Abstract
Background and Aims:
Several opportunistic infections have increased, including cytomegalovirus (CMV) in coronavirus disease 2019 (COVID-19) patients. The literature has reported few cases, and the authors are not aware of any systematic review of CMV infection reactivation associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. This review aims to assess the incidence of CMV reactivation and its outcomes among patients diagnosed with SARS-CoV-2 infection.
Methods:
We searched several electronic literature databases for studies published between 1 November 2019 and 7 February 2022. We then selected all available English-language articles (a total of 15 case reports and case studies) that reported underlying conditions, hospitalisation courses, treatment specifics and clinical outcomes of hospitalised patients coinfected with SARS-CoV-2 and CMV viruses. Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines were followed.
Results:
Out of 1304 screened articles, this review included 15 articles, representing 34 patients. Among those, five were case series and 10 were case reports with patients’ ages ranging from 42 to 92 years. Other parameters evaluated included gender, comorbid conditions, intensive care unit (ICU) length of stay (ranging from 15 to 40.5 days), use of immunosuppressants, other treatments administered and clinical outcome (death for 19 of the 34 patients).
Conclusion:
Our findings showed that besides immune system dysregulation due to severe COVID-19 infection itself, underlying conditions and the use of immunosuppressants substantially contributed to the reactivation of CMV in hospitalised COVID-19 patients. Additionally, CMV reactivation was linked with poor clinical outcomes, including increased mortality.
Introduction
The World Health Organisation (WHO) declared coronavirus disease 2019 (COVID-19) a pandemic on 11 March 2020.[1] It is now known that the majority of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infected people are either asymptomatic (accounting for an estimated 40%–50% of transmissions) or develop mild-to-moderate symptoms.[2,3] However, severe infection occurs in 20% of patients, with 15% developing severe pneumonia and 5% developing acute respiratory distress syndrome, septic shock or multiorgan failure requiring hospitalisation and respiratory support.[2,3]
Cytomegalovirus (CMV) is one of the largest known double-stranded DNA viral genomes.[4] Most cases of symptomatic CMV infection occur in immunocompromised patients, such as organ transplant patients, primarily kidney transplants,[5] or patients on immunosuppressive medications.[6] However, about 50-80% of healthy individuals also face the risk of developing symptoms related to CMV infection,[5] a scenario that is most prevalent when CMV reactivation occurs in critically ill patients. CMV infection can affect multiple organ systems, with the gastrointestinal (GI) system accounting for the majority of infections common.[7] GI symptoms such as diarrhoea, nausea and abdominal pain may also be associated with COVID-19 infection,[8] making CMV reactivation difficult to recognise in such patients.
CMV is an important component of the elderly’s immunological risk profile, and CMV infection is associated with increased two-year mortality from diagnosis.[9] It has been confirmed by more research, which shows that as people get older, more CMV-specific memory and effector T cells are made.[10,11] CMV co-infection is common in the severely ill due to multifactorial immunosuppression from a variety of sources.[11]
The full impact of CMV coinfection on the prognosis of COVID-19 infection remains unknown, despite the recent linkage of severe COVID-19 to substantial immunological suppression, which may affect CMV reactivation and contribute to the clinical outcome.[12] This systematic review aims to find an association between COVID-19 and the risk of CMV reactivation, as well as characterise any risk factors associated with CMV reactivation in such patients.
Methods
Database Search Strategy
The reporting of this systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines [Figure 1].[13] We systematically searched three different electronic databases – PubMed, ScienceDirect and Google Scholar – for relevant case reports and case series from early 2019 to 7 February 2021. Keywords used to conduct the search included ‘COVID-19’, ‘SARS-CoV-2’, ‘CMV’, ‘cytomegalovirus’, ‘reactivation’ and the medical subject headings (MESH) terms ‘SARS-CoV-2’ and ‘COVID-19’.
