Abstract
Background and Aim:
Diabetes is a chronic metabolic disorder with increasing prevalence and mortality. Historically it has been considered as a controllable but incurable disease. In recent years, the concept of diabetes remission has gained significant attention due to its potential benefits in achieving normoglycemia without any medication. This review aims to explore the understanding of diabetes remission, its mechanism and its clinical implications for managing Type 2 diabetes.
Methodology:
The information and data regarding the topic were acquired from different platforms such as PubMed, Google Scholar and clinical trials, including meta-analysis, multicentric trials, observational studies and systematic reviews.
Results:
Emerging studies revealed that significant changes in lifestyle, intensive and regular physical activity, early insulin therapy and surgical interventions can lead to remission in individuals with Type 2 diabetes. Further, the rate of remission was high in patients who underwent early-stage interventions. The underlying mechanisms of remission are recovery of pancreatic β-cell function, improvement in insulin sensitivity and weight loss. Even though remission is a new hope for diabetes patients, it is not a permanent cure and relapse can occur. Current research is evolving to incorporate remission as a therapeutic goal.
Conclusion:
Research on diabetes remission is continuously evolving, and it could be a transformative approach to diabetes management. Remission can shift the concept from lifelong manageable disease to short-term management. Achieving remission depends on early intervention and lifestyle modifications with regular monitoring.
Introduction
Diabetes mellitus (DM) is a chronic progressive metabolic disorder characterised by persistent hyperglycaemia, posing a significant threat to individual well-being and the healthcare system.[1] It affects 537 million people worldwide and its prevalence is expected to increase 643 million by 2030.[2] Type 2 diabetes (T2D) accounts for 90%-95% of all diabetes cases, and is strongly linked to lifestyle factors such as poor diet, stress, physical inactivity and obesity. The progressive nature of T2D gives rise to various micro and macrovascular complications like diabetic nephropathy, neuropathy, retinopathy and cardiovascular disease that result in increased mortality.[3]
