Abstract
Introduction:
Maternal, placental and foetal factors together or in isolation influence the well-being of the foetus. The two most important causes of intrauterine growth retardation (IUGR) and intrauterine death (IUD) are placental factors and hypoxemia.
Material & Methods:
The present study was a prospective study conducted in the Department of Pathology. Placentas of IUD and IUGR cases were collected and routinely processed.
Results:
There were a total of 52 placentas, 40 from IUD cases and 12 from IUGR cases. Gross infarction was identified in 13.46% cases and microscopic infarction in 23.1% cases which were the most common findings.
Conclusion:
Chorioamnionitis was the most common histological finding in both IUGR and IUD cases. Careful examination of the placenta is necessary in IUD and IUGR cases.
Keywords
Introduction
Placenta forms a functional unit between mother and foetus. Foetal growth and viability depend on supply of nutrients and oxygen through the placenta into umbilical circulation. Any disturbance in maternal health will affect placenta and placental circulation thereby leading to foetal morbidity and mortality. Placental factors and hypoxia are key to intrauterine growth retardation (IUGR) and intrauterine death (IUD). IUGR is an adaptive response associated with placental insufficiency. Failure of these adaptive changes leads to an IUD. IUGR may be caused by foetal, maternal or placental factors. Conditions resulting in placental insufficiency can be recurrent. So placenta can provide valuable information relating to perinatal morbidity and mortality. The present study was done to identify the morphological changes of placenta in IUD and IUGR foetuses.
Material and Methods
The present study was a prospective study done in the Department of Pathology over a period of two years. Inclusion criteria were placentas of IUGR and IUD cases which were diagnosed clinically and radiologically and with gestational age >24 weeks of the foetus. Exclusion criteria were placentas of IUGR and IUD cases with gestational age <24 weeks.
Prior approval was taken from an institutional ethics committee. Consent was taken from all the mothers after explaining the study details. A proforma was maintained which included detailed obstetric and medical history along with relevant investigations.
After delivery, placentas were collected in a clean tray, blood was removed gently and they were washed thoroughly under tap water. They were then placed in 10% formalin and transported to the Pathology Department. After fixation for 24 hours, the placentas were examined grossly for dimensions, shape, number of lobes, changes in maternal surface and foetal surface, cut section and weight. Membranes were observed for colour, transparency, oedematous changes, and blood clots. Length of the cord, cord thickness, coils in the cord, insertion of the cord, number of vessels were noted. Photographs were taken wherever necessary. Parallel slices were made 2cm apart and representative sections were submitted.
Data was analysed using MS Excel and Statistical Package for Social Sciences (SPSS) version 22. Data was represented using descriptive statistics like frequencies and percentages for categorical variables.
Results
A total of 52 placentas were included in the study, of which there were 40 IUD cases, 12 IUGR cases. Ten normal cases were also included. History of maternal/foetal/both risk factors were present in 27 (52%) cases and no such history of risk factors in 25 (48%) cases [Table 1]. In cases with no risk factors, placental abnormalities were still found in 13 cases. Low placental weights were found in 7 (13.46%) cases. The maternal risk factors most commonly seen in the present study were pregnancy-induced hypertension (PIH) (11.53%) and maternal heart disease (3.85%).
