Abstract
Background and Aims:
Platelet-rich fibrin (PRF), rich in growth factors and cytokines, found useful for bone healing in animal studies, has paucity of literature on open fractures in humans. This randomised comparative interventional study aimed to find out the role of PRF in wound healing and fracture union in open fractures of tibia.
Material and Methods:
Random computerised allocation of 14 open tibial shaft fractures managed with an external fixator were done into PRF and non-PRF groups. Autologous PRF injected at fracture site, with PRF gel over open wounds in the PRF group, was compared with conventional standard treatment in the non-PRF group. Wound assessment on day 3, day 7 and weekly till complete wound healing and radiographs for fracture union at 3, 6, and 12 months were done in both groups. The statistical analysis was done using an unpaired t-test (wound size assessment) and chi-squared test (fracture union), using SPSS version 25.
Results:
Fractures union in PRF group 7/8 (87.5%) was better than 3/6 (50%) in non-PRF group. External fixators were removed 4 weeks earlier in PRF group. Wound healing time, though not statistically significant, was faster in PRF group (36.25±3.03 days) compared to non-PRF (42±7 days), even with larger wound size in PRF group (7.87±1.43 cm 2 ) than non-PRF group (7.5±1.5 cm 2 ).
Conclusions:
Results of injectable PRF at fracture site and PRF gel over the wound are encouraging, found to be safe, with faster wound healing and better fracture union in the small sample size of this pilot study.
Introduction
Tibial fractures are the most common long-bone fractures with around 25% being open fractures.[1] Majority of these fractures result from high-velocity injury like road traffic crashes and fall from height. The management of these fractures can be complex due to relative lack of soft tissue coverage and blood supply of the tibial shaft.[2] Avoidance of infection and promotion of fracture union remain challenges to the clinician in open tibial diaphyseal fractures. Open fractures are also associated with poorer outcomes than closed fractures including longer time to union, longer hospital stay, higher reoperation rate and higher incidence of infection when compared to closed fractures. Controversy continues as to the ideal treatment of these fractures. External fixators have been used for open fractures for over 100 years. Modern fixators can adjust fracture alignment over time and allow early weight bearing.[3]
With developments made in the field of molecular biology and genetics, much attention has been recently placed on the healing environment at the molecular level. New research is focusing on the use of autologous growth factors to increase the effect of bone and soft tissue healing while decreasing the length of healing time. Platelets have been demonstrated to be the natural storage for several growth factors and cytokines that promote blood coagulation, tissue repair and the process of bone mineralisation.[4,5] Though the use of platelet-rich plasma (PRP) has been reported in fracture unions, there is a paucity of literature concerning platelet-rich fibrin (PRF). Compared to PRP, PRF is simple and cost-effective with the advantages of avoiding the biochemical handling of blood or the use of bovine thrombin and anticoagulants. PRF results in favourable healing due to slow polymerisation, more efficient cell migration and proliferation in addition to haemostasis and a supportive effect on the immune system.[6] PRF mixed with bone graft has been extensively used for filling bony alveolar defects in orthodontics,[7] but has not been reported to have been clinically used in the treatment of open fractures in orthopaedics. The present prospective study aims to evaluate the role of autologous PRF in wound healing and fracture union in open fractures of the tibia.
Material and Methods
This randomised comparative interventional study was carried out on 14 patients randomly allotted into two groups (PRF and non-PRF) by a simple randomisation process by Microsoft Excel. The study included open fractures of the tibia stabilised by an external fixator between October 2019 and April 2021 and were followed up till January 2024. After due approval of the Institutional Ethics Committee, the study was registered in the Clinical Trials Registry – India (CTRI) (CTRI number-CTRI/2020/05/025401). Due to the paucity of published literature on the use of autologous PRF in long-bone fractures or open fractures, the sample size was calculated based on the study of nine patients by O’Connell et al., where significant result was seen with the use of autologous PRF matrix in the healing of chronic lower-extremity ulcer.[8] In our pilot study, the sample size in the interventional PRF group was eight patients with open tibial fractures, while the non-PRF group had six patients.
General Data of Patients
The strict inclusion criteria of our cases ensured a similar type of open diaphyseal tibia fractures, Gustilo-Anderson classification of grade 2 (six cases); 3A (five cases) with wound size ranging from 1 to 10 cm 2 (mean size 7.68 cm 2 ) and grade 3B where the wound could be closed (three cases). The age of the patients ranged between 20 and 50 years (mean age of 26.5 years), with a platelet count of more than 2 lac/mm 3 . On comparing the two groups, there were no statistically significant differences concerning distribution of age, gender, type and location of the fractures. The mean time interval (hours) between injury and surgery was 26.63±4.75 and 11.50±3.94, respectively, in PRF and non-PRF groups, which was not statistically significant. There was no statistically significant difference in platelets count between the two groups (p = .69).
The cases excluded were those with grossly contaminated wounds from farmyard or sewage, fractures with bone loss, comminution, segmental fractures, fractures with intra-articular extension and pathological fractures or any open fracture where the bone remained exposed devoid of its periosteal cover which could not be covered by any fascio-cutaneous or myo-cutaneous flaps.
Treatment
After routine investigations, X-rays and pre-anaesthetic clearance, all patients were taken up for surgery in the emergency/elective operation theatre.
Surgery was performed under spinal or general anaesthesia. After standard wound debridement and washing with povidone-iodine and saline, a tubular external fixator (AO type) was used to stabilise the fracture as per standard orthopaedics guidelines. Subsequently, the patients were treated in one of the two ways mentioned below. In the PRF group, the fracture site was injected with liquid PRF referred to as injectable PRF (i-PRF) through a wide-bore needle or an infant feeding tube, while the dressing of the open wound was done using PRF gel (matrix) or liquid PRF soaked sterile dressings. The non-PRF (control) group cases were treated conventionally without the application of PRF.
Preparation of PRF
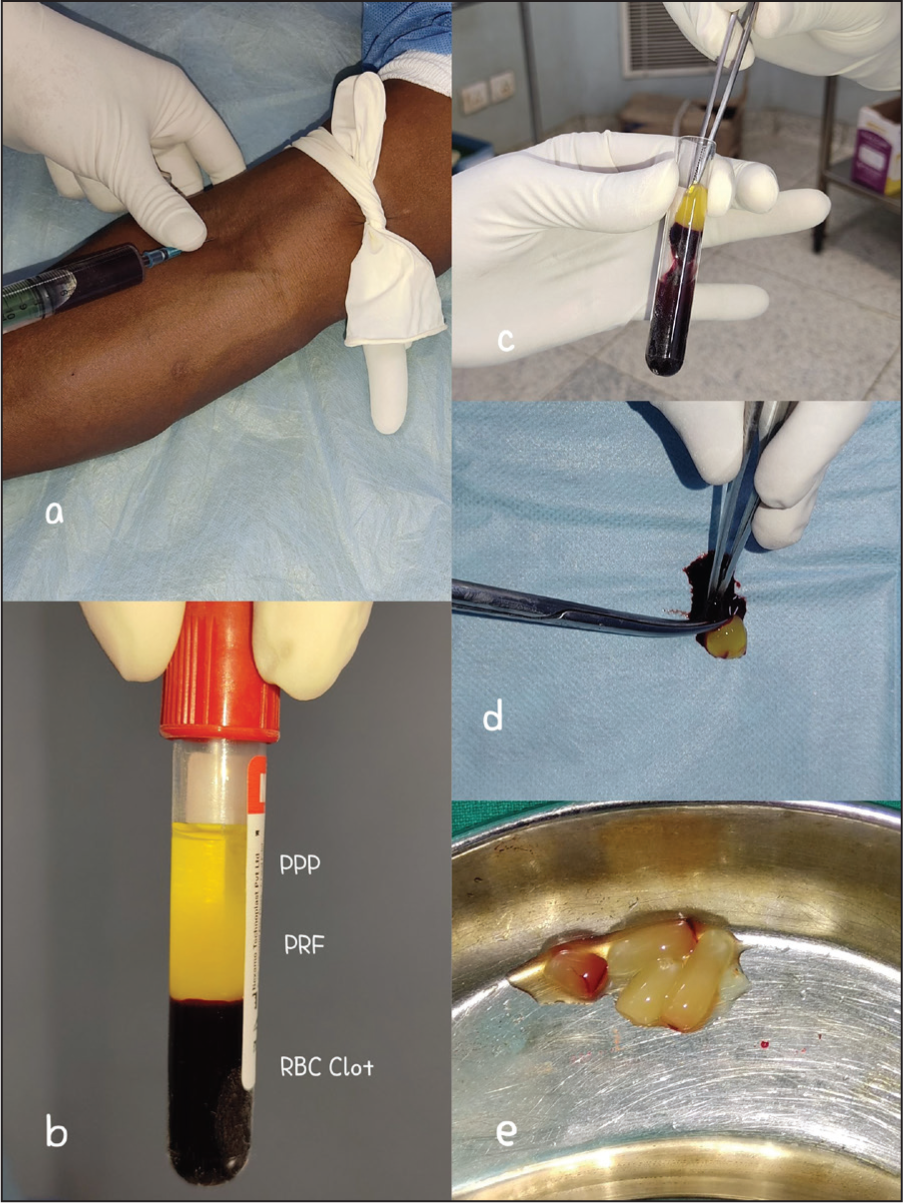
Using all aseptic precautions, 10–20 mL of venous blood was withdrawn using an 18-gauze sterile needle and was quickly transferred to a plain centrifuge vial (without any anti-coagulant) and placed in a centrifuge machine within a minute or two. The PRF was prepared in two forms, a liquid or injectable form and a gel or matrix form [Figure 1]. For the matrix form, the blood was centrifuged for 10 minutes at 3000 rpm. This was followed by a standing period of 10 minutes in the centrifuge itself. The resultant derivative consisted of three layers, an uppermost layer of acellular plasma, a middle layer of PRF and a bottom layer of red blood cell (RBC) clot, out of which the middle fibrin layer along with the RBC clot was taken out of the vial in a sterile manner with the help of blunt forceps. The fibrin layer was separated from the RBC clot with the help of scissors or surgical blades to form the PRF matrix or gel.[9] This fibrin matrix was used for wound dressing [Figure 2c].
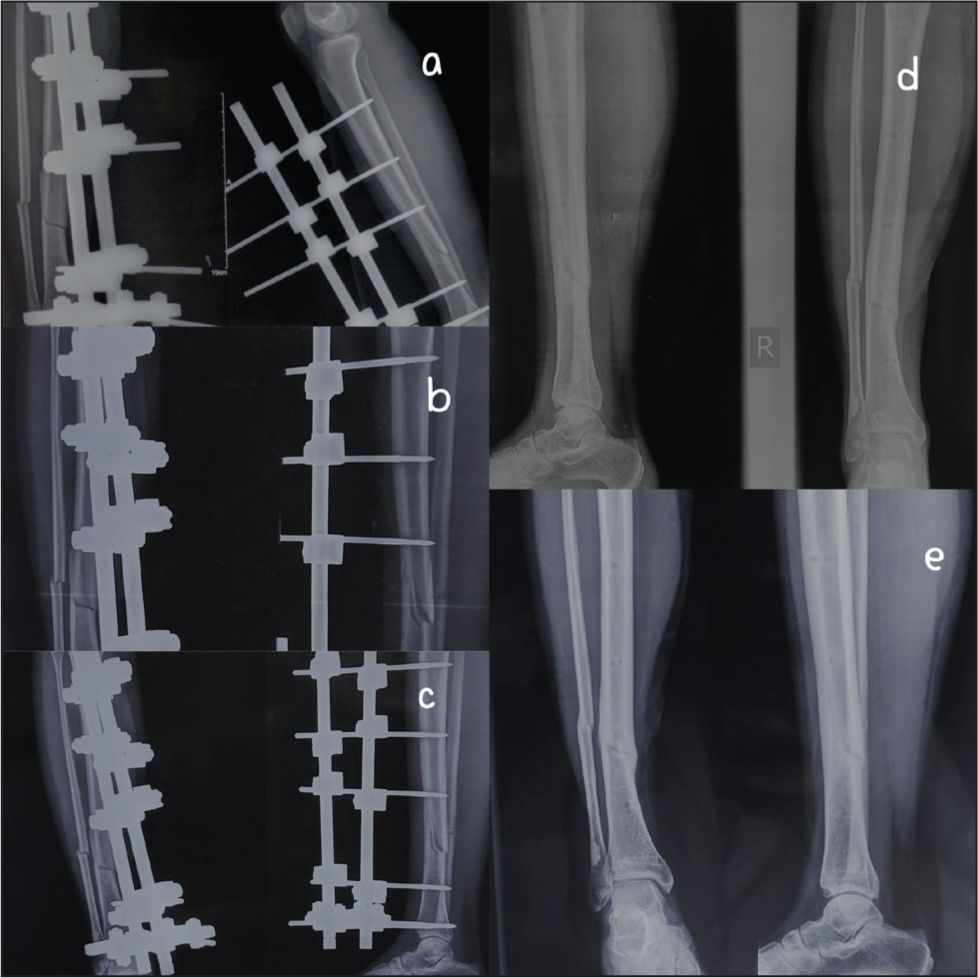
Radiographs of fracture union of a case of non-PRF group, (a) immediate post-op, (b) 2 months, (c) 6 months, (d) 1 year
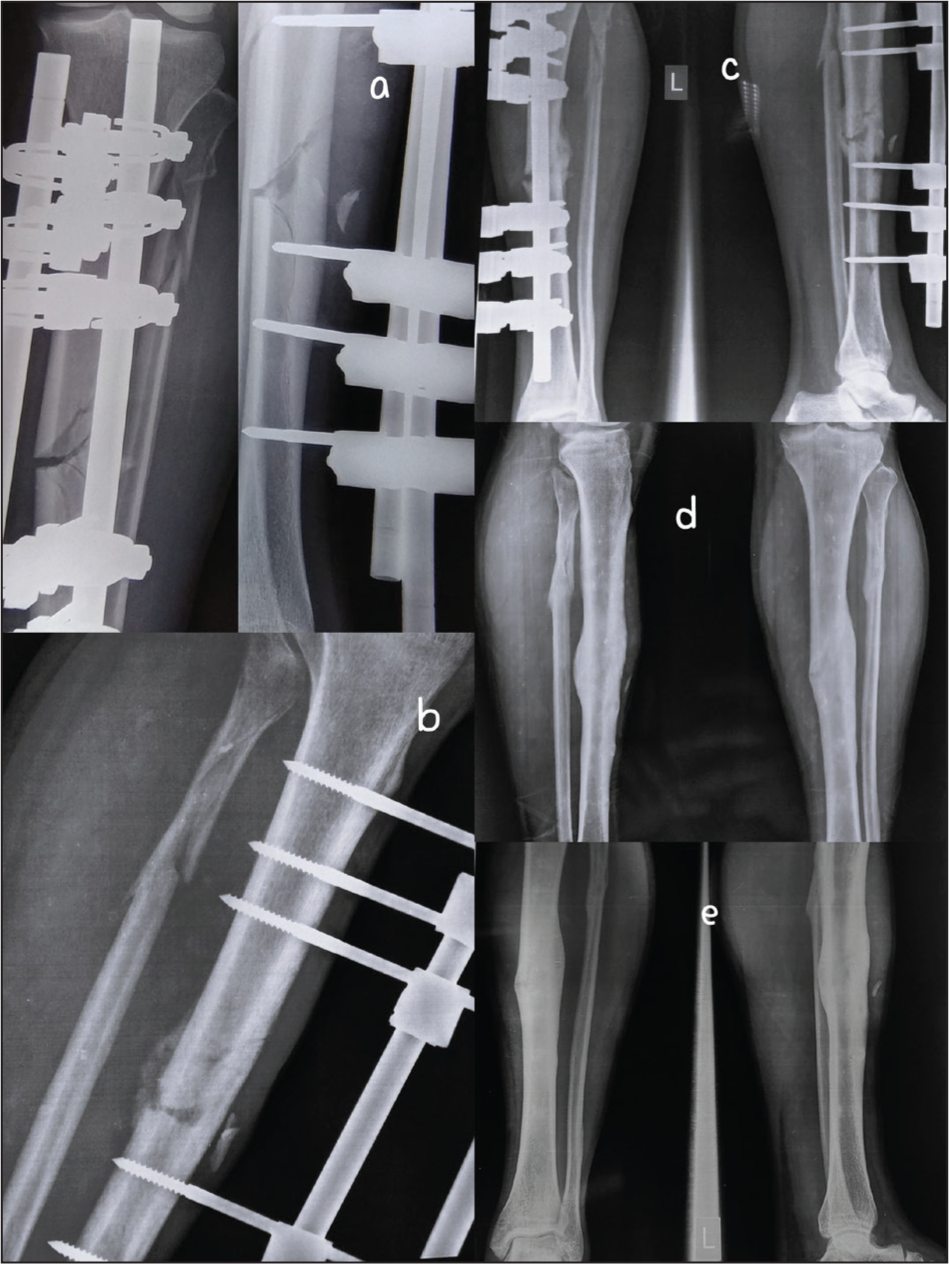
Radiographs of fracture union of another case of PRF group: (a) immediate post-op, (b) 2 months, (c) 3 months, (d) 6 months, (e) 1 year
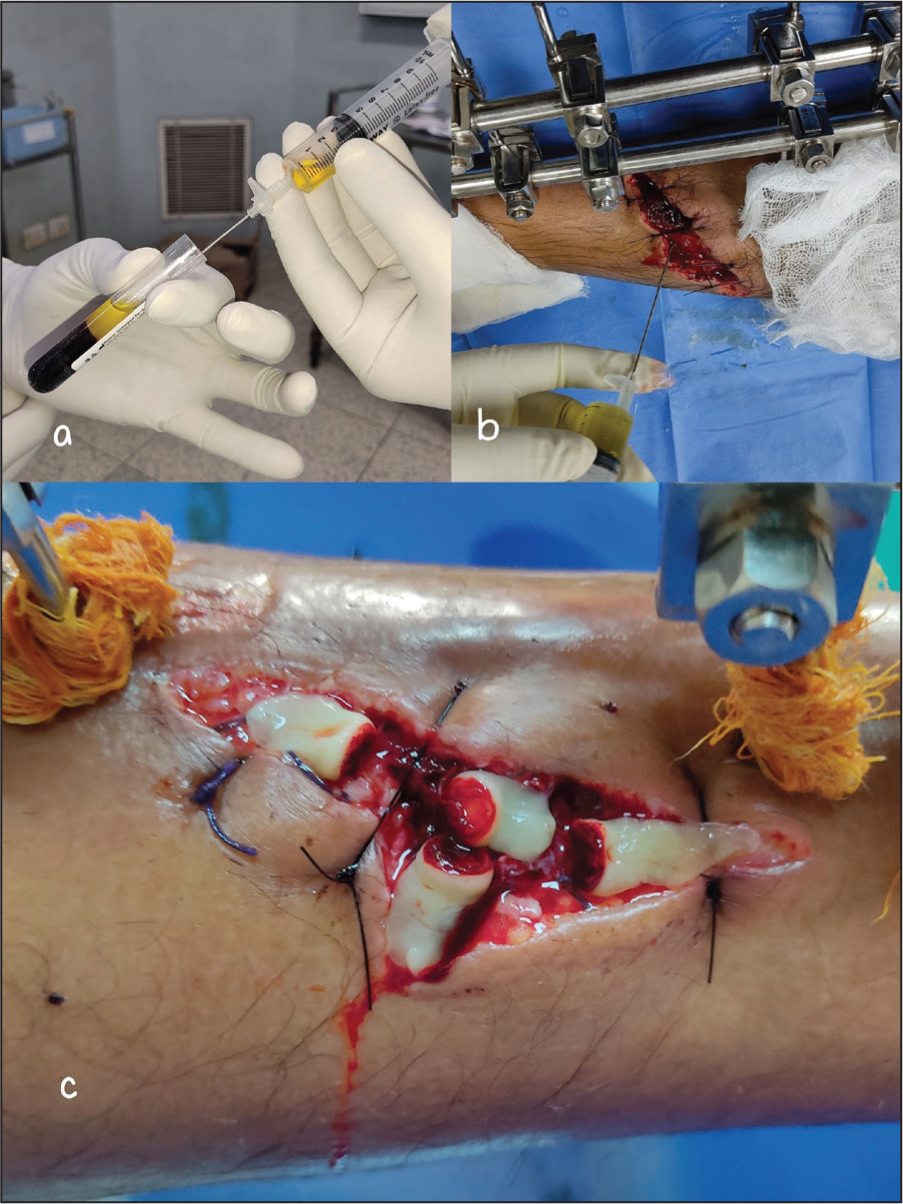
For preparing the liquid or injectable PRF [Figure 2a and b], the blood was centrifuged at 3300 rpm for 2 minutes. The resultant derivative consisted of only two layers, a liquid layer of PRF and the RBC clot. Not allowing a standing period, the liquid PRF was quickly aspirated using a 16–18 gauze needle in a sterile manner.[10] The resultant aspirate of i-PRF was injected at the fracture site.
Follow-up
In the PRF group, the dressings were done on day 3, day 7 and then biweekly till complete wound healing. In the non-PRF group, dressings were done using standard dressing protocol and were changed daily or on alternate days till complete healing. In either group, wound assessment was done on day 3, day 7 and then every week till complete wound healing and was compared between groups. Radiological assessment was done at 4 weeks, 2 months, 6 months and 1 year. The fracture union was defined by the formation of a bridging callus in three out of four cortices in anteroposterior (AP) and lateral views. The AO-type tubular external fixator was used as definitive fixation till the fractures showed clinical–radiological signs of the union. In those cases where the union was not achieved by the tubular external fixator, other modalities of fixations including ring fixator or intramedullary nailing with bone grafting were used subsequently.
