Abstract
Background and Aims:
Auditory Neuropathy Spectrum Disorder (ANSD) is a condition that affects the auditory pathway, specifically the transmission of sound signals from the ear to the brain. In ANSD, the outer hair cells (OHCs) in the cochlea, which are responsible for amplifying sound, typically function normally. However, there is an issue in the transmission of sound signals from the cochlea to the brain. ANSD is often due to problems in the inner hair cells (IHCs), the auditory nerve fibres, or the synaptic connections between them. Prematurity, ototoxic medication therapy, hyperbilirubinemia and positive family history are risk factors for ANSD.
Methods:
The search for published articles on ANSD was done with databases like PubMed, Scopus, Medline and Google Scholar. This article discusses on prevalence, etiopathogenesis, clinical manifestations, diagnosis and present treatment of ANSD.
Results:
ANSD encompasses a variety of hearing disorders that are typified by poor speech perception even though pure tone detection thresholds are largely intact. The afflicted patients exhibit speech-discriminating issues that are disproportionate to their pure tone detection thresholds, especially when there is background noise. Acoustic reflexes, otoacoustic emissions (OAEs), cochlear microphonics (CMs) and auditory brainstem evoked responses (ABERs) are missing or significantly reduced in patients with ANSD. While cochlear implants are the primary option for people with substantial hearing loss, especially in cases with IHCs presynaptic diseases or when hearing aids have failed, hearing aids are beneficial for ANSD patients with mild to moderate hearing loss.
Conclusion:
As OAEs and/or CMs of neonates with ANSD are normal, auditory evoked responses should be added to screening tests for high-risk neonates in order to prevent false-negative results.
Keywords
Introduction
The word auditory neuropathy was coined by Arnold Starr, MD, and colleagues in 1996 as hearing impairment by a defect in the auditory nerve.[1] This definition was expanded later on to include loss of inner hair cells (IHCs) of the cochlea or damage between the auditory nerve and brain.[2] In 2008, an expert panel at the International Newborn Hearing Screening Conference in Italy suggested adopting the term ‘auditory neuropathy spectrum disorder’ (ANSD) because of its heterogeneous and multifactorial nature.[3] This is why auditory neuropathy has been subsequently renamed as ‘auditory neuropathy spectrum disorder’. ANSD is a condition in which the transmission of auditory signals from the cochlea to the auditory nerve and brainstem is impaired. ANSD is a disorder of hearing impairment where cochlear outer hair cells (OHC) are spared, but afferent neural transmission is affected.[4] According to the site of the lesion, a patient can have a clinical presentation ranging from mild hearing loss to functionally deaf. The criteria to diagnose ANSD for any person of any age who fulfils most or all of the following criteria: (a) recorded otoacoustic emissions (OAEs) or cochlear microphonic (CM); (b) absent or atypical auditory brainstem response (ABR); (c) understanding of speech ‘worse’ than would anticipate from the behavioural pure tone audiogram; and (d) Hearing loss of variable severity and configuration.[5] Patients of ANSD present with hearing deficits with preserved OAEs and absent or severely abnormal ABRs. Individuals diagnosed with ANSD frequently report difficulty understanding speech, especially in noisy environments. Two auditory function investigations such as OAE and brainstem potentials are able to detect the function of the VIII nerve and OHCs.[6] The diagnosis of ANSD is made on the evidence of altered neural processing, such as abnormal ABRs with absent or diminished wave V, even though OHC responses, such as OAEs and/or CM, remain intact.[7]
Methods of Literature Search
Several techniques were employed to locate research articles on ANSD. A preliminary online search was performed across several databases, including Scopus, PubMed, Medline and Google Scholar. The search strategy was developed based on PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines to ensure a comprehensive and systematic review of the relevant literature. Besides manually obtaining research publications through citations, our search method also included reviewing the abstracts of published studies. The eligibility criteria included randomised controlled trials, observational studies, comparative studies, case series and case reports that offered a comprehensive assessment of ANSD. A total of 56 papers were included: 14 case reports, 17 case series and 25 research articles [Figure 1]. This article covers the history, etiopathology, clinical presentations, diagnosis and current treatment of ANSD. It provides a foundation for future prospective trials and has the potential to drive early diagnosis and treatment of ANSD, an area with currently limited studies.
