Abstract
Introduction:
Lymphomas of the central nervous system (CNS) are rare, accounting for 3.1% of all primary brain tumours. They may be either secondary to systemic lymphoma or primary without the evidence of systemic involvement. In the past two decades, an increased incidence has been reported in patients aged >60 years. The study was undertaken to analyse the morphological and immunohistochemical profile of CNS lymphoma.
Methods:
The study included all lymphoma cases involving the CNS between 2014 and 2023. Two histopathologists examined the slides independently. For immunophenotyping, indirect immunohistochemistry (IHC) was used on paraffin-embedded tissue.
Results:
A total of 50 cases were diagnosed during this period with confirmed immunophenotyping. The age range was 11-76 years with male: female distribution of 3:2. HIV serology was available only for 30 cases and was negative in all except for one. The frontal lobe was the most common site of involvement. IHC was done in all 50 cases, of which 47 cases were large B-cell lymphoma, one peripheral T cell lymphoma, one Burkitt lymphoma, and one post-transplant primary CNS lymphoma (PCNSL). Among large B-cell lymphomas, two cases showed dual expression and cyclin D1 aberrant expression was detected in one case.
Conclusion:
This morphological and immunohistochemical study solidifies the fact that diffuse large B-cell lymphoma (DLBCL) was the most common immunophenotype observed among PCNSL.
Introduction
Lymphomas of the central nervous system (CNS) are rare. They may be either secondary to systemic lymphoma or could be primary without evidence of systemic involvement. Among Central Nervous System Lymphoma, Diffuse Large B-cell Lymphoma (DLBCL) accounts for 2.4%-3% of all brain tumours and 4%-6% of all extra-nodal lymphoma.[1,2] The peak incidence is between the fifth and seventh decades of life, with a median age of 66 years. The male-to-female ratio was 3:2, with slight male preponderance.[2]
The definitive pathophysiology of CNS lymphoma is still unclear. However, several hypotheses, as reviewed in the article by Nakamura et al.,[3] have been put forth, which include:
May arise from a systemic lymphoma that seeds multiple organs, including the brain. Though the immune system can find and eradicate systemic tumours, an immunologically privileged site like the brain gives sanctuary to the malignant lymphocytes within the CNS, resulting in the development of tumours. However, this is improbable, as there is no evidence of associated lymphoma in other immunologically privileged sites, like the testis. In the events of trauma or infectious processes that may attract peripheral blood lymphoid cells which get stimulated to proliferate locally and/or to undergo clonal selection and progress to a monoclonal neoplastic state. Though it is a plausible scenario, at present, however, we do not know which exogenous or endogenous antigenic stimulus might trigger an antigen-dependent clonal proliferation. In addition, inflammatory diseases mostly attract T-lymphocytes, while primary central nervous system lymphoma (PCNSL) is usually of B-cell origin. In addition, there is no increased incidence of PCNSL in patients with inflammatory CNS diseases. Lymphomatous cells generated in other tissues might acquire a “homing” for cerebral endothelium after developing adhesion molecules. However, a comparison of adhesion molecules and other surface antigens expressed by both PCNSL and systemic non-Hodgkin lymphoma (NHL) showed no significant differences.[4]
No data exist to support or refute any of these potential mechanisms. However, the basis of molecular pathogenesis is that the tumour cells exhibit increased self-reactivity/polyreactivity during a dysregulated germinal centre reaction, which enables them to bind to multiple CNS antigens. This could be the reason for the lymphoma cells’ affinity for and confinement to the CNS microenvironment.[2] These tumour cells also carry rearranged and somatically mutated Immunoglobulin genes with evidence of ongoing somatic hypermutation and show persistent BCL6 activity. The process of somatic hypermutation is not only confined to its physiological targets but also to other genes, which include BCL2, MYC, PAX5, PIM1, KLHL14, RHOH, OSBPL10, and SUSD22. These genes are implicated in tumorigenesis.
Current WHO, fifth edition, 2021, classifies CNS lymphoma under the following categories: Primary DLBCL, Immunodeficiency-associated CNS lymphoma, Lymphomatoid granulomatosis, Intravascular large B-cell lymphoma, MALT lymphoma of the dura, Other low-grade lymphomas of the CNS, Anaplastic large cell lymphoma (ALK+/ALK-), T cell lymphoma and NK/T cell lymphomas. Since CNS lymphoma is rare, we aim to study the morphological and immunohistochemical pattern of lymphomas involving the CNS diagnosed in a tertiary care centre in India between 2014 and 2023.