Flowchart of the study selection process
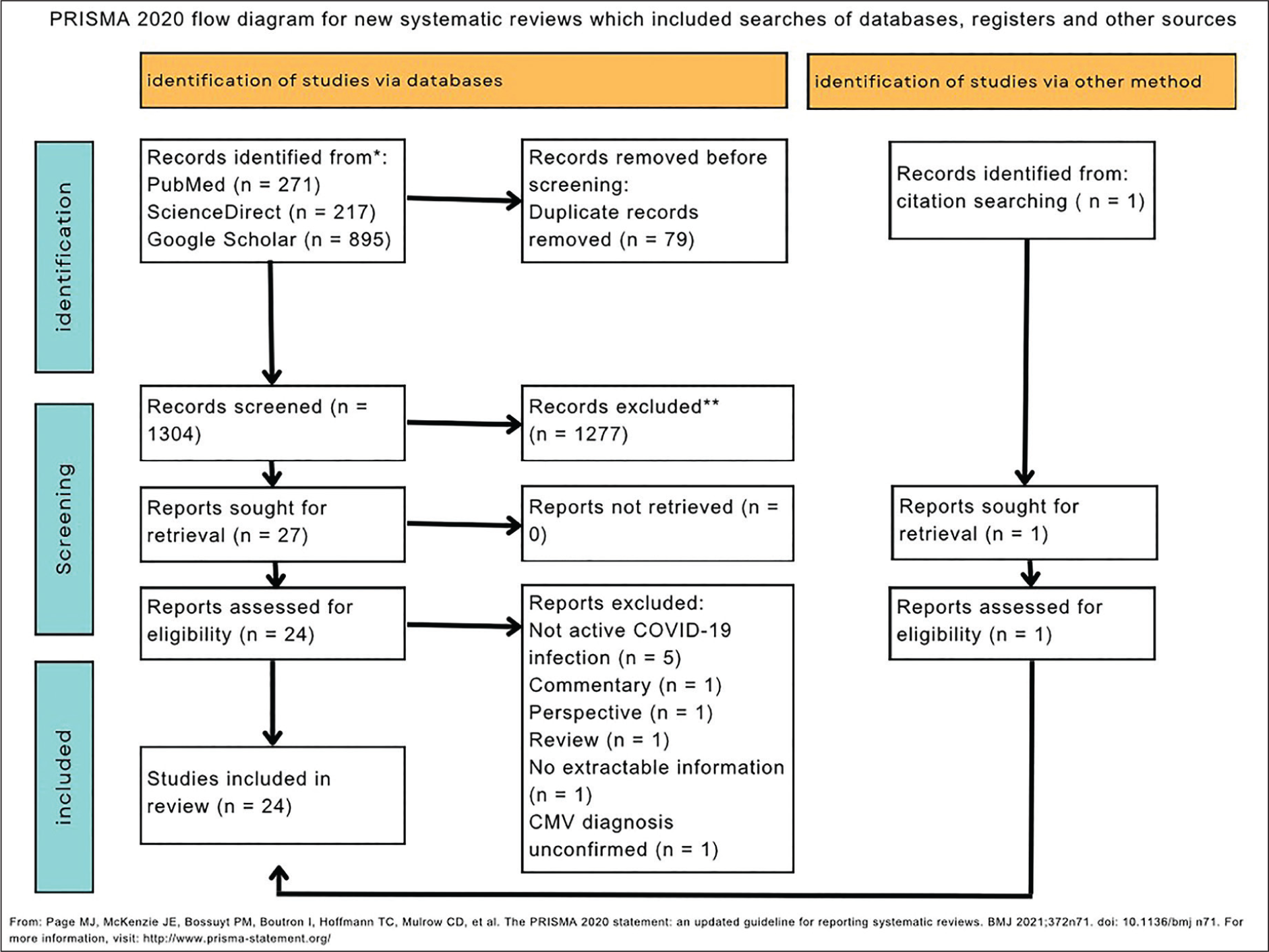
Study Selection & Quality Assessment
In order to find relevant studies, two authors (HC and AN) independently screened titles and abstracts, evaluated them in full text and included them if they met the inclusion criteria. The authors resolved conflicts by consensus. We did not assess the risk of bias because the aim of this review was to compile the characteristics of patients with SARS-CoV-2 and CMV. coinfection, not to pool the results from separate studies. The inclusion criteria consisted of (a) case reports and case series, (b) articles published in English, (c) patients over the age of 18 and (d) laboratory evidence of both infections (CMV and SARS-CoV-2).
Data Extraction
We used an Excel→ spreadsheet to extract the data. The extracted data included (a) author’s name and publication year, (b) study location, (c) study design, (d) number of patients, (e) population characteristics (gender, age, underlying conditions and medications) and (f) hospitalisation course (disease management, intensive care unit (ICU) admission and stay and clinical outcome).
Outcomes of Interest
The primary focus of this review is finding the associations between active COVID-19 infection and CMV reactivation. We aim to characterise any risk factors that could increase the risk of CMV reactivation.
Results
Literature Search
A total of 271 articles from PubMed, 217 from ScienceDirect and 895 from Google Scholar underwent the initial screening. After 79 duplicates were removed, 1304 articles were subjected to title and abstract screening. We retrieved a total of 24 studies from the 1304 articles for full-text screening. Ten articles were excluded for failing to satisfy the inclusion criteria. Additionally, one article was identified through reference screening. Therefore, the final analysis included a total of 15 articles that met the selection criteria. Figure 2 provides a detailed description of the screening process.
Graphical abstract representation
Study and Patient Characteristics
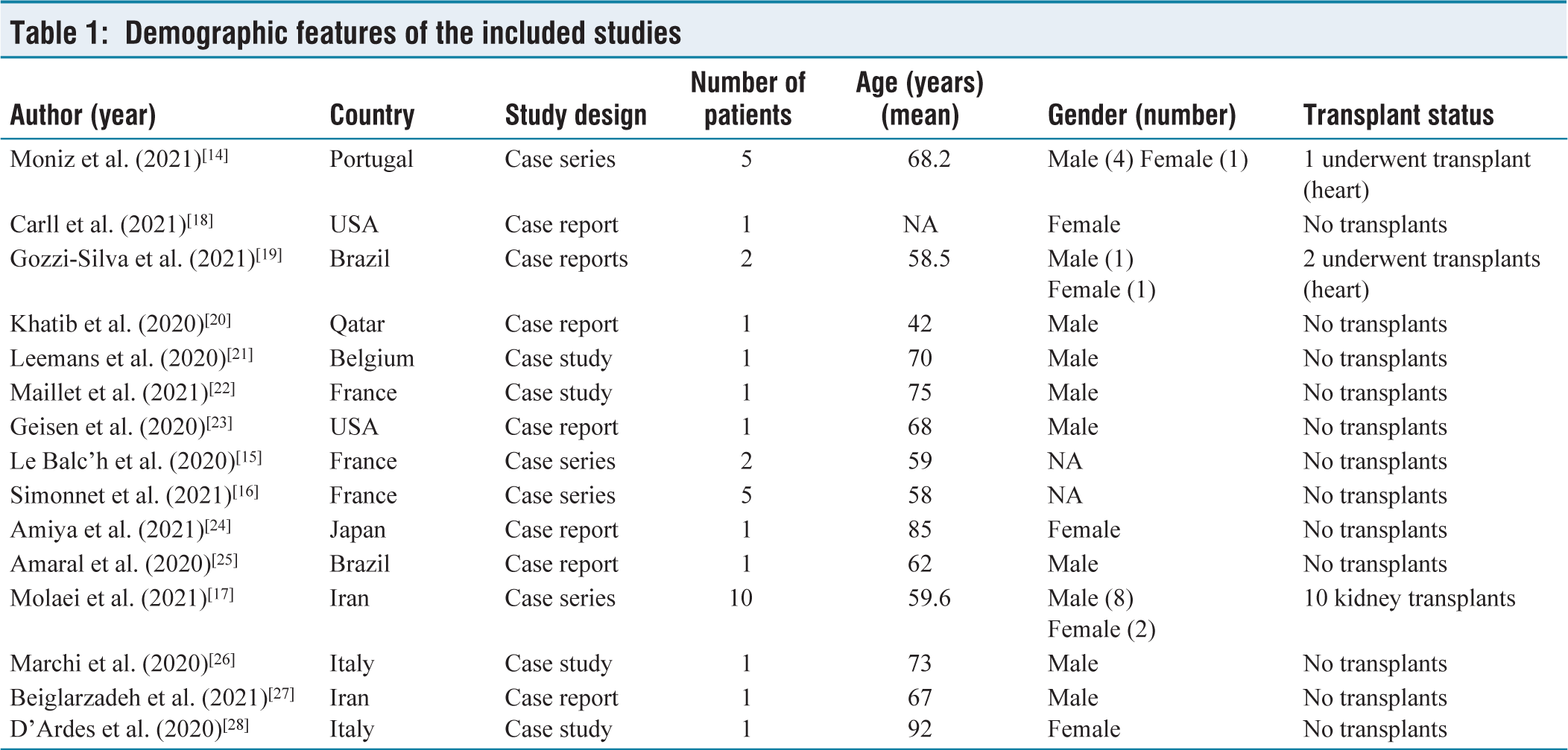
The included 15 studies were all observational; four were case series.[14-17] and the remaining 11 articles are case reports with a total of 34 patients. Out of 34 patients, 7 were female and 20 were male, with two case series not specifying the gender of their subjects.[15,16] We included studies from multiple countries, with patients ranging in age from 42 to 92 years. Table 1 displays the demographic features and study design of the included studies.
