Abstract
Introduction:
One of the most prevalent and major causes of cancer-related mortality is colorectal cancer (CRC). Unfortunately, despite being labelled as low-risk cancers based on their tumour, node, metastasis (TNM) stage, many tumours behave poorly. The independent forecasting tool for CRC, particularly in node-negative illness, is tumour budding (TB). The term ‘tumour buds’ (TB) refers to detached (epithelial) tumour cells, which are single or clusters (less than or equal to five cells) at the maximum invasive front. The aim of this study was to evaluate TB using pan-cytokeratin immunostain and comparing it with haematoxylin and eosin (H&E) staining and clinicopathological parameters.
Methods:
Resected specimens without presurgical therapy were analysed in the Department of Pathology for one year. Among histologically proven colorectal carcinoma, TB scores on H&E and pan-cytokeratin immunostained slides were compared.
Results:
Among the various clinicopathological parameters evaluated in 83 cases of CRC, we emphasise from our study that there was a strong correlation of TB score with tumour type, grade of tumour and metastasis. There is a definitive chance of upgradation of TB score by cytokeratin immunostain.
Conclusion:
TB is a negative prognostic factor responsible for adverse outcomes in CRC patients. Regardless of the scoring method used, cytokeratin-based assessment of TB is superior compared with the TB score assessed by H&E alone.
Keywords
Introduction
One of the most prevalent and major causes of cancer-related mortality is colorectal cancer (CRC).[1] In 1950, CRC was relatively uncommon, but today, it is the most common cancer in Western nations, accounting for 10% of all cancer-related deaths. The tumour, node, metastasis (TNM) staging system’s classification of the disease severity serves as the foundation for the prognosis and therapy of CRC.[2] Unfortunately, despite being labelled as low-risk cancers based on their TNM stage, many tumours behave poorly. The independent forecasting tool for CRC, particularly in node-negative illness, is tumour budding (TB). This is the most encouraging finding among the histopathological variables examined.[3] The term ‘tumour buds’ refers to detached (epithelial) tumour cells that, in direct contact with their milieu,[4] undergo at least partial transformation into mesenchymal stem-like phenotype.[5] During the process, the TB loses specific epithelial properties and gains those associated with enhanced cell motility.[6] TB is visually and physiologically connected to poorly differentiated clusters (PDC), which are single or big tumour cell clusters (less than or equal to five cells) without gland development and are also found near the tumour’s invasive front.[7] The crucial interface where tumour development and tumour cell dispersion occur, together with the loss of cytological markers of differentiation (dedifferentiation of CRC cells), is the tumour–host interaction at the invasive front of CRC.[8] Epithelial mesenchymal transition (EMT) is thought to occur physiologically throughout embryological development and is expected to occur pathologically during fibrosis and tumour invasion, which is reflected in TB.[9] Tumour cells are attempting to spread outside of the primary tumour mass. It is considered a robust predictor of nodal metastases and advanced cancer stages. Infiltrating growth patterns, apparent variations at the invasive front and unexplained tumour interaction with the surrounding stroma are all indicators of TB in standard H&E staining. TB and cytoplasmic pseudo-fragmentation, although they have heterogeneous mechanisms, are potentially stated as markers for increased aggressiveness. Fibroblastic proliferation, peritumoral inflammatory cells and cytoplasmic pseudofragmentation can occasionally be confused for buds, and single-cell buds are typically undetectable and frequently overlooked during H&E evaluation. Pan-cytokeratin immunostaining is a good option in routine diagnostic practice to address all these diagnostic challenges and evaluate TB.
Advantages of cytokeratin over H&E: The biology of cancer at the invasive front is better reflected by immunostaining, which unmasks three to six times more TB than H&E.[10] The increased interobserver agreement that can be attained by immunohistochemistry (IHC) with cytokeratin in contrast to H&E is an objective measure of reliability.[11] Typing of the tumour margins, determination of the degree of TB and the degree of cytoplasmic pseudo-fragmentation will be performed more accurately on the pan-cytokeratin immunostained slides.[12] The prospective study was carried out by evaluating TB using pan-cytokeratin immunostain and comparing it with H&E staining.