Materials and Methods
This retrospective study was conducted from 2014 to 2023 at our hospital. Only cases of CNS lymphoma diagnosed at our hospital with complete clinical details and immunophenotype-confirmed diagnosis were considered for inclusion. Cases with incomplete clinical data or insufficient tissue samples for immunophenotyping were excluded. Among cases eligible for analysis, a total of 50 cases met the eligibility criteria. Archived histopathological slides and formalin-fixed paraffin-embedded (FFPE) tissue blocks were retrieved for all eligible cases. Immunophenotyping was performed using standard diagnostic protocols to ensure accurate classification of CNS lymphoma subtypes. In this study, we aimed to find the morphological and immunohistochemical pattern of lymphomas involving the CNS diagnosed in a tertiary care centre in India.
Methodology
The specimens were received in 10% neutral-buffered formalin (NBF) and fixed for at least six hours. Fixation was done at room temperature (15°C-25°C). The specimens were then examined and grossed accordingly, followed by which the sections were treated using graded alcohols, cleansed in xylene, impregnated, and embedded in paraffin wax using standard techniques. A standard rotary microtome was used to cut three to five microns thick sections. The sections were then placed onto standard glass slides and stained using routine Harris Haematoxylin and Eosin stain.
Two independent pathologists reviewed the stained slides for various histological parameters, such as diffuse infiltration of brain parenchyma, with angiocentric growth pattern forming collars or perivascular cuffing, invasion of blood vessels, reactive T-lymphocytes infiltrating the tumours, reactive astrocytes, and areas of necrosis.
In all cases, immunostaining was performed in a Ventana autostainer. The following immunohistochemistry (IHC) antibodies, with their corresponding clones, were used.
Statistical Analysis
The summary of the statistics was presented with frequency (%) and mean ± SD for the categorical and continuous factors, respectively. The basic statistics were performed with SPSS software (IBM 28, .0).
Results
A total of 50 cases of CNS lymphoma were diagnosed in our department between the years 2014 and 2023.
