Abstract
Background and Aims:
The overarching goal of this study is to conduct a robust risk assessment using the Failure Modes and Effects Analysis (FMEA) approach. By identifying potential failure modes and their potential impacts on patient care and hospital operations, this research seeks to provide actionable insights and mitigation strategies for the safe and effective implementation of the ZAP-X Radiosurgery System. The swift integration of advanced medical technologies, exemplified by the ZAP-X→ Gyroscopic Stereotactic Radiosurgery System, a computer-controlled machine that delivers radiation to an intended target in the brain, necessitates a comprehensive proactive risk assessment before the launch of the system to ensure the safety of patients, staff and operational processes.
Method:
Applying the FMEA methodology, we meticulously mapped all processes and sub-processes associated with the ZAP-X Radiosurgery System implementation. Our multidisciplinary team, through a discussion on ‘what can go wrong’, identified failure modes. Risks were prioritised based on severity, likelihood and detectability giving them a Risk Priority Number (RPN). The team proposed mitigation strategies, deploying controls to enhance safety and reliability.
Keywords
Introduction
The rapid evolution of medical technologies has ushered in a new era of innovation and sophistication in healthcare. Among these advancements, the ZAP-X Radiosurgery System stands out as a cutting-edge solution for precise and non-invasive medical interventions. The ZAP-X→ Gyroscopic Radiosurgery System is a technological innovation with data to support its medical results, safety and efficiency.
This technology redefines precision with minimal radiation exposure, enabling new standards in effectiveness and patient comfort. Unlike conventional methods, ZAP-X utilises a self-shielded, gyroscopic linear accelerator design, to direct radiosurgical beams from thousands of potential angles, accurately focusing radiation on the intended tumour or target. This therapeutic radiation device is intended for stereotactic radiosurgery (SRS) treatment for a variety of diseases such as tumours, lesions and neurological conditions in the brain and neck. It offers non-invasive, pain-free sessions.[1] Using a linear accelerator, it generates a combination of beams that can precisely target and eliminate the abnormal objects down to the nanometer leaving the adjacent areas unharmed and healthy.
ZAP-X’s clinical capabilities will enable neurologists and neurosurgeons to treat primary and metastatic brain tumours, arteriovenous malformations (AVMs), trigeminal neuralgias, movement disorders such as Parkinson’s disease, epilepsy and other intracranial lesions such as meningiomas, acoustic neuromas and pituitary adenomas, with better accuracy and fewer side effects to patients. It achieves over 95% control rate in 10 years with minimal facial or trigeminal side effects. ZAP-X shows an exceptional 99.4% control rate in five years for small, defined tumours.[1]
With the increasing deployment, complexity and sophistication of the equipment and related processes within the clinical environment, system and process failures are more likely to occur. These failures may have varying effects on the patient, ranging from no harm, including near misses, to devastating harm be this temporary or permanent, or even loss of life. Patient safety ideally should focus on preventing harm to the patient, rather than on reacting to an adverse event or near-miss event. Furthermore, staff including are more likely to embrace and accept prospective analysis because it does not focus on whom to blame, as can be the case with incident reporting. Prospective analysis is a more positive approach to a problem because it uses a person’s knowledge and competencies, rather than highlighting a person’s weaknesses or faults. Predicting the likelihood of failures allows steps to be put in place to prevent or mitigate the occurrence of failures and their consequences. There are a handful of risk management tools, including FMECA (Failure Mode, Effects and Criticality Analysis) and FTA (Fault Tree Analysis).[2] One of these prospective management processes is Failure Mode and Effect Analysis (FMEA), a tool that can be used to study and prioritise the consequences and the frequency of occurrence of failures. FMEA can be used (a) during the initial conceptual design and development phase of a process or (b) for subsequent process control as a component of continuous improvement.[3]
With a focus on quality and safety being paramount, Joint Commission International requires its accredited organisations to ensure ‘An ongoing program of risk management is used to identify and to proactively reduce unanticipated adverse events and other safety risks to patients and staff’. through its standards of Quality and Patient Safety (QPS) in the 7th Edition of Accreditation Standards[4] for Hospitals stressing the need for a proactive and comprehensive risk assessment.
This article delves into the specific processes and sub-processes of the ZAP-X Gyroscopic Stereotactic Radiosurgery System within the setting of a quaternary care hospital. As the efficacy of the ZAP-X system is reliant not only on technological precision but also on the seamless coordination of healthcare professionals, engineers and administrators, a comprehensive risk assessment becomes paramount.
The overarching goal of this study is to conduct a robust risk assessment using the FMEA approach. By identifying potential failure modes and their potential impacts on patient care and hospital operations, this research seeks to provide actionable insights and mitigation strategies for the safe and effective implementation of the ZAP-X Radiosurgery System.
