Abstract
Background and Aims:
Spondylolysis refers to a stress fracture of the pars interarticularis. We investigated if there is a correlation between a novel measurement, the sagittal pars inclination angle and the incidence of spondylolysis (pars defect) in adolescent and young adult patients.
Methods:
A retrospective cohort study was conducted at the Royal Orthopaedic Hospital, UK (a tertiary referral orthopaedic centre) over two years identifying 37 Adult patients under the age of 30 years who had a computed tomography (CT) scan of the lumbar spine. In total 255 pars interarticularis, from levels L2 to L5, were analysed in 37 patients (19 male, 18 female). The pars interarticularis were divided into two groups: (1) pars intact (N = 217) and (2) pars defect (N = 38). Statistical analysis of each group and vertebral level was performed including load measurements.
Results:
In the pars intact group (N = 217), the sagittal mean ± standard deviation (SD) pars inclination angle for the pars intact group was 17.91 ± 5.62°. In the pars defect group (N = 38), the mean ± SD sagittal pars inclination angle was 35.95 ± 6.42°. A two-tailed P value was less than .0001. There was an increase in the normal sagittal pars angle from L2 to L5.
Conclusion:
In this study, an increased sagittal pars inclination angle demonstrated a significant association with spondylolysis. Its implications in the management of athletic individuals, surgical planning and indeed a true causative relationship are yet to be evaluated in future studies.
Introduction
Spondylolysis refers to a stress fracture of the pars interarticularis. A precursor to this is an unusually high-stress reaction in the neural arch of a vertebral body. The reaction and the fracture can be either unilateral or bilateral and at one or more levels. It is observed radiographically in 6% of the general adult population with a male-to-female ratio of 2:1.[1] Spondylolysis occurs most commonly at L5 (90%) followed by L4 (10%) and may co-exist with other spinal anomalies including spina bifida, transitional vertebrae and scoliosis.[2] It is not always symptomatic.[3]
Spondylolysis may lead to a forward slippage of the upper vertebra on the lower level. This anterior translation is termed spondylolisthesis. It is categorised by the Wiltse classification.[4] One of the types that is most common is isthmic (type II) spondylolysis, an acquired defect in the pars interarticularis. Although for many readers this lesion may come as an incidental observation, it is a common finding attributed to lower back pain in active adolescents and children. The incidence of spondylolysis is reported in up to 47% of symptomatic adolescent athletes in comparison to only 5% of symptomatic adults in the same case series.[5,6]
Historically, spondylolysis was diagnosed as a discontinuity in the pars interarticularis visible on lateral and oblique (‘Scottie dog view’) radiographs of the lumbar spine. With the contemporary and widespread use of magnetic resonance imaging (MRI) in the assessment of lower back pain, especially given the radiation dose limitation for adolescent patients, spondylolysis has since been considered on a spectrum from stress reaction (appearing as focal oedema) to fracture (appearing as a sclerotic line) and has been classified as such by Hollenberg et al.[7,8] There are a large number of radiological parameters used to assess the severity and prognosis of spondylolisthesis. These include the (i) lumbar index which measures the percentage slip of one vertebra on the subjacent vertebra, and (ii) the slip angle which is the angle between the end plates of the inferior and superior vertebra. Other factors such as a trapezoid-shaped L5 vertebral body and a cranially domed sacrum have also been implicated albeit disputed.[9]
Studies have suggested that an increased lumbar lordosis and sacral tilt angle are individual risk factors in the development of spondylolysis.[10,11] To our knowledge, the sagittal pars inclination angle represents a novel measurement which has not yet been investigated. This study aims to investigate if there is a correlation between the sagittal pars inclination angle (the pars orientation to the adjacent vertebral body) with the incidence of spondylolysis (pars defect) in adolescent and young adult patients.
Patients and Methods
Study Design
A retrospective cohort study was conducted at the Royal Orthopaedic Hospital, UK (a tertiary referral orthopaedic centre) over two years. Following local hospital committee ethical approval, we performed a retrospective evaluation of the Radiology Information System (RIS), Picture Archiving and Communication System (PACS), Clinical Record Interactive Search System (CRIS) computer system database to identify 37 Adult patients under the age of 30 years that had a computed tomography (CT) scan that included the lumbar spine.
Inclusion and Exclusion Criteria
Patients under the age of 30 years who had a CT of the lumbar spine for back pain with suspected pars defect were included. Patients with previous surgery, infection and tumour were excluded. In total 255 pars interarticularis were analysed in 37 patients.