Methods and Materials
This prospective study was conducted at a quaternary care centre between June 2022 and May 2023, with a sample size of 83 cases of CRC. Resected specimen was analysed in the Department of Histopathology. The study aimed to elucidate the utility of pan-cytokeratin in evaluating TB scores in CRC patients. All resection cases of CRC were included in the study, and all cases of CRC with preoperative chemotherapy, radiotherapy and combined chemo-radiation were excluded. The study objectives encompassed an exploration of practical issues related to TB, an examination of challenges associated with definitional heterogeneity in its assessment, and a comparison between TB scores obtained by conventional H&E-stained slides and pan-cytokeratin IHC slides.
Methodology
From the computerised medical records and histology requisition forms, clinical details were obtained. The excision biopsy specimens were fixed in 10% neutral buffered formalin for at least 8 hours and grossed according to the protocol. The tissue was processed overnight, 3–5 micron-thick sections were cut, and the slides were stained with H&E. Light microscopy study of the morphology of the tumour was done and classified according to the TNM staging system along with the College of American Pathologists (CAP) protocol reporting format, which incorporates TB score. Quantification of TB score should be reported by selecting a ‘hotspot’. Hotspot was selected after a review of all slides with invasive tumours. A total number of TB should be reported in an area measuring 0.785 mm2 and corresponding to 20x field in all the microscopes.
Appropriate tumour blocks were selected for the immunohistochemistry study. Pan-cytokeratin immunostaining was performed using the VENTANA Benchmark XT System. Primary antibody ZYTOMED prediluted PANCYTOKERATIN Clone AE1/AE3 was used. This antibody produces membrane and cytoplasmic staining. The TB score was compared between routine H & E staining and pan-cytokeratin immunostaining.
Statistical Analysis
Data analysis was carried out by IBM SPSS for Windows Version 28.0 (Armonk, NY: IBM Corp). All the continuous variables were represented as mean ± SD (standard deviation). Skewed variables were represented as medians (interquartile range [IQR]). Categorical variables were represented as percentages. A comparison of categorical variables was made using the chi-squared test or Fisher’s exact test. The coefficient of variation was calculated. Kappa statistics were used to determine the agreement between the TB score by conventional H&E and the TB score by pan-cytokeratin immunostain.
Results
In this study, 83 cases of CRC from resection specimens were chosen. TB score on H&E was compared with cytokeratin immunostain. Of the 83 cases received during the study period of one year, cases with low, intermediate and high TB score on H&E were 59.0% (n = 49), 38.6% (n = 32), and 2.4% (n = 2) respectively. Cases with low, intermediate and high TB score on IHC were 48.2% (n = 40), 37.3% (n = 31) and 14.5% (n = 12) respectively. The P value was determined by comparing the TB score obtained on IHC to all clinical and histomorphological characteristics. Of the clinicopathological parameters evaluated, there was a strong correlation of TB score with histologic tumour type (P value < .004), histologic grade of the tumour (P value < .001) and metastasis (P value < .001). Other clinicopathological parameters such as age, gender, type of resection specimen, location of the tumour, tumour size, tumour infiltration, lymphovascular invasion, perineural invasion, regional lymph node status, margin and stage were not statistically significant [Table 1].
Comparison of the clinicopathological parameters with tumour budding score on IHC
Tumour Budding Score on H&E Compared with Tumour Budding Score on IHC
The IHC scores for low, intermediate and high TB cases were 48.2% (n = 40), 37.3% (n = 31) and 14.5% (n = 12) of the 83 cases respectively. Among the 48.2% (n = 40) cases with low TB scores, on IHC, 97.5% (n = 39) demonstrated concordance with the H&E-derived TB scores. Similar to this, 74.2% (n = 23) of the 37.3% (n = 31) cases among intermediate TB score matched the TB score determined by H&E, while only 16.7% (n = 2) of the 14.5% (n = 12) cases among high TB score matched the TB score determined by H&E. Therefore, out of 49 cases among low TB score by H&E, 2 cases upgraded to high TB score and 8 cases upgraded to intermediate TB score by IHC. Similarly, among 32 cases of intermediate TB score by H&E, 8 cases upgraded to high TB score and a single case to low TB score by IHC. There is a definitive chance of upgradation of TB score by cytokeratin immunostain [Table 2].