Method
Setting
The FMEA was performed at Indraprastha Apollo Hospital, Mathura Road, Jasola Vihar, Delhi between February and May 2023. The stages of conducting an FMEA were followed [Figure 1].
Stages of failure mode and effect analysis (FMEA)
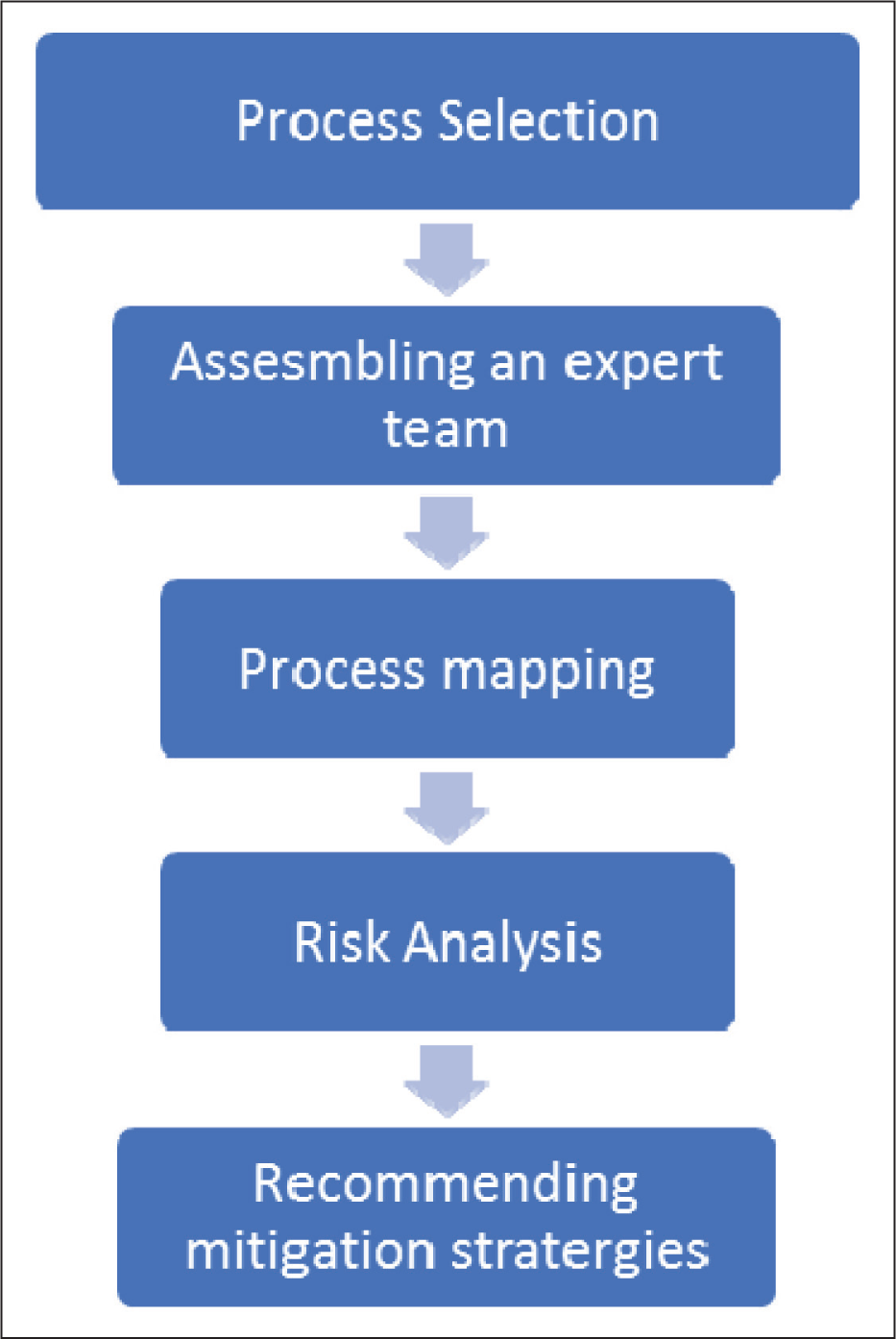
Process Selection
The ZAP-X installation and patient treatment process were chosen as the focus for the FMEA due to their critical importance and the stage of development of the systems involved.
Team Assembly
As the FMEA methodology is driven by the knowledge, expertise and experience of the involved team members working on various parts of the process and systems to get insights into the potential ways a process may fail. Our FMEA team included representatives from medical services, medical physics and radiation safety, bio-medical engineering, quality department, nursing department, neuro surgery, medical operations, anaesthesia, fire safety and the top management. The team was briefed about the FMEA methodology and risk assessment.