Interventions and Outcome of Included Studies
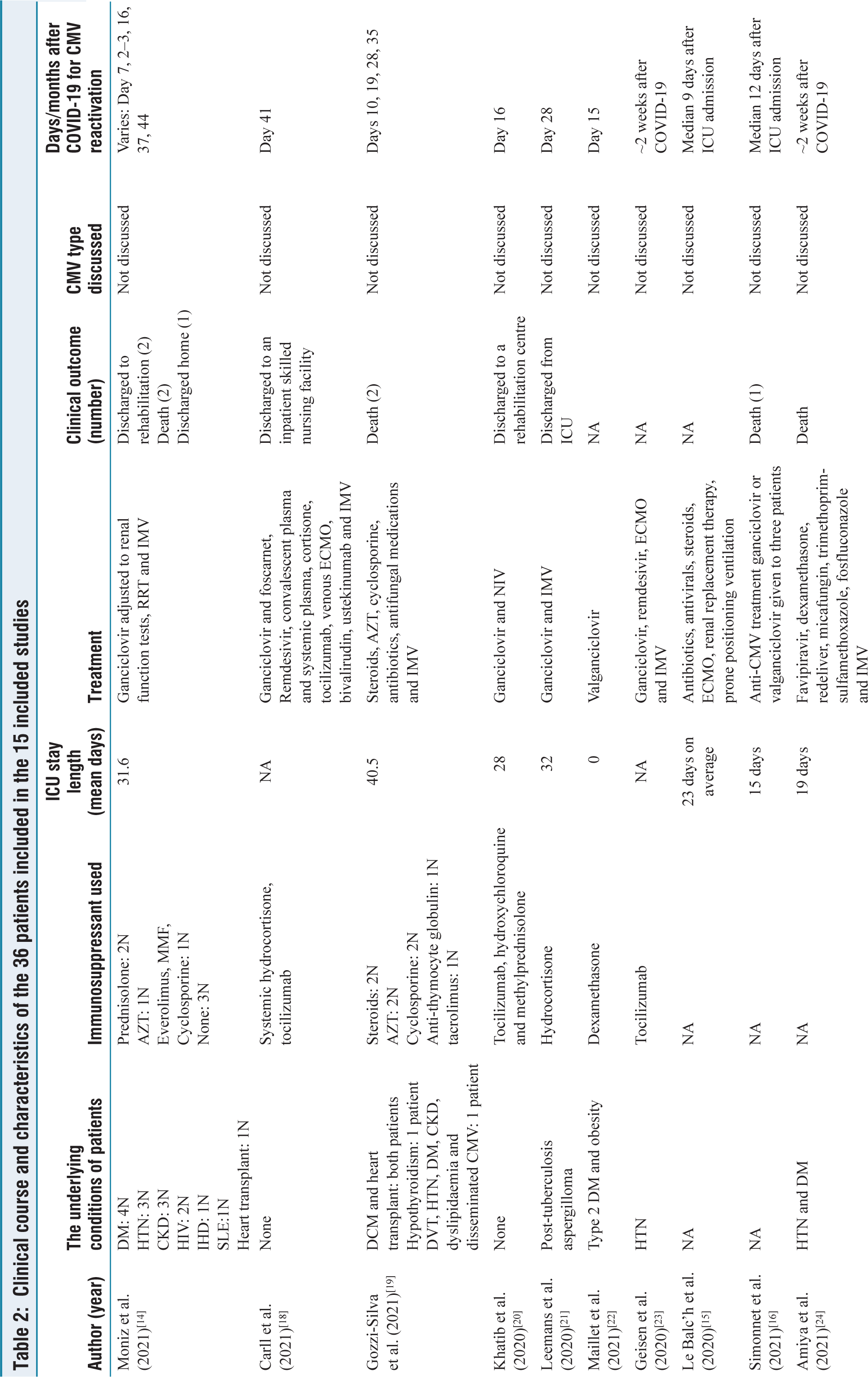
Most patients included in this review had underlying chronic medical conditions such as hypertension, diabetes mellitus and chronic kidney disease. Only two case reports dealt with patients without any chronic illness.[18,20] Antiviral medications and mechanical ventilation treated both patients in those studies, leading to their eventual recovery and hospital discharge.[18,20] Gozzi-Silva et al. treated two heart transplant recipients[19] with invasive mechanical ventilation, antivirals and immune modulation, but these patients succumbed to the illness after a prolonged ICU stay. All patients in our review received either ganciclovir or valganciclovir to treat their CMV infection. While some of the reports contain no indication of the final clinical outcome of the included patients,[15,22,23] the clinical outcome of 19 patients was death. All these 19 patients had at least one underlying chronic medical condition in addition to contracting COVID-19 infection and suffering from CMV virus reactivation. The ICU admitted most of the patients, who needed invasive mechanical ventilation for respiratory support. Table 2 displays the underlying conditions, treatment plans and clinical outcomes of the patients from the 15 included studies.