Image Analysis
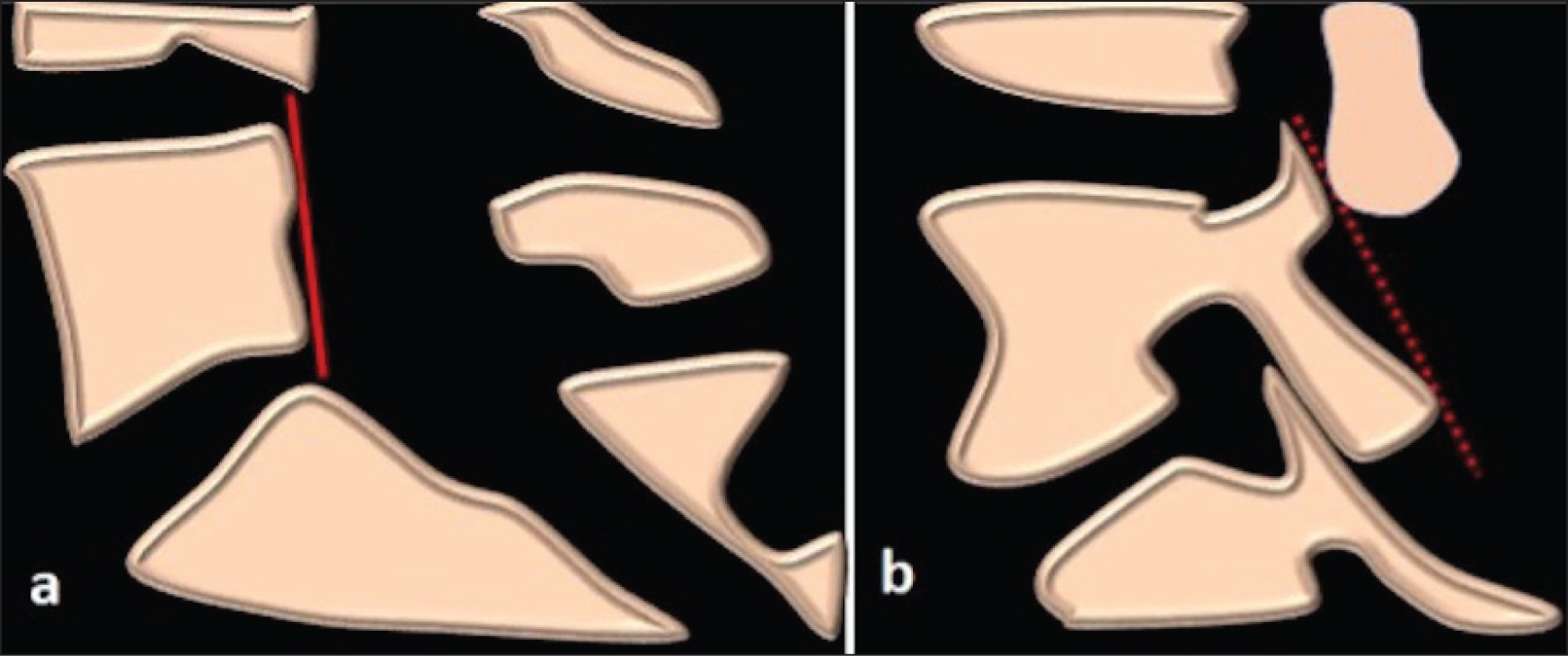
CT was analysed by two fellowship-trained, experienced musculoskeletal radiologists with over 15 years of experience with one reader repeating the measurements after one week. Spondylolysis was identified as a cortical breach in the pars interarticularis on sagittal reconstructed images. This was either documented as unilateral or bilateral. The sagittal pars inclination angle was calculated at all levels imaged on the CT by drawing two reproducible lines. The first was a vertical line along the posterior wall of the vertebral body. The second line was tangent to the posterior cortex of the pars interarticularis for a particular side. The sagittal pars inclination angle was calculated as the angle between these two lines [Figure 1].
Schematic demonstrating lines used to measure the sagittal pars inclination angle. (a) Vertical line along the posterior wall of the vertebral body. (b) The line tangent to the posterior cortex of the pars interarticularis for a particular side. The sagittal pars inclination angle was calculated as the angle between these two lines
Data Collection
Patients were divided into two groups: (a) pars intact and (b) pars defect. A Microsoft Excel data sheet was used for data collection. For each pars interarticularis included in the study, the sagittal pars inclination angle, vertebral level and patient age were recorded.