Process Mapping
At the time of the study, the ZAP-X installation process was still in the conceptual design and development phase, making it crucial to identify potential failure modes and risks early on.
This process encompasses various activities, including equipment setup, calibration and integration with existing hospital infrastructure. Identifying potential failure modes in the installation process is essential for ensuring the smooth implementation and functionality of the ZAP-X system within our hospital environment.
Similarly, patient treatment with the ZAP-X system was selected for FMEA due to its significance in delivering effective medical care and the inherent risks associated with radiation therapy. As this process involves direct interaction with patients and complex technological systems, understanding and mitigating potential failure modes are paramount to ensure patient safety and treatment efficacy.
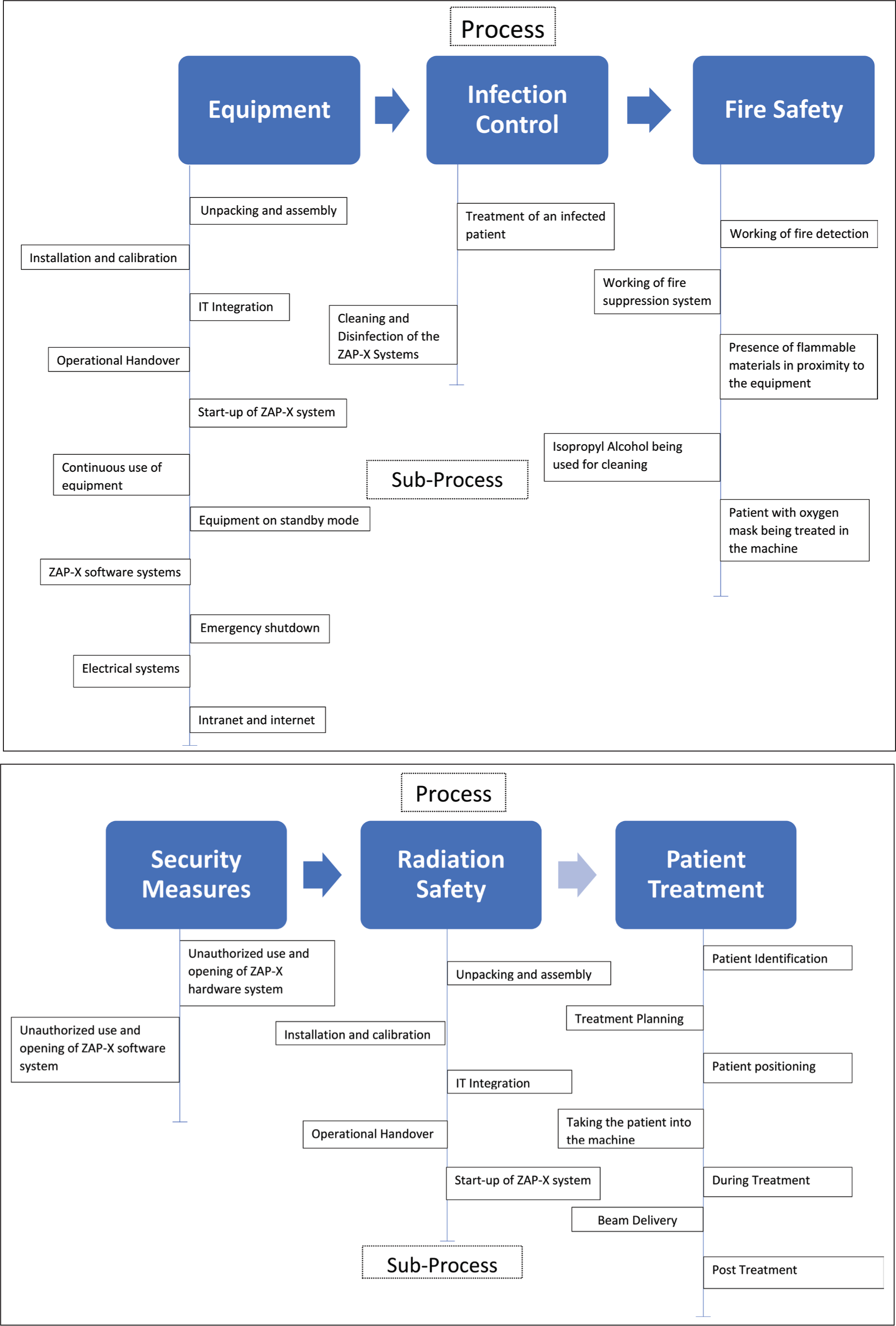
Hence, processes and sub-processes that circumvented the ZAP-X systems [Figure 2] were constructed starting from the equipment and followed by infection control, fire safety, security, radiation safety and the patient treatment process.
