Abstract
Background and Aims:
A noninvasive form of fungal rhinosinusitis is called allergic fungal sinusitis (AFS). It is characterised by pathognomonic eosinophilic mucin-containing hyphae, which are helpful in the diagnosis combined with certain histopathological and imaging characteristics. Most patients with AFS present with nasal obstruction, nasal discharge with eosinophilic mucin and headache.
Methods:
The search for published articles on AFS was done with databases like PubMed, Scopus, Medline and Google Scholar. This review article discusses the prevalence, etiopathology, clinical features, diagnosis, management and prognosis of AFS.
Results:
In AFS, computed tomography (CT) scans of the paranasal sinuses typically show abnormalities indicative of chronic rhinosinusitis. These include central regions with increased contrast (hyperattenuating) in the affected sinuses, which are associated with fungal elements and allergic mucin. In most cases of AFS, effective treatment typically involves trans-nasal endoscopic surgery, topical nasal steroids, postoperative nasal saline irrigations and endoscopic cleaning in the office. Additional medical treatments may include systemic steroids, oral antifungals, a combination of systemic corticosteroids and oral antifungals or, in some cases, immunotherapy.
Conclusion:
AFS tends to recur more often than chronic bacterial sinusitis. Managing recurrence remains a major challenge in AFS treatment. A multidisciplinary approach by otolaryngologists, ophthalmologists and neurosurgeons is recommended for AFS because of its slow and locally destructive disease.
Keywords
Introduction
Allergic fungal sinusitis (AFS) is a persistent inflammatory condition of the paranasal sinuses that can be divided into invasive and noninvasive categories.[1] It results from an allergic response to airborne environmental fungi, typically from dematiaceous species, in individuals with a normal immune system.[1] AFS falls under the noninvasive type of fungal rhinosinusitis.[2] It is marked by nasal polyps, eosinophilic mucin and the enlargement of the affected sinus cavities. AFS results from a type I hypersensitivity reaction.[2] Patients with AFS may present with unilateral or bilateral nasal polyps, and diagnosis is confirmed through distinctive histopathological features.[3] If left untreated, AFS can lead to complications such as erosion and remodelling of the bone along the orbital walls and the base of the skull.[4] AFS is usually managed by trans-nasal endoscopic sinus surgery. Endoscopic debridement is a fundamental treatment approach for most types of fungal sinusitis. However, when allergic fungal infections invade the intracranial or intraorbital regions, it presents significant surgical challenges. A key difficulty in managing AFS is the potential for recurrence.[1] The current strategy in the treatment of AFS to reduce the recurrence is the complete removal of antigenic material such as allergic mucin and fungal debris by performing adequate surgical procedures.[5] Postoperative medical treatment with systemic corticosteroids is helpful in reducing intranasal inflammation and recurrence. Recurrence of AFS is not uncommon following surgical intervention. To prevent recurrence, anti-inflammatory medications and immunotherapy are typically used.[5]
Methods of Literature Search
We conducted a search for research articles on AFS using various methods. Initially, we searched online databases including Scopus, PubMed, Medline and Google Scholar. We developed a search strategy based on PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines. This strategy helped identify relevant abstracts of published articles, and additional articles were found manually through citations. We assessed randomised controlled trials, observational studies, comparative studies, case series and case reports to determine their suitability for inclusion in this review. There were 65 articles (20 case reports; 14 case series; 31 original articles) were found on different databases and 42 were included in this review (Figure #1). This article reviews the details of AFS. This review article discusses the epidemiology, pathophysiology, clinical manifestations, prognosis, diagnosis and treatment of AFS.