The conventional therapeutic approach for T2D has focused on lifelong management, prioritising blood glucose control and reducing complications with the combination of medications and lifestyle changes.[4] However, as diabetes progress, it requires intensive treatment to maintain glycaemic control. Despite significant advancement in therapeutic modalities, it remains a chronic and incurable disease requiring continuous care. Recently, there has been growing interest in diabetes remission which continuously evolves to achieve cure for T2D or freedom from the disease.[5] This review is focused to provide an overview of the current understanding of disease progression, diabetes remission, mechanisms, treatment strategies and clinical implications.
Chronic and Progressive Nature of Type 2 Diabetes
T2D is a chronic and progressive disease that starts with insulin resistance and compensatory hyperinsulinemia. As gradually β-cell dysfunction sets in, there is a relative insulin deficiency and this results in hyperglycaemia. The progression from normal condition to diabetes involves a combination of genetic predisposition, metabolic disturbances and environmental factors.[6,7] The disease can be broadly divided into two stages (Figure 1).
Stages of Type 2 diabetes development and progression
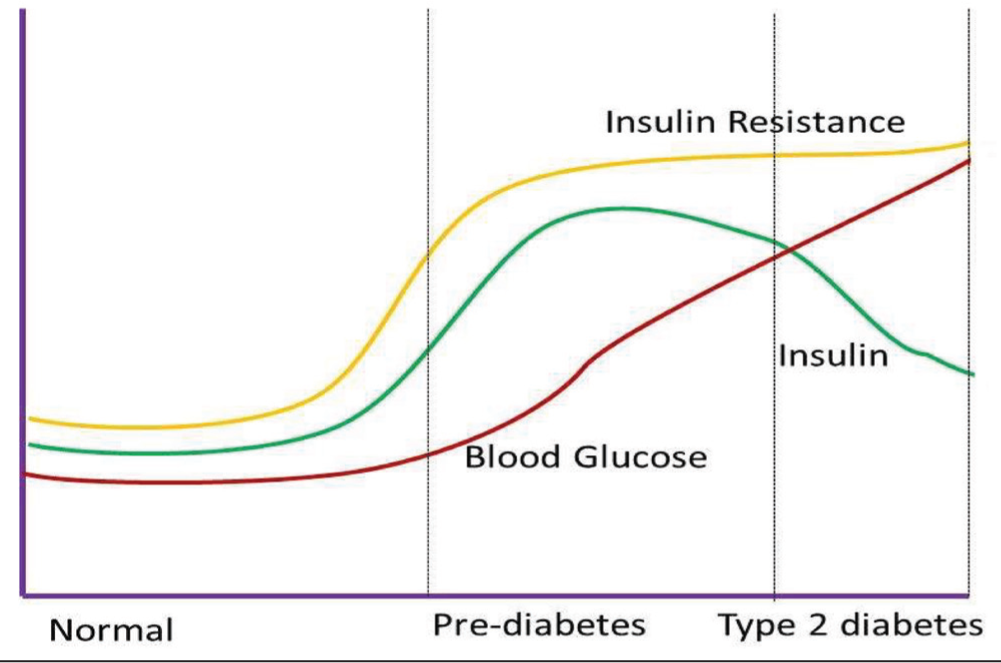
Pre-diabetes is an early stage, individuals may have normal fasting glucose levels, but their muscle, liver and fat cells become less responsive to insulin. It is often referred to as insulin resistance or the pre-diabetic stage. In this stage, the pancreas compensates by producing excess insulin to maintain normal glucose levels resulting in hyperinsulinemia.