Process selected for ZAP-X failure mode effect analysis and sub-processes
Risk Analysis
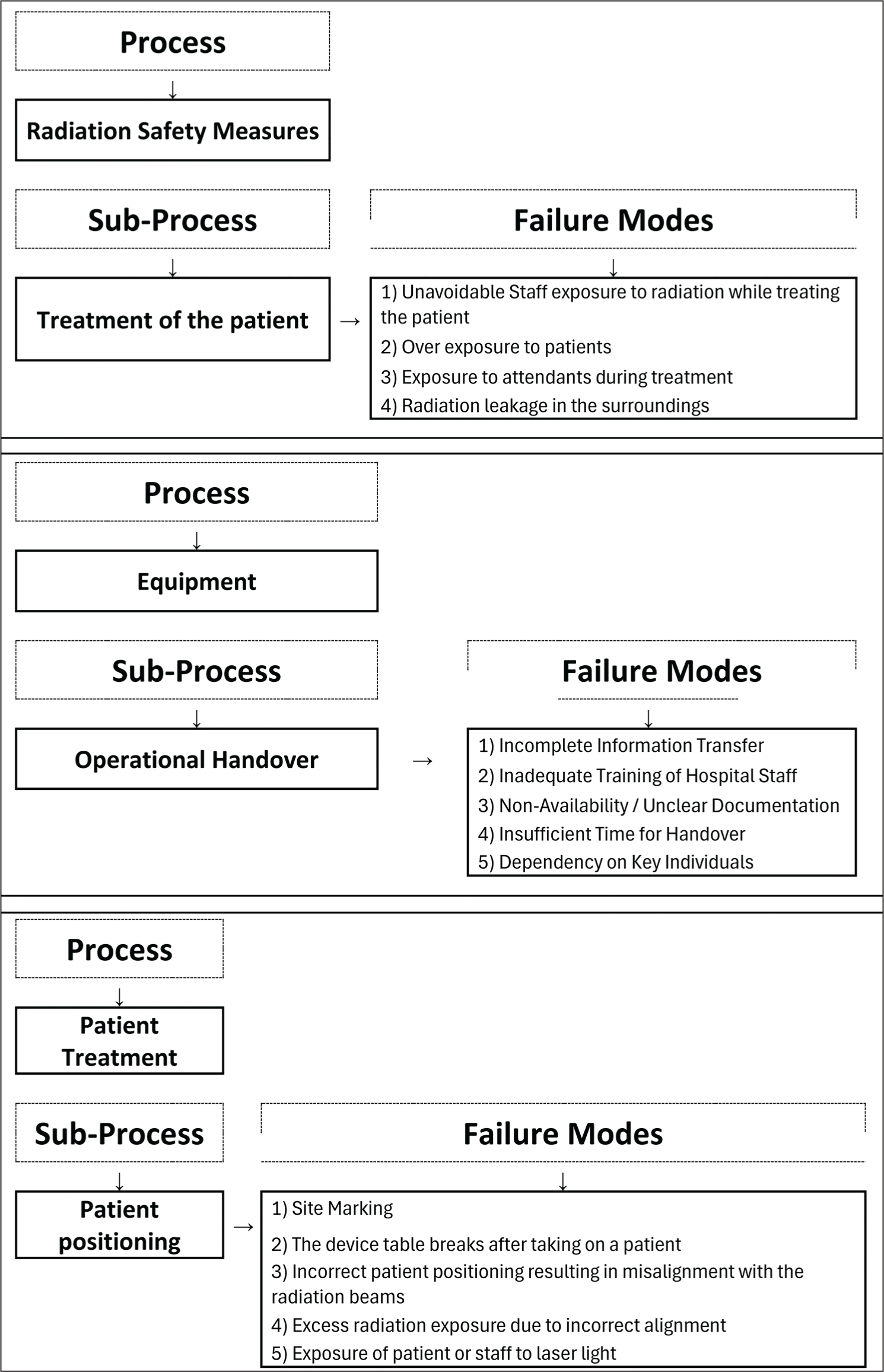
Potential failure modes and their Risk Priority Number (RPN) were identified at each process step [Figure 3]; the RPN helps us to determine the criticality linked to each of these failure modes. A RPN is a numerical score calculated by multiplying the severity X probability X detectability of failures according to a defined rating scale [Figure 4]. The