Method of literature search

Prevalence
ANSD is not an uncommon cause of hearing disorders. Approximately 1 in 7,000 neonates evaluated by newborn screening is detected as having abnormal auditory nerve function.[8] Although studies have shown prevalences ranging from less than 1%-10% of patients with hearing impairments, the exact prevalence of ANSD is still unknown.[9] The wide range of clinical profile heterogeneity among ANSD patients is reflected in the variety of prevalence. The prevalence of sensorineural hearing loss in the neonatal intensive care unit (NICU) discharged population is approximately 1/150, as opposed to 1/1000 for normal-term neonates.[10] About 10%–15% of newborns who are discharged from the NICU have a higher prevalence of SNHL, especially ANSD.[11] Up to 30% of all SNHL in children discharged from the NICU are caused by ANSD.[12]
Etiopathology
A wide spectrum of aetiologies are documented such as prenatal causes (genetic, cochlear nerve malformation, foetal infections), and postnatal factors (genetic with delayed manifestations, prematurity, perinatal disorders).[13] Prenatal infections like measles, mumps, or cytomegalovirus can cause ANSD.[14] Perinatal disorders like ototoxic drugs, hypoxia, icterus and kernicterus, septicaemia and meningitis can manifest ANSD.[15] ANSD can result from a variety of factors, including both syndromic and non-syndromic genetic disorders, environmental influences and structural abnormalities. These factors may involve hypoplasia or aplasia of the cochlear nerve, and lesions at different locations.[15] The exact location of the lesion in ANSD remains uncertain. However, potential sites for the lesion include IHCs, synapses between inner and/or OHCs, bipolar neurons of the auditory nerve, spiral ganglion cells, or the brainstem.[16] The IHCs function as mechanosensory cells, converting mechanical deflections of their hair bundles into molecular signals that initiate the electrical firing of neuronal fibres. When the hair bundle of IHCs is deflected by the vibration of the tectorial membrane, it causes a cation influx into the IHCs. The resulting depolarising potential leads to a calcium influx through one or two voltage-gated calcium channels. At the presynaptic area of ribbon synapse, the activation of these calcium channels enhances the fusion of synaptic vesicles, triggering release of glutamate into synaptic cleft. This presynaptic signalling occurs in a graded fashion to maintain the rate coding of action potentials in the spiral ganglion neuron fibres. The increased release of neurotransmitters at the synapse generates an excitatory postsynaptic potential, which activates calcium-sensitive receptors, specifically AMPA receptors. This activation leads to the generation of neural spikes in the spiral ganglion neurons (SGNs), which then travel along axon to the SGN cell body. ANSD may be seen in association with other syndromes or neurological lesions. Harding disease is a condition similar to multiple sclerosis, caused by a mutation in the 11778mtFNA gene. It is marked by Leber hereditary optic neuropathy and symptoms associated with ANSD.[17] Gaucher disease, a neuropathic condition, is marked by neurological abnormalities caused by the toxicity of glucocerebroside. Audiologically, this disease is associated with a progressive decline in ABRs, while OAEs remain preserved.[18] Waardenburg syndrome and Friedreich ataxia, a genetic neurological illness, are also linked to ANSD.[19] Consanguineous marriage is practised among subpopulations of Israel.[20] ANSD is one of several congenital conditions linked to consanguinity.[20]
Risk Factors for ANSD
The risk factors of ANSD are multifactorial. Various factors linked to ANSD include hyperbilirubinemia, ototoxicity, premature birth and a positive family history.[6] There are several risk factors for causing ANSD such as hypoxia in newborn babies during or prior to birth, jaundice, premature birth, low birth weight and dietary deficiency of thiamine. Apgar scores, respiratory distress, cytomegalovirus infections, meningitis, sepsis, asphyxia and family history of hearing impairment are important factors for causing ANSD.[21] Neonates with hyperbilirubinemia who develop kernicterus have been led to irreversible damage to the brainstem and cranial nerve nuclei. However, hearing impairment which is associated with hyperbilirubinemia is considered to be signal conduction defects along the auditory nerve, as substantiated by ABR findings in these cases.[22] A study found that hearing-impaired infants with hyperbilirubinemia (serum level >20 mg/dl) who received a single blood transfusion as treatment experienced a reversal of hearing loss at the three-month follow-up.[23]
Clinical Presentations
The understanding of speech among patients of ANSD is worse than predicted from the behavioural pure tone audiogram. The hearing loss of patients with ANSD is variable in severity. ANSD imitates the typical signs of retro-cochlear hearing loss, particularly the damage to type 1 afferent neurons. The site of this damage could be as peripheral as the synapse between the IHCs and the spiral ganglion cells, which, although not retro-cochlear, is intracochlear. The individuals with ANSD present with speech discrimination problems, especially in noisy environments, that are disproportionately greater than what would be expected based on their pure tone detection thresholds.[24] The patient often struggles significantly with understanding spoken words but is able to comprehend speech through visual cues from lip and facial movements. Individuals with ANSD frequently have trouble communicating over the phone. They may also be disturbed by loud sounds. Hearing fatigue can occur at higher sound intensities and/or with prolonged tonal stimuli, potentially hindering threshold assessment.[25] Patients of ANSD have poor sound localisation.[26] In older children, hearing levels in ANSD have been observed to vary with fluctuations in core body temperature.[27] This fluctuation of core body temperature affecting hearing level may be due to conduction blockage in the auditory nerve as in demyelinating lesions.