Clinical Features
The mean (SD) age was 52(15) years, ranging from 11 to 76 years, with most patients falling in the group between 50 and 70 years (23 out of 50 cases, 46%). Of these 50 cases, 21 (42%) were female, and 29 (58%) were male, with a male-to-female ratio of 3:2. Clinically, most patients presented with signs of increased intracranial tension like headache and vomiting, followed by seizures and memory disturbance. HIV serology was available only in 30 cases, all of which were negative except for one. We had one patient of a post-renal transplantation (immunodeficient state). The most common site of the presentation was the frontal lobe (28%).
Histology and Immunohistochemical Analysis
Detailed slide reviews and IHC were performed on all 50 patients. All cases showed features of NHL in which tumour cells were arranged in a diffuse sheet pattern or dispersed pattern. The individual tumour cells were large with a vesicular nucleus, 1-3 conspicuous nucleoli, and a moderate amount of pale to eosinophilic cytoplasm. Mitotic activity was high in the majority of the cases with areas of necrosis. Characteristic perivascular or angiocentric pattern splaying apart the reticulin framework of blood vessels was seen in most of the tumours. In a few cases, the tumour cells were noted to extend along the Virchow Robin spaces.
IHC was performed in all 50 cases and the initial panel comprised of CD3, CD20, and Ki67. Additional markers like GFAP, BCL2, BCL6, MUM-1, SOX11, CYCLIN D1, C-MYC, EBER, etc. were done as the secondary panel [Table 2].
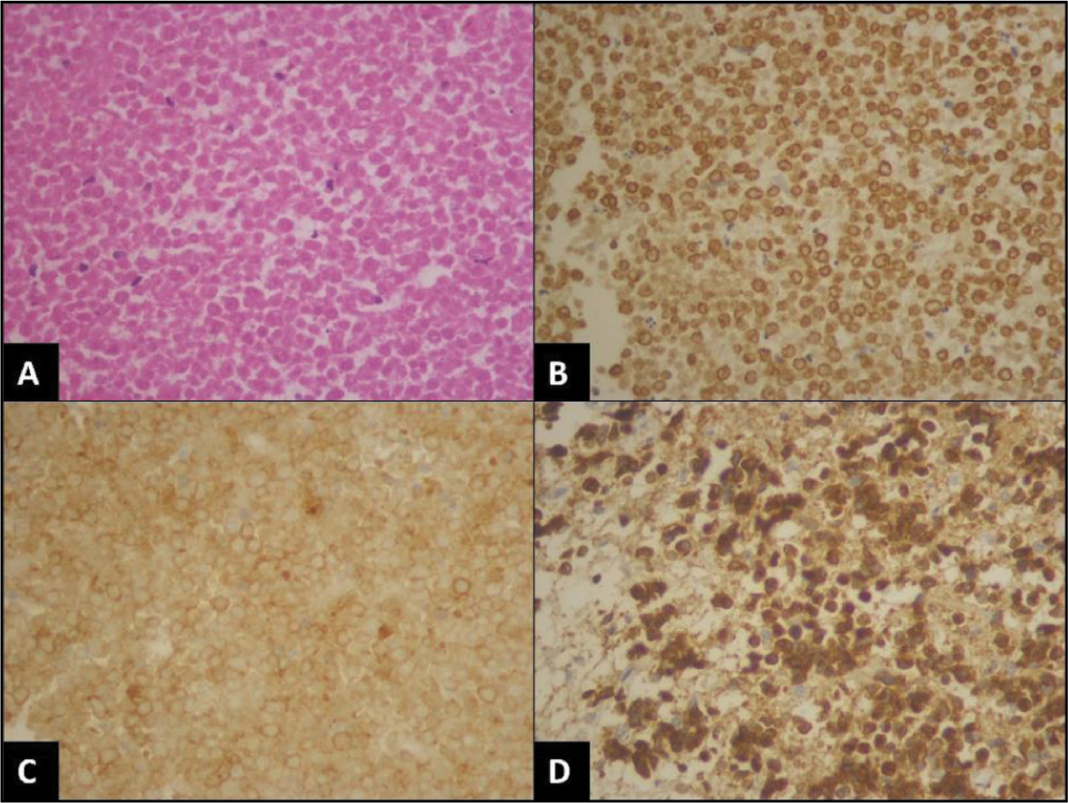
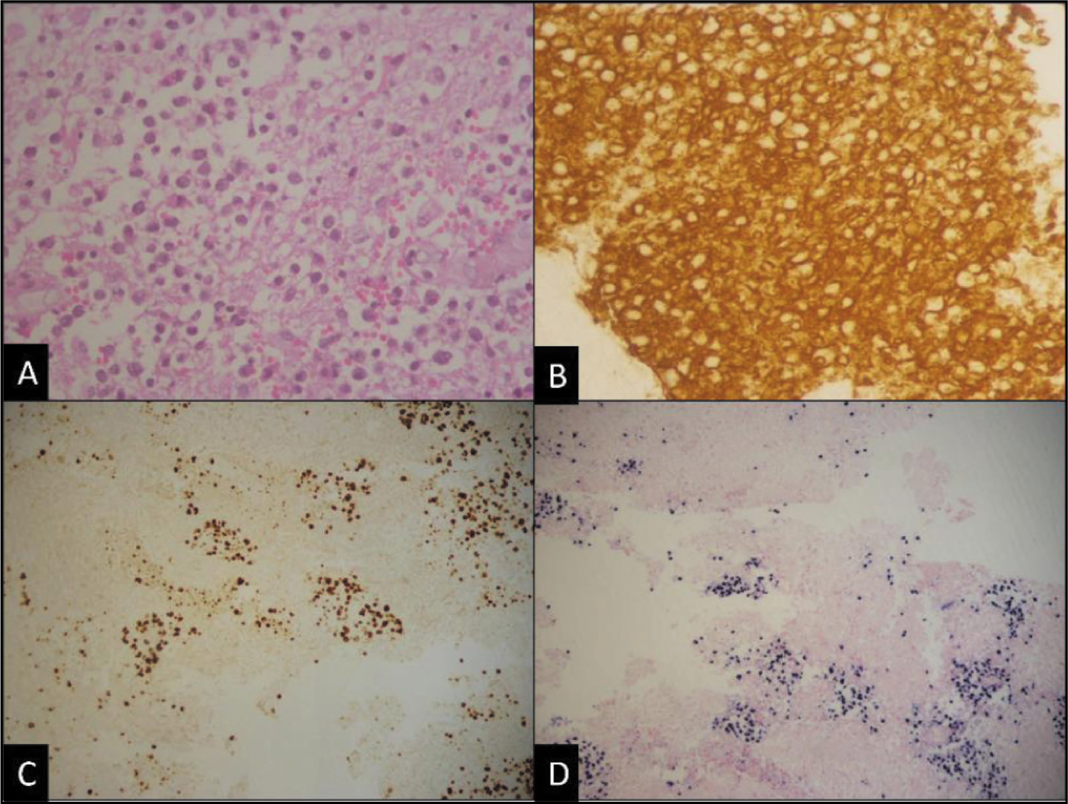
Almost all cases were B-cell lymphoma (49 out of 50 cases, 98%) with diffuse and strong CD20 positivity and CD3 negative except for one, which was CD3 and CD4 strong positive with high proliferative index (70%-80%) and diagnosed as peripheral T cell lymphoma [Figure 1A-1D]. Among B-cell lymphomas, the most common type was found to be DLBCL (48 out of 50 cases), with one case of post-renal-transplant CNS lymphoma [Figure 2A-2D] which was diagnosed as primary DLBCL since there was no systemic involvement, one case was Burkitt lymphoma and one case of DLBCL revealed double expression and one aberrant cyclin D1 expression [Figure 3A-3C].