rating scale is based on the Healthcare Failure Mode and Effect Analysis (HFMEA) Guidebook by VHA National Centre for Patient Safety (January 2021).[5]
Figure showing examples of processes, their sub-processes along with the possible failure modes
Healthcare failure mode and effect analysis (HFMEA) guidebook by VHA National Centre for Patient Safety[5]
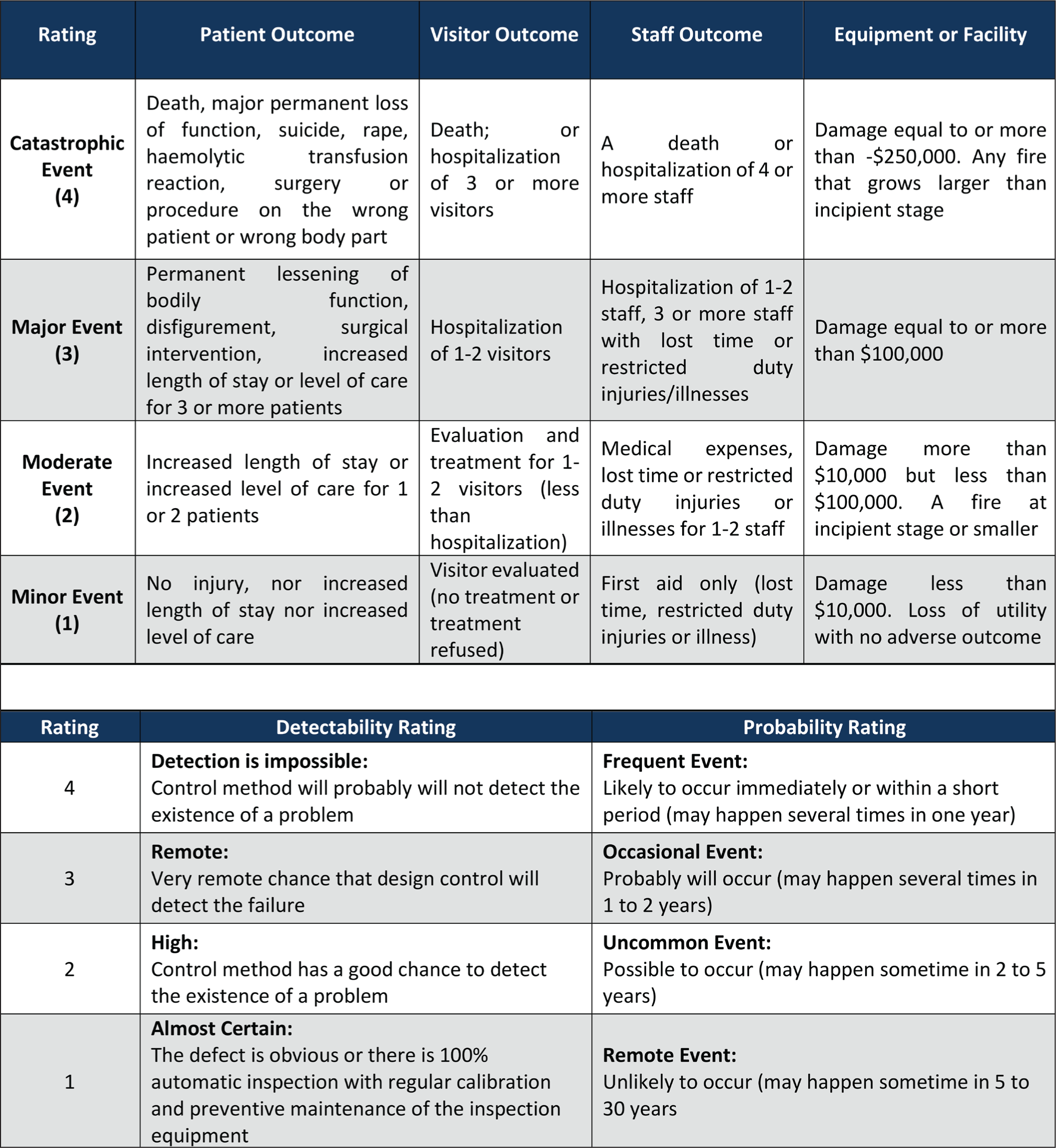
The highest possible RPN as per the scale is 64 and the lowest possible RPN is 1.