Statistical Analysis
At the end of the study, data was collected and analysed by appropriate statistical tests including unpaired t-test and chi-squared test. All the assessments were done by the authors.
Results
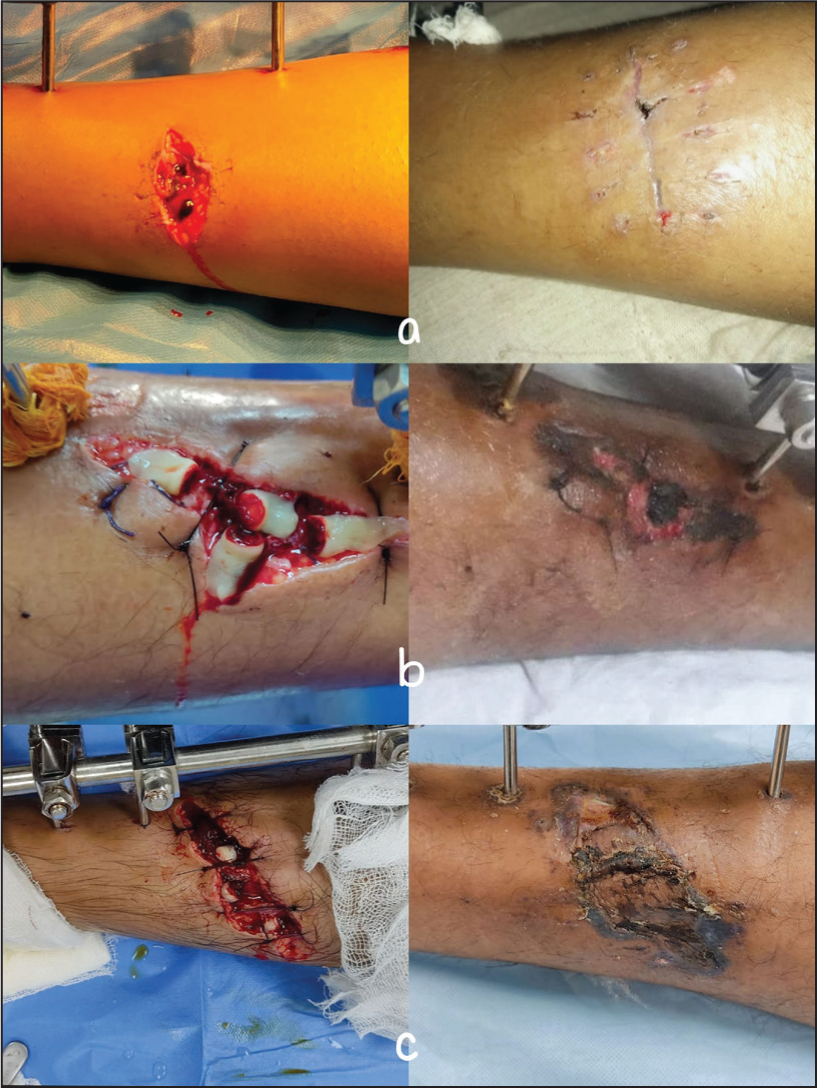
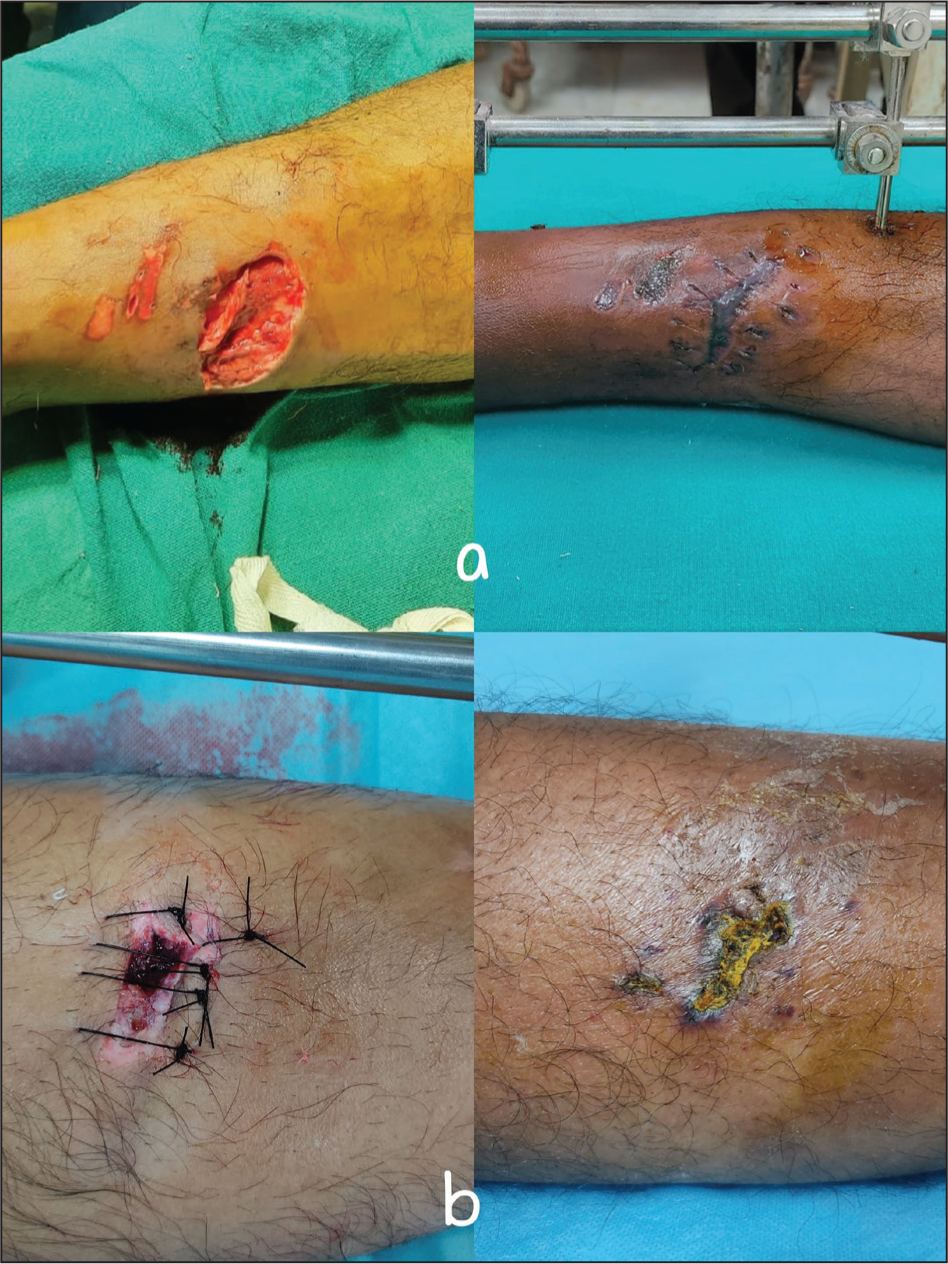
At the time of surgery, primary closure of the wound was possible in four out of eight patients in the PRF group and four out of six patients in the non-PRF group. The initial area of the wound where primary closure was not possible was slightly larger in the PRF group (7.87±1.43 cm 2 ) compared to the non-PRF group (7.5±1.5 cm 2 ), which was not statistically significant (p = .64). All wounds healed well and there was no local site infection in either group [Figures 3 and 4]. The time required for complete healing of those wounds, which had been primarily closed, was slightly faster (14.75±2.16 days) in the PRF group compared to (16±1.58 days) in the non-PRF group, though not statistically significant. The mean time for complete healing of the wound gaps was also slightly faster (36.25±3.03 days) in the PRF group compared to (42±7 days) in the non-PRF group, which was also not statistically significant. There were pin tract infections but no statistically significant difference between the groups.