Risk Factors Identified for IUD/IUGR in the Present Study
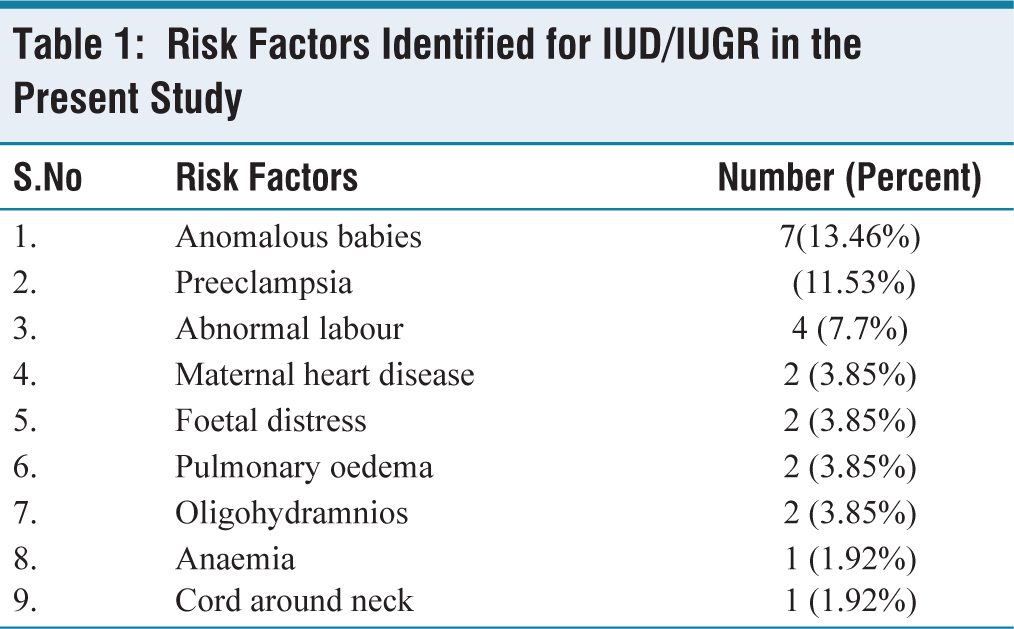
Gross abnormalities found in the present study include infarcts (13.46%), hypercoiling (3.85%), single umbilical artery (1.92%), cord constriction (1.92%), hypocoiling (1.92%), false knots (1.92%) and supernumerary vessels (1.92%) [Figures 1a-1d, 2a-2d and Table 2].
(a). Gross infarcted area. (b) Cut section of infarcted placenta. (c) Single umbilical artery. (d) Constriction of cord
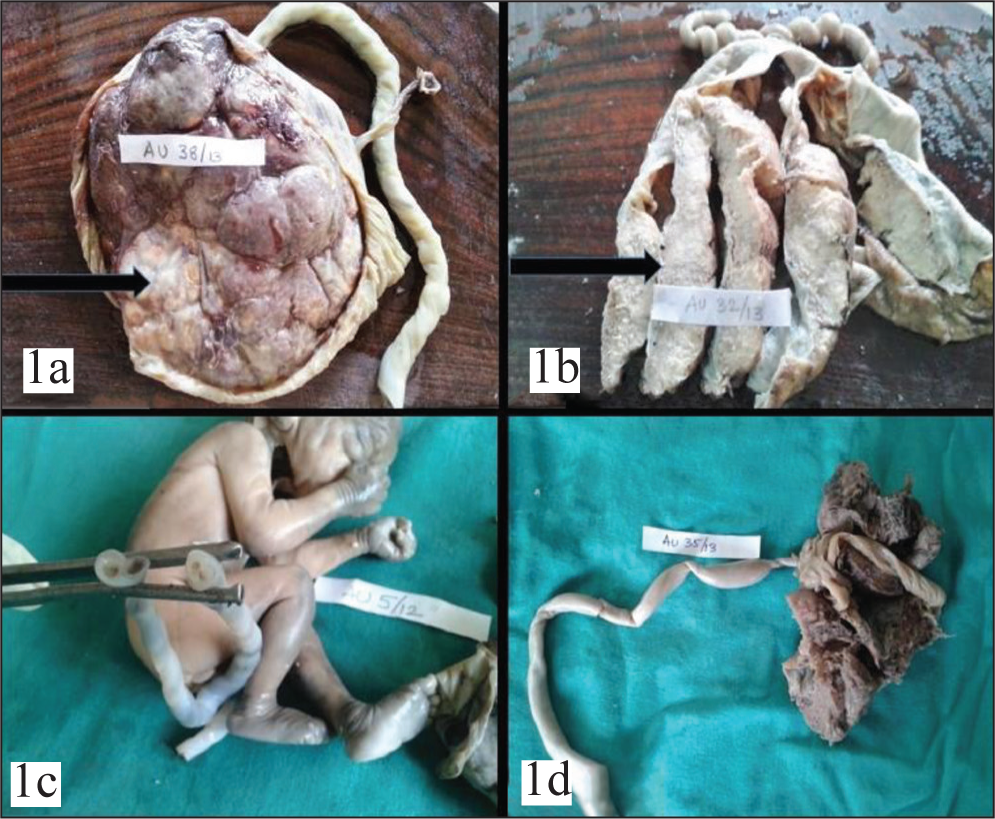
(a) Hypercoiling. (b) Hypocoiling. (c) False knots. (d) Supernumerary vessels
Gross Abnormalities Identified in the Present Study

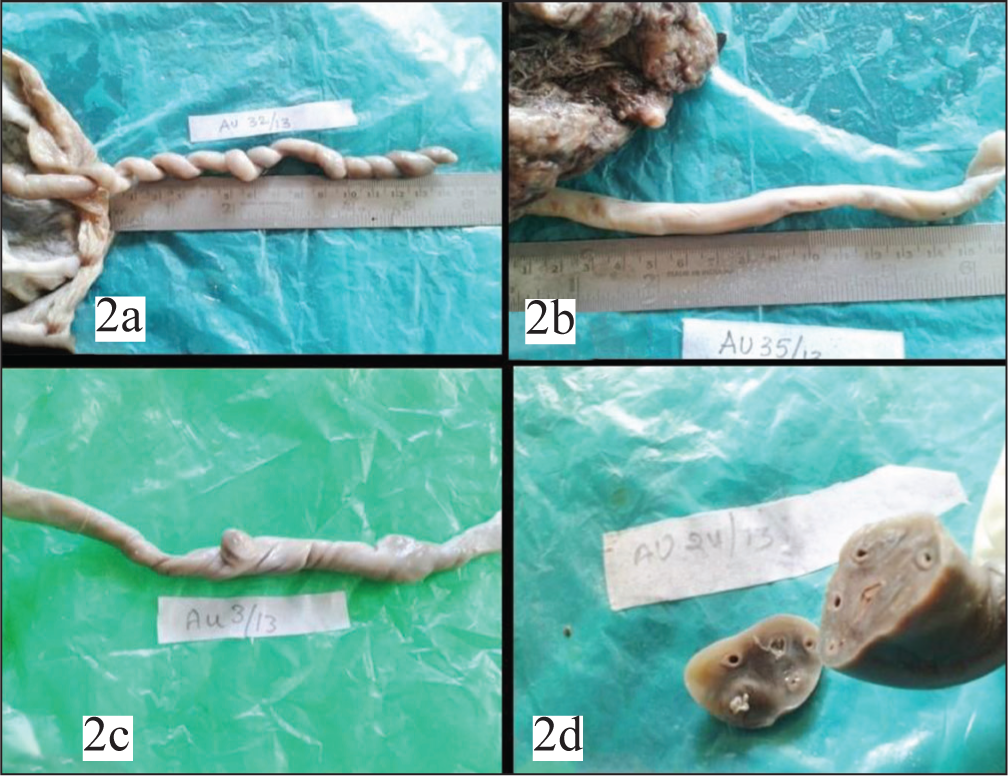
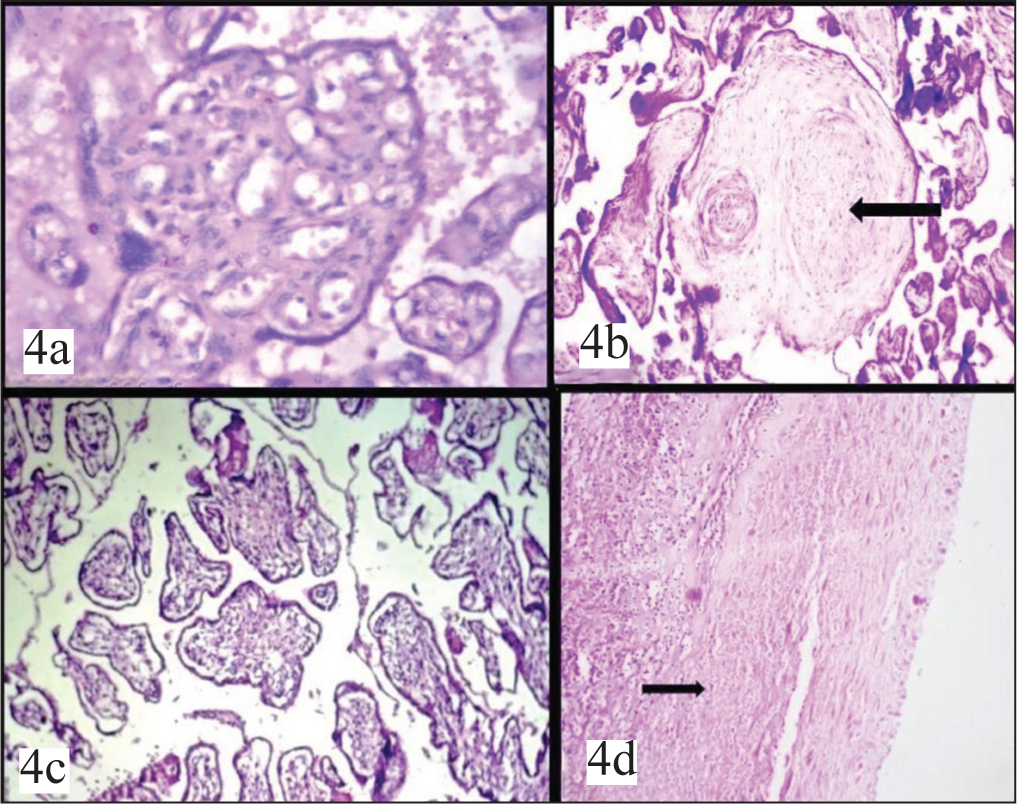
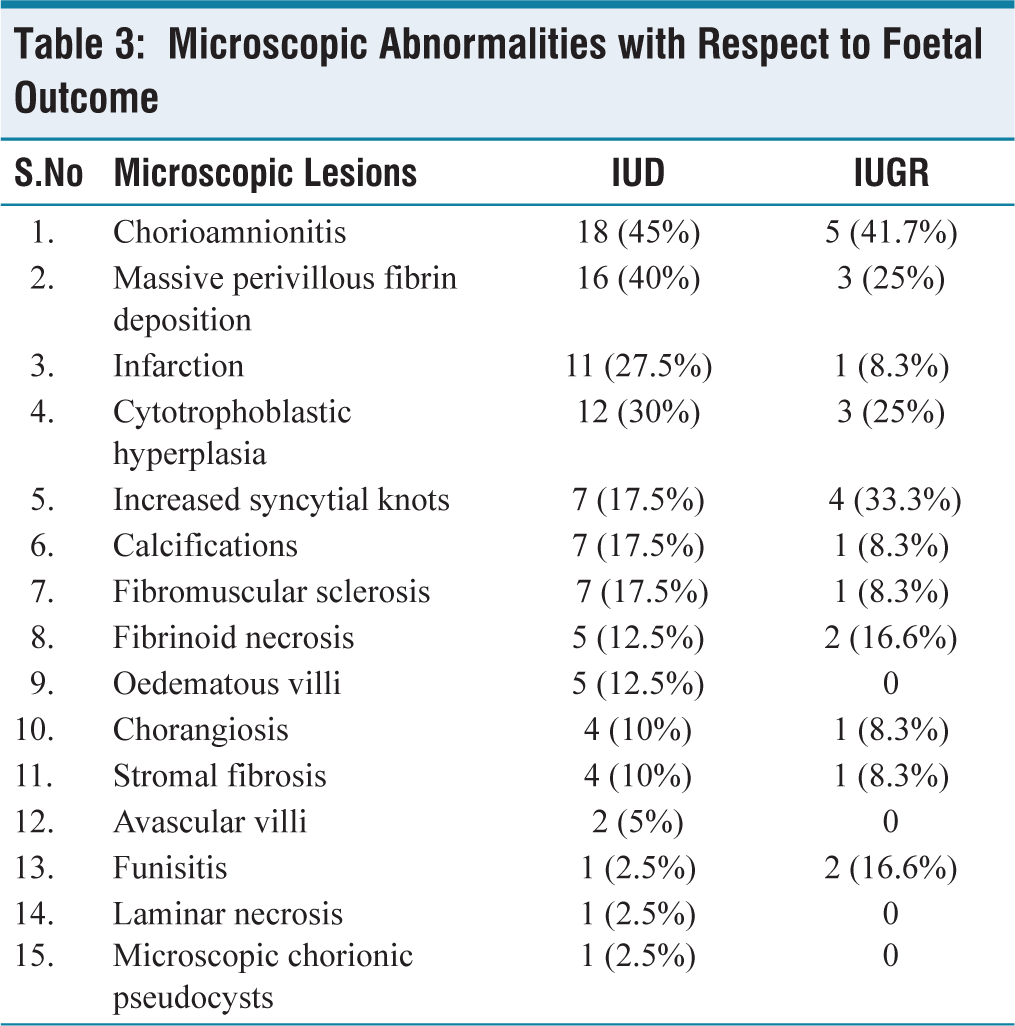
On microscopic examination, pathological changes ranged from the most commonly seen chorioamnionitis to rarer entities like laminar necrosis, microscopic chorionic pseudocysts [Table 3]. Both IUD and IUGR cases were commonly associated with chorioamnionitis on microscopic