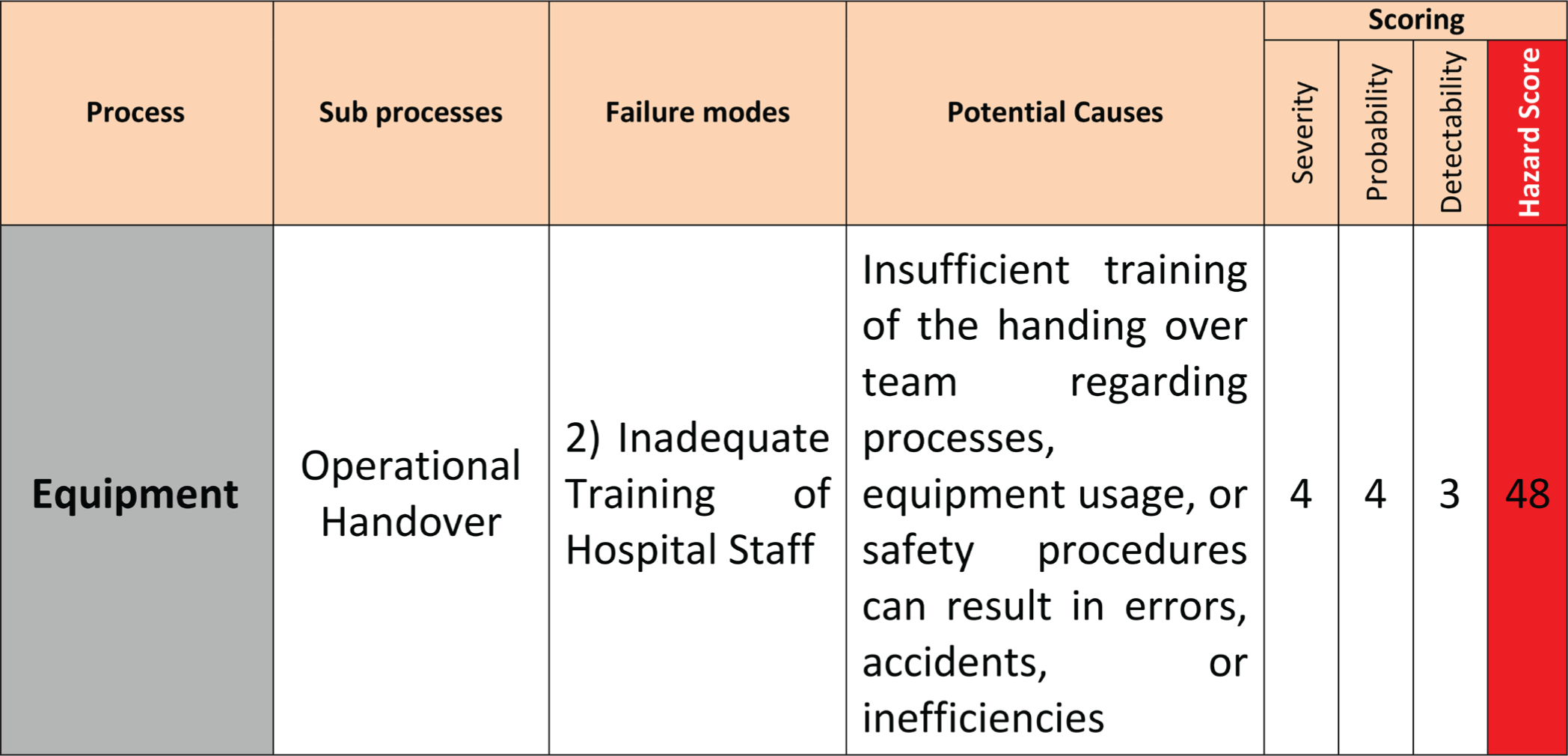
The failure modes were ranked by the team on severity, probability and detectability, as shown in the risk scoring example in Figure 5, on the basis of day-to-day operation, team expertise, any incidents/occurrences of the given failure mode in other processes of the hospital and brainstorming. Existing controls were also taken into account while determining the RPN for the failure modes.
Example of risk scoring
Recommending Mitigation Strategies
Once the failure modes were identified, control measures were brainstormed by the FMEA team. The expected outcome of the control measure was documented and the person responsible for implementing these measures was identified.
Results
The FMEA revealed a total of 78 failure modes distributed across 6 processes, encompassing 33 sub-processes. RPNs varied widely, with the highest reaching 48 and the lowest recorded at 1.
Process: Equipment
Within the equipment process, the FMEA identified the highest RPN (48) associated with the operational handover sub-process out of the 12 identified sub-processes. The critical failure mode identified was ‘Inadequate training of hospital staff’. Potential causes included insufficient training of the handover team regarding equipment usage and safety procedures, leading to errors or accidents. The absence of effective controls exacerbated the risk associated with this failure mode.[6]
Conversely, the lowest RPNs (2) were observed in the installation and calibration of equipment sub-processes. Issues such as lack of stability and performance degradation during integration with the hospital’s IT infrastructure were identified. However, the presence of effective controls, including installation by a trained manufacturer’s team and tested downtime protocols, mitigated these risks, resulting in reduced overall RPNs.[6]
Process: Infection Control
Within the infection control process, the average RPN was 8 for a total of 2 sub-processes, indicating moderate risk levels. Notably, both identified failure modes were effectively controlled by existing measures in place.