Radiographs of fracture healing in the case of PRF group, (a) immediate post-op, (b) 2 months, (c) 3 months, (d) 6 months, (e) 2 years
Wound healing in non-PRF group
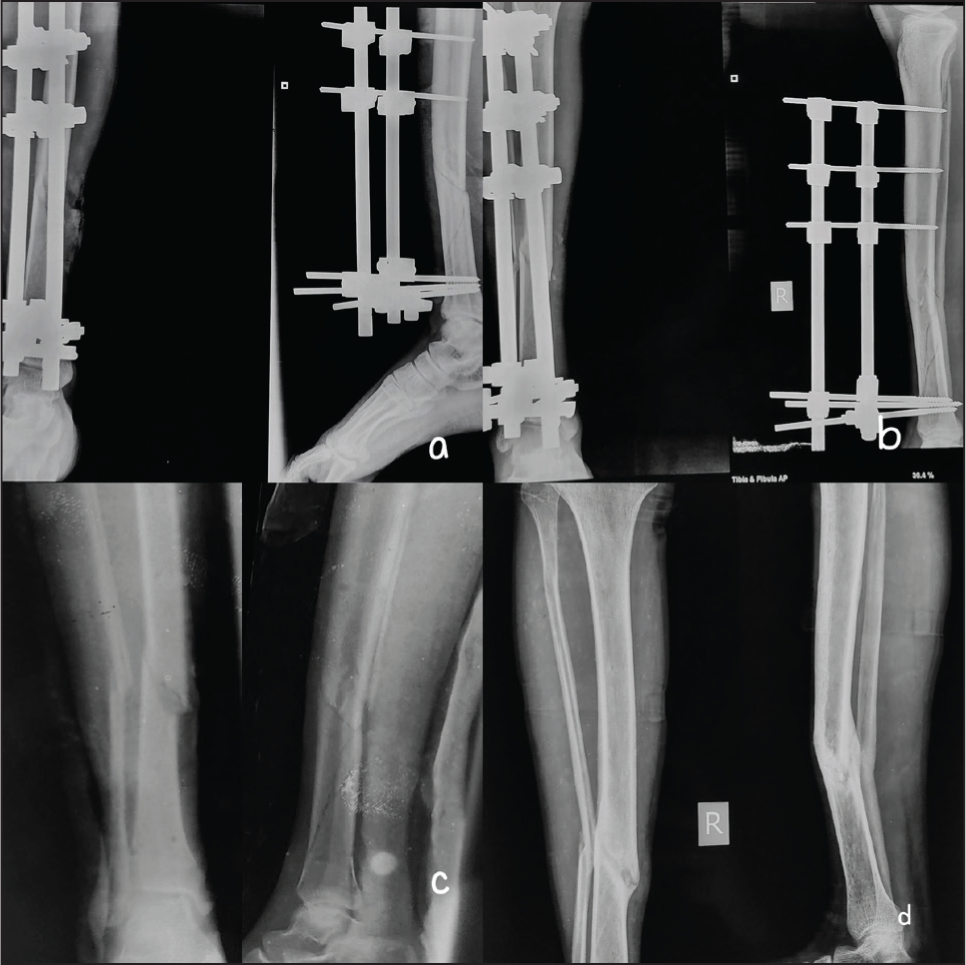
There was no fracture union till 3rd month in either group; however, one case in the PRF group showed a good amount of callous at 2 months [Figure 5]. At 6 months, seven out of eight (87.5%) fractures united in the PRF group [Figures 5 and 6] compared to three out of six (50%) in the non-PRF group [Figure 7] (p = .24). The remaining fracture of the PRF group united at 9 months using intramedullary nailing with autologous bone grafting. In the non-PRF group, three fractures which were un-united subsequently united after treatment by ring fixator with bone graft. The mean time for fixator removal was slightly faster (19.5±2.39 weeks) in the PRF group compared to (22.66±3.77) in the non-PRF group (p = .10). The fixators were removed just after the fractures showed radiological union. The results are summarised in Table 1.
