Abstract
Background and Aims:
Clinical trials are gold standard for research and evaluating the efficacy and safety of healthcare interventions. Teaching about clinical trials is part of the pharmacology curriculum for undergraduate medical students. The aim of this study is to evaluate the knowledge, attitude and perception (KAP) of undergraduate medical students towards clinical trial.
Methodology:
An observational cross-sectional study was conducted at a medical college of Ahmedabad, using standardised self-administered questionnaires having total 25 questions regarding KAP, prepared by the authors. The pre- and post-KAP questionnaires (after educational intervention) were offered to the participants after taking an informed consent. Analysis was done using SPSS software, version 26. P value <.05 was considered as significant.
Results:
We surveyed 212 medical students; there was an improvement in the overall scores of knowledge domain from 79.67 to 92.19%, about 31.44% improvement in the attitude toward clinical trial and 78.17-91.12% improvement in the perception. There was improvement in median score of 84.76% in having ‘sufficient’ knowledge, 91.18% in having ‘positive’ attitude and 91.63% of respondents having ‘favourable’ perception, regarding clinical trials.
Conclusion:
The results of this study depict the intervention has enhanced KAP regarding clinical trial among the medical students. The medical undergraduates are future innovators, clinicians and scientific explorers. It would be better if they are trained at earlier days of learning about clinical trials/research and medical ethics to ensure that future medical students receive better training in understanding and conducting clinical trials.
Keywords
Introduction
Clinical trials of drugs are prospective ethically designed investigation in human subjects to objectively discover, verify or compare the results of two or more therapeutic measures.[1] They are designed to acquire information about the pharmacokinetic and pharmacodynamic properties of a candidate drug.[2] Information from clinical trial helps patients, clinicians, policymakers and funding agencies to make clinical and health policy decisions.[3,4]
Clinical trials contribute significantly to healthcare improvement and are conducted according to good clinical practice (GCP) standards. Most medical institutes participate, often with doctors as principal investigators. Teaching informed consent and trial procedures is part of the pharmacology curriculum for undergraduate medical students.[5] Encouraging research-specific skills, including communication, teamwork and critical thinking, among these students is recommended by various authorities. In the past five years, clinical research in India has seen significant growth due to its robust medical infrastructure, the availability of diverse and treatment-naive patient populations, increasing awareness of GCP among investigators and cost-effective operations.[6] India aims to become an attractive destination for clinical research. To achieve this, emerging contract research organizations (CROs) must uphold high ethical standards and adhere to GCP guidelines to support this effort.[7] The clinical development of drugs through clinical trials is essential for healthcare progress, with the goal of benefiting patients and society.[8] However, trials carry inherent risks and require careful interpretation of results, which can be misunderstood by the public. Recent ethical concerns have complicated matters, ensuring that clinical trial subjects are not treated as ‘guinea pigs’ and recognising the importance of pharmaceutical industry involvement can be challenging for the public to grasp.[9]
Within the medical community, doubts about trial integrity and usefulness have arisen, which are concerning, especially in India’s scientific context. Over the past 25 years, there has been a decline in medical graduates pursuing clinician–scientist careers, making it important to promote research-integrated analytical skills among medical students.[10-14] Evidence-based medicine plays a significant role in medical practice and its advancement. Hence, every clinician should cultivate adequate knowledge and develop right attitude for clinical research and trials. The medical students in medical colleges are the cornerstone of next-generation medical fraternity. Hence, they also should have a basic idea about clinical trial to improve the existing medical education system and thus to foster research culture.[15] However, data regarding the knowledge and perception of clinical trial among doctors and medical students are scanty, thus it promotes the need for conduct of more such studies. Medical students should understand clinical research as it offers a strong career path. Some may choose careers in clinical trials, making it crucial to prepare future investigators.[16]
This study aims to evaluate the knowledge, attitude and perception (KAP) of undergraduate medical students towards clinical trials before and after an educational intervention in a medical college of Ahmedabad, India.
Materials and Methods
Study Design
This study was conducted at a Medical College in Ahmedabad, India. Institutional review board approval was obtained prior to the initiation of the study. This was an observational cross-sectional, questionnaire-based study, focusing on the KAP regarding clinical trials. Third term MBBS students (n = 212) were selected as the study participants.