Proper protocols were not followed while taking up an infected patient for treatment.
Spread of infections or hospital-associated infections to patients and/or staff.
Process: Fire Safety
The fire safety process encompasses five sub-processes, each contributing to the overall fire safety measures within the hospital.
Under the sub-process of fire suppression, a notable failure mode emerged with an associated RPN of 36. This failure mode pertained to the failure of the fire suppression system, attributed to the absence of an appropriate system tailored for the ZAP-X equipment.
Conversely, the lowest RPN within this process, standing at 6, was linked to the failure mode of a patient wearing an oxygen mask while being treated with the ZAP-X machine. This scenario posed a potential fire hazard due to factors such as untrained or inattentive staff and the lack of appropriate protocols. However, existing control measures, including halting oxygen supply for patients undergoing ZAP-X treatment and staff training, effectively mitigated this risk.[6]
Process: Security
The security process is paramount for maintaining the integrity and safety of the ZAP-X hardware and software systems.
Divided into two sub-processes, it focuses on preventing unauthorised access and safeguarding sensitive information and equipment. The sub-processes scored a moderate RPN or 4 and 8, respectively.
The sub-process addresses unauthorised access to ZAP-X systems both hardware and software, identifying potential risks such as emission of lethal doses of radiation, delivery of high voltage electric shocks, unexpected system operation, failure in startup or functioning and patient information leakage.[6]
These risks stem from factors like untrained operation, unauthorised personnel usage, unauthorised system changes by both authorised and unauthorised staff and cyber threats. However, existing control measures including safety interlocks, warning notifications, limited access, physical key and button usage, built-in firewall and Transport Layer Security (TLS) encryption effectively mitigate these risks.[6]
Process: Radiation Safety
Legal and statutory compliance:
The sub-process focusing on legal and statutory compliance revealed the highest RPN of 32. This was attributed to the failure mode of the machine not complying with regulatory standards.
Patient treatment safety:
Conversely, the sub-process addressing safety measures during patient treatment exhibited the lowest RPN of 1. The identified failure mode involved attendants being exposed to radiation during treatment. However, effective controls, such as restricting attendant access to the treatment area and the self-shielding nature of the machine, effectively mitigated this risk. These measures ensured that attendants remained shielded from radiation exposure, thereby eliminating the potential hazard associated with this failure mode.[6]
Process: Patient Treatment
The patient treatment process exhibits an average RPN of 6 across 7 sub-processes, indicating a moderate risk level. These sub-processes include patient identification, treatment planning, patient positioning, taking the patient into the machine, during treatment, beam delivery and post-treatment care. Several critical failure modes were identified within these sub-processes, including excess dose delivery to the patient, erratic radiation delivery, high voltage passed onto the patient during treatment, incorrect patient alignment and incorrect site marking.[6]
To mitigate these risks, various control measures have been implemented[6]:
The system generates an emergency stop (E-Stop) if the measured beam intensity deviates from the expected amount. Real-time monitoring of the patient is conducted throughout the treatment process. Dose verification systems ensure the appropriate dose is delivered to the patient. Regular maintenance checks are performed to ensure optimal functioning of ZAP-X systems. High voltage is only enabled under preset safety conditions, with a physical key and button required for activation. Software controls prevent accidental activation of high voltage.
These comprehensive control measures, combined with the inherent safety features of the ZAP-X machine, contribute to maintaining the lowest possible RPNs within the patient treatment process, ensuring the safety and well-being of patients undergoing treatment.
Discussion
The identified failure modes across different processes, ranging from equipment operation to patient treatment, highlight the diverse array of risks inherent in the integration of the ZAP-X system. Notably, the following failure modes—‘Unauthorized access to hardware and software systems’, ‘Inadequate training of hospital staff', and ‘Non-compliance with regulatory guidelines’—were scored as the highest risks in the FMEA. However, the implementation of robust control measures, including safety interlocks, warning notifications and stringent access controls, played a crucial role in mitigating these risks.
The application of the FMEA approach especially as a proactive measure for risk assessment proved instrumental in systematically identifying and prioritising potential failure modes, enabling targeted mitigation efforts.
The modes especially the higher RPN failure modes were given targeted mitigation measures which helped in reducing the overall risks associated with those measures. However, it is important to continuously monitor and evaluate control measures to adapt to evolving risks and operational challenges. Regular maintenance checks, real-time monitoring systems and cross-checking procedures by healthcare professionals are integral components of maintaining the safety and reliability.