examination [Figure 3a]. Microscopic features like fibrin deposition, infarction, and calcifications were more commonly associated with IUD rather than IUGR whereas cytotrophoblastic hyperplasia, syncytial knots, fibrinoid necrosis, funisitis were commonly associated with IUGR compared to IUD [Figures 3b-3d and 4a-4d].
(a) Chorioamnionitis H&E 100X. (b) Infarction H&E 100X. (c) Fibrin deposition H&E 100X. (d) Syncytial knots H&E 400X
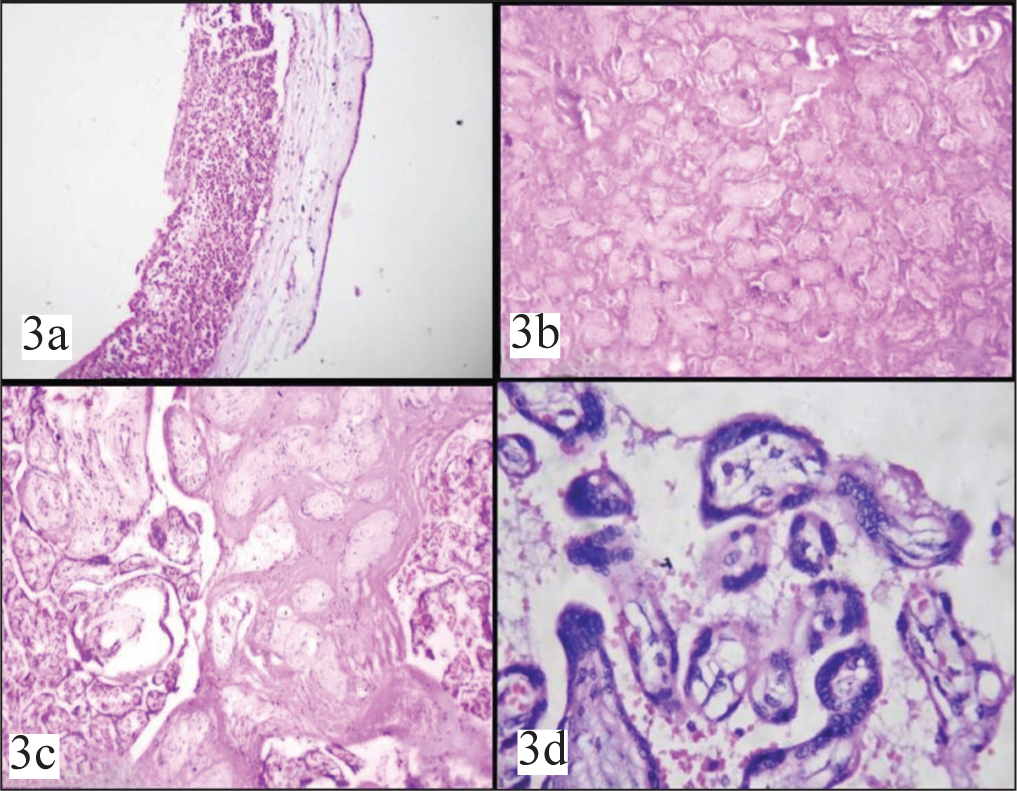
(a) Chorangiosis H&E 40X. (b) Fibrous obliteration of villi H&E 100X. (c) Villous oedema H&E 100X. (d) Laminar necrosis H&E 100X
Microscopic Abnormalities with Respect to Foetal Outcome
Discussion
In chromosomally normal gestations, small placentas usually result from conditions leading to prolonged reduction in uteroplacental perfusion. As maternal blood nourishes the placenta as well as the foetus, maternal vascular diseases such as hypertension, preeclampsia and autoimmune disorders complicated by vascular disease may induce a variety of pathologic placental changes, such as placental infarction and abruption in addition to overall diminished placental growth.
Constriction or stricture of the cord consists of focal depletion of Wharton’s jelly with narrowing of vessels. Constriction can lead to foetal death because of chronic ischaemia superimposed by a catastrophic vascular event. Cord constriction was present in 4.1% of IUD cases in a study by Singh et al., whereas it was found in 8.3% cases of IUD in the present study.[1]
Placental infarction can be observed in normal pregnancies also. It is usually of no significance unless it affects more than 10%-20% of the placental volume.[2] In a study by Khajuria et al., placental infarction was detected in 53.3% cases of IUGR.[3] In the present study, microscopic infarcts were found in 11 cases (27.5%) of IUD and in one case (8.3%) of IUGR. Infarcts were more commonly associated with PIH as five out of six cases showed infarcts. Other causes of infarcts in the study were abnormal foetal presentations, anencephaly etc.
In a study by Ujwala et al. perivillous fibrin deposition was seen in 3 (11.1%) cases of IUD and 1 (3.7%) case of IUGR.[4] In the present study, massive perivillous fibrin was found in 40% of IUD and 25% of IUGR cases. Fibrinoid necrosis was found in 5 (12.5%) cases of IUD and 2 (16.6%) cases of IUGR. It was associated with pre-eclampsia in the present study. Iiker et al., identified fibrinoid necrosis in 4% of IUGR cases and 3% of IUD cases.[5]
Fibromuscular sclerosis results in abnormal foetal stem vessels due to thickening of the muscular wall of vessels and progressive obliteration of lumen with loss of endothelial lining.[6] Vessel wall thickening was found in 45% of 120 consecutive deliveries in a study by Ramachandran et al.[7] In the present study, it was found in 7 (17.5%) IUD cases and 1 (8.33%) IUGR cases.
Karmakar et al. identified syncytial knotting in 42% of 150 random deliveries and most of them were associated with maternal hypertension and diabetes.[8] In the present study, it was seen in 7 (17.5%) IUD cases and 4 (33.33%) IUGR cases of which there were three cases of PIH and one case of anaemia.
Foci of calcifications are normally seen in the term placenta. Ramachandran et al. found calcification in 22.5% of placentas while it was seen in 7 (17.5%) IUD cases and 1 (8.33%) IUGR cases in the present study.[7]
Focal villous oedema has been associated with premature delivery, neonatal hypoxia and neonatal death.[9] It is characterised by a split artefact between trophoblastic shell and villous core and can be differentiated from hypocellular villi which do not show this split artefact.[10] It was found in 12.5% cases of IUD in the present study. Iiker et al. found it in 12% cases of IUD and 12 % of IUGR cases.[5]
Cytotrophoblastic hyperplasia is seen in chronic hypoxic conditions that inhibit trophoblast fusion, differentiation and stimulate proliferation as seen in preeclampsia. In the present study, it was found in 12 (30%) cases of IUD and three cases (25%) of IUGR. Association with preeclampsia was seen in one case. In most of the cases, no significant risk factors could be identified to explain its causation.