Demographic features of the included studies
Clinical course and characteristics of the 36 patients included in the 15 included studies

Discussion
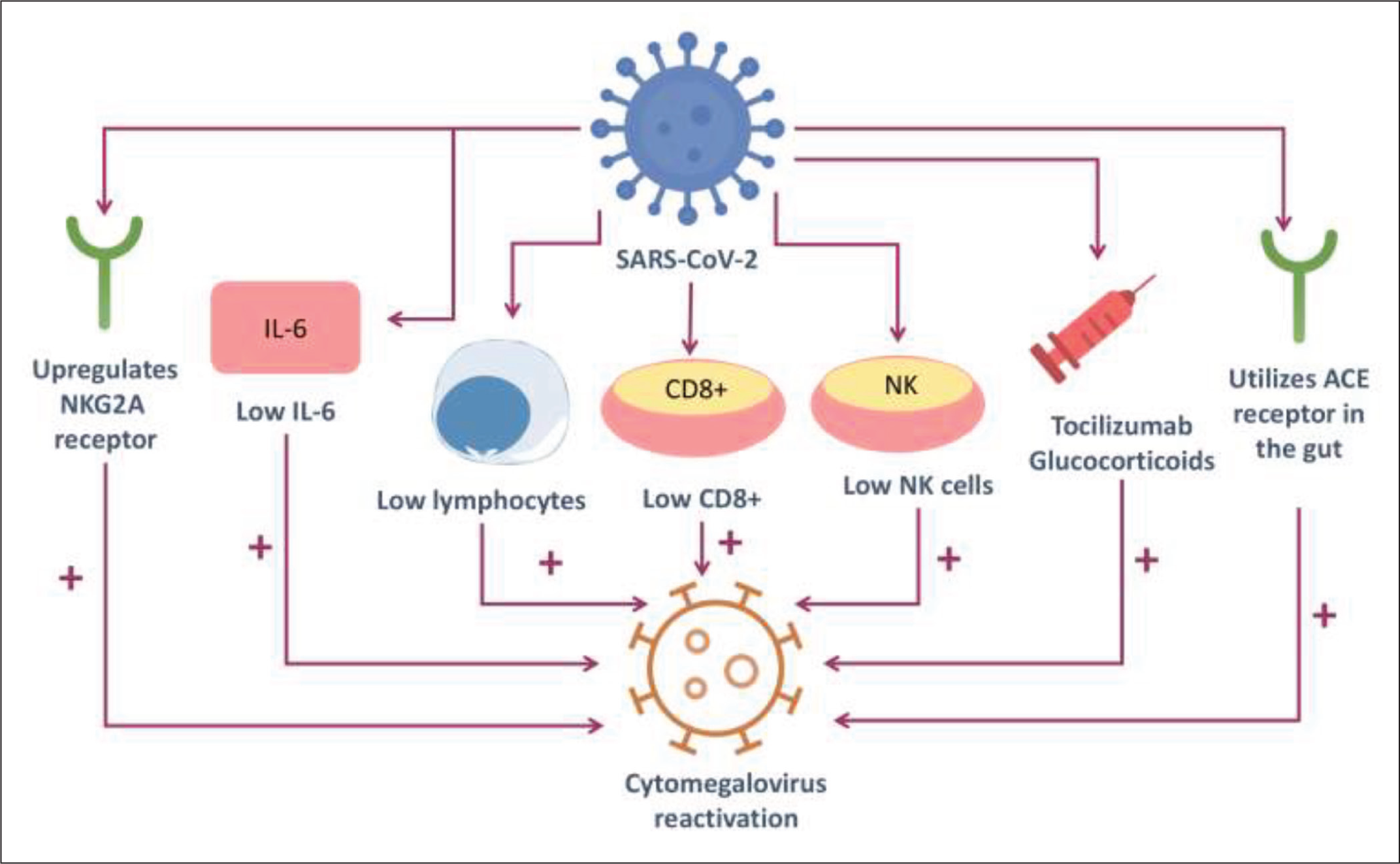
This systematic review highlights the relationship between moderate-to-severe COVID-19 and CMV reactivation. Severe COVID-19, often marked by lymphocytopenia and innate immune dysfunction, results in immune dysregulation, which can trigger CMV reactivation.[26] CMV reactivation may further exacerbate immune dysfunction, as both SARS-CoV-2 and CMV target the immune system. A key factor is the overexpression of the NKG2A receptor in COVID-19 patients, which reduces the function of natural killer (NK) cells and CD8+ T cells, making it difficult for the immune system to control viral infections like CMV.[29,30]
Immunosuppressive therapies, such as corticosteroids and tocilizumab—commonly administered to critically ill COVID-19 patients—also contribute to CMV reactivation.[16,25] Tocilizumab, for example, alters the immune response by interfering with cytokine regulation, which may trigger CMV reactivation in vulnerable patients.[23,33] Lymphocytopenia, a characteristic of severe COVID-19, further impairs cellular immunity and increases susceptibility to CMV reactivation.[18]
Our findings suggest that older patients, especially those with underlying chronic conditions such as cardiovascular disease and diabetes, are more prone to CMV reactivation.[35] The presence of chronic illnesses in COVID-19 patients likely amplifies the risk of immune system failure and viral co-infection. Additionally, men appear to have a higher risk of mortality from CMV reactivation, which is consistent with broader trends of worse outcomes in male COVID-19 patients.[34] These observations point to the combined impact of age, gender and comorbidities on the risk of CMV reactivation in COVID-19 patients.
Gastrointestinal involvement may also play a role in CMV reactivation, as both SARS-CoV-2 and CMV can affect the gastrointestinal tract, potentially increasing the vulnerability of these tissues to co-infection.
Anti-CMV treatments, such as ganciclovir and valganciclovir, effectively treated many patients included in this review.[16] However, the overall mortality rate (55%) remains high, indicating that CMV reactivation is a serious complication in COVID-19 patients. Timely diagnosis and treatment of CMV are crucial for improving patient outcomes.