Methods of literature search

Prevalence
There is no clear consensus on the definition of AFS, and the disease’s relative rarity leads to a variable epidemiological profile. Its prevalence varies between 5 and 27% among cases of refractory chronic rhinosinusitis, with 6–9% of rhinosinusitis cases requiring surgery and 9–12% of sinonasal polyposis cases associated with AFS.[6] The highest incidence of AFS has been reported in India, where its prevalence is estimated to be between 56 and 79% of all chronic rhinosinusitis cases.[7] Additionally, another study found that AFS accounts for 11.2% of chronic sinusitis cases that require surgical intervention.[8] Geographical factors influence the incidence of AFS, with higher rates observed in more humid regions. An epidemiological study indicated that only 1–2% of patients with nasal polyposis had a positive skin prick test for allergies.[9] This suggests limited evidence to support the idea that allergies predispose individuals to nasal polyposis, given the presence of watery nasal discharge, mucosal swelling and eosinophils in nasal secretions.[10]
Etiopathology
Most published research indicates that the cause of AFS is not clearly understood. The primary fungi involved in human diseases are moulds and yeasts, with moulds being the most common cause of fungal sinusitis.[5] Fungi are widespread and typically do not cause harm or illness in the sinonasal tract. Colonisation of the paranasal sinuses by fungi is quite common and usually does not lead to any disease. The term AFS was first coined by Robson et al. in 1989.[11] The fungi most frequently associated with AFS are Dematiaceous fungi, such as Curvularia lunata, Bipolaris spicifera, Exserohilum rostratum, Alternaria, Drechslera, Helminthosporium, Chrysosporium, Rhizopus, and Fusarium.[12] These fungi are responsible for about 84% of AFS cases, with Aspergillus species being the next most common.[13] The developments of polyps in AFS occurs due to chronic inflammation of the sinonasal tract.
Immunopathology
The most extensive series of AFS cases has been observed in patients with inhalant allergies, including type I immediate hypersensitivity to the moulds responsible for AFS.[14] Other immunological characteristics of AFS include a strong mucosal inflammatory response, which is typically marked by eosinophilic and lymphocytic inflammation, and may also involve atopic or allergic reactions, as well as asthma.[14] The presence of thickened allergic mucin outside the mucosal surface, along with eosinophilic degranulation products, further defines the eosinophilic aspect of this immunological condition.[15] These characteristics support the immunopathogenesis of AFS. Immunoglobulin E (IgE) is crucial in the development of AFS, with elevated total serum IgE levels commonly observed, indicating the role of immune mechanisms in the condition. Additionally, immunoglobulin G (IgG) is involved in the immunopathogenesis of AFS, as changes in antigen-specific IgG levels to the moulds responsible for AFS can predict the clinical status of the disease.[16]
Clinical Features
AFS most frequently occurs in adolescents and young adults during their second decade of life. Both males and females are equally affected, with a ratio of 1:1.[3] Most AFS patients have a history of allergic rhinitis. Typically, they present with chronic rhinosinusitis accompanied by nasal polyps, inhalant allergies, elevated total serum IgE levels and sinus blockages due to thick, ‘peanut buttery’ visco-elastic eosinophil-rich material known as ‘allergic mucin’, which contains a few fungal hyphae. AFS primarily affects the unilateral paranasal sinuses. Common symptoms associated with AFS include nasal obstruction, nasal discharge, postnasal drip, reduced or loss of smell (hyposmia/anosmia), facial pain, proptosis and headache.[17] In one study, patients with allergic fungal rhinosinusitis (AFRS) exhibited more frequent bilateral involvement of the paranasal sinuses, with an incidence of 60.0%. Additionally, 91.4% of these patients had involvement of two or more paranasal sinuses, with the maxillary sinus being the most commonly affected, followed by the ethmoid, frontal and sphenoid sinuses.