Data Analysis and Statistical Methods
A Microsoft Excel data sheet and SPSS 24.0 software (SPSS Inc. Chicago, Illinois, USA) were used for data analysis. Mean and standard deviation (SD) were used to summarise the data for continuous variables and frequency/percentage for categorical variables. The 95% confidence interval (CI) was calculated in each group. Statistical analysis was performed using a student t-test. A two-tailed P value was calculated and a P value of less than .5 was considered to be significant. The interclass correlation coefficient was calculated.
Results
A total of 255 pars interarticularis was examined in 37 patients (male = 19, female = 18) with a mean age of 17.9 (range = 10–29). A total of 217 intact pars were observed in 9 males and 9 females with a mean age of 17.5 years (range = 10–28). A total of 38 pars defects were identified in 10 males and 9 females with a mean age of 18.3 years (range = 12–29). Of the pars defect group, 17 patients demonstrated bilateral L5 pars defects, 2 patients demonstrated unilateral L5 pars defects and 1 patient demonstrated bilateral L4 pars defects. No pars defects were identified at L2 and L3 vertebral levels.
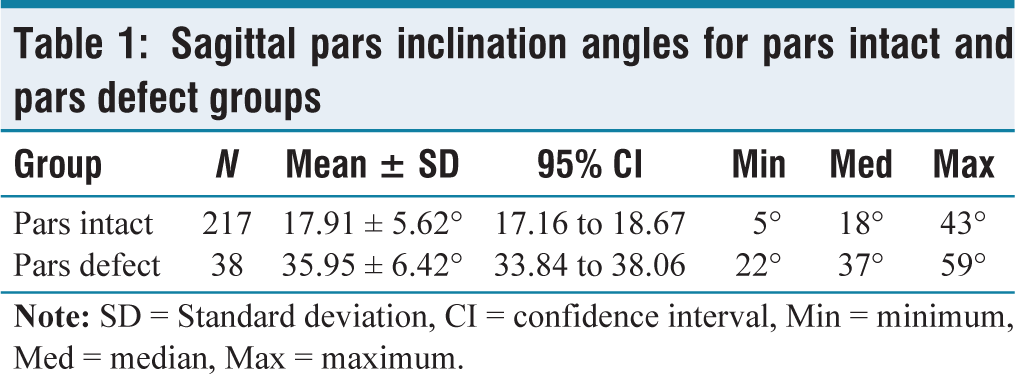
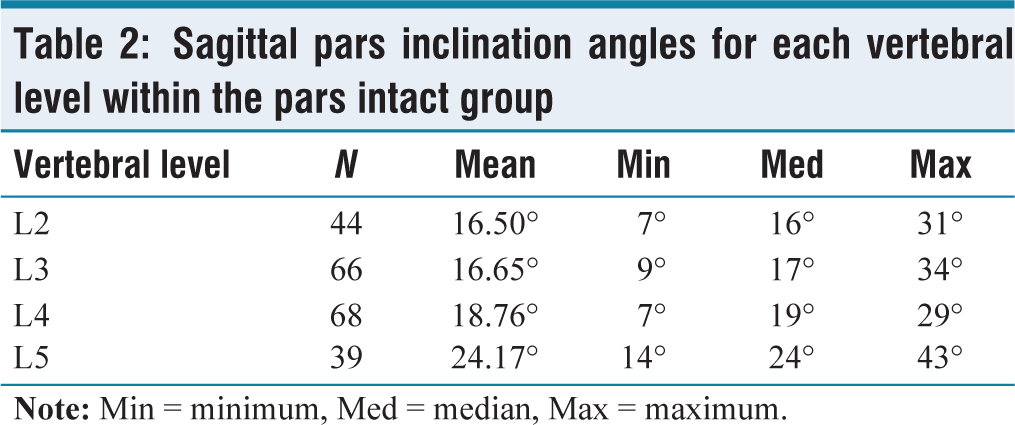
In the pars intact group (N = 217), the sagittal mean ± SD pars inclination angle for the pars intact group was 17.91 ± 5.62°, 95% CI [17.16, 18.67] [Figure 2]. The minimum value was 5°, median 18° and maximum 43°. In the pars defect group (N = 38), the mean ± SD sagittal pars inclination angle was 35.95 ± 6.42°, 95% CI [33.84, 38.06] [Figure 3]. The minimum value was 22°, median 37° and maximum 59°. A mean difference of 18.03°, 95% CI [16.04, 20.03] was identified between the pars intact and pars defect groups (standard error of difference = 1.011). The interclass correlation coefficient was 0.8. A two-tailed P value was less than .0001 and, by conventional criteria, deemed to be extremely statistically significant. The results demonstrated a significant correlation between the size of the sagittal pars inclination angle and the presence of spondylolysis [Table 1]. The mean angle of the pars interarticularis at L2 (N = 44) was 16.50° (range = 7–31), at L3 (N = 66) was 16.65° (range = 9–34), L4 (N = 68) was 18.76° (range = 7–29) and L5 (N = 39) was 24.17° (range = 14–43) [Table 2].