Tumour budding score on H&E compared with tumour budding score on IHC

In this study of 83 cases, 77.1% (n = 64) cases show no variation between the TB score assessed by conventional H&E and the TB score assessed by pan-cytokeratin immunostain. About 22.9% (n = 19) of cases show variation between TB score assessed by conventional H&E and TB score assessed by pan-cytokeratin immunostain. Of which 42.1% (n = 8) cases were reported as low TB score by H&E, but pan-cytokeratin immunostain had an intermediate TB score. About 10.5% (n = 2) of cases were reported as low TB scores by conventional H&E, but pan-cytokeratin immunostain turned out to be high TB score. About 42.1% (n = 8) cases reported as intermediate TB score by conventional H&E, but by pan-cytokeratin immunostain, turned out to have a high TB score. One case (5.3%) was reported as an intermediate score by conventional H&E, but pan-cytokeratin immunostain turned out to be a low TB score.
Agreement Between Tumour Budding Assessed on Conventional H&E Sections and Tumour Budding Assessed on Pan-Cytokeratin Immunostained Sections in CRC
Intermediate and high TB scores are summed up together as high scores. Among these, 48.2% (n = 40) and 51.8% (n = 43) cases were of low and high scores by IHC. Of 40 cases, 97.5% (n = 39) cases were concordant with the TB score obtained by H&E; of the 43 cases, only 76.7% (n = 33) cases were concordant with the TB score obtained by H&E. Kappa value (0.737) strongly suggests that the assessment of TB score by pan-cytokeratin immunostain is reliable when compared with the TB score assessed by routine H&E stain. Therefore, from our study, the TB score assessed by IHC was reliable and reproducible [Table 3].
Agreement between tumour budding assessed on conventional H&E sections and tumour budding assessed on pan-cytokeratin immunostained sections in CRC

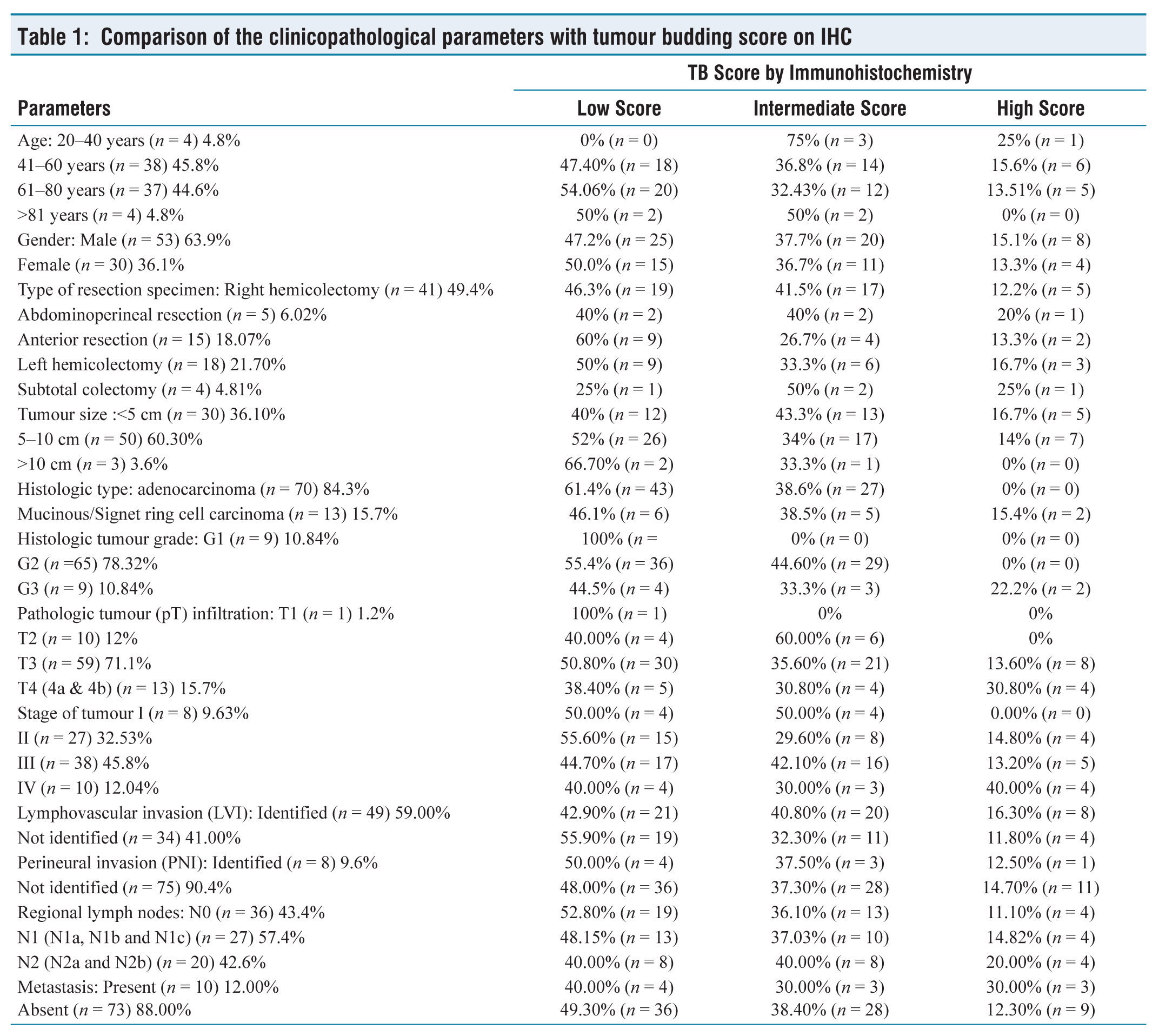
Tumour budding score by H&E and IHC respectively. A, B (20X): low score, C, D (20X): intermediate score and E, F (20X): high score
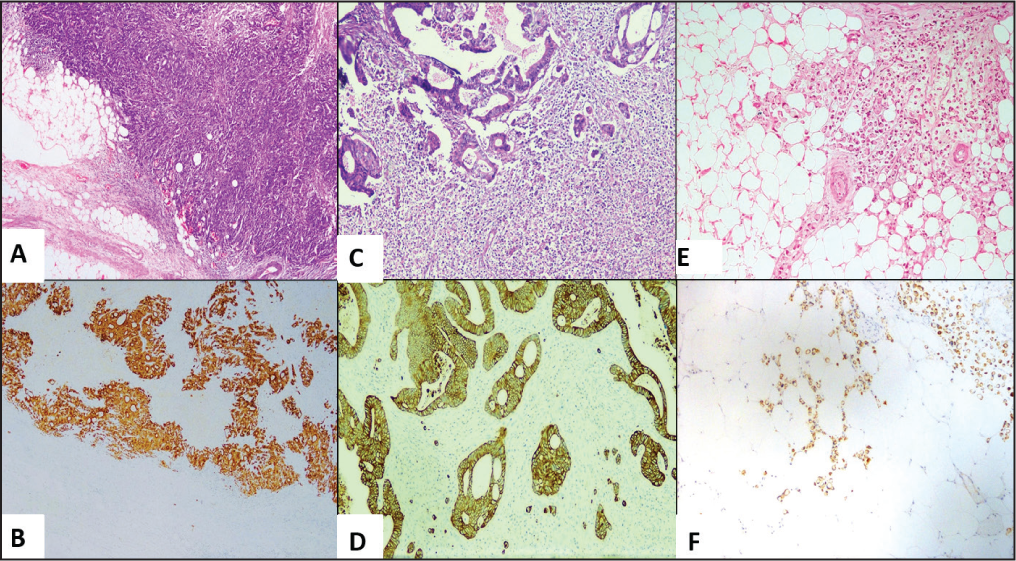
CK-Positive mimickers. A (20X): H&E (peritumoral inflammation and fibroblastic proliferation) and B (20X): IHC
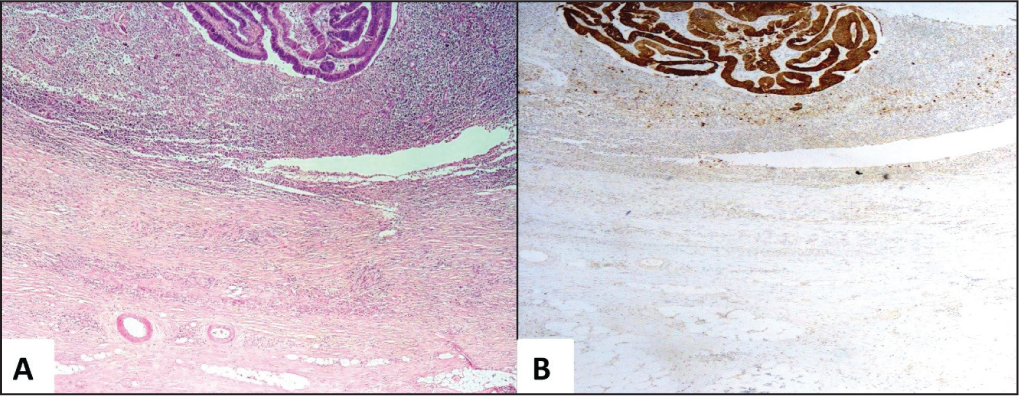
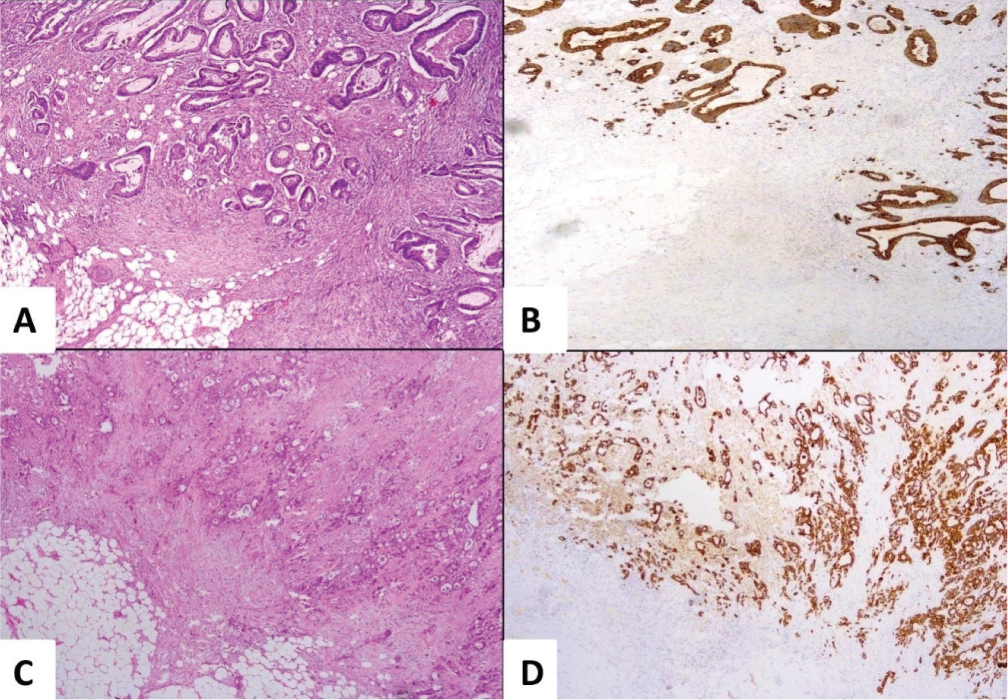
