Abstract
Background and Aims:
Neuropathic pain refers to the illness that affects the somatosensory nerve system which is characterised by spontaneous pain and pathologically increased sensitivity to both noxious and benign stimuli. The purpose of this study was to compare the short-term therapy of young adults with chronic neuropathic pain with gabapentin in order to assess the safety and effectiveness of pregabalin.
Materials and Methods:
In cooperation with the Department of Pharmacology at a tertiary care facility in India, the outpatient departments of neurology, orthopaedics, general medicine, and psychiatry undertook this prospective, comparative, randomised, open-label, single-centre cross-sectional study. One hundred and thirty-five patients in Group A received 75 mg of pregabalin and were then given 150 mg of pregabalin twice a day. One hundred thirty-five participants in Group B received 300 mg of gabapentin. The Numeric Pain Rating Scale (NPRS) was used to quantify pain intensity at baseline, 1 month, and 2 months later in order to compare the negative effects of pregabalin to those of gabapentin.
Results:
When comparing the two groups’ NPRS scores at baseline, 1 month, and 2 months, Group A (pregabalin) had a comparatively lower NