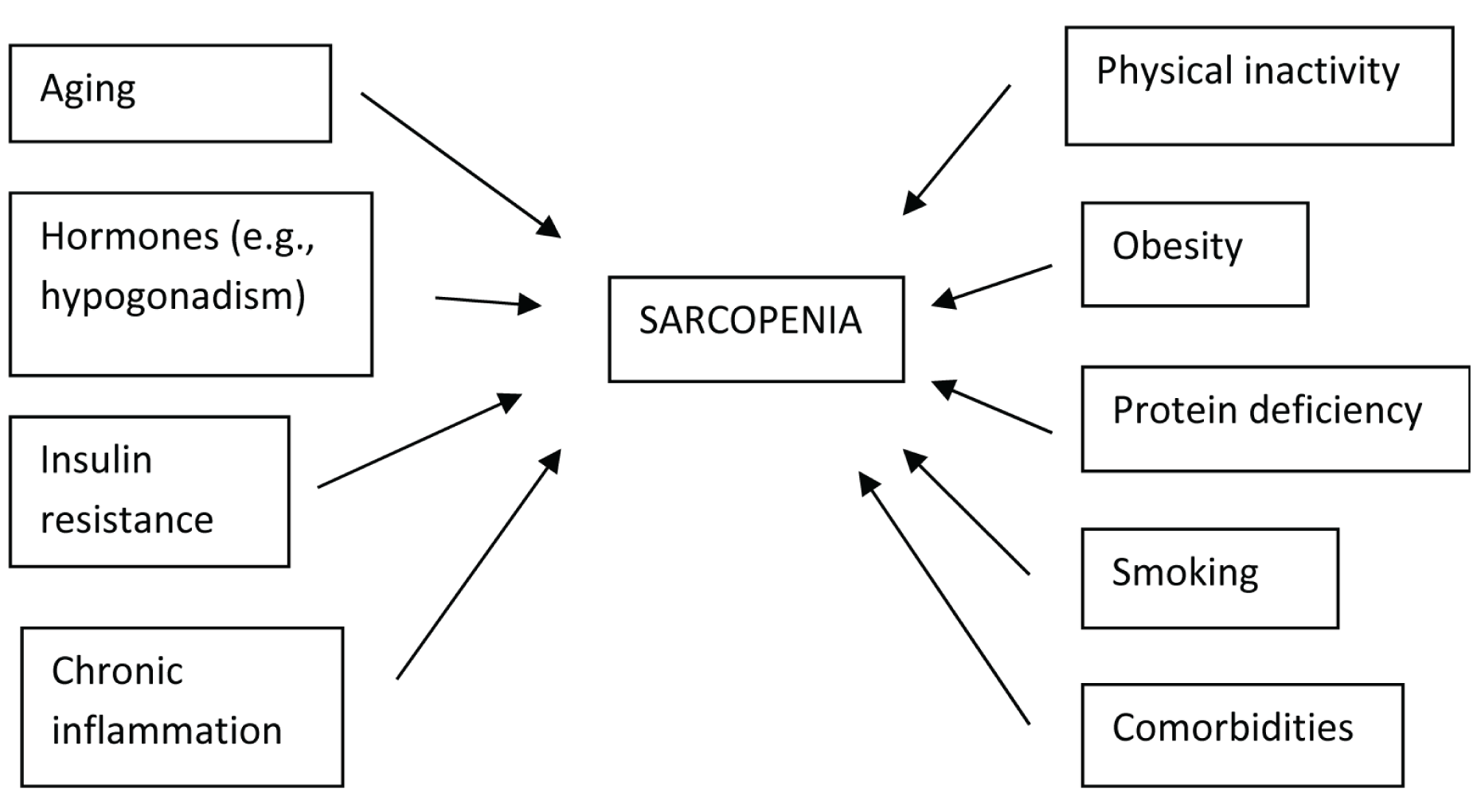
Loss of skeletal muscle mass contributes to increasing insulin resistance and increasing blood glucose. Impaired insulin action in skeletal muscle may interfere with protein synthesis, leading to a decrease in muscle mass.[23] Elevated glucose levels may lead to muscle wasting via the ubiquitin proteasome activation.[24] Insulin resistance inhibits the mammalian target of rapamycin (mTOR) pathway leading to an increase in muscle catabolism. Myostatin, which belongs to the transforming growth factor β (TGF-β) superfamily, decreases mTOR signalling. This leads to the activation of the AMPKα2 pathway, leading to proteolysis. Myostatin could also promote catabolism via the p38-caspase pathway. Insulin resistance in T2DM leads to hyperinsulinemia and increases myostatin activity, thereby causing muscle loss.[25]
An increase in the adipose tissue depots within and surrounding skeletal muscle is called myosteatosis. Myosteatosis increases with aging, obesity and lower physical activity, which contribute to cellular senescence and mitochondrial dysfunction.[26] It is a significant risk factor contributing to both sarcopenia and T2DM. Insulin resistance associated with myosteatosis is a key defected leading to T2DM. Higher intermuscular adipose tissue (IMAT) leads to increased local inflammation and impaired contractility in skeletal muscle.[27] Increased insulin resistance per se increases ectopic fat accumulation, thereby promoting local inflammation.[25]
T2DM is characterised by a chronic inflammatory state. This inflammation, acting via the PI3K-Akt pathway, leads to a decrease in protein anabolism combined with an increase in muscle catabolism. An increase in inflammatory markers such as interleukin 6 (IL-6), tumour necrosis factor alpha (TNF-α) is seen in patients with T2DM and insulin resistance. These inflammatory mediators adversely affect skeletal muscle.[28] IL-6 acts on muscle cells directly and via the janus kinase-signal transducer and activator of transcription (JAK/STAT), nuclear factor kappa β (NF-κβ) pathways and leads to muscle catabolism. TNF-α, IL-1 and IL-10 act via other signalling pathways and induce mitophagy.[29]
Hyperglycaemia in T2DM triggers the production of reactive oxygen species (ROS) leading to oxidative stress. ROS activates the ubiquitin proteasome pathway and leads to muscle catabolism.[30] ROS inhibits the Akt/mTOR pathway and inhibits protein synthesis and muscle repair, thereby promoting muscle atrophy.[31] Hyperglycaemia increases the production of advanced glycation end-products (AGEs), which accumulate in tissues such as muscles and bone. AGEs bind to the AGE receptor on muscle cells and increase inflammation, oxidative stress. AGEs can interfere with muscle contractility by increasing protein cross-linking.[32]
The decline in exercise ability with age is a major contributor to muscle loss in the elderly. Muscle tissue needs constant stimulation and load bearing in order to maintain proper function. Lack of exercise, which is compounded by a fear of falling, will result in a decrease in muscle mass.[33] Sedentary lifestyle with sedentary time exceeding two hours leads to a higher risk of sarcopenia.
Management
Dietary Interventions
There is evidence to suggest that increasing protein intake benefits both T2DM and sarcopenia. A high protein diet has been shown to improve insulin sensitivity.[34] It also decreases postmeal glycaemic fluctuations in patients with T2DM by slowing down the absorption of glucose. Most guidelines for T2DM currently recommend protein intake of 0.8 g/kg-1.5 g/kg body weight per day in patients with normal kidney function.[35] However, increasing the daily protein intake to 1.2 g/kg-1.6 g/kg of body weight may help maintain muscle mass and strength in older adults.[36] Using protein elements to reach an intake of 1.2g/kg body weight per day is beneficial, especially in those with low basal protein intakes. An randomised controlled trial (RCT) studying whey protein supplementation found a significant increase in muscle mass, with no significant change in either upper or lower limb strength.[37] However, the combination of protein supplementation and resistance exercise was found to increase both muscle mass and muscle strength.[37] A meta-analysis of studies looking into the benefits of animal versus plant proteins did not find any difference between the two with regard to increase in total muscle mass or muscle strength, though animal protein was found to be produce a higher percentage gain in muscle mass.[38,39] However, data about benefits of animal versus plant protein remain unclear and need further research.