Questionnaire
To prepare for the study, the questionnaire used in the study was developed with minor modifications from similar previous studies.[16-22] Experts who actively participate in clinical trials analysed and validated the questionnaire. Before the main study, a pilot study was conducted among 20 participants to assess the questionnaire’s readability, comprehensiveness and understanding. The reliability of the questionnaire was assessed by Cronbach’s alfa (α=0.741) and Kuder–Richardson (KR20) coefficient (0.732) for binary responses. This ensured that the study protocol was well-prepared and ready for implementation.
All students were provided with an explanation regarding the study’s nature well in advance. The QR code for pre-KAP Google Form questionnaire was shared to the participants through WhatsApp. Only those who had given online consent were granted access to the questionnaire and were allowed to fill the questionnaire. The pre-KAP questionnaire consisted of 24 questions related to clinical trials. Out of these, 15 questions were designed to assess knowledge, 5 questions to evaluate attitude and 4 questions for perception. An intervention in the form of a lecture, presented using a PowerPoint presentation by deserving faculty, was delivered to all students to convey key concepts about clinical trials. Following the lecture, all students were requested to respond to the post-KAP questionnaire for which another QR code was shared, which also contained the same 24 questions, to check the impact of the intervention.
Analysis
The collected information was recorded, kept confidential and subsequently analysed. Likert scale was employed to evaluate attitude-based questions, and for ease of analysis, responses were categorised as agree/neutral/disagree. Knowledge and perception questions were presented with options of yes/no, and responses were then categorised as correct or incorrect, +1 for correct response and –1 for incorrect response. More than 50% of improvement in the score in each domain was considered as sufficient knowledge, positive attitude and favourable perception. The complete data was entered in Microsoft Excel 2021, paired t-test was applied and was analysed using SPSS software version 26. The P value < .05 was considered statistically significant.
Results
In a survey of 212 participants who were in their third year of medical school (MBBS), various questions were posed to assess their knowledge of clinical trials. The researchers analysed the responses both before and after participants received education intervention and used a statistical test to determine the significance of the changes, indicated by a P value < .05.
Prior to the intervention, we observed 28.8% (61) respondents knew the necessity of reporting serious adverse events affecting clinical trial participants within 14 days to the Drug Controller General of India (DCGI), Post-intervention, 97.6% (207) respondents recognised the importance of this reporting. There was a substantial improvement regarding Phase I clinical studies: 94.5% (200) respondents believed that the majority of these studies are structured as double-blind, randomised controlled trials which after the intervention increased to 98.6% (209). Regarding the use of blinding as a technique in clinical studies to prevent bias, there was a modest increase from 88.4 (187) to 91.1% (193). An improvement in awareness was noted in understanding that clinical trials typically involve two groups for comparison that one receiving the test drug and the other a placebo this increased from 91.8 (195) to 96.2%(204). Participants’ understanding of the need for approval from the DCGI for novel medications also saw improvement, rising from 76.7 (162) to 82.8% (176). Knowledge regarding involvement of a diverse group of participants showed improvement from 91.2 (193) to 97.6% (207). The importance of informed consent in clinical trials showed recognition increasing from 26.1 (55) to 85.7%(177). Similarly, regarding the informed consent form should include the contact details of the principal investigator and the ethics committee improved significantly from 88.7 (188) to 94.4% (200). Understanding of the inclusion of treatment devices in clinical trials rose significantly from 82.1 (174) to 97.6% (207). Recognition of Phase IV clinical trials as post-marketing surveillance increased from 82.6 (175) to 93.8% (199). Awareness of reporting adverse events to multiple entities, including institutional ethics committee, regulatory authority and the sponsor, rose from 85.4 (181) to 94.3% (200). The definition of significant adverse events, including those resulting in death or long-term harm, saw an increase from 87.2 (185) to 92.4% (196). Understanding that a Phase-I trial testing a new drug for cancer may include a cancer patient as a participant significantly rose from 54.3 (115) to 98.1% (208). Investigational new drug definition showed improvement from 85.6 (182) to 91.3% (193). Understanding of participants right to leave the trial before its completion showed improvement to 92.2 (195) from 88.1% (187) (Table 1).