Upgradation of tumour budding score by IHC (low score on H&E, upgraded to intermediate and high score on IHC). A, C (20x): low score by H&E, B (20X): high score by IHC and D (20X): intermediate score by IHC
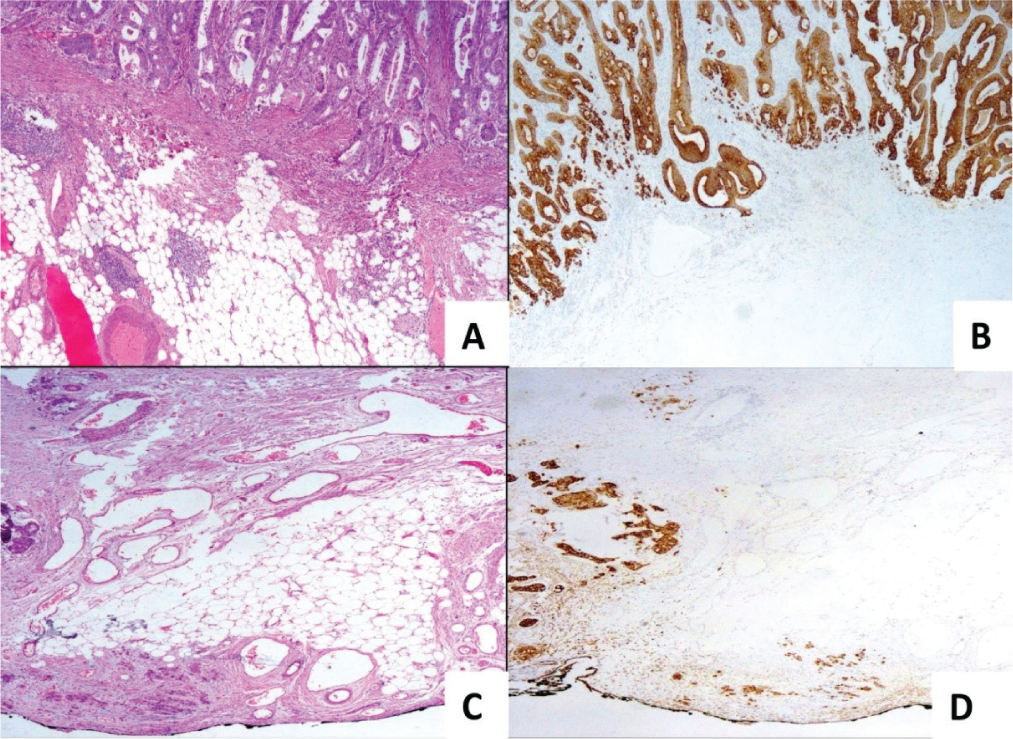
Upgradation of tumour budding score by IHC (intermediate score on H&E, upgraded to high score on IHC). A, C (20X): intermediate score by H & E and B, D (20X): high score by IHC
Discussion
CRC is a heterogeneous disease with complex aetiology.[13] In CRC, expansile and infiltrative tumour growth patterns at the invasive front are usually distinguished.[14] Further investigations in CRC, identified two morphological variations of cancer cell dissociation, namely TB and PDCs, which act as independent prognostic markers and upon which novel grading systems for CRC have been developed.[15] While the experienced pathologist can identify TB reliably and reproducibly, according to Kai et al. (2016),[16] cytokeratin immunohistochemistry may reduce interobserver variability. Later important questions to be answered are how many TBs are significant and how they shall be enumerated (definitional heterogeneity in TB). Several authors have proposed a wide range of scoring systems, including categorical and quantitative ones. To be relevant for prognosis and therapeutic decision-making in clinical practice, a reproducible method with a definite cut point must be used. Accordingly, in 2016, ITBCC (International et al. Conference) defined TB and scored it accordingly, which is incorporated in the reporting format of the CAP protocol of colon.[17] In our study, we followed the ITBCC method for counting TB. Shinto et al.[18] recently proposed that there is a morphological connection between TB and tumour cell migration at the invasive margin by pseudopod formation –CK-positive mimickers. The ability to clearly see a cell nucleus is crucial for TB since it enables viewers to identify TB from the CK above positives.[19] The major objective of this work is to investigate TB cells utilising pan-cytokeratin CK immunostain. We used AE1/AE3 CK staining following prior findings. In contrast to CK17 and CK18.25, the keratin cocktail AE1/AE3 can recognise CK1–6, CK8, CK10, CK14–16 and CK19. It is essential to choose a broad-spectrum anti-CK because different molecular subgroups of CRC may have different cytokine profiles.