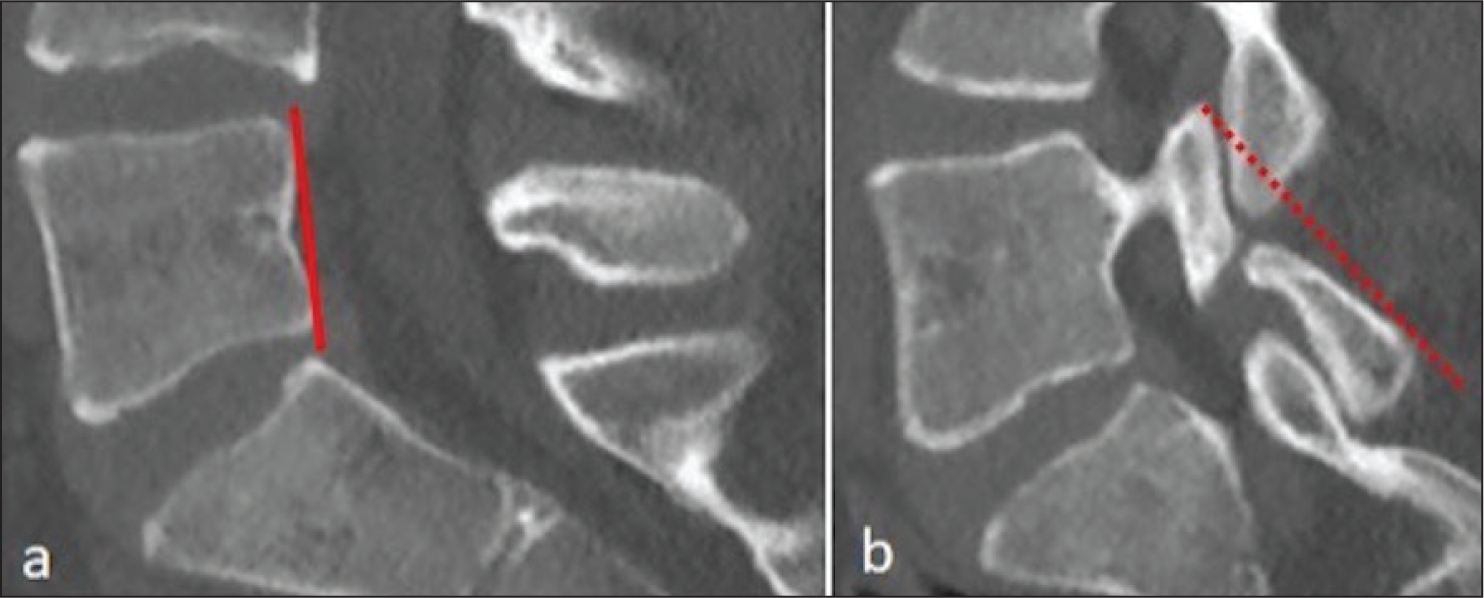
Sagittal pars inclination angle at the level of L3 in patients within the pars intact group. (a) Vertical line along the posterior wall of the vertebral body. (b) The line tangent to the posterior cortex of the pars interarticularis for a particular side. The sagittal pars inclination angle was calculated as the angle between these two lines. The angle is 22 degrees
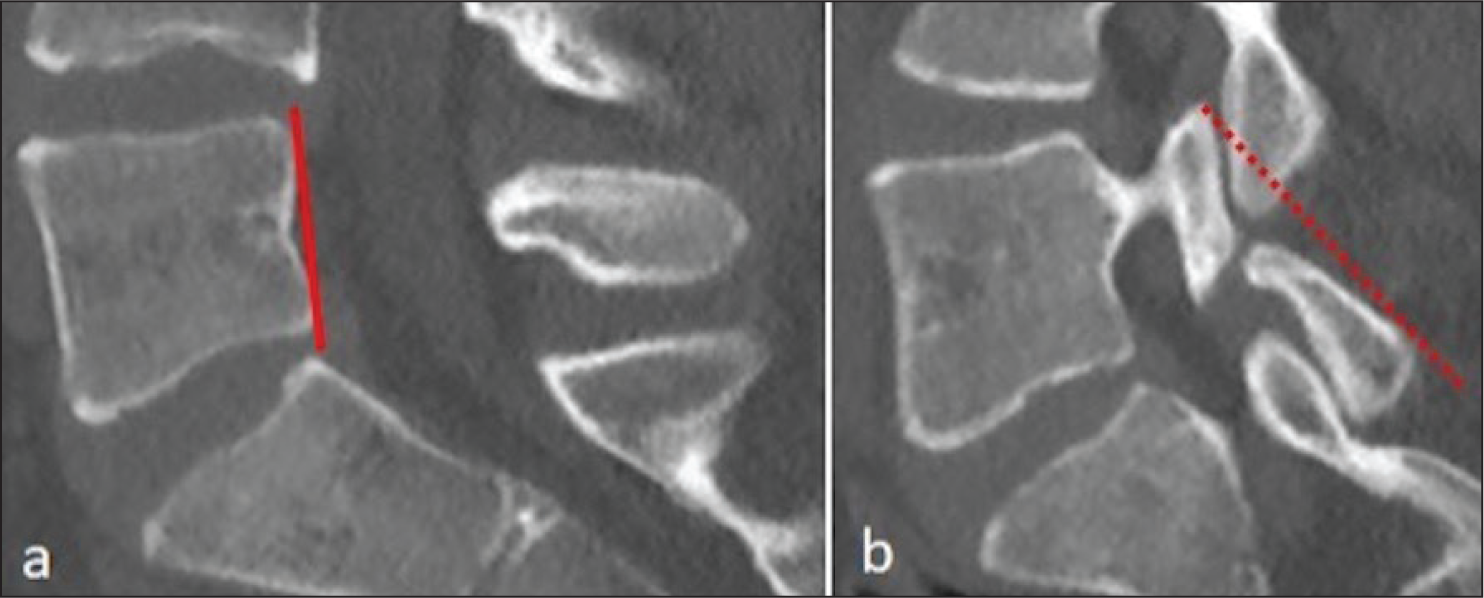
Sagittal pars inclination angle at the level of L5 in patient within the pars defect group. (a) Vertical line along the posterior wall of the vertebral body. (b) The line tangent to the posterior cortex of the pars interarticularis for a particular side. The sagittal pars inclination angle was calculated as the angle between these two lines. The angle is 43 degrees
Sagittal pars inclination angles for pars intact and pars defect groups
Sagittal pars inclination angles for each vertebral level within the pars intact group
The relative force across the pars interarticularis was calculated using a mathematical formula: Force (Newton) = mg cos theta (mg being the weight, theta being the angle). Assuming weight is 1 kilogram, the force across L2 was 0.13N, L3 was 0.10.5N, L4 was 0.11N and L5 was −4.9N. The force across L5 pars was negative implying that the stress is resulting in relatively more strain and deformity of the pars interarticularis at this level.
Discussion
Our study investigated the sagittal pars inclination angle in 37 individuals, under the age of 30, in whom 255 pars interarticularis were examined. All cases of spondylolysis occurred at L5 apart from in one patient where it occurred at L4. The mean sagittal pars inclination angle in the pars intact group was 17.91° compared with the pars defect group which was 35.95°. The difference of 18.03° was deemed statistically significant according to a two-tailed P value and student t-test statistical analysis. We also found that there was a variation in the sagittal pars inclination angle at the different vertebral body levels summarised in [Table 2]. There was no apparent age or gender predilection, although the patient population was small. The study did not select for an individual’s activity levels or physical characteristics. The small population of this study reflected the local institutional practice of performing T1 VIBE imaging as a replacement for CT in the cross-sectional assessment of the lumbar spine to reduce radiation burden in this young population.