Wound healing in PRF group. (c) skin grafting was done after early wound coverage with PRF matrix
(a) Aspiration of liquid injectable PRF (i-PRF) after centrifugating blood at 3300 rpm for 2 minutes, (b) liquid injectable PRF (i-PRF) being injected into the fracture site, (c) dressing of the wound with the PRF matrix (PRF gel)
Preparation of PRF. (a) Withdrawing of venous blood, (b) after centrifugation, vial showing three layers: uppermost layer of platelet-poor plasma (PPP), middle layer of PRF and lowermost layer of RBC clot, (c) after discarding the uppermost layer of PPP, PRF matrix is being taken out of the vial by using a blunt forceps, (d) PRF matrix is being separated from RBC clot, (e) PRF matrix (PRF gel) after separating from RBC clot
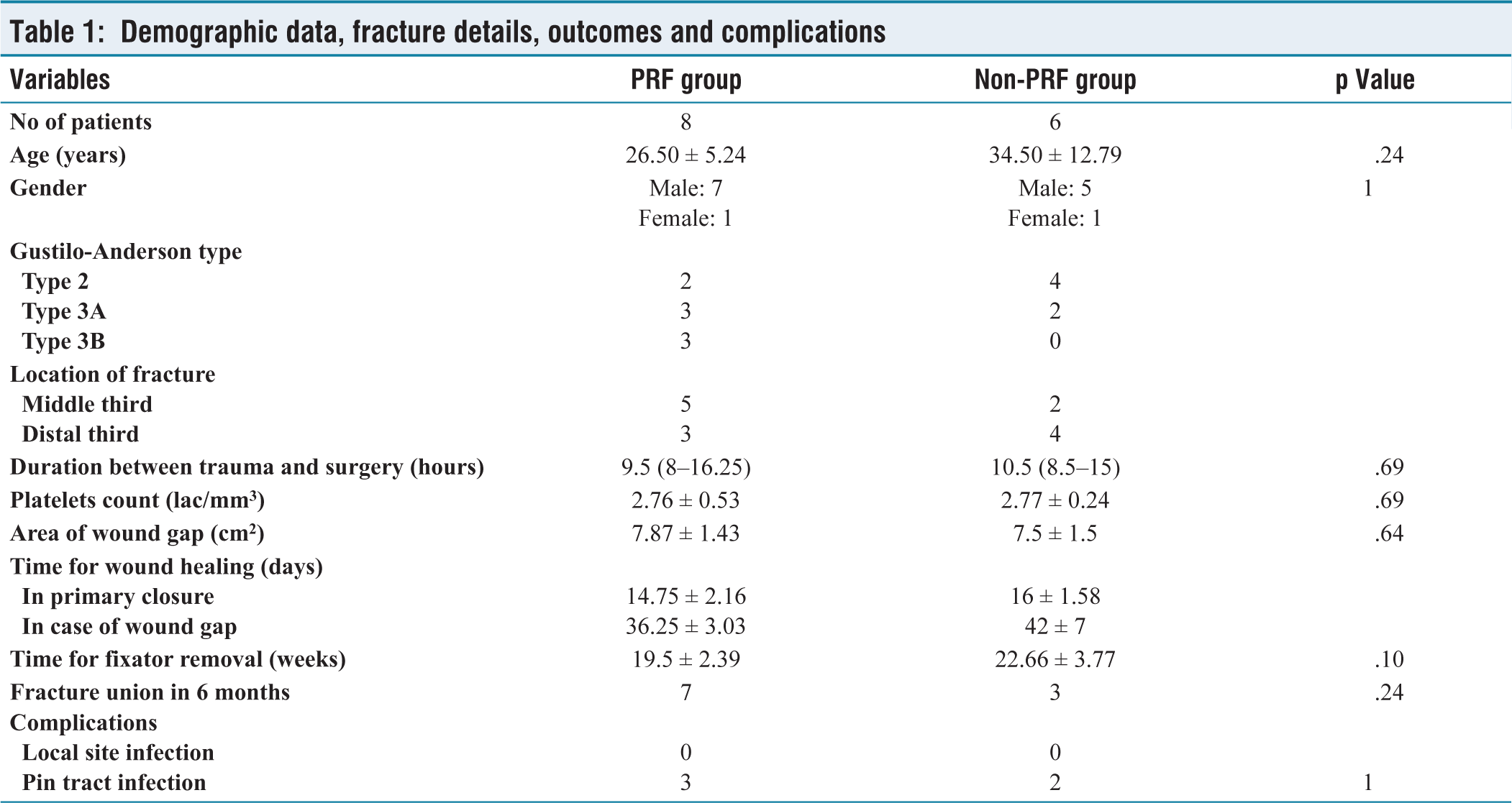
Demographic data, fracture details, outcomes and complications
Discussion
Management of soft tissue injuries and promotion of union remains a challenge in open fractures of the tibia. New studies are focusing on the use of autologous growth factors to increase the effect of wound healing and fracture union. The positive results obtained initially with PRP and then PRF correlate with the fact that platelets are active growth factor-secreting cells that initiate the wound healing process specifically connective tissue healing, bone healing, fibroblast mitogenesis, macrophage activation and cell proliferation.[11] Multiple studies reported positive results regarding the use of the PRF in oral and maxillofacial surgery,[12–15] for the treatment of cartilage injuries,[16] rotator cuff repair[17] and meniscal lesions.[17] O’Connell et al. showed complete healing in a majority of patients with severe non-healing venous ulcers treated with an autologous PRF matrix.[8] For the treatment of atrophic non-unions of upper limbs, Mazzotta et al. reported that the addition of bone marrow aspirate concentrate and PRF to lyophilised bone chips was safe and helped to accelerate healing time.[18]
Singh et al. used PRP in the acute femoral shaft fractures and showed accelerated healing in the initial phase of the union.[19] However, one of the disadvantages of PRP is the process of its preparation with anticoagulants which further requires thrombin for activation. Besides PRP causes a massive but short-lasting release of growth factors.[18] On the contrary, PRF is prepared without the addition of anticoagulants and presents a much slower and more natural activation, so that the release of growth factors and cytokines, such as platelet derived growth factor, transforming growth factor β-1 and insulin like growth factor-1 accompanies bone healing more physiologically. In addition, the PRF gel or matrix has a more elastic three-dimensional structure compared with PRP, which favours cell migration and retention of circulating growth factors.[20] There is a paucity of published studies showing the use of autologous PRF in acute long-bone fractures. In a recent study, Taufik et al. in June 2022 reported accelerated healing but only in three cases, that too with the additional use of bovine hydroxyapatite xenograft along with PRF, of which only one was of an open fracture of the distal humerus, the second was of an initially open distal femur fracture with subsequent healed bone defect and the third was a case of delayed union of tibia.[21]
With this background, this pilot study done to assess the effect of PRF in acute open fractures of the tibia concerning fracture union and wound healing found a positive result. In our study, we found that suture removal was earlier in PRF than non-PRF group, in those cases where the wounds were primarily closed. Also, though not statistically significant, in cases where the wound could not be closed, the healing time in the PRF group was marginally faster, even though the size of the wounds in the PRF group was larger. In the PRF group, the mean time for complete healing of wounds was 5.17 weeks with a mean initial wound size of 7.87 cm 2 , while in the non-PRF group, the mean complete healing time was 6 weeks with a mean initial wound size of 7.5 cm 2 . Thus, the larger wounds of the PRF group healed faster than the smaller wounds of the non-PRF group. There were no superficial or deep soft tissue infection in either group in our study. There were pin tract infections in three patients of the PRF group and two patients of the non-PRF group. All the pin tract infections in our study were of grade 1 (minor infection: slight redness, little discharge) and it got resolved by improved pin site care within 1 week in both groups.