In this study, TB score by pan-cytokeratin immunostain strongly correlated with tumour type, histologic grade and metastasis.
The reason behind the discordance between TB score by conventional H&E and TB score by pan-cytokeratin immunostain, based on our experiences, substantiates
Fibroblast proliferation Dense peritumoral inflammatory cell infiltrates Infiltrative growth pattern. Fragmented tumour cells floating in mucin. Retraction artefact surrounding the fragmented glands.
This was in agreement with Mitrovic et al.’s findings (2012).[20]
In this study, 3/83 cases were presented as dual tumours, and 1/83 were presented as triple tumours. In all dual and triple tumours, there was no variation in TB score assessed by H&E and TB score assessed by IHC.
In this study, 10/83 cases presented with metastasis to adjacent organs. Sites involved were fallopian tubes, ovary, liver, spleen, pancreas and jejunum. The TB score assessed by H&E was compared with the TB score assessed by pan-cytokeratin immunostain. Accordingly, intermediate and high TB scores were summed together to find out the reliability among these. Thus, the assessment of TB score by H&E stain (59%, n = 49) and 41%, n = 34) was of the low and high scores (intermediate and high TB scores summed up). Similarly, the assessment of the TB score by IHC stain (48%, n = 40 and 52%, n = 43) was of low and high TB scores (intermediate and high TB scores summed up). Among 40 cases under a low score by IHC, 97.5% (n = 39) cases were concordant with the TB score assessed by H&E. Similarly, among 43 cases of high TB score by IHC only 76.7% (n = 33) cases were concordant with the TB score assessed by H&E; however, discrepancies were observed between TB score assessed by IHC and H&E in remaining cases, these discrepancies cases shows that there was an upgradation of TB score by IHC stain. We conclude our study by stating that there is a fair agreement between the TB score assessed by H&E and the TB score assessed by IHC stain with a Kappa value of 0.737 and P value < .001. Cytokeratin-based assessment of TB is superior compared with the TB score assessed by H&E alone.
Limitations of the Study
Only patients without presurgical therapy were included in the study.
Recommendations
Educational initiatives on the accuracy and reproducibility of TB score assessment are required. Since there is a significant change in the scores between H&E and IHC interpretation of TB, assessment by H&E and IHC should be employed regularly in histopathology (HPE) reports of CRC.
Conclusion
TB is a negative prognostic factor responsible for adverse outcomes in CRC patients. Regardless of the scoring method, TB assessed by cytokeratin is feasible and significant to overcome the diagnostic challenges faced while evaluating TB score on H&E, leading to expected associations with clinicopathological features. TB is essential for refining criteria to identify patients with stage II disease who may benefit from postoperative therapy.
Footnotes
Acknowledgements
Thanks to all technical staff of Histopathology Department, Apollo Hospitals, Chennai, Tamil Nadu, India.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
All authors can attest that in the submitted research article, only non-identifiable images are depicted. Approval was obtained from the Institutional Review Board (Ethics Committee) at Apollo Hospitals, Chennai, Tamil Nadu, India. (Institutional Ethics Committee (IEC). Application Number ASH-DNB- 010/05-22).
Informed consent
Informed consent has been obtained.
Credit author statement
KM participated in conceptualisation, methodology, data curation, analysis and investigation, manuscript preparation and manuscript editing.
SN participated in the data analysis, investigation, manuscript preparation and editing.
AS participated in conceptualisation and supervision. All the authors have reviewed and approved the manuscript.
Data availability
Raw data supporting the findings of this study are available from the corresponding author upon reasonable request.
Use of artificial intelligence
Nil.