[8,9] A major challenge in insulin resistance is that most people are asymptomatic and difficult to detect unless it’s specifically looked into. Moreover, there is an increase in hepatic glucose output due to an increase in glucagon secretion, contributing to postprandial hyperglycaemia. Further, impaired incretin response also interrupts glucose homeostasis, thereby exacerbating metabolic dysregulation.[10,11]
Current medications for T2D primarily focus on decreasing blood glucose levels, either by augmenting insulin sensitivity, enhancing insulin secretion from residual β-cells or exogenous insulin therapy. Though new drugs like incretin based therapy, sodium glucose cotransporter-2 inhibitors (SGLT2i) and glucagon like peptide-1 receptor agonist (GLP1-RA) have drastically changed the spectrum of diabetes management, they are yet to halt the progression of disease.[14,15] A key challenge in T2D management is identifying therapeutic strategies that can mitigate the progressive decline in β-cell function and mass, which prevents disease progression and if possible reversal of the disease.
Remission Definition and Its Importance
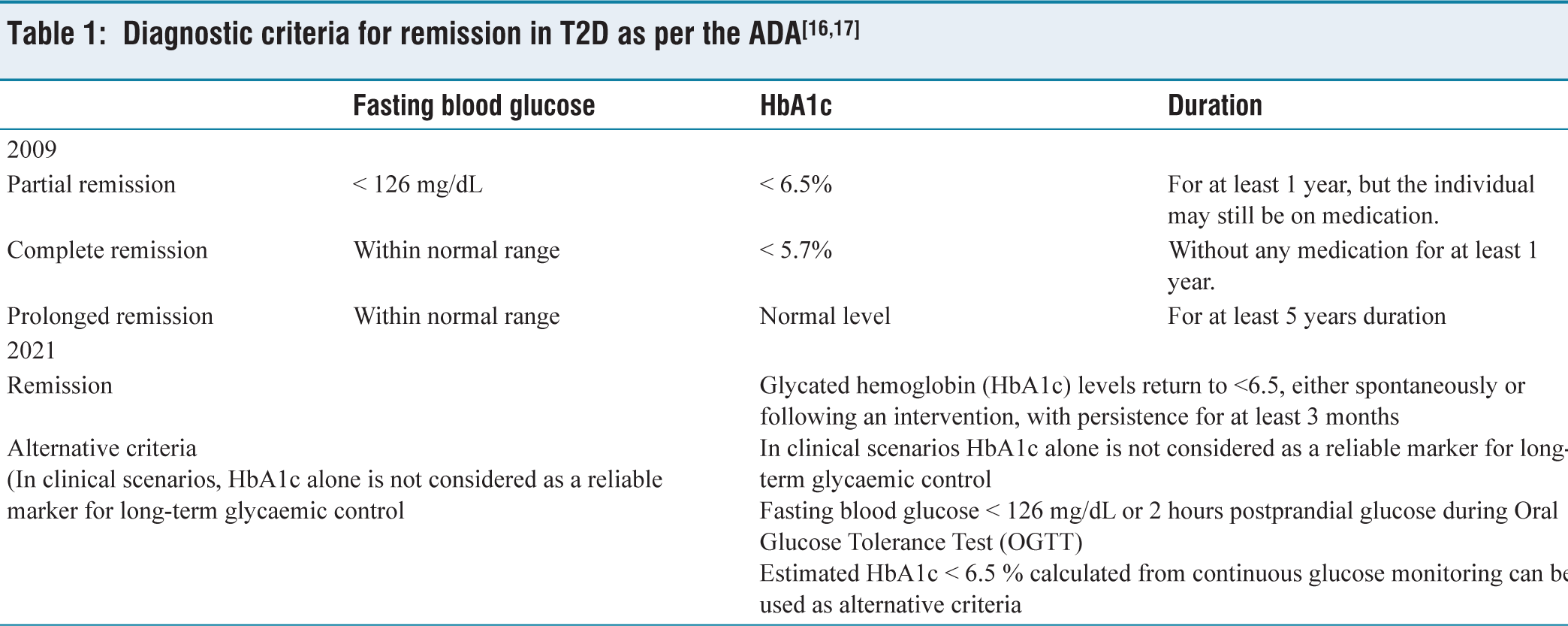
According to the American Diabetes Association (ADA), remission in the context of T2D is defined as a period when blood glucose level is below the diagnostic threshold without the need for medications. The criteria for defining T2D remission are outlined in Table 1.[16,17]
Diagnostic criteria for remission in T2D as per the ADA[16,17]
The ADA’s 2025 Standards of Care in Diabetes do not specifically address diabetes remission, but the diagnostic and management recommendations could help individuals to achieve remission.[18] Remission refers to the absence of disease manifestation and a reduced a risk of emerging complications. It is an innovative concept for people living with T2D, as it challenges the long-term notion that T2D was a permanent, inevitably progressive disease. Remission implies that diabetes progression has been partially reversed or arrested, but the risk of relapse remains if lifestyle changes are not maintained. It is distinct from a ‘cure’ where the disease is completely eradicated and pancreatic function restored to normal.[19]