Peripheral T cell lymphoma of the central nervous system. (A) Tumour cells are arranged in a diffuse sheeted pattern (haematoxylin and eosin [H&E], 400×). (B and C) shows CD3, and CD4 positive tumour cells respectively [immunostain, 400×]. (D) Ki-67 proliferative index is 70%-80% [immunostain, 400×]
Post-transplant primary central nervous system lymphoma (PT-PCNSL). (A) Shows scattered loose clusters of atypical lymphoid infiltrates [H&E, 400×]. (B) CD20 shows diffuse strong membrane positivity [immunostain, 400×]. (C) Shows a Ki-67 proliferative index of 40%-45% [immunostain, 200×]. (D) The neoplastic cells are positive for Epstein–Barr encoding region [EBER] on in situ hybridisation studies, [EBER ISH, 200×]
DLBCL of CNS (A) Sheets of intermediate to large mononuclear cells [H&E, 400×]. (B) CD20 shows diffuse membrane positivity [immunostain, 400×]. (C) Aberrant Cyclin D1 expression [immunostain, 400×]. (D-F) DLBCL of CNS, a non-GCB subtype with (D) tumour cells in sheeted pattern [H&E, 400×], (E) CD20 shows strong membrane positivity [immunostain, 400×], and (F) shows the strong nuclear expression of MUM-1 [immunostain, 400×] respectively. (G) DLBCL of CNS, GCB subtype, shows the characteristic angiocentric pattern of infiltration, [H&E, 400×] with (H) strong CD20 membrane positivity, [immunostain, 400×] and (I) Ki-67 proliferative index of 70%-80%, respectively
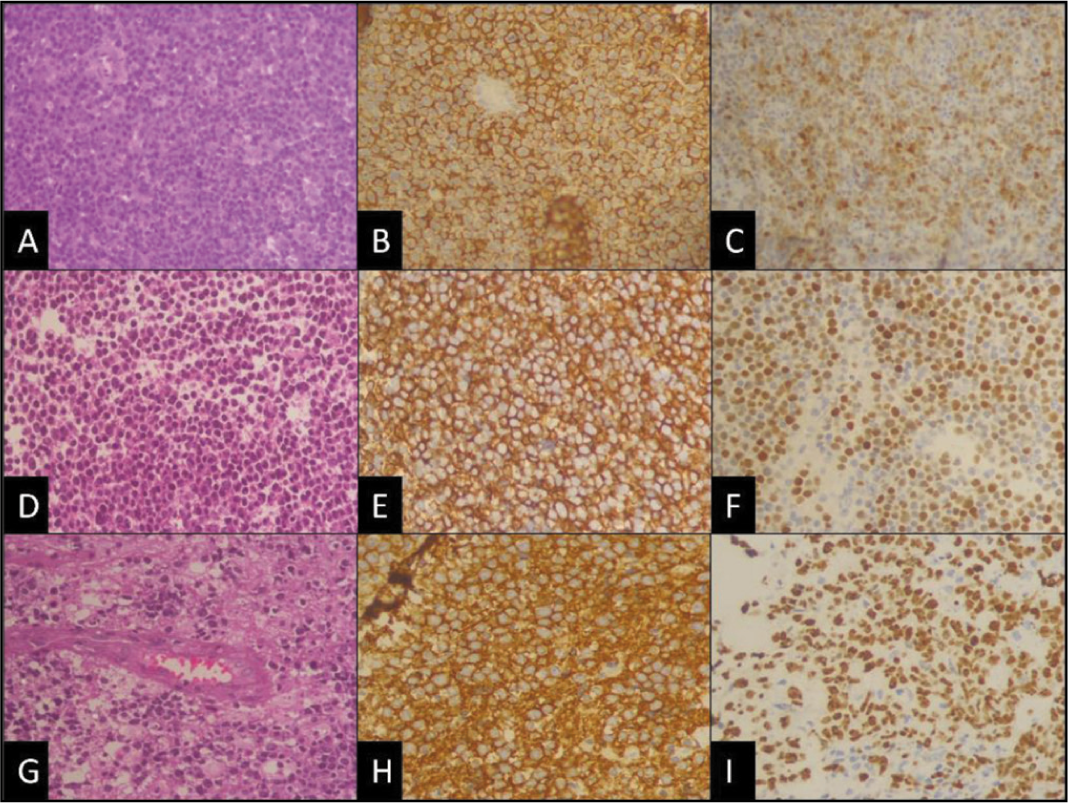
In a few DLBCL cases where sufficient additional markers were done, we were able to subclassify them into non-Germinal Centre B-cell phenotype (non-GCB) [Figure 3D-3F] or GCB [Figure 3G-3I] subgroups, though diagnostically, they are not important.
For the classification of DLBCL into two immunophenotypic subgroups, the Hans algorithm was used in this study. The Hans algorithm uses the expression pattern of CD10, BCL-6, and MUM-1 immunostain as given below [Flowchart 1] to classify DLBCL into GCB and non-GCB subtypes.
The Hans algorithm for the classification of diffuse large B-cell lymphoma
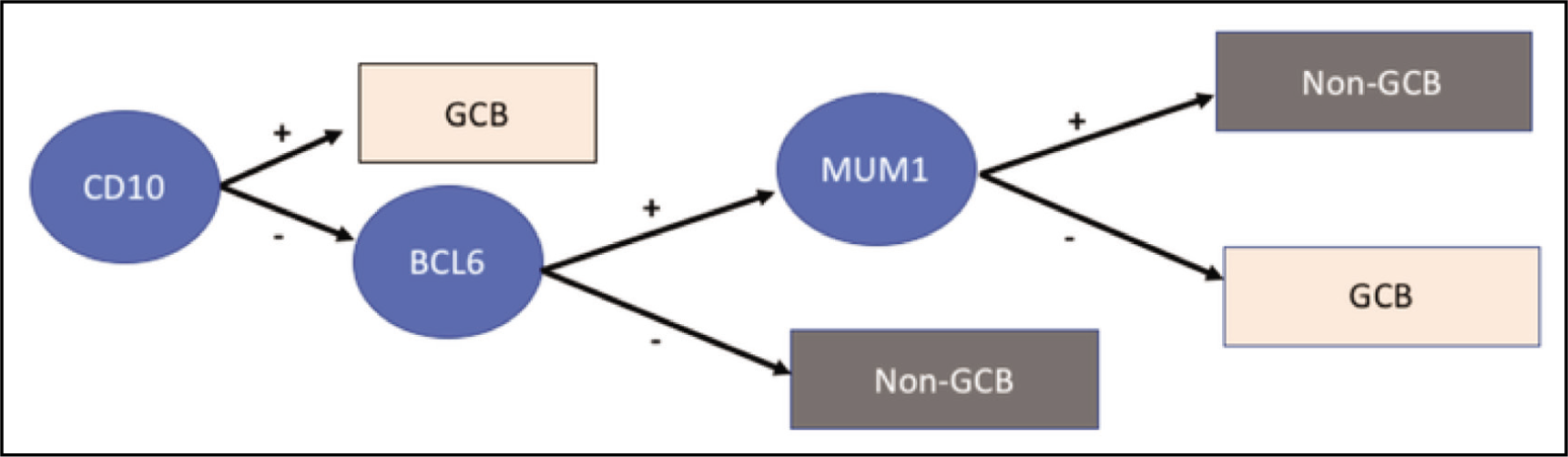
Positive expression of CD10, BCL-6, and MUM-1 was considered when >30% of tumour cells stained positive for CD10, BC6, and MUM-1, respectively. Membrane and cytoplasmic staining for CD10 and diffuse or granular nuclear staining in the case of BCL-6 and MUM-1 were considered positive.
(A & B) Post-contrast MRI of one of the central nervous system lymphomas showed enhancing lesions in the left temporoparietal and right posterior parietal cortices, radiologically resembling glioblastoma. FLAIR (Fluid attenuated inversion recovery) showed significant perilesional oedema
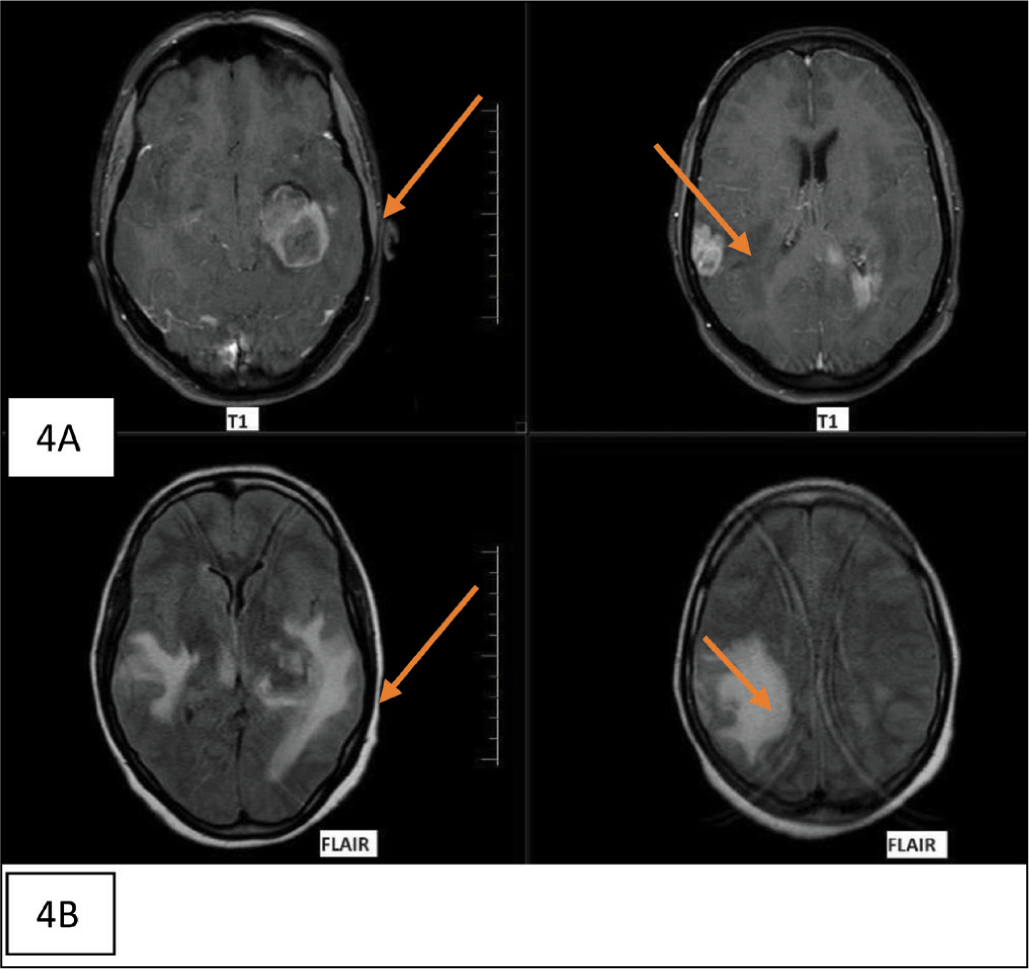
Discussion
This study was done to observe the histopathological and immunohistochemical profile of CNS lymphoma seen in our tertiary care centre. CNS lymphoma can occur in all age groups. However, the peak incidence of occurrence inferred from our study was between the 5th and 6th decade, which was similar to studies done by Song KW et al.,[10] Ambroise, M. Moses et al.[11]; however, a decade young when compared with Western literature, Bataille et al.,[12] Raoux et al.[13] This can be attributed to the difference in age structure in India (only approximately 10% of the population is older than 60) compared to Western and other Asian countries. Our study observed a slight male preponderance identical to studies done by Patel et al.,[5] Powari et al.,[6] Sarkar et al.,[7] [Table 1].
Comparison of clinicopathological parameters of CNS lymphoma among different studies
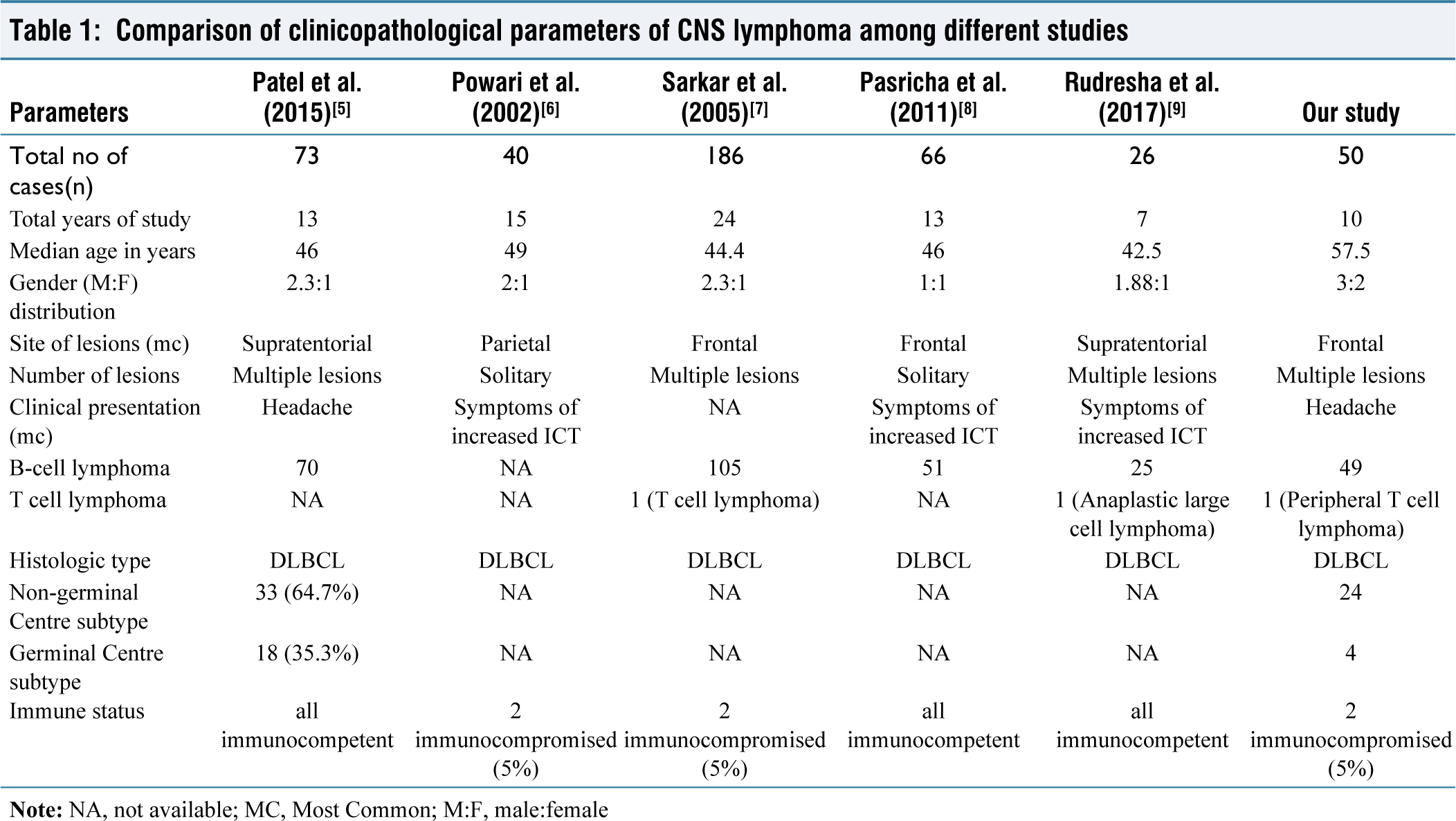
Clinical presentation depends upon the site of involvement in the CNS. In our study, the most common presentation was headache (45%), followed by seizures (25%), focal neurological deficient (18%) neuropsychiatric symptoms like sleep and memory disturbance (12%), which harmonise with studies done by Powari et al.,[6] Pasricha et al.,[8] and Rudresha et al.[9] [Table 1] where signs of increased intracranial tension namely headache and vomiting were the most common presentation.
It was also discovered that the commonest site of involvement was the frontal lobe with multiple areas of brain involvement, which is further supported by the research done by Sarkar et al.[7] Pasricha et al.[8] [Table 1].
In neuroradiologic studies, CNS lymphoma predominantly manifests as either a single mass or multiple homogeneously enhancing lesions surrounded by oedema.
Our study had 50 cases of diagnosed CNS lymphoma, mostly B-cell lymphoma (98%), in which DLBCL (96%) was the most common histological type and subtype categorised, respectively. This conclusion was identical to analyses done by Patel et al.,[5] Sarkar et al.,[7] Pasricha et al.,[8] and Rudresha et al.[9] in their respective studies [Table 1], where they concluded DLBCL was the most common CNS lymphoma.
Antibodies clones with their corresponding company were used in our study
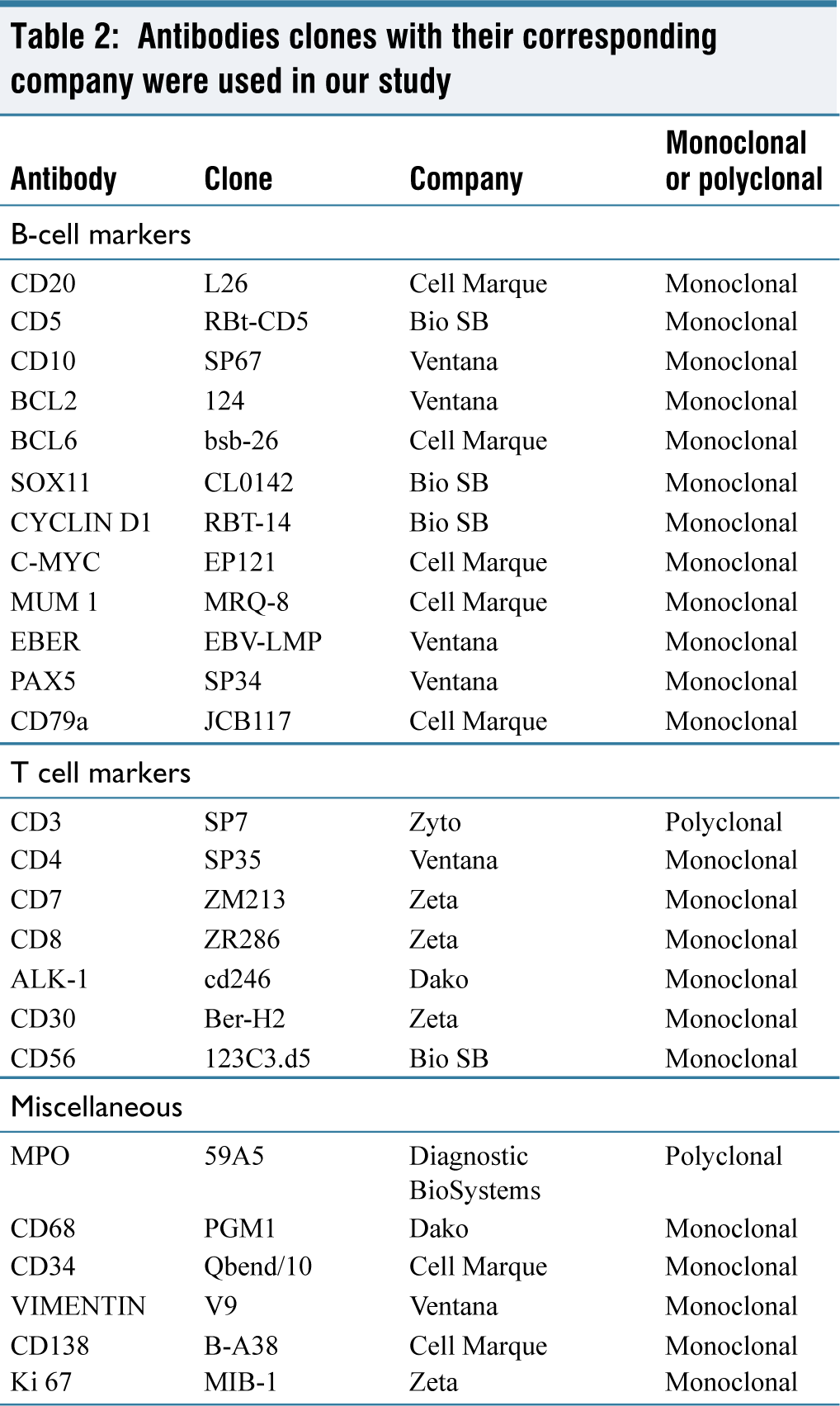
In the literature, several algorithms, Hans et al.,[14] Choi et al.,[15] Meyer et al.,[16] have been used to subdivide DLBCL. However, the most widely used algorithm is Hans [Flowchart 1], which was also used in our study further to subdivide DLBCL into non-GCB and GCB. In our study of the cases in which IHC was done, the non-germinal centre subtype was found to be the most common, which concurs with the other research done by Bhagavathi et al.,[17] Aki et al.,[18] Hattab et al.,[19] Camilleri-Broet et al.,[20] Cheng et al.,[21] and Levy et al.[22]
Few studies analysed the prognostic significance of PCNSL with different immunostains, one such study was done by Lin et al.,[23] in which they concluded that Expression of BCL-6 represented a favourable prognostic marker for patients with PCNSL and Bhagavathi et al.,[17] concluded that PCNS DLBCL shows predominantly homogenous Activated B-cell (ABC) immunophenotype with high Bcl-2 positivity and MIB-1 index which could be associated with a worse prognosis in PCNS DLBCL. There is limited literature available to state the prognostic significance of different immunostains and overall survival post-treatment for CNS lymphoma. Hence, a more elaborate study has to be done.
All our patients were immunocompetent except for two of which one was seropositive and the other case was post-renal-transplant. Despite several limitations related to this retrospective study design, one among these includes the non-availability of serology status in every patient, as a result in which the incidence of CNS lymphoma among immunocompromised versus immunocompetent could not be studied. The overall survival rate, the difference between primary or secondary CNS lymphoma, could not be summarised due to a lack of detailed information.