Knowledge of clinical trials before and after educational intervention – ‘YES’ response

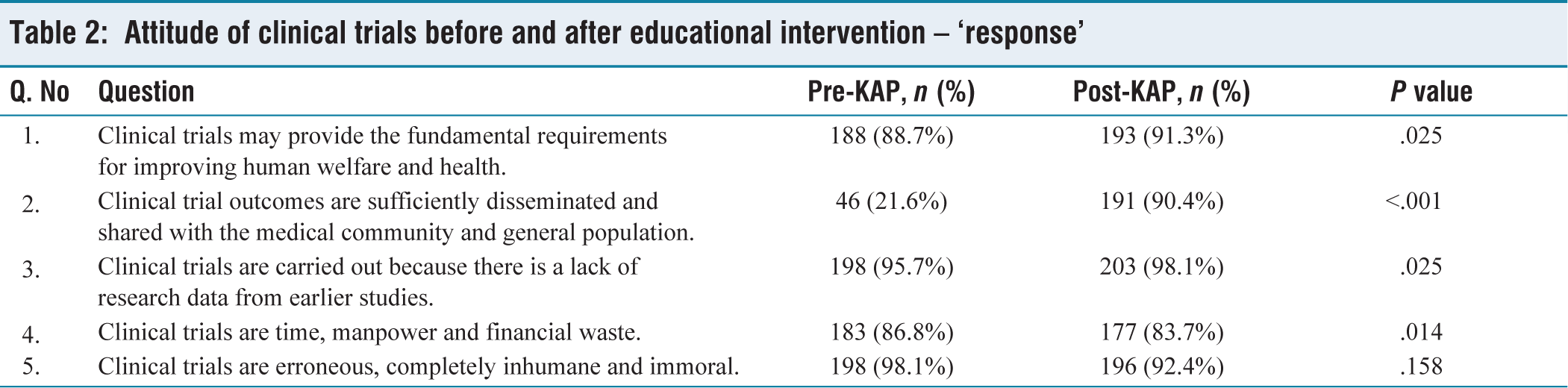
Initially, 88.7% (188) respondents agreed that clinical trials contribute to human welfare and health; post-intervention, this rose to 91.3%(193). Pre-intervention, only 21.6% (46) thought clinical trial outcomes were sufficiently disseminated; post-intervention, this belief significantly increased to 90.4% (191). Initially, 95.7% (198) believed clinical trials are carried out due to lack of research data, increasing to 98.1% (203). Pre-intervention, 86.8% (183) viewed clinical trials as a waste of time, manpower and finances; this decreased to 83.7% (177). Belief in clinical trials being erroneous and inhumane decreased from 98.1 (198) to 92.4% (196) (Table 2).
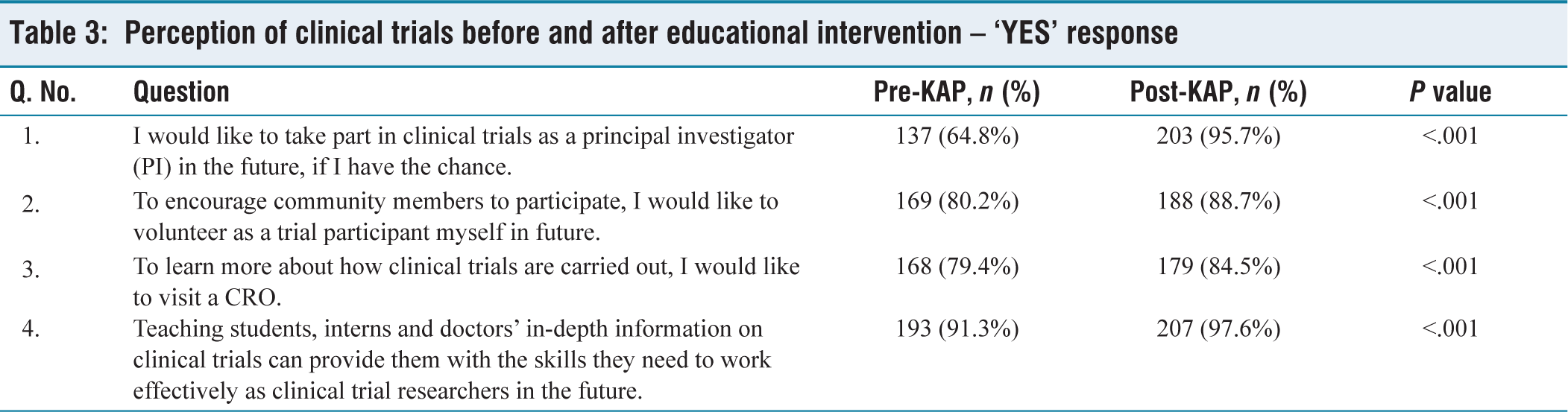
Before the knowledge intervention, 64.8% (137) respondents were interested in becoming a principal investigator in future clinical trials; this increased significantly to 95.7%(203). Willingness to volunteer as a trial participant increased from 80.2 (169) to 88.7%(188). Pre-intervention, 79.4% (168) respondents were interested in visiting a CRO, rising to 84.5% (179) post-intervention. Initially, 91.3% (193) respondents believed that it is important to teach students about clinical trials; post-intervention, this belief rose to 97.6% (207) (Table 3).