Diagnosis
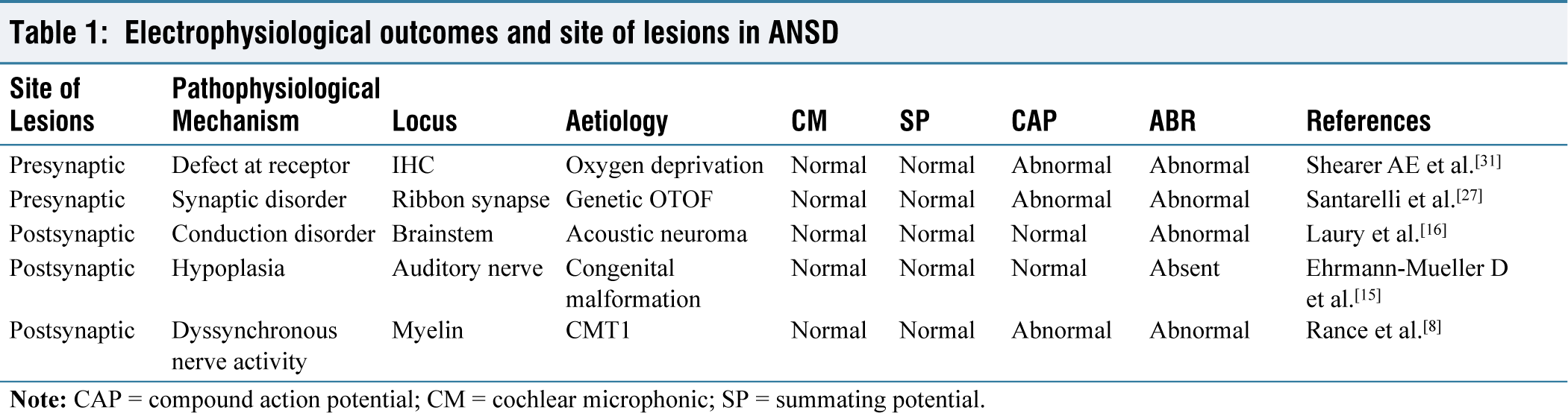
ANSD is a condition in which the transmission of auditory signals from the cochlea to the auditory nerve and brainstem is disrupted. Both OAEs and CM serve as physiological indicators of OHC activity. Therefore, the presence of CM is essential for diagnosing ANSD. Hearing impairment in ANSD can vary, ranging from mild to profound.[7] ANSD is characterised by the normal function of OHCs in the cochlea with disruption of auditory nerve function.[1] Diagnosis of ANSD is based on objective neurophysiological measurement of cochlear hair cells and auditory nerve functions; imaging of auditory nerve/brain stem; and behavioural audiological tests. The defining characteristic of ANSD is the presence of normal OHC activity alongside abnormal afferent and efferent auditory neural functions, potentially affecting the eighth cranial nerve and brainstem. The activation of OHCs is assessed through the acoustic energy emitted from the inner ear (OAEs) and the electrical response from the CM. The conduction and threshold of the auditory nerve are evaluated using ABRs. OHCs primarily contribute to the generation of the CM, an early evoked response with a short latency that occurs before wave I of the ABRs. The CM is generated by the mechano-sensitive transduction channels in the stereocilia of both IHCs and OHCs, with OHCs contributing more due to their larger quantity. This compound potential is produced by the opening and closing of these transduction channels in the hair bundle, driven by the movement of the basilar membrane.[28] Since Evoked Otoacoustic Emissions (EOAEs) and CMs rely on the integrity of cochlear OHCs and are perineural events, they remain present and normal in individuals with ANSD. In these patients, pure tone audiometry may show hearing loss that can range from normal to profound, without following a consistent pattern. The hearing impairment profiles can be quite variable, showing anything from flat thresholds to more pronounced loss at either low or high frequencies. ANSD is bilateral in approximately 75% of cases and unilateral in about 25% of cases.[29] Speech perception tests often show significantly poor speech discrimination abilities in individuals with ANSD. The impairment in speech perception is typically much greater than what would be expected based on tonal hearing thresholds. Additionally, background noise exacerbates the difficulty, further reducing the ability to discriminate speech.[30] Stapedial reflexes and EOAE suppression, whether elicited sounds given to the ipsilateral or contralateral ear, are absent in cases of ANSD.[31] Individuals with the involvement of auditory nerve show low outcomes in cochlear implants as the neural transmission of an electrical signal from a cochlear implant is affected.[31] In electrocochleography (ECochG), the origin of SP is more debatable and unclear in ANSD, but it is more acceptable than summating potential (SP) which usually reflects the activity of IHCs.[32] Another component of the Electrocochleography (ECochG) response that reflects neural activity is the ANSD. This response is generated by auditory nerve’s reaction to low-frequency stimuli.[33] This is often challenging to differentiate from CM. Abnormal or absent cortical auditory evoked potentials have been seen in approximately one-third of paediatric patients with ANSD.[34] Patients with ANSD have audiological findings that include preservation of OAEs and increasing deterioration of ABERs. Electrophysiological outcomes and site of lesions in ANSD are given in Table 1.