When it comes to the method of diagnosis, histology is still the gold standard for diagnosis. However, biopsy itself presents certain limitations, including the risks associated with the procedure and difficulties in diagnosing the tumour from typically small fragments of tissue. Stereotactic biopsy is the most effective method for obtaining tissue because lesions frequently involve deep and critical structures. Unlike malignant gliomas, gross total resection has no therapeutic benefit and does not affect disease control or survival. It is also widely known that giving corticosteroids before biopsy can result in tissue necrosis and/or rapid apoptosis, which can lead to non-diagnostic biopsies. Despite being more readily accessible, dural-based lesions can be challenging to diagnose because of crushed artefacts brought on by dense fibrous tissue.
Other brain tumours that histologically mimic 24 CNS lymphoma include metastatic carcinoma, diffuse glioma, germinoma, inflammatory myofibroblastic tumours, vasculitis, and infections. Hence, detailed evaluation is crucial for tissue biopsy, using a wide range of immunohistochemical markers (augmented, if possible, by molecular diagnostics, fluorescent in situ hybridisation [FISH] studies, and cytogenetics) and review by experienced pathologists for standard diagnosis and proper treatment.[24]
Conclusion
The study concludes that all diagnosed CNS lymphoma patients, regardless of age, present with a high disease burden. Hence, early diagnosis and timely treatment could improve the overall outcome.
Footnotes
Acknowledgements
To Dr Shivashankari-research assistant, and Mr Logesh-Biostatistician, Research department, Apollo Hospital Chennai.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Institutional ethical committee approval has been obtained [ASH-C-S-042/12-23].
Informed consent
Informed consent has been obtained from all the patients who met the eligibility criteria.
Credit author statement
LM Participated in data acquisition, literature search, investigations, data analysis and manuscript preparation. SLN Participated in conceptualization, investigation, supervision, data analysis, and manuscript editing.
Both the authors reviewed and approved the manuscript.
Data availability
Data presenting the study’s findings is available with the corresponding author.
Use of artificial intelligence
The use of artificial intelligence is not relevant to this article, as it does not involve AI technologies or methodologies in its analysis or conclusions.