[18] The features of complications in AFS include visual symptoms, proptosis, headache and increased nasal symptoms, particularly in associated bony erosion on paranasal sinus computed tomography (CT) scans.[19] Patients with advanced cases of AFS manifest with bilateral involvement of the paranasal sinuses and are prone to intracranial or intraorbital extension. The bone erosion in AFS may mimic aggressive sinonasal neoplasms.[19] Intracranial involvement of AFS may present with neurological symptoms like cranial nerve palsy and seizure. The abducent cranial nerve is often affected in AFS with intracranial involvement followed by the oculomotor nerve and trochlear nerve.[20] The range of clinical manifestations in AFS is from nasal obstruction to polyposis with gross facial disfigurement and orbital abnormalities.[20]
Diagnosis
The diagnosis of AFS is done by assessing the clinical features, radiological findings, endoscopic features with the presence of mucin, microbiological and pathological findings.[21] The diagnosis of AFS depends on several key criteria: evidence of type 1 hypersensitivity, nasal polyps, the presence of eosinophilic mucin, positive fungal staining, and characteristic findings on CT scans. Increased internal attenuation on unenhanced CT scans (Figure 2) can help distinguish AFS from more common sinonasal tumours. Additionally, minor criteria such as unilateral disease, asthma and the presence of Charcot–Leyden crystals in the mucin can support the diagnosis. Patients often have distinctive dark-coloured, rubbery nasal casts.[22] AFS is characterised by specific histopathological features, including the presence of allergic mucin, eosinophils and Charcot–Leyden crystals.[23] The diagnosis of AFS primarily relies on histopathological findings (see Table 1). Examination of thick mucin reveals inflammatory cells embedded in an amorphous eosinophilic mucin and necrotic cellular debris. Charcot–Leyden crystals, derived from degranulated eosinophils, may also be present.[24] Special stains, such as Gomori methenamine silver, may be necessary to identify fungal elements that are not always visible with routine haematoxylin and eosin stains. Histopathological and/or gross examination of surgically obtained allergic mucin is crucial for diagnosis. This mucin should show positivity for fungal hyphae upon fungal staining, and fungal cultures from the sinus should be positive in patients with AFS. There should be no evidence of mucosal invasion in histopathological studies. Other histopathological features should not include signs of tissue invasion such as mucosal necrosis, granuloma formation or giant cells. It is also important to exclude other types of fungal rhinosinusitis as the primary diagnosis. CT scans of the paranasal sinuses typically reveal areas of hyperattenuation, which is primarily due to the presence of allergic mucin.[25] The radiological criteria for AFS include typical opacification with central hyperattenuation on CT scans, often involving multiple sinuses. On magnetic resonance imaging (MRI), eosinophilic mucin is characteristically identified by central low signal intensity on both T1- and T2-weighted images due to its high protein concentration and low levels of free water and mineral content.[26] Additionally, bony erosion, including erosion of the skull base and/or the orbit, is relatively common in AFS (up to 56%) and is considered a distinguishing feature compared to non-AFS conditions.[21] Grossly, allergic mucin appears as peanut buttery or viscoelastic material. Histopathologically, on haematoxylin and eosin (H&E) stains, it is seen as strongly staining laminated concretions of pyknotic and degranulated eosinophils, surrounded by lightly staining mucin and scattered Charcot–Leyden crystals.[14] Laboratory investigations for AFS typically reveal elevated total serum IgE levels (>500 IU/mL) and increased levels of fungal-specific IgE. High IgE levels are generally a good indicator of disease activity. However, AFS can occasionally occur with normal total serum IgE levels. Peripheral eosinophilia may be observed but is less common. Patients with AFS often show positive skin test reactions and in vitro radioallergosorbent test (RAST) responses to both fungal and nonfungal allergens. Gell and Coombs type I hypersensitivity in AFS is indicated by elevated serum levels of fungal-specific IgE and positive skin tests for both fungal and nonfungal antigens. However, this hypersensitivity reaction is not specific to fungi.