Remission can be easily achieved in naive T2D with early lifestyle interventions, intensive insulin treatment and surgery by preserving β-cell function. However, achieving remission becomes more challenging in long standing T2D due to progressive β-cell decline. The attainment of diabetes remission offers significant benefits in multiple facets of an individual’s life beyond improved glycaemic control. Remission of T2D can avoid or delay diabetes associated complications. Further, it eliminates the need for medication, fosters self-empowerment and also reduces the emotional, physical and financial burdens associated with the condition.[20,21]
β-cell Dynamics
The reversal of T2D is an intricate and multifaceted process, and achieving complete remission is a challenging and elusive goal. To investigate the reversal of T2D, a comprehensive understanding of β- cell dynamics is essential.[22] β-cell dysfunction plays a vital role in the development of T2D, as it causes impaired glucose homeostasis. Emerging evidence suggests that alteration in β-cell identity and functional β-cell mass reduction in individuals with T2D also contributes to the impairment of β-cell function. The β-cell mass is maintained by a dynamic equilibrium between β-cell proliferation, (via replication and neogenesis) and β-cell loss through apoptosis[23,24] (Figure 2).
Regulation of β-cell mass: A balance between β-cell proliferation and loss
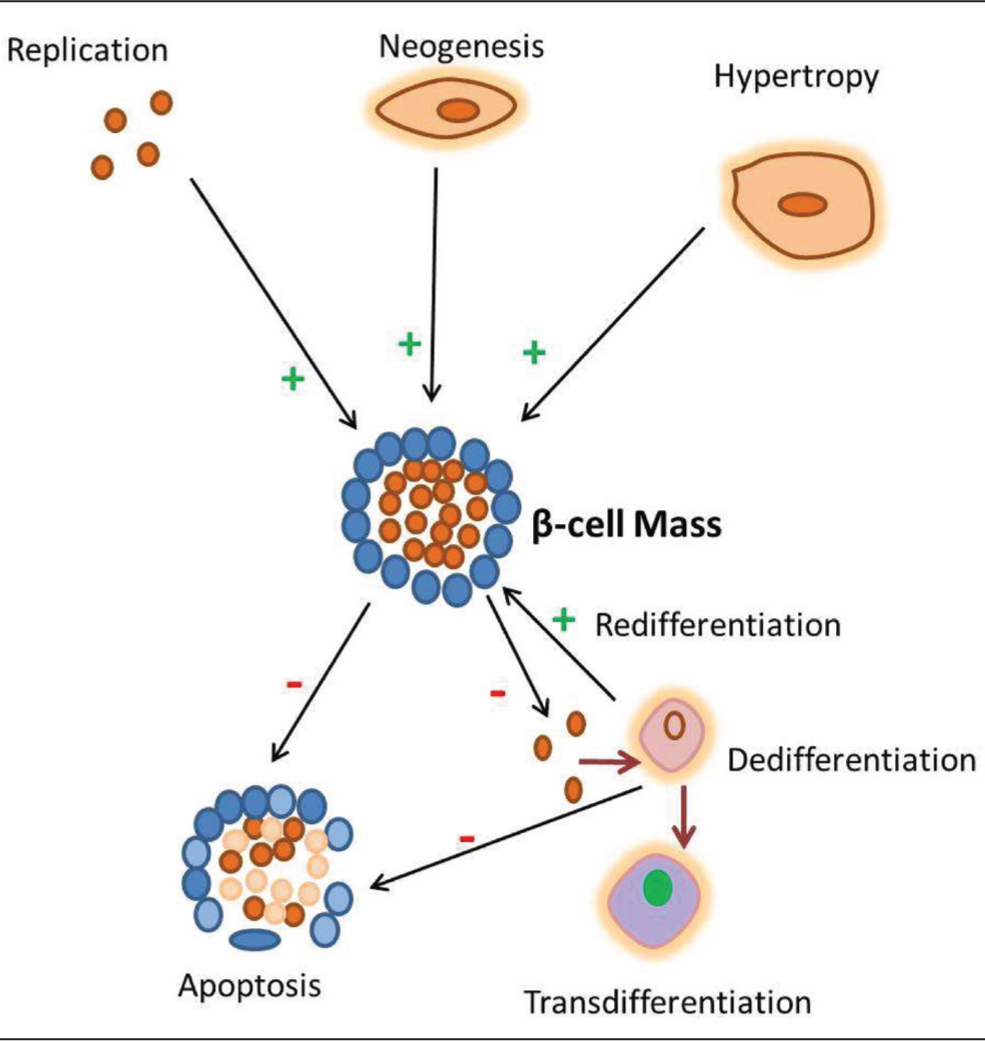
The identity of β-cell altered in two ways: dedifferentiation and trans-differentiation that contribute to the functional β-cell loss in diabetes. The dedifferentiated β-cells do not undergo apoptosis immediately, which only exhibit the loss of cellular identity and function. Targeting the dedifferentiated β-cells and converting them into functional β-cell could be a potent target to achieve remission in diabetes. Furthermore, the dedifferentiated β-cells undergoes trans-differentiation, where the β-cells changed their characteristics and converted into other islet cell types, it worsens hyperglycaemia.[25-27]