Hypovitaminosis D is associated with a decrease in muscle mass and physical function.[40] Vitamin D supplementation to maintain adequate vitamin D levels, especially in patients at risk for vitamin D deficiency, helps improve musculoskeletal health in this population.[41] Combining a physical exercise programme, with whey protein and leucine supplementation, in addition to vitamin D optimisation showed significant improvement in muscle mass and function in a meta-analysis including 637 patients.[37]
Observational and cross-sectional studies have shown a positive association between higher magnesium intake and muscle mass, muscle strength, especially in women.[42] Similarly, cross-sectional studies of calcium intake found a correlation between lower calcium intake and the presence of sarcopenia.[43]
Weight Management, Dietary Interventions in Older Adults with Type 2 DM
Weight loss achieved through hypocaloric diets and exercise is a first-line treatment approach in overweight patients with T2DM. On the other hand, higher mortality rate has been reported in older adults with lower BMIs. The lowest mortality risk was reported between BMI of 24 kg/m2 and 30.9 kg/m2.[44] Weight loss, while effective for glycaemic control, leads to loss of muscle and bone mass, especially in older adults.[45] Very low calorie diets are, therefore, not appropriate for older adults in the management of T2DM. Adequate energy and protein intake helps prevent muscle loss. It is important to maintain a balanced diet and adequate protein intake of at least 1g/kg body weight in older adults. Additionally, the daily negative calorie balance should not exceed 500 calories.[46] The Mediterranean diet with an emphasis on whole grains, legumes, vegetables and lean sources of protein is a widely accepted approach. Essential amino acid supplementation has been studied in T2DM and sarcopenia. Supplementation with branched-chain amino acids such as leucine, isoleucine, valine has shown benefit in reducing muscle loss.[47] Leucine also acts as an insulin secretagogue. Omega-3 fatty acid supplementation in combination with exercise shows benefit in some studies as well.[48] Omega-3 fatty acids also reduce inflammation.
Combining exercise with dietary approaches increases insulin sensitivity and provides further improvement in glycaemic control.[49] Exercise can also favourably influence changes in muscle mass that occur with dietary interventions. Studies in older overweight patients on weight-reducing diets have shown that the addition of exercise produces greater improvement in physical function.[50] The addition of resistance exercise showed the most benefit in these patients.
Physical Activity
Physical activity plays an important role in the management of both T2DM and sarcopenia. Exercise can delay muscle loss by counteracting NF-κβ.[51] The American Diabetes Association (ADA) recommends at least 150 minutes of moderate to vigorous intensity aerobic exercise per week, with no more than two consecutive days without exercise. The ADA also recommends resistance exercises at least two to three times per week. Aerobic exercise can improve insulin resistance, blood glucose levels and muscle strength. Resistance exercise decreases insulin resistance, improves mitochondrial function and increases muscle strength.[52] Functional balance and strength training, reduce frailty, falls and fragility fractures in the elderly. Both the ADA and the World Health Organisation (WHO) guidelines for older adults with chronic conditions place an emphasis on the reduction in sedentary time, and to break up periods of sitting every 30 minutes.[53] A majority of older adults spend up to 80% of their waking day sedentary and do not meet the minimum recommended goals for physical exercise. This pattern of behaviour is associated with declining muscle strength, incident T2DM and worse health outcomes.[54]
In the look action for health in diabetes (look AHEAD) trial, adults with T2DM and overweight or obesity were randomised to either intensive lifestyle intervention (weight loss ≥10% and physical activity goal of ≥150 minutes per week in the first month, increasing to ≥175 minutes per week by the 6th month) or diabetes support and education.[55] The intensive lifestyle group was supported by dedicated lifestyle coaches throughout the study. At one to eight years after randomisation, the intensive lifestyle intervention group had better subjective and objective scores of physical performance as compared to the controls.[56] Reducing time spent sitting that is sedentary behaviour has also been shown to improve muscle health.
Exercise interventions can be either aerobic exercise or resistance exercise. Aerobic exercise is typically performed at moderate intensity for prolonged periods of time, with examples including walking, jogging and swimming. Resistance or muscle strengthening exercise involves repetitive muscle contractions against external resistance such as lifting weights. Several studies have investigated whether exercise interventions can improve metabolic health in older T2DM patients. The benefits of resistance training for improving muscle function in older adults with sarcopenia have been proven in a number of studies.[57] The type of resistance training, intensity and frequency of exercise influences the benefits obtained. A meta-analysis of 15 studies involving older adults found that improvements in muscle strength tended to be greater when higher loads were used.[58]
A practical and easy approach in sedentary older adults involves isolated short bouts of exercise, lasting ≤1 minute, performed multiple times throughout the day. The benefits of such an approach for glycaemic control have been demonstrated.[59] A pilot study in older adults using short bouts (9 min) of body weight exercises reported significant improvements in muscle power and muscle mass.[60] Positive changes in muscle strength were observed in frail patients, even with short bouts of less intense strength training.
