Abstract
Introduction:
Acute coronary syndrome (ACS) should be identified from various cardiac and noncardiac causes of chest pain. Non-ST segment elevation (NSTE-ACS) diagnosis can be done immediately in the case of typical changes in the electrocardiogram (ECG) and/or increased levels of myocardial markers, the absence of such variation does not always exclude NSTE-ACS. NSTE-ACS in its early stages difficult to diagnose. To stratify the risk of chest pain, aid early diagnosis, and prediction of outcome HEART score was designed. Components of the HEART score are history, ECG, age, risk factors, and troponin. The objectives of the study are risk stratification of chest pain in ED, prediction of MACE, validate the HEART Score in risk stratification of chest pain, and deciding on the discharge of patients.
Materials and methods:
This was a hospital-based, prospective study involving 220 patients who were admitted and discharged according to risk stratification using the HEART score. Patients are followed up over six weeks for prediction of MACE. MACE includes AMI, PCI, CABG, or death. The collected data were analysed with SPSS version 26.0, IBM Corp, Chicago, Illinois, USA. P value ≤.05 is considered as significant.
Results:
Out of 220 patients, 51 reached MACE. Low-risk HEART scores were seen in 25% of patients and No MACE occurred in these patients. Intermediate risk HEART scores were seen in 53% of patients and MACE occurred in 9.3% of patients. High-risk HEART scores were seen in 22% of patients and MACE occurred in 83% of patients.
Conclusion:
HEART score is helpful in risk stratification of chest pain in our study. It also aids in early diagnosis of intermediate and high-risk groups. HEART score also helps in decision-making to discharge patients. Thus, HEART score provides a quick and reliable prediction of outcomes in chest pain and should be used in ED.
Introduction
Chest pain is a leading cause of emergency room visits globally, and cardiovascular diseases (CVDs) are the primary cause of death worldwide. In 2016, CVDs were responsible for 31% of all global deaths, amounting to approximately 17.9 million fatalities, predominantly due to heart attacks and strokes.[1] This issue is particularly acute in India, where the age-standardised CVD death rate of 272 per 100,000 population as of April 2016 exceeds the global average of 235 per 100,000.[2]
In emergency departments (EDs), distinguishing acute coronary syndrome (ACS) from other causes of chest pain presents a significant challenge. The potential causes are diverse, encompassing cardiac issues like angina and myocardial infarction, vascular conditions such as aortic dissection, pulmonary diseases, gastrointestinal disorders, musculoskeletal problems, infectious diseases, and psychological conditions.[3-6]
Diagnosing non-ST-elevation ACS (nSTE-ACS), previously known as ‘unstable angina’, is particularly complex. While typical changes in the electrocardiogram (ECG) and elevated myocardial markers can immediately suggest nSTE-ACS, their absence does not necessarily rule it out. This complexity can lead to misdiagnoses, resulting in inappropriate patient discharge and potentially severe consequences, including sudden cardiac death or progression to myocardial infarction, underscoring the need for rapid and accurate diagnosis.[7]
Traditional diagnostic approaches, including physical examination, history taking, and reference standards, often fall short of accurately diagnosing ACS. A normal ECG reading cannot exclude unstable angina or NSTEMI, and initial ECGs fail to diagnose acute myocardial infarction in 20%-50% of cases.[8] Additionally, two-thirds of ischaemic episodes are clinically silent and therefore likely to be missed by ECGs.[9] Troponin I and T tests also have their limitations, as they typically do not show increased levels until 6-12 hours after symptom onset.[8]
Given these challenges, there has been a drive to develop more effective predictive tools. These include logistic regression models,[10-12] computer-derived protocols,[13,14] and artificial neural networks.[15] However, their clinical adoption has been limited due to complexity and the lack of inclusion of cardiac troponins in most models. While established scoring systems like TIMI, GRACE, FRISC, and PURSUIT have been effective in prognosticating acute coronary events, they do not adequately address the need for early risk stratification of chest pain in ED patients.
Addressing this gap, Six et al.[7] introduced the HEART Score in 2008 in the Netherlands. This method provides a rapid and resource-efficient way to stratify risk, taking into account History, ECG findings, Age, Risk factors, and Troponin levels at the patient’s initial presentation. This score has been validated through prospective studies. The current study leverages the HEART Score to stratify the risk of chest pain in the ED, predict major adverse cardiac events, validate its effectiveness in this setting, and inform decisions regarding patient discharge.
Materials and Methods
After obtaining institutional ethics clearance, a hospital-based, prospective study was performed from January 2018 to August 2019 with patients presenting with chest pain and admitted to the Emergency Department of S.S. Institute of Medical Sciences and Research Centre Hospital, Davangere. Patients with chest pain who are admitted, and discharged according to risk stratification HEART score are followed up over six weeks for prediction of major adverse cardiac events. Follow-up with patients using digital devices and written patient records, including discharge letters, revascularisation reports, and any other relevant documentation.
Patients who were presented and admitted to the emergency room due to chest pain aged >18 years were included in the study. Patients with ST-elevation acute myocardial infarction (STEMI) were excluded from the study. A total of 220 patients were admitted and discharged according to risk stratification using the HEART score. Patients are followed up over six weeks for prediction of MACE [AMI, PCI, CABG, or death].
The data was collected in the master chart obtained in the Microsoft Excel format. The collected data were analysed with IBM.SPSS statistics software 25.0 Version, IBM Corp, Chicago, Illinois, USA. To provide insight into the data, descriptive statistics such as frequency and percentage analysis were utilised for categorical variables, while mean and standard deviation were employed for continuous variables. Sensitivity, specificity, PPV and NPV, as well as diagnostic accuracy using the Receiver Operating Curve (ROC), were utilised to assess the efficacy of the HEART Score. In the aforementioned statistical tool, a probability value of 0.05 is deemed significant.
Results
Out of 220 patients studied, 143 (65%) were males and 77 (33%) were females. The minimum age of the patients included in the study was 18 years and the maximum age was 85 years, the range was 67 and the mean age of the study subjects was 47.97 years. The characteristics of study participants are tabulated in Table 1. Of the 220 subjects, 56 had a HEART Score from 0 to 3, accounting for 25%, 116 had a score from 4 to 6, accounting for 53%, and, 48 had a score from 7 to 10, accounting for 22% as mentioned in Table 2. The consolidated clinical characteristics and outcomes of the study participants are tabulated in Table 3. HEART score analysis & predictive value and the admission data about HEART score are tabulated in Table 4.
Combined gender and demographic data

Comprehensive age-related data

Consolidated clinical characteristics and outcomes
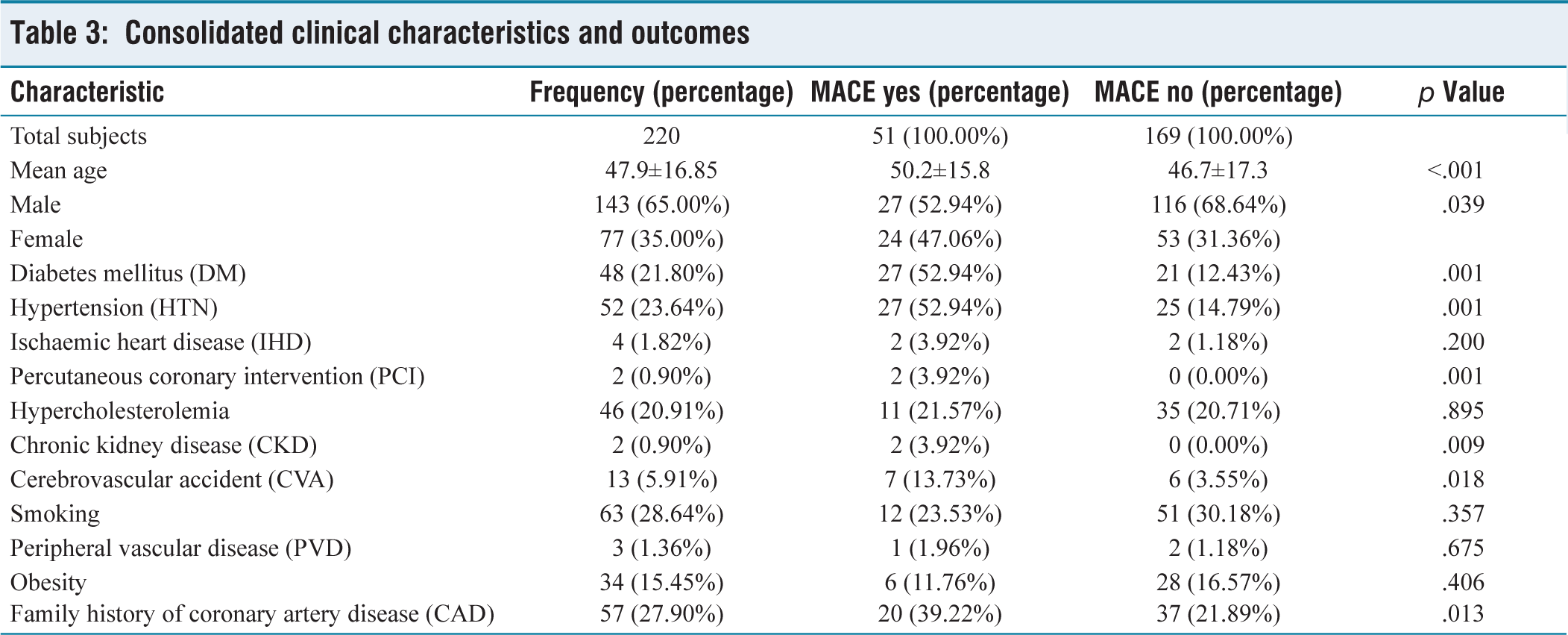
HEART score analysis and predictive value
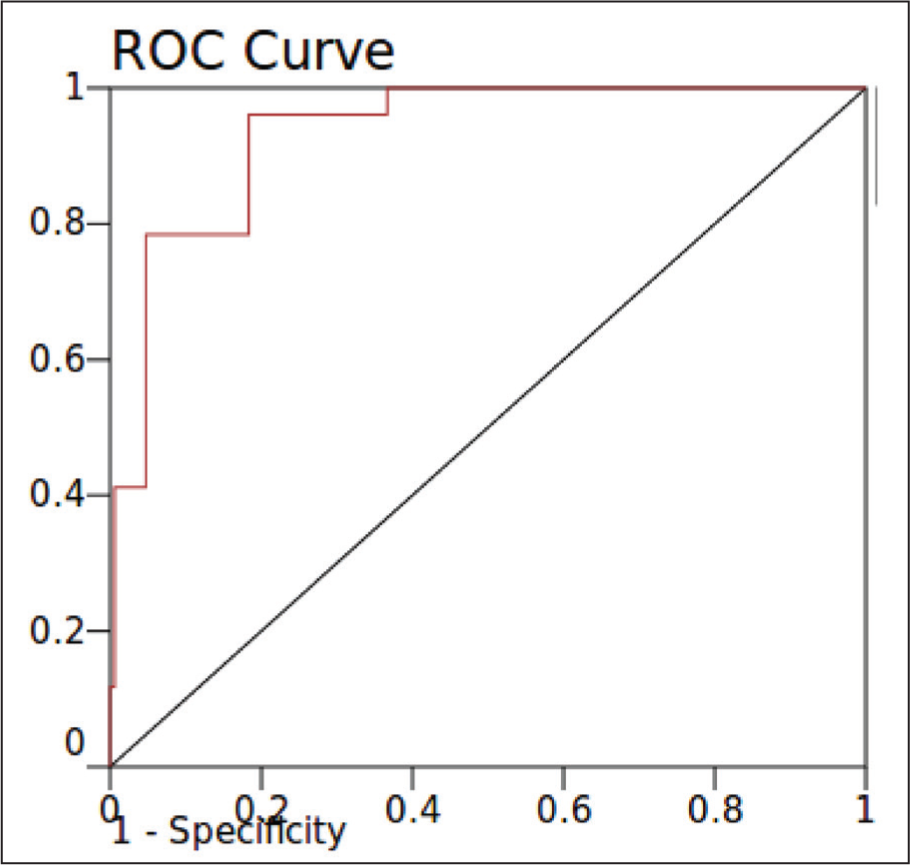
The area under the ROC curve is 0.96 and the standard error is 0.01, determined with a confidence interval of 95% (p value = .005), which is shown in Table 5 and Figure 1. Under the nonparametric assumption with a null hypothesis stating a true area of 0.5, the obtained p value is highly significant at p ≤ .01. The study’s collected data, employing a HEART Score cutoff of 6 to differentiate between low and high scores, revealed a specificity of 82% and a sensitivity of 96%. Among the 220 subjects examined, 56 exhibited low HEART scores (0–3), 116 had intermediate scores (4–6), and 48 had high scores (7–10). Notably, all 48 subjects with high scores experienced Major Adverse Cardiac Events (MACE) within a six-week observation period. In contrast, among the 56 subjects with low scores, none experienced MACE. Of the 116 subjects with intermediate scores, 11 experienced MACE, while 105 did not. This distribution corresponds to a sensitivity of 78% and a specificity of 95% for the high score category, with a positive predictive value (PPV) of 83% and a negative predictive value (NPV) of 93%. Therefore, using the HEART score categories, the predictive capacity for MACE is 83% for positive cases in the high-risk group and 93% for negative cases in the low-risk group.
Area under the curve (HEART score)

ROC curve
Discussion
ACS, which includes Unstable Angina (UA), Non-ST Segment Elevation Myocardial Infarction (NSTEMI), and STEMI, is a prevalent cause of chest pain encountered in medical practice. The diagnostic challenge lies in the early phase of Non-ST Segment ACS (NSTACS), where ECG and cardiac biomarkers may not show definitive signs of ischaemia, necessitating serial ECGs and leading to precautionary hospital admissions based on clinical judgement. This approach, while aimed at reducing the risk of missing an ongoing coronary event, has led to the observation that a significant number of patients subjected to coronary angiograms do not demonstrate significant coronary arterial disease, thus inflating healthcare costs and resource utilisation.[16]
The development of the HEART score was motivated by the need for a reliable tool to stratify chest pain patients in the emergency room (ER) into low, intermediate, and high-risk categories for adverse cardiac events. Our study of 220 patients with chest pain sought to evaluate the HEART score’s effectiveness and compare our findings with existing literature, particularly the study by Six et al.,[7] which also assessed the HEART score’s value in managing chest pain in the ER. In our cohort, the distribution of genders and the prevalence of MACE were similar to those reported by Six et al.,[7] indicating a consistent pattern of ACS presentation and outcomes across different populations.
In our analysis, we identified several patient characteristics as significant predictors of MACE, including Diabetes Mellitus (DM), Hypertension (HTN), history of Percutaneous Coronary Intervention (PCI), Chronic Kidney Disease (CKD), Cerebrovascular Accident (CVA) history, and a family history of Coronary Artery Disease (CAD), each demonstrating a varying degree of risk association with MACE (p ≤ .00001 for DM and HTN, p ≤ .0097 for PCI, p ≤ .009 for CKD, p ≤ .0181 for CVA, and .0133 for family history of CAD). These findings highlight the multifactorial nature of ACS risk, suggesting that a comprehensive evaluation of patient history and comorbidities is crucial in the risk stratification process.
Gender-based analysis within our study revealed that males had a higher incidence of MACE compared to females, though the percentage of females experiencing MACE was greater due to their lower representation in the study population. This observation aligns with the study by Bank et al.,[17] which explored sex-based differences in the performance of the HEART score, underscoring the importance of considering gender in the assessment of ACS risk.
Age was another critical factor influencing MACE risk, with our findings indicating an increased incidence of MACE among older patients. This age-related risk increment is consistent with previous literature, including the study by Six et al.,[7] reinforcing the need to consider age as a significant determinant in the evaluation of chest pain and potential ACS.
The HEART score’s predictive value for MACE was underscored by our findings, where patients with higher HEART scores exhibited a significantly increased risk of MACE. This stratification capability facilitates targeted management strategies, allowing clinicians to tailor their approach based on the patient’s risk category. Importantly, our study suggested that patients with low HEART scores could potentially be safely discharged early, reducing unnecessary hospital admissions and optimising healthcare resource use.
The specificity and predictive values of the HEART score across different risk thresholds further validated its utility in clinical practice. For instance, the high sensitivity and specificity of the HEART score in predicting MACE among high-risk patients highlight its role in identifying individuals who would benefit from more aggressive management or emergency invasive procedures.
Our study, while reinforcing the value of the HEART score in stratifying chest pain patients in the ER, also contributes to the ongoing discussion regarding the optimisation of healthcare resources in the management of ACS. By comparing our results with those from studies such as those by Six et al.[7] and Six et al.,[18] underscore the HEART score’s robustness as a risk assessment tool, capable of guiding clinical decision-making processes in the acute setting.
In light of our findings and the corroborative evidence from the literature, it becomes evident that the HEART score serves not only as a diagnostic aid but also as a strategic tool in the management of chest pain, potentially leading to a more efficient allocation of healthcare resources and a reduction in unnecessary diagnostic procedures. The application of the HEART score in clinical practice promises to refine patient care pathways, ensuring that individuals at genuine risk of ACS are identified promptly and managed appropriately, while those with low-risk scores are spared the anxiety, expense, and potential complications of unwarranted hospital admissions and invasive testing.
This study possesses several notable strengths. First, its prospective design allowed for systematic patient follow-up over six weeks, minimising recall bias and facilitating the collection of real-time data on MACE. Conducted in a hospital-based emergency department, it simulated real-world conditions, thereby enhancing the applicability of findings to typical clinical practice. The study employed the HEART score, a comprehensive risk stratification tool that integrates patient history, ECG, age, risk factors, and troponin levels, enabling a robust evaluation of patients presenting with chest pain. Additionally, the study validated the effectiveness of the HEART score in categorising patients into low, intermediate, and high-risk groups for MACE, supporting its use as a reliable risk assessment tool in alignment with existing literature. The inclusion of 220 patients provided a sufficiently large sample size to yield statistically significant results, increasing the reliability of the conclusions. Moreover, the detailed statistical analysis, including ROC curve analysis, sensitivity, specificity, PPV, and NPV, allowed for a comprehensive evaluation of the HEART score’s diagnostic performance.
Despite its strengths, this study has several limitations. Conducted in a single hospital, the findings’ generalizability may be limited due to differences in patient demographics, healthcare settings, and clinical practices across regions. The six-week follow-up period, while informative, is relatively short and does not provide insight into the long-term predictive capacity of the HEART score for cardiac events. Furthermore, the exclusion of patients with STEMI restricts the applicability of the results to the broader spectrum of ACS. Observer bias is another concern, as subjective elements within the HEART score, such as patient history and ECG interpretation, could have influenced outcomes. The limited ethnic diversity of the study population also affects its generalizability to other populations, given the potential variation in ACS presentation and outcomes across different ethnic groups. Additionally, the study’s focus solely on the HEART score, without comparison to other risk stratification tools like the TIMI or GRACE scores, limits the understanding of its relative effectiveness. The reliance on digital and written records for follow-up poses the risk of underreporting MACE if patients sought care elsewhere. Moreover, the study does not explore the specific interventional outcomes or the impact of HEART score stratification on clinical decision-making. Finally, the absence of adjustments for potential confounding factors, such as lifestyle habits or medication adherence, could influence patient outcomes and affect the study’s conclusions.
Conclusion
This prospective study demonstrates the utility of the HEART score in risk stratification of chest pain in the emergency department. The HEART score, incorporating History, ECG findings, Age, Risk factors, and Troponin levels, proved to be a reliable tool for predicting MACE in patients presenting with chest pain. Out of 220 patients, those with high HEART scores showed a significantly higher incidence of MACE compared to those with low or intermediate scores. The study indicates that a HEART score higher than 6 is a strong predictor of MACE, suggesting the need for aggressive management and potential hospital admission. Conversely, patients with a score lower than 6, particularly in the low-risk category (0-3), had a negligible incidence of MACE, implying that they might be suitable for early discharge with less intensive follow-up. These findings support the implementation of the HEART score in emergency departments for efficient and effective management of patients with chest pain, optimising patient outcomes while potentially reducing unnecessary hospital admissions and healthcare resource utilisation.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
S.S Institute of Medical Sciences and Research Centre Hospital, Davangere – IERB no. 166-2017 dated 22.11.2017.
Informed consent
Obtained from all the participants in the study.
Credit author statement
Conceptualisation - RR
Data collection and analysis - RR, AKCS
Manuscript writing - AKCS, SNR
Manuscript revision - RR, SNR
Figures acquisition - AKCS
Supervision - RR
Data availability
Data is included in the manuscript.
Use of artificial intelligence
The use of artificial intelligence is not relevant to this article, as it does not involve AI technologies or methodologies in its analysis or conclusions.