Mechanism of Diabetes Remission
Diabetes remission requires a multifaceted approach that includes interventions like weight loss through diet, lifestyle modifications, medication, bariatric surgery, fasting and caloric restriction (Figure 3). Further, remission has been correlated with reduced ectopic fat deposition in the liver and pancreas, accompanied by restored insulin secretory function. These interventions target key mechanisms that facilitate remission and avoid long-term complications.[28-30]
Lifestyle Modifications
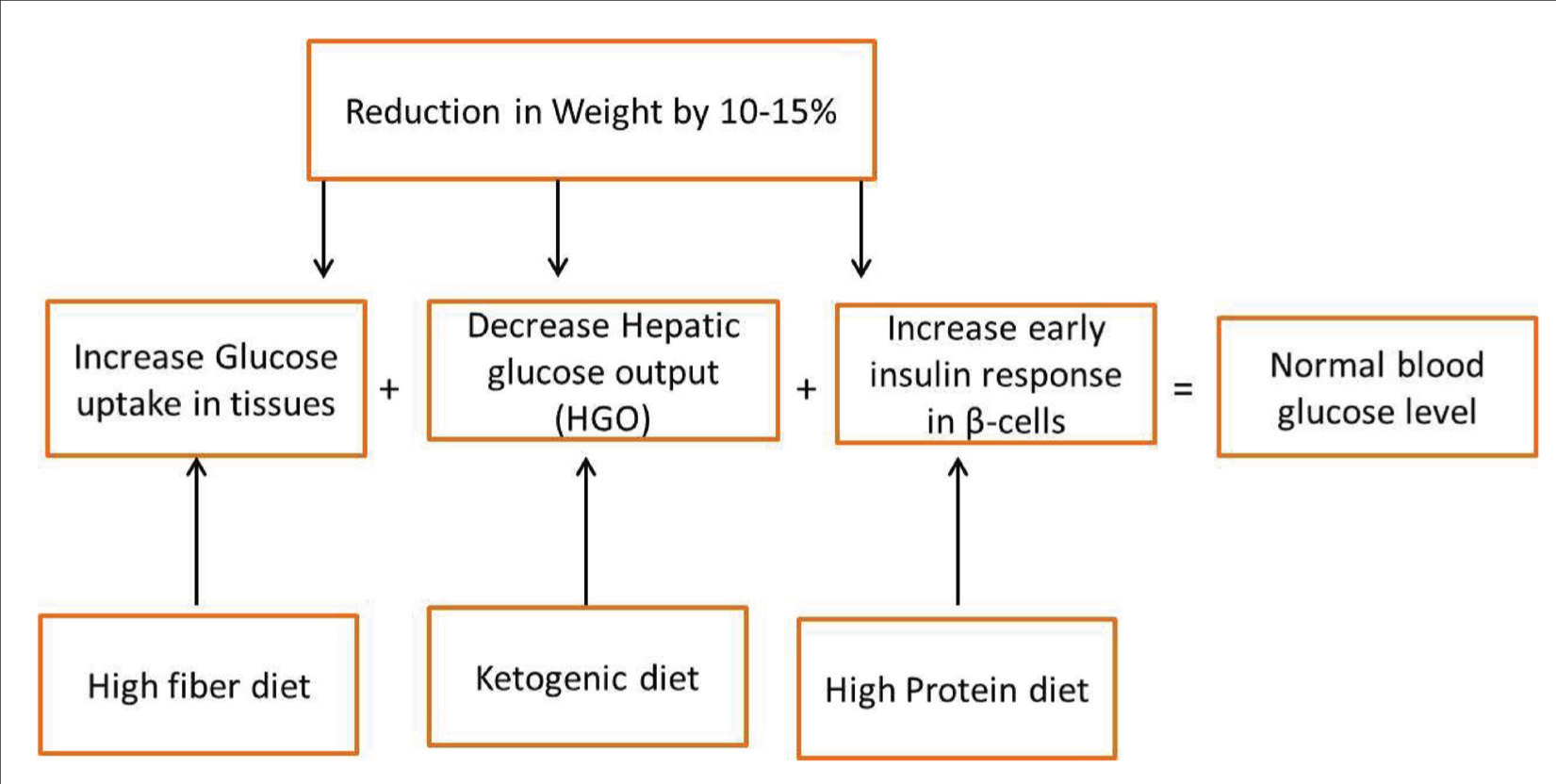
Lifestyle modification plays a vital role in the management and remission of T2D, with dietary changes, weight loss and exercise being key factors.[31,32] Remission of T2D is possible with > 10% weight loss, but sustaining long-term weight loss is very difficult. Hence, it requires modified dietary interventions to maintain weight and achieve remission[33] (Figure 4). Low-calorie diet, especially 800-1000 calories per day, promotes weight loss and improves insulin sensitivity, which are the two essential components for diabetes remission. A low-carbohydrate diet, particularly a person reducing the intake of refined carbohydrates and sugars, helps regulate blood glucose levels by enhancing insulin sensitivity.[34] Further keto diet helps in blood glucose reduction by switching body’s primary energy source from carbohydrate to fats. Saslow et al.[35] reported that 60% of individuals who followed ketogenic diet discontinued their medication and also observed significant reduction in HbA1c. Additionally, the Mediterranean diet rich in healthy fibre, fats and antioxidants reduces inflammation and supports glycaemic control.[36] Meanwhile, plant-based diets that emphasise legumes, vegetables and whole grains lower the risk of major complications and increase insulin sensitivity.[37] However, it is essential to consult a healthcare provider before starting these interventions, especially patients receiving antidiabetic therapy, due to the risk of hypoglycaemia.