While this review provides insights into the clinical impact of CMV reactivation in COVID-19 patients, several mechanisms underlying CMV reactivation remain unclear. Immune dysregulation caused by severe COVID-19, combined with the use of immunosuppressive therapies and prolonged ICU stays, likely contributes to the risk. Further research is needed to understand the precise mechanisms of CMV reactivation in COVID-19 patients and to develop preventive strategies to mitigate this risk.
The review is limited by the small sample size and reliance on case reports and case series, which may introduce selection bias. As COVID-19 continues to evolve, more comprehensive data are needed to confirm these findings and develop effective strategies for managing CMV reactivation in COVID-19 patients.
Conclusion
The interplay between SARS-CoV-2 infection and CMV coinfection remains to be completely understood. Multiple confounding variables associated with immunosuppression, such as older patients’ immune-senescence, secondary immune system dysregulation due to severe COVID-19 itself and immunosuppressive therapy, could explain CMV reactivation. [31,32] CMV reactivation and coinfection appear to be associated with COVID-19 severity and poor outcomes. Our findings underscore the importance of early diagnosis and prophylactic strategies for CMV in high-risk COVID-19 patients, particularly those with underlying conditions, admitted to the ICU, mechanically ventilated and receiving immunosuppressants.
Footnotes
Acknowledgements
We would like to acknowledge the collaborative efforts of both authors in conducting this systematic review. This work was completed solely by the authors, without any external funding or support. Each author contributed significantly to the conception, design and writing of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Credit author statement
HC contributed to conceptualisation, methodology, writing – original draft, supervision, writing – review & editing.
AN helped in data curation and formal analysis.
Data availability
Data availability pertains to publicly accessible sources used in the study.
Use of artificial intelligence
The study did not employ artificial intelligence tools or algorithms in data analysis or writing.