Chorangiosis appears to be a nonspecific change related to a variety of conditions including maternal diabetes, hypertension, infections, anamolies, IUD and IUGR.[11] In a study on hypoxic patterns of placental injury, Stanek et al. found chorangiosis as a manifestation of hypoxic injury in 16.8% of cases.[10] In the present study, chorangiosis was found in 4 (10%) of IUD cases and 2 (16.6%) of IUGR cases, though there were no risk factors in all six cases. One placenta from normal delivery also showed chorangiosis.
In the study by Gupta et al., >5% of hypovascular villi were present in 57.1% of IUGR placentas.[12] Such villi reflect chronically abnormal uteroplacental vascular perfusion and may impair placental growth. They can be diffuse and associated with prolonged stillbirths or focal as in foetal thrombotic vasculopathy. In the present study, avascular villi were found in 2 (5%) of IUD cases, one of which was associated with maternal heart disease and other with preeclampsia.
Chorioamnionitis frequently precedes membrane rupture causing preterm labour. In a study by Khajuria et al. chorioaminionitis was found in two cases (6.6%) of IUGR and none in IUD cases.[3] In the present study, chorioamnionitis was found in 18 (45%) cases of IUD and 5 (41.66%) cases of IUGR.
Stanek et al. observed laminar necrosis in 27% of placentas from mothers with hypertensive disorders and was linked to various other maternal, foetal, neonatal and placental conditions associated with perinatal hypoxia.[13] We encountered one case in IUD.
MCP’s were strongly associated with pre-eclampsia and diabetes mellitus with their incidence mirroring the incidence of pre-eclampsia throughout gestation, peaking in the early third trimester.[14] They were seen in one case of IUD which was associated with PIH in the mother.
Conclusion
Gross abnormalities like infarcts, cord abnormalities etc were found in 26.9% cases. Microscopic abnormalities like chorioamnionitis, infarction were associated with IUD and IUGR. Strict criteria are essential to attribute various lesions as a cause of poor foetal outcomes as they can also be identified in normal placentas sometimes. Since pathological examination of placentas gives insight into the causes, placental examination should be routinely performed in all the cases associated with adverse foetal outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional Ethical Committee Approval Number
Institutional Ethical Committee Approval was obtained prior to the commencement of the study, ensuring adherence to ethical guidelines and patient safety.
Informed consent
Informed consent was obtained from the patient for participation in the study, ensuring full understanding of the purpose, procedures, and potential risks involved.
Data availability
The authors confirm that the data supporting the findings of the study are available within the article and its supplementary files. Raw data that supports these findings are available with the corresponding author, upon reasonable request.
Credit author statement
Chitturi Ramya: Design, Definition of intellectual content, Literature search, Data acquisition, Data analysis, Statistical analysis, Manuscript preparation, Manuscript editing, Manuscript review, Guarantor
Chavali Venkata Lakshmi: Design, Definition of intellectual content, Literature search, Data acquisition, Data analysis, Statistical analysis, Manuscript preparation, Manuscript review
Madana Padma: Literature search, Data acquisition, Data analysis, Manuscript preparation, Manuscript editing, Manuscript review
Gayatri Devi Parasa: Literature search, Data acquisition, Data analysis, Manuscript preparation, Manuscript editing, Manuscript review
Pathivada Sucharitha: Literature search, Data acquisition, Data analysis, Statistical analysis, Manuscript preparation, Manuscript editing, Manuscript review
Suratha Sivajyosthna: Literature search, Data acquisition, Data analysis, Statistical analysis, Manuscript preparation, Manuscript editing, Manuscript review
Use of Artificial Intelligence
Nil.