Therapeutic strategies and underlying mechanism for achieving Type 2 diabetes
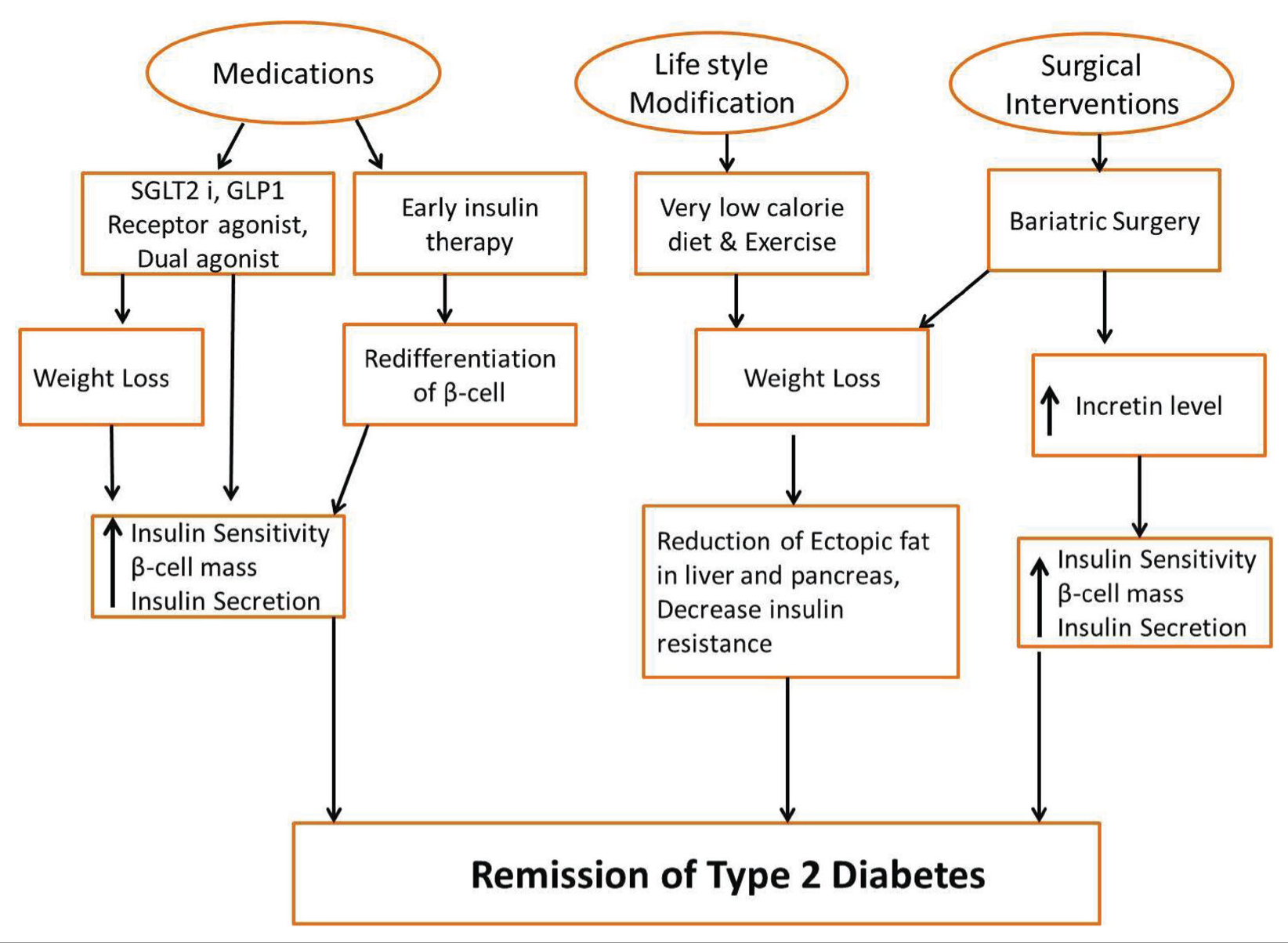
Mechanistic pathways linking weight loss and dietary interventions to remission
In the DiRECT trial, 306 participants with T2D were randomly assigned to either a total diet replacement with calorie restricted diet (800 kcal/day) intervention or standard care. After 12 months, 24% of the intervention group achieved a weight loss of 15 kg or more, compared to none in the control group. Moreover, 46% of participants achieved remission in the intervention group whereas only 4% in the control group. Further, the continued support group had a significantly higher proportion of sustained diabetes remission, with 36% of participants in the intervention group maintaining remission at five years, compared with only 3% in the control group. This study emphasises that a structured weight loss program for long-term supports weight management and could be a potential strategy for achieving glycaemic control and reversing early stage T2D.[38]
Regular physical activity is a pivotal factor in simplifying diabetes remission, as it reduces insulin resistance, improves metabolic homeostasis and contributes to weight management. Regular exercise, including aerobic and strength training activities, makes the body use more glucose effectively, thereby reducing blood glucose levels. During physical activity, skeletal muscle exhibits increased glucose uptake from the bloodstream and diminishes the insulin-mediated glucose uptake. This adaptive response can be particularly beneficial for persons with T2D or prediabetes.[39] Furthermore, substantial weight loss, especially a person with obesity, is one of the effective strategies to achieve diabetes remission. Reduction in 5%-10% of body weight can improve glycaemic control and insulin sensitivity, potentially reversing the disease progression in its early stages. Further, Diabetes Prevention Programs (DPP) combined with exercise, lifestyle counselling and dietary changes can delay or prevent the progression from prediabetes to T2D and may even lead to diabetes remission. The lifestyle intervention targeted to maintain ≥7% weight loss, resulted in 58% reduction in the incidence of diabetes.[40]
Medications
Pharmacological interventions are an important component in inducing and sustaining T2D remission, but their efficacy alone is not sufficient to achieve remission. In some cases, medications may contribute to achieving normal blood glucose levels and eliminate the necessity of medication over time.[41] However, the role of medications in remission is complicated and depends on various factors such as duration of diabetes, type of drugs and individual patient characteristics, including comorbidities, genetic predisposition and metabolic profile. Early insulin therapy has shown to reduce glucotoxicity that contributes to the remission of diabetes.[42] Certain classes of drugs, including metformin, SGLT2i and GLP1-RAs, are reported to support the mechanism that induces diabetes remission.[43]
Early intensive insulin therapy has been demonstrated to achieve remission in newly diagnosed T2D patients with severe hyperglycaemia. Studies have shown that intensive insulin therapy can induce remission lasting over two years; it suggests that prompt initiation of treatment after diagnosis is a critical determinant of remission success. Further, the efficacy of early insulin therapy has been found to conserve β-cell function and reduce glucotoxicity, while patients treated with short-term insulin therapy showed improved β-cell function and long-term β-cell preservation.