Overall, the findings of this risk assessment contribute valuable insights to enable a safe integration of advanced medical technologies like ZAP-X into healthcare facilities. By identifying potential risks and implementing effective control measures healthcare organisations can enhance patient safety, optimise operational efficiency and ensure the successful adoption of innovative medical technologies like the ZAP-X Radiosurgery System.
Conclusion
In conclusion, this study provides a comprehensive risk assessment for the implementation of the ZAP-X Radiosurgery System in a quaternary care hospital setting. Through the application of the FMEA approach, potential failure modes and associated risks across various processes were systematically identified and evaluated. The findings underscore the critical importance of proactive risk management in ensuring the safety and effectiveness of advanced medical technologies within healthcare environments.
The study identified a range of failure modes, from equipment operation to patient treatment, highlighting the diverse array of risks inherent in the integration of any new process or technology into the healthcare system. However, the implementation of robust control measures, including safety interlocks, warning notifications and stringent access controls, played a crucial role in mitigating these risks.
While the FMEA identified many risks and failure modes in our current setting, it’s important to recognise that each healthcare organisation, department and facility is unique. While major aspects of these identified risks may be applicable to other healthcare settings, further analysis and brainstorming involving organisational staff will be necessary to adapt them to other units or facilities. Nonetheless, the approach and methodology outlined in this study can serve as a valuable framework for conducting risk assessments in various contexts.
Overall, this study contributes valuable insights to the ongoing dialogue surrounding the safe integration of advanced medical technologies into healthcare practices. By identifying potential risks and implementing effective control measures, healthcare institutions can enhance patient safety, optimise operational efficiency and ensure the successful adoption of innovative medical technologies like the ZAP-X Radiosurgery System
Footnotes
Acknowledgements
The authors extend their sincere gratitude to the leadership of Indraprastha Apollo Hospital and the Apollo Hospitals group for their visionary initiative in bringing the ZAP-X gyroscopic radiosurgery platform to South Asia for the first time. Their unwavering commitment to advancing healthcare technology has paved the way for groundbreaking advancements in patient care.
Special thanks are also due to Mr. Saji Oommen, Chief Consultant Medical Physicist and Radiation Safety Officer, Indraprastha Apollo Hospital for his invaluable time and expert technical knowledge. His guidance and expertise were instrumental in ensuring the success of this study.
The authors are also deeply grateful to Dr Gaurav Tyagi, Neurosurgeon, Indraprastha Apollo Hospital for generously sharing his expert clinical insights and valuable time. His contributions greatly enriched the clinical perspective of the study.
Dr Fauzia Khan, Deputy Manager – Clinical Interface Operations, Indraprastha Apollo Hospital deserves special recognition for her invaluable contributions in interpreting and coordinating the ZAP-X process. Her dedication and coordination were instrumental in facilitating seamless collaboration among stakeholders.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this.
Institutional ethical committee approval number
This study did not require formal ethical approval, as it did not involve human participants, or any patient-identifiable data. The focus of the research was on the assessment of processes and systems using the Failure Modes and Effects Analysis (FMEA) methodology. All data used in the study were related to equipment and operational risk management, with no direct interaction or impact on patient care at the individual level.
Credit author statement
Libin K. Babu played a pivotal role in formulating the Failure Mode Effect Analysis (FMEA) risk assessment, conducting in-depth research on the subject of ZAP-X, and synthesising information from ZAP-X manuals. With the technology being new and emerging, Libin invested considerable effort in coordinating clinical and technical expertise to compile relevant data. Additionally, Libin conceptualised the article, prepared materials, conducted analysis and drafted the manuscript.
Satish Kumar contributed his expert knowledge in risk assessment during the FMEA phase, providing invaluable insights into identifying and evaluating potential failure modes.
Yogmaya Nayak brought her expertise in Failure Mode Effect Analysis to the project, offering valuable insights and guidance throughout the risk assessment process, especially constructing the framework for the FMEA.
Arpita Malani contributed to the project by vetting the FMEA methodology, reviewing the manuscript and providing valuable ideas for improvement.
Collectively, the contributions of Libin K. Babu, Satish Kumar, Yogmaya Nayak and Arpita Malani were essential in conducting the risk assessment, preparing the article and ultimately publishing the findings. Their dedication, expertise and collaborative spirit have greatly contributed to the success of this endeavour.
Data availability
The FMEA data that support the findings of this study are available from the corresponding author, LB, upon reasonable request.
Use of artificial intelligence
No artificial intelligence was used for the preparation of this article.