Control of Blood Glucose Level
Uncontrolled blood glucose levels decrease mitochondrial activity, impair energy metabolism and predispose to sarcopenia. Impaired insulin signalling secondary to hyperglycaemia increases insulin resistance and predisposes to muscle atrophy.[61] Stable insulin levels in people with well controlled T2DM contribute to muscle anabolism. Skeletal muscle in turn aids in glucose disposal and helps control diabetes.
Medication
There are no approved pharmacological therapies for the management of sarcopenia. Inhibitors of myostatin have been found to increase muscle mass in animal models.[62] However, they have shown no effect on physical function. Testosterone treatment improves muscle mass and glycaemic control in hypogonadal men.[63] There is no definite evidence to support the use of testosterone in men without hypogonadism. Selective androgen receptor modulators (SARMs) have recently been investigated in the management of muscle wasting. SARMs are non-steroidal ligands that bind to androgen receptors with tissue selective androgen signalling. However, clinical efficacy of this group of drugs remains to be seen.
Nandrolone is a synthetic anabolic androgen with a higher myotropic: androgenic ratio. Nandrolone has been shown to increase lean body mass in chronic conditions such as HIV and chronic renal failure.[64] Nandrolone combined with resistance training was found to improve insulin sensitivity and lipid parameters in a study involving HIV-infected men.[65] However, there is currently insufficient evidence to recommend its use in the elderly with sarcopenia.
The drugs used to treat T2DM can have different types of effects on muscle function. Metformin, which is one of the oldest drugs used in the management of diabetes, has shown beneficial effects on skeletal muscle.[66] Thiazolidinediones improve insulin sensitivity, decrease intramuscular fat and may be beneficial to muscle.[67] However, they increase the risk of fractures and congestive cardiac failure in high-risk populations. Sulfonylureas can aggravate muscle loss in T2DM and should be used with caution in sarcopenic patients.[68] dipeptidyl peptidase 4 (DPP-4) inhibitors act on the incretin pathway and have some minor beneficial effects on muscle mass. The effects of sodium glucose co-transporter 2 (SGLT-2) inhibitors on muscle mass are inconclusive and need further study. One study of dapagliflozin significantly decreased skeletal muscle mass (−2.1%) compared to placebo over 24 weeks.[69] Two studies with Luseogliflozin showed significant reduction in skeletal muscle indices (−1.9% and −2.9%).[70,71] However, other trials did not report any changes.[72,73] A six-month study of glucagon like peptide -1 (GLP-1) analogue dulaglutide, in patients on haemodialysis showed a significant (−3.8%) decrease in skeletal muscle mass.[74] Studies of liraglutide attributed 33%-77% weight loss to loss of fat free mass.[75] The newest generation of incretin-based medications such as semaglutide (GLP-1 analogue) and tirzepatide (dual GIP and GLP-1 agonist) have shown a lot of promise in the management of T2DM as well as obesity. However, a decrease in absolute lean mass, with preservation of relative muscle mass, has been observed with this group of medications. Studies of semaglutide, reported fat-free mass to comprise 40% of weight lost(6.92 kg lean mass in 68 weeks or 13.2%), which correlates with the loss seen in diet induced weight loss or bariatric surgery.[76] The pivotal study of tirzepatide also showed a loss 10.9% of lean mass during the 72 weeks of the study.[77] Measures to preserve muscle mass and function should, therefore, be taken while initiating treatment with these agents.
Conclusion
Sarcopenia represents a progressive degenerative muscle disease affecting a significant number of older adults in the community. Type 2 diabetes is a metabolic disorder which has increasing prevalence with age. There is a complex interaction between these two conditions with each potentiating the other. Lifestyle factors, age, insulin resistance and elevated blood glucose can decrease muscle mass in diabetic patients. Sarcopenia leads to a poor quality of life with an increased morbidity and mortality in diabetic patients. Early detection helps initiate measures to address sarcopenia and improve quality of life.
Muscle health can be preserved by paying attention to nutrition, physical exercise and hormonal considerations. A diet rich in protein, essential amino acids, vitamin D, calcium and omega-3 fatty acids with adequate calories needs to be encouraged. A customised exercise plan encompassing aerobic and resistance exercises needs to be given to each patient. While prescribing diabetic medications, the impact of the drug on skeletal muscle also needs to considered in addition to efficacy of glycaemic control.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not applicable.
Informed consent
Not applicable.
Credit author statement
SS conceived the article and designed the layout. SS wrote the original draft and revised and edited the final manuscript.
Data availability
Not applicable.
Use of artificial intelligence
Not applicable.