[44] In multicentric randomised clinical trial, intensive insulin treatment and combined oral antidiabetic drug therapy were compared for their effects on long-term glycaemic control and β-cell function. Both treatments achieved short-term glycaemic control, but the intensive insulin treatment group showed significantly better glycaemic control with lifestyle medication alone and a 52.5% lower risk of failing to maintain drug-free remission than the combined oral antidiabetic drug therapy.[45] Li et al.[46] confirmed that short-term continuous subcutaneous insulin infusion achieved optimal glycaemic control within seven days and the remission rate was 72.6% at 3rd month and 42.3% at 24th month. Patients with sustained remission showed greater recovery of β-cell function and improved insulin secretion.
Metformin is the most commonly used first-line therapy for T2D. It improves insulin sensitivity and reduces hepatic glucose production by suppressing the action of glucagon. Metformin does not typically induce remission, but it can play a role in maintaining normal blood glucose levels during the remission periods after intensive lifestyle modifications or bariatric surgery.[47] Hence, metformin helps in sustaining remission, particularly if combined with weight loss. SGLT-2 inhibitors work through a unique mechanism that is independent of insulin action and secretion (e.g., empagliflozin and canagliflozin). These inhibitors block the SGLT2 protein in the proximal convoluted tubule and reduce the glucose reabsorption in the kidneys. Clinical evidence suggests that it also promote weight loss, lowers blood pressure and improves cardiovascular and renal function. SGLT-2 inhibitors are not directly involved in remission; they improve overall metabolism and maintain remission with other strategies.[48]
GLP-1-RAs mimic the action of the natural hormone GLP-1, which enhances insulin secretion when blood glucose levels are high. Further, it inhibits glucagon secretion and promotes weight loss by increasing satiety and slowing gastric emptying. These characteristics can help to achieve remission in individuals with T2D, especially with lifestyle interventions.[49] Moreover, analogues of GLP-1 also increase glucose stimulated insulin secretion and expression of Islet duodenal homeobox 1 (IDX-1) in β-cells that promotes β-cell redifferentiation.[50] A dual glucose-dependent insulinotropic polypeptide (GIP)/ glucagon-like peptide-1 receptor co-agonist (GLP-1 RA) regulates food intake through stimulating neurons in the brain satiety centre. In addition to regulating glucose homeostasis, GIP and GLP-1 also stimulate insulin release from pancreatic β-cells. However, their effects on glucagon production in pancreatic α-cells diverge: GIP enhances glucagon secretion during hypoglycaemia, whereas GLP-1 inhibits glucagon release during hyperglycaemia. Both hormones play key roles in maintaining glucose homeostasis and regulating insulin secretion, and their combined activation may have a significant impact on reversing or ‘remitting’ T2D, especially when used early in the disease process. A multicentric trials result showed that tirzepatide reduced HbA1c and body weight compared to other drugs and resulted in a remission rate of 66%-81% over 52 weeks depending on the dose.[51]
Surgical Intervention
Bariatric surgery, particularly weight loss methods such as Roux-en-Y gastric bypass, adjustable gastric banding and sleeve gastrectomy are powerful procedures to achieve long-term remission in T2D with severe obesity. The mechanism behind these interventions includes a significant reduction in body weight, improved insulin sensitivity, altered gut hormone dynamics and improved insulin sensitivity. Bariatric surgery can reverse the metabolic changes associated with T2D, in patients diagnosed with disease within the initial six years of onset. Bariatric surgery achieves optimal therapeutic outcomes for individuals with class III obesity (body mass index (BMI) ≥ 35), where the rate of diabetes remission is highest.[52]