Electrophysiological outcomes and site of lesions in ANSD
Psychoacoustic Test
The psychophysical assessment of ANSD should involve modified behavioural pure tone audiometry and speech discrimination testing both in quiet and noisy environments. Tone decay and frequency discrimination tests are also essential for a comprehensive audiological evaluation. Additionally, the assessment should encompass language abilities, overall motor and cognitive development among children, as well as, attention levels and psychological profiles in patients with ANSD.
Treatment
Children with AN or ANSD are managed by a team of specialists such as audiologists, otolaryngologists, speech-language therapists and education specialists. Rehabilitation of ANSD consists of hearing aid and speech auditory training. The treatment of this disorder depends on the child’s age, the lesion’s site at the auditory pathway, and how much hearing loss. Most of the children diagnosed with AN or ANSD benefit from assistive listening devices. These are most effective when used alongside continuous speech-language therapy to enhance speech and hearing abilities. The frequency modulation system minimises background noise and amplifies the speaker’s voice. For example, the teacher uses a microphone and transmitter that sends an electrical signal to a wireless receiver worn by the child, either in their ear or built into a hearing aid. The frequency modulation system is portable and can be useful in classrooms and other noisy places. A hearing aid is helpful to amplify sounds coming into the ear. Hearing aids are helpful among some children with AN or ANSD, particularly when used with a frequency modulation system. Only hearing aid use is not helpful to children with ANSD or AN as these only make the disorganised sound louder. Patients who do not demonstrate adequate progress are suggested for a cochlear implant.[35] There is ongoing debate about whether to offer hearing devices like hearing aids or cochlear implants to children with ANSD, as well as, whether to focus on aural-oral or visual-manual approaches to rehabilitation.[36,37] Currently cochlear implantation is the preferred treatment for patient option for ANSD. Multi-channel cochlear implantation is a surgical procedure in which electrodes are inserted into the scala tympani of the cochlea to provide stimulation.[38] Cochlear implants deliver electrical stimulation that exceeds normal physiological levels to the auditory nerve, which may help improve the synchronisation of neural activity.[39] The electrical current flowing between the electrodes stimulates the auditory nerve at various locations, including myelinated dendrites, auditory ganglion cells and auditory axons. Many patients with ANSD experience improvements, such as the ability to use the telephone effectively, and children often show typical rates of speech, language development and academic performance.[40] After implantation, there is enhanced audiological performance, along with strong implant-evoked brainstem responses and Neural Response Telemetry.[41]
Conclusion
The defining characteristic of ANSD is the presence of normal function of OHC, alongside defective auditory nerve and brainstem activity. ANSD is a significant subtype of sensorineural hearing loss, marked by the absence or dysfunction of ABERs while OAEs remain intact. This can result in false-negative outcomes during neonatal hearing screenings. To prevent false-negative results, screening tests for high-risk newborns should be supplemented with auditory evoked responses. Since ANSD is a retro-cochlear disorder with normal OHC function, hearing aids are typically not effective. While the pathology primarily affects the cochlear nerve, cochlear implantation is considered a viable option when conventional rehabilitation methods fail.
Footnotes
Acknowledgements
The authors are thankful to Swagatika Swain for the language corrections of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Informed consent
Consent was not applicable, as this is a review article compiled from various research articles and guidelines and not from patients directly.
Credit author statement
SKS: Conception, design, data analysis, data interpretation, drafting and final approval of the manuscript.
Data availability
Data openly available in a public repository that issues datasets with DOIs.
Use of artificial intelligence
No artificial intelligence was used by authors for the draft compilation. The authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