None of the patients of either group showed signs of radiological union at 4 weeks; however, one case in the PRF group had callus at 2 months. At 6 months among all the patients, 10 patients showed signs of fracture union, 7/8 (87.5%) patients in the PRF group and 3/6 (50.0%) patients in the non-PRF group. Mazzotta et al. reported a higher radiographic score at 6 months, in patients treated with PRF in non-unions of the upper limb.[18] In our study of open tibial fractures, only one patient of the PRF group subsequently required intramedullary nailing with autologous bone graft, whereas in the non-PRF group, three patients needed conversion to ring fixator with bone grafting. Comparing the two groups, it was found that radiological union was earlier in the PRF group than in the non-PRF group by about 3.8 weeks. Even the external fixator was removed much earlier in the PRF group compared to the non-PRF group.
Though the strength of our study was computer-generated randomisation into two groups, the main limitation was the small sample size. This was done as a pilot study, due to a paucity of literature mentioning the effect of PRF in wound healing and fracture union in open fractures of the tibia. As the efficacy and safety were not known, the sample size was kept smaller, considering that open fractures of the tibia are prone to infected non-unions. After completion of the recruitment of cases of our study by 2021, a recent study in 2022 by Taufik et al. reported only one case of acute open distal humerus fracture out of the total three cases, where PRF was used along with bony hydroxyapatite matrix.[21]
Despite the small sample size limitation, this study is of clinical relevance showing promising results of PRF, both in wound healing and fracture union and adds to the growing literature on the use of PRF with an encouraging and safe outcome. Probably this study is the first in the English literature showing the effect of PRF in acute open fractures of tibia. It is recommended to conduct more multi-centric studies with a larger sample size to be able to highlight a statistically significant result.
Conclusion
The use of injectable as well as matrix PRF was found to be safe in open fractures without causing superficial or deep infection, immediate or late. Larger wounds healed with the PRF matrix slightly faster than the smaller wounds of the non-PRF group. More number of fractures united in the PRF group and all of them united earlier than the non-PRF group.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional ethical committee approval number
Ethical approval was taken prior to the study from the Institutional Ethics Committee, Maulana Azad Medical College and associated hospitals, Delhi (Approval number: F.1/IEC/MAMC/70/05/2019/No528).
Informed consent
Informed consent was taken from all the patients.
Credit author statement
S.S. was involved in conceptualization, designing and manuscript editing.
S.M. contributed to literature search, data acquisition, data analysis, statistical analysis and manuscript preparation.
The manuscript has been read and approved by all the authors. All the requirements for authorship as mentioned have been met. All authors believe that the manuscript represents honest work.
Data availability
The corresponding author has all the data provided in this article.
Use of artificial intelligence
Artificial intelligence has not been used.