CT scan of paranasal sinus in AFS
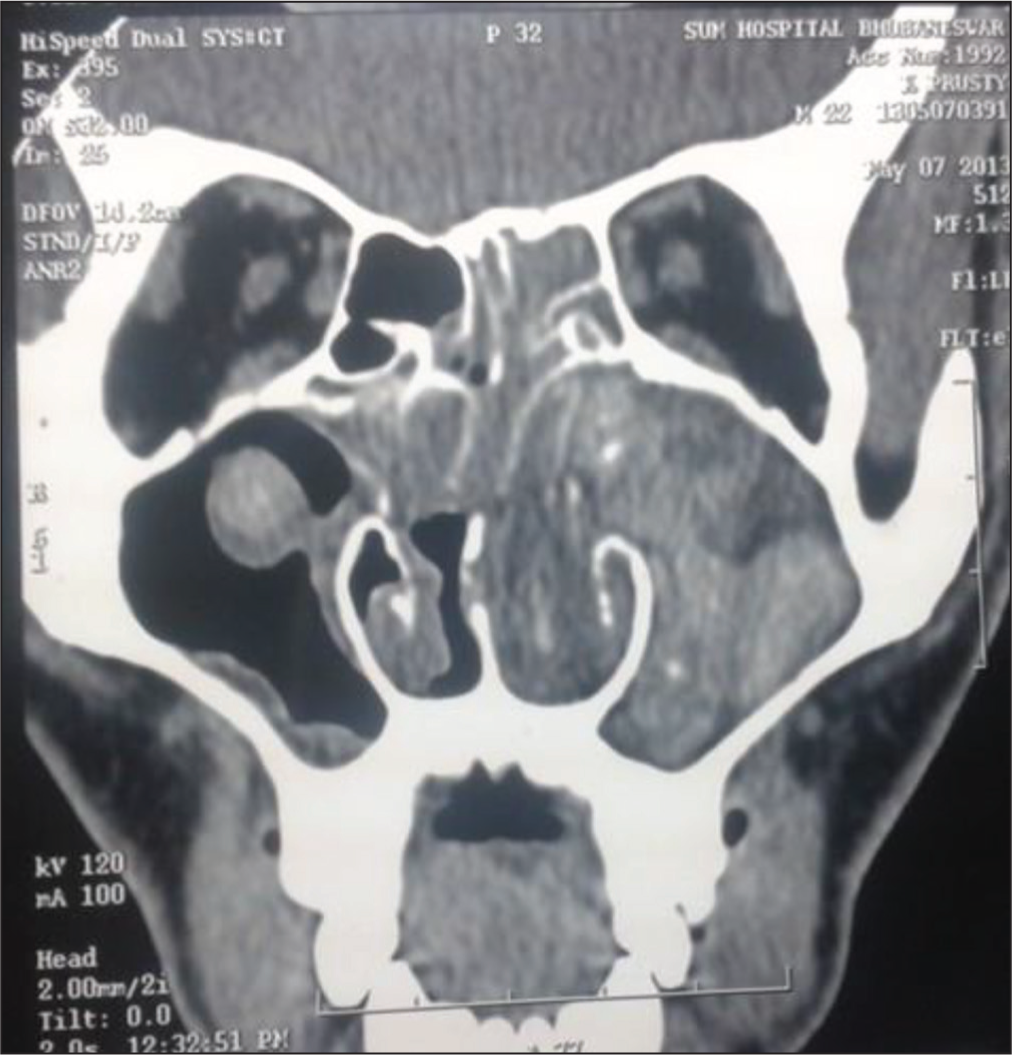
Diagnostic features of allergic fungal sinusitis

Treatment
Following the advancement in the treatment of AFS, more emphasis is now done on surgery, medical therapy and immunotherapy for the complete clearance of diseases and preventing recurrence.[27] Effective treatment of AFS relies on several key factors: thorough preoperative evaluation and mapping using CT and MRI scans, the use of appropriate surgical tools by experienced surgeons and careful postoperative endoscopic follow-up to detect and address any recurrences.
Surgical Treatment
The initial treatment for AFS involves endoscopic sinus surgery to remove all obstructing thickened allergic mucin and any diseased, hypertrophic sinonasal mucosa.[28] The goals of surgical treatment for AFS are to completely remove all allergic mucin and fungal debris, ensure permanent drainage and ventilation of the affected sinuses and preserve the integrity of the underlying sinonasal mucosa. Inferior turbinectomy and partial middle turbinectomy may be performed during functional endoscopic sinus surgery to reduce the recurrence rate and improve the management of any postoperative recurrences. The goals of surgical treatment for AFS are to completely remove all allergic mucin and fungal debris, ensure permanent drainage and ventilation of the affected sinuses and preserve the integrity of the underlying sinonasal mucosa. Inferior turbinectomy and partial middle turbinectomy may be performed during functional endoscopic sinus surgery to reduce the recurrence rate and improve the management of any postoperative recurrences.[29] If there is suspicion of dural invasion or extensive involvement of the intracranial and/or intraorbital regions, a craniotomy may be necessary to remove the disease. Inadequate surgical clearance of sinus disease increases the likelihood of AFS recurrence. Postoperative oral corticosteroids can help reduce overall disease activity, including symptoms and recurrence rates.[29]
Medical Treatment
The current approach to treating AFS focuses on minimising recurrence after the complete surgical removal of antigenic materials, such as allergic mucin and debris. Oral corticosteroids can be administered to patients with acute and chronic fungal sinusitis to alleviate symptoms by reducing inflammation and lowering circulating IgE levels.[30] There is no standardised regimen for corticosteroid use in AFS. However, one commonly accepted preoperative approach is to start systemic steroids, such as prednisone at 0.5-1 mg/kg/day, one week before surgery to help reduce intranasal inflammation.[31] In addition to steroids, preoperative antibiotics are prescribed to reduce the concomitant postobstructive bacterial sinusitis. Topical corticosteroids can be used in combination with systemic steroids.[32] One study demonstrated that using a combination of topical and intermittent oral steroids for two years effectively decreased the recurrence rate of AFS.[33] In some cases, endoscopic debridement followed by systemic antifungal therapy can help prevent the recurrence of AFS.[34] Amphotericin and azole antifungals are often beneficial in these situations. However, topical antifungals generally offer limited success and are not recommended.[34]
Immunotherapy
Despite standard treatment options, including surgery and allergy management, AFS patients often experience a high rate of recurrence. Subcutaneous immunotherapy has been explored as an adjunctive treatment for AFS. The presence of immunoglobulins specific to fungal elements suggests that immunotherapy might help prevent the host’s inflammatory response and reduce recurrence.[35] A study on AFS patients indicated that allergen immunotherapy targeting aeroallergens resulted in improved outcomes.[25] Allergen immunotherapy targeting all relevant aeroallergens, including any known etiologic moulds, can reduce sinonasal allergic reactivity to the lowest possible levels.[25] The rationale is that treatments aimed at reducing the allergic inflammatory environment in AFS are beneficial. Therefore, topical nasal steroids, antihistamines, and even antileukotrienes should be considered as part of managing this immunological process.[34] Fungal immunotherapy serves as an adjunct treatment for AFS by desensitizing the body to fungal antigens, helping to reduce the allergic response to fungi.[30] The short-term benefits of fungal immunotherapy are more pronounced compared to its long-term benefits. The fungal immunotherapy is expensive. It reduces the requirement of corticosteroids and so prevents the side effects of long-term corticosteroids.[36] In certain cases, fungal immunotherapy for AFS has demonstrated improvements in nasal crust formation, polyp development, reduced need for systemic corticosteroids and enhanced quality of life. Additionally, sublingual immunotherapy has been studied and shown to be a safe option for managing AFS, potentially leading to better patient outcomes.[37]
Prognosis
AFS has a high chance of recurrence.[38] Despite the availability of various treatment options for AFS, including endoscopic sinus surgery, corticosteroids, antifungal drugs and immunotherapy, achieving satisfactory results often require multiple surgical interventions, and success can still be challenging to attain.[39] Misdiagnosis or delayed diagnosis of AFS can lead to severe complications, including loss of smell, loss of vision, cranial nerve involvement, intracranial abscesses or cavernous sinus syndrome.[40] In severe cases, invasive fungal infections can occur, potentially resulting in patient death.[40] Surgical intervention for AFS without postoperative medical treatment results in a recurrence rate of as high as 100%.[41] The use of oral corticosteroids after surgery for AFS often yields positive results, similar to their effectiveness observed in allergic bronchopulmonary aspergillosis.[41] AFS may involve bone and cause erosion that extends into the anterior skull base and orbit. Intraorbital and intracranial involvement may occur in AFS although rare. A multidisciplinary approach involving otorhinolaryngologists, ophthalmologists and neurosurgeons is recommended for managing AFS due to its locally destructive nature. AFS with complications requires effective treatment by trans-nasal endoscopic sinus surgery, orbitotomy and craniotomy.
Conclusion
AFS is usually seen in immunocompetent, young males, belonging to low socioeconomic group. Patients of AFS often present with nasal discharge, nasal polyposis, nasal obstruction and postnasal drip. The combination of relevant clinical, biological, radiological, pathological and microbiological signs is important for getting the diagnosis of AFS. Adequate sinus surgery is considered the essential first step in treating AFS. The goal of the surgery is to thoroughly remove allergic mucin and fungal debris, ensure permanent drainage and ventilation of the affected paranasal sinuses and preserve the mucosal lining. Managing recurrence remains a major challenge in AFS treatment. Current strategies focus on controlling the high likelihood of recurrence after endoscopic sinus surgery.
Footnotes
Acknowledgements
The author is thankful to all faculties of the Department of Otorhinolaryngology and Head and Neck Surgery, AIIMS, Bhubaneswar, for their valuable suggestions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Not applicable.
Credit author statement
Santosh Kumar Swain contributed to concept, literature search, data acquisition, manuscript preparation, manuscript editing and approval of the final version to be published.
Yash Mittal helped in literature search, manuscript preparation and drafting.
Data Availability
Data are available in a public, open-access repository.
Use of artificial intelligence
Not applicable.