STAMPEDE (Surgical Treatment and Medications Potentially Eradicate Diabetes Efficiently) is a 12-month randomised clinical trial compared the three interventions for T2D patients with BMI 30-40: Roux-en-Y gastric bypass, laparoscopic adjustable gastric banding and lifestyle weight loss intervention. Roux-en-Y gastric bypass showed significantly higher weight loss (27.0%) compared to 17.3% with laparoscopic adjustable gastric banding and 10.2% with lifestyle weight loss intervention. Diabetes remission rates were 50% partial, 17% complete in Roux-en-Y gastric bypass and 23% complete for laparoscopic adjustable gastric banding. No remission occurred in lifestyle weight loss intervention.[53]
Cummings et al.[54] reported that Roux-en-Y gastric bypass in patients with mild-to-moderate obesity showed significant decrease in HbA1c from 7.7% to 6.4%, weight loss (25.8%) and remission rate was 60% after one year. A randomised controlled trial with newly diagnosed T2D obese patients found that laparoscopic adjustable gastric banding resulted in significantly better outcomes compared to conventional therapy. At two years, 73% of that laparoscopic adjustable gastric banding patients achieved diabetes remission (fasting glucose <126 mg/dL, HbA1c <6.2%) than 13% in the conventional therapy group. Further, vertical sleeve gastrectomy induces fast β-cell redifferentiation and improves their function within two weeks of surgery.[55,56]
Factors That Predict Remission of Diabetes
Several clinical factors have been identified as predictors for T2D remission. These include age, duration of diabetes, weight loss, dietary habits, physical activity, HbA1c, C-peptide and HOMA-IR which determine the efficacy of interventions and treatment responsiveness (Table 2). Evidence suggests that higher C-peptide, lower HbA1c and homeostasis model assessment of insulin resistance (HOMA-IR) are independent predictors for complete remission of diabetes.[57] In addition to these factors, other features such as ethnicity, genetic predisposition and the presence of comorbid conditions (e.g., dyslipidaemia, hypertension) may influence remission outcomes.[58,59]
Predictors of Type 2 diabetes remission

Conclusion
In conclusion, understanding diabetes remission offers a significant solution in the management of T2D, from a chronic disease to one of potential remission. Recent studies demonstrate that remission is achievable in many cases through lifestyle intervention, weight loss and pharmacological strategies. However, the process of remission sustaining is multifaceted, and requires a personalised approach to address the underlying cause of the disease. Emerging research into genetic, environmental and psychological factors that influence the remission of diabetes, along with the innovation of new therapies, will pave the way for a future to reverse rather than simply manage. The concept of diabetes remission gives hope to millions of people worldwide and highlights the need for ongoing research and innovation in diabetes care. A deeper understanding of diabetes remission and its mechanism can revolutionise diabetes treatment modalities, thereby improving the patient’s health and quality of life. Early interventions are crucial for maximising the potential of diabetes remission and time is a key for successful remission, particularly in T2D.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not applicable.
Credit author statement
KV: Conception, Editing and Final approval GP: Literature Search, Manuscript Drafting and Editing, BR: Literature Search and Editing.
Data availability
Data is available within the manuscript.
Use of artificial intelligence
Not used.