Attitude of clinical trials before and after educational intervention – ‘response’
Perception of clinical trials before and after educational intervention – ‘YES’ response
In this study, more than 50% of improvement in the median score of each domain was considered as sufficient knowledge, positive attitude and favourable perception. About 84.76% participants have ‘sufficient’ knowledge, 91.18% of responders have ‘positive’ attitude and 91.63% of responders have ‘favourable’ perception regarding clinical trials (Figure 1).
Median score of each domain: Knowledge, attitude and perception
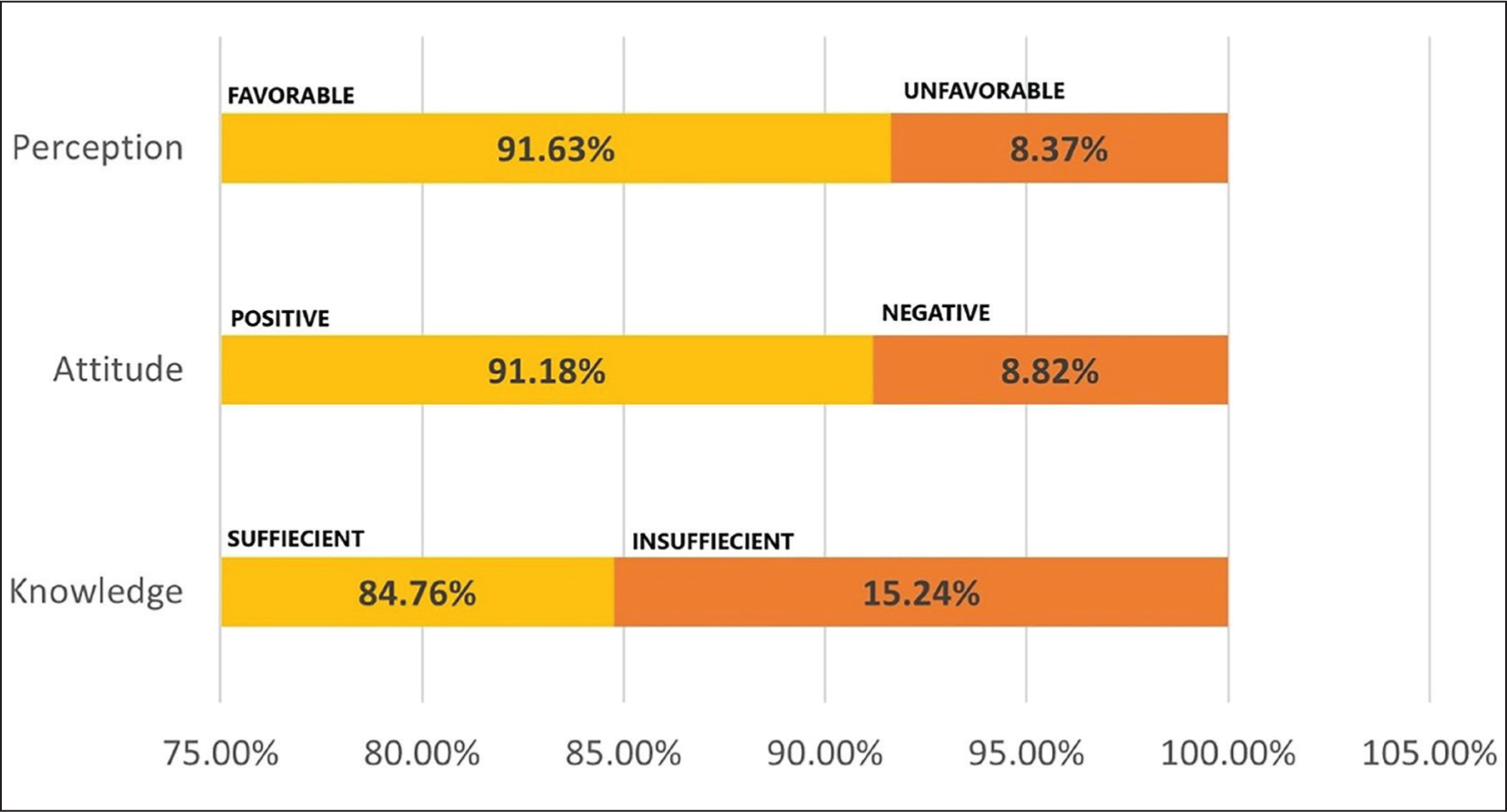
Figure 2 shows mean and standard deviation (SD) pre- and post-intervention.
Mean and standard deviation (SD) pre- and post-intervention
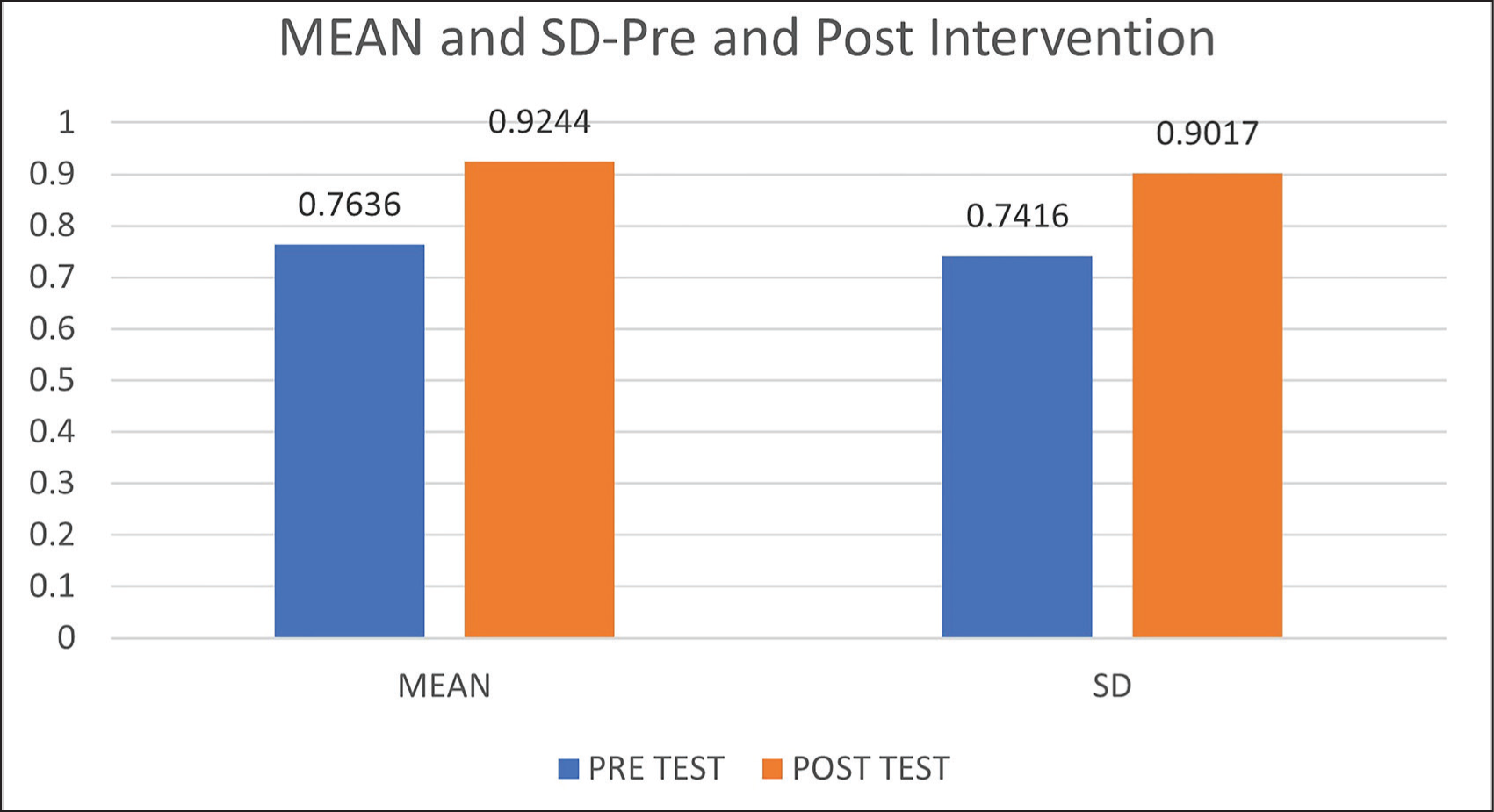
We found P value < .05 for majority of the parameters in post-KAP scores in all domains of KAP, showing positive impact of the educational intervention.
Discussion
Modern medical education and clinical practice are increasingly adopting evidence-based approaches, with a focus on clinical trials as a fundamental element of this transformation. The findings from research and trials significantly influence disease prevention, diagnosis and treatment strategies, driving substantial changes in both medical education and clinical practice.[19] The medical students in medical colleges are the cornerstone of next-generation medical fraternity, making it important to promote research-integrated analytical skills among medical students.
Exhibiting the knowledge helps the medical students in having clear idea regarding clinical trial. According to the current survey, students now understand the importance of reporting the adverse events to the DCGI, with a 97.6% comprehension rate, comparing the results to the Deodurg et al.[16] study, which had scores of 76.5%, lower than those of our study group; this contrast highlights the value of clinical trial education at the undergraduate level. The level of understanding about the purpose of blinding of participants in clinical trials to prevent bias improved to 98.6%, while Vodopivec et al.[17] had scores of 80%; the higher score indicated that our intervention was effective in elucidating the significance of this method in preserving trial integrity. We observed 96.2% improvement in knowledge about how participants were divided into two treatment groups in clinical trial which is nearly identical to the findings of the Deodurg et al.[16] study, suggesting that participants may have possessed prior awareness regarding this information. Participants showcased an enhanced appreciation for the role of clinical trials in generating robust evidence for medical decision-making, reflecting a paradigm shift towards a more evidence-based approach to healthcare. According to our research, the significance of DCGI approval for selling or importing medications has increased to 82.8%, while 90% in Deodurg et al.’s study,[16] which is somewhat similar to our findings. To include participants of different ages, genders, ethnicities, socio-economic backgrounds, healthcare professionals like doctors, interns, nurses and so on, making trial outcomes more applicable to a broader population and providing insights into varied responses to treatments. The study results indicate that, after the intervention, around 85% of undergraduate students now show awareness regarding ethics committee and informed consent after the intervention; conversely, Kiruthika et al.[20] found that participants in their study had a score of 33.8%, indicating a significantly lower understanding of concepts related to institutional ethics committees and clinical trials. This could be explained by the fact that Kiruthika et al. study included members of the nursing community also which could have led to lower scores. As evidence of the educational session’s beneficial outcomes, participants’ understanding of questions on major adverse events improved to 92.4% compared to 88.7% in the Vodopivec et al.[17] study. The educational initiative contributed to a heightened awareness of the regulatory frameworks governing clinical trials, equipping students with the knowledge necessary to navigate and adhere to established guidelines. To include a cancer patient in Phase I trial testing a cancer drug showed significant improvement of 98.1% consistent with the NIH guidelines. The knowledge score of 94.3% about reporting adverse events to the sponsor and authorities is similar to the findings of studies conducted by Vodopivec et al.[17] and Deodurg et al.[16] Approximately 92.2% of our study participants understand the right to withdraw from the trial without any consequences, as per our study, compared to Deodurg et al.[16] participants, who had an 86.9% score that was somewhat lower than ours, indicates that participants’ understanding had improved, maybe primarily a result of the educational session’s focus on participant rights and ethical considerations.
The study has been successful in changing the attitude of medical students. Our study’s results indicate that students’ attitudes towards the potential benefits of clinical trials to improve human welfare and health have improved to 91.3%; these findings are consistent with those of Makoul et al.[18]. According to our study, 90.4% of medical students have a positive attitude towards disclosing clinical trial results to the medical community and the general public; on the other hand, Mello M et al.[19] score was only about 37% when clinical trial volunteers were included in the study. The intervention instilled a sense of appreciation among students for their potential role in advancing medical knowledge through participation in clinical research, fostering a positive outlook on the impact of their future contributions. In this study, 90% of students now have a positive attitude, believing that clinical trials are conducted ethically and do not waste resources. This finding aligns with the Burt et al. study.[7] The positive change in students’ attitude toward these benefits can be linked to the specific educational intervention implemented in our study.
The recruitment and retention of participants in a trial are critical to its success, and as such, the participant’s perception of clinical trial plays a significant role in these processes. About 95.7% of our study’s post-intervention participants said they would like to be the principal investigator in the future, similar to 90.4% of participants in a study by Deodurg et al.[16] However, according to Chu et al.[21] only 25% of respondents indicated a willingness to participate in clinical trials. Students exhibited a shift in their perceived role from passive observers to active contributors in the realm of clinical trial, showcasing an emerging sense of responsibility for shaping the future of medical science. Students’ favourable perception following educational intervention demonstrated by the fact that 84.5% of them wanted to visit a CRO, compared to 85.2% in Deodurg et al’s study.[16] Students’ perception regarding teaching them about clinical trial at undergraduate level showed score of 97.6% which is consistent with the findings of Lee et al.’s[27] study report. To ignite interest in participating in clinical trials, it’s crucial to explain the technology in a way that’s easy to understand (Figure 3).
Why is research important for undergraduate students?

Table 4 shows comparative evaluation and inferences of similar studies done by various authors.
Comparative evaluation of similar studies

Strengths
The study’s design allows for a before-and-after comparison, enabling a clear assessment of the intervention’s effectiveness in improving knowledge and perceptions. By focusing on medical students, the study targets a crucial demographic that will play a vital role in future clinical research.
Limitations
The study was limited by the representativeness of the sample, as only third term MBBS from a single medical college and, which makes it difficult to directly generalise results of this study. Also, they may have limited clinical exposure, which can affect the depth of their understanding of clinical trial and their responses.
Conclusion
The results of this study depict the intervention has enhanced KAP regarding clinical trials among the medical students. The medical undergraduates are future innovators, clinicians and scientific explorers. It would be better if they are trained at earlier days of learning about clinical trial and medical ethics to ensure that future medical students receive better training in understanding and conducting clinical trials. Embracing evidence-based practices and cultivating a deep appreciation for clinical trials among the medical fraternity will ultimately lead to improved patient care and advancement in the field of medicine. The study’s findings can inform medical schools about areas in the curriculum that need improvement in terms of clinical trial education for better understanding of clinical trials. Also, the study may be extended to include resident doctors, registered medical practitioners, para-medical individuals and even the general population in the future.
Footnotes
Acknowledgements
The authors would like to thank all the third-term medical students for their enthusiasm in their participation and the resident doctors of the Department of Pharmacology.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
IRB letter attached during submission, as no specific approval number is given.
Informed consent
Only participants who provided informed consent online were granted access to and allowed to complete the questionnaire.
Credit author statement
Conceptualization: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Bhumika Bhavsar, Supriya Malhotra.
Methodology: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Bhumika Bhavsar, Supriya Malhotra.
Formal analysis: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Bhumika Bhavsar, Supriya Malhotra.
Investigation: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra,, Supriya Malhotra.
Data curation: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Supriya Malhotra.
Writing - original draft: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Supriya Malhotra.
Writing - review & editing: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Bhumika Bhavsar, Supriya Malhotra.
Visualization: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Bhumika Bhavsar, Supriya Malhotra.
Supervision: Vipul Dhanjibhai Prajapati, Supriya Malhotra.
Project administration: Vipul Dhanjibhai Prajapati, Mehta Yash Dharmendra, Supriya Malhotra.
Data availability
Data is available upon request.
Use of artificial intelligence
No artificial intelligence tools were used in the research or manuscript preparation.