Although there is some debate regarding the definitive aetiology of isthmic spondylolysis, it is most thought of as an acquired entity which develops in the immature skeleton of children and adolescents. The normal pivot point for axial motion centres over the vertebral body. When repetitive extreme hyperextension and rotational forces are applied to the lower back, the force is applied through the facet joints, leading to closure and limitation by the facet joints. Extension beyond this physiological point must occur through the posterior neural arch with resultant microtrauma to the pars interarticularis. The presence of a significantly larger sagittal pars angle found in patients with spondylolysis in our study could suggest an increased susceptibility to the aforementioned microtrauma from repeated hyperextension, owing to the more horizontal course of the pars interarticularis. Similarly, an increased lumbar lordotic angle and pelvic inclination angle have also been suggested as individual risk factors for spondylolysis.[11]
Low-grade injuries may be treated conservatively with activity reduction, rehabilitation and splinting. These represent the majority of cases. Stress fractures in other parts of the body demonstrate a natural course of healing following immobilisation. Such immobilisation may not occur in individuals with spondylolysis due to absent, mild or non-specific symptoms and subsequent delayed radiological diagnosis. In early pars stress injury, the utilisation of an anti-lordotic brace has demonstrated healing and resolution of symptoms, suggesting early identification and instigation of treatment may reverse the process.[12] However, other authors have found a poor correlation between pain and spondylolysis and as such dispute recommending activity restriction as a treatment option.[3] A prospective study similarly reported that pars defects follow a clinical course similar to that of the general population, although it did not discriminate against individuals with an athletic lifestyle or those presenting with pain.[13] The ultimate management will depend on clinical presentation, patient/parent expectations and physician preference.[14]
High incidences of spondylolysis with or without spondylolisthesis have been described in gymnasts, divers, wrestlers, weight lifters, cricketers, footballers and those engaging in racket sports such as tennis.[15,16] The defect typically occurs in childhood and adolescence; it rarely develops or progresses after skeletal maturity. It is not observed in newborn infants or a large case series of non-ambulatory patients, further supporting the idea that it is acquired.[3,17] Other established predisposing factors include genetics (>20% incidence among first-degree relatives) and Indigenous Alaskan ancestry.[1,18] One of the largest case series of 4243 patients conducted by Rossi highlighted the differential occurrence of pars defects in adolescent athletes.[15] The highest incidence was identified in divers (40.35%), wrestlers (25%) and weight lifters (22.32%) followed by footballers (16.25%) and gymnasts (16.64%) and lower in rugby players (10.76%), basketball players (9.7%) and rowers (7.72%).
In the context of the findings from our study, which showed a significant association between a large sagittal pars inclination angle and incidence of spondylolysis, we propose that a large sagittal pars inclination angle may be used as an indicator of whether an athletic adolescent may (a) develop or (b) progress a known spondylolysis.[7] The value would be in assessing a young athlete who is excelling in multiple sports—for example, both rowing and weight lifting, in whom a high pars inclination angle is identified, allowing him/her to pursue the lower risk option as a lifetime career. A high pars inclination angle may also help coaches and sports scientists schedule training plans with more time for recovery to accommodate load management. The angle may also be measured on MRI, rather than CT to reduce radiation burden in this young population. Further prospective studies are required to assess whether a large pars angle predisposes individuals to spondylolysis, whether spondylolysis increases the sagittal pars angle or in fact if they occur in tandem.
In highly active sportspeople, those with high-grade injuries or injuries with evidence of significant interval progression to spondylolisthesis, surgery may be indicated. The sagittal pars inclination angle has been reported in one study utilising Buck’s technique of direct spondylolysis repair as an alternative to conventional fusion surgery.[19,20] This may be automated by artificial intelligence software with additional computed algorithms providing the ideal corrected pars inclination angle specific to an individual patient based on (a) physical characteristics, for example, size, (b) age and (c) the vertebral level in question. Further studies with larger data sets are required for this to be possible.
Conclusion
In this study, an increased sagittal pars inclination angle demonstrated a significant association with spondylolysis. Its implications in the management of athletic individuals and surgical planning need to be evaluated further in future studies.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
No patient is involved in the study. An Institutional review board approval was obtained.
Credit author statement
Hussein M and Botchu R conceptualised the study and helped in resources and data curation and methodology and writing—original draft.
Hussein M, Iyengar KP, Mehta J and Botchu R helped in formal analysis.
Hussein M, Azzopardi C, Bendi SR, Hassan F, Bhogal G, Iyengar KP, Mehta J and Botchu R contributed to writing—review and editing.
Data Availability
Nil.
Use of Artificial Intelligence
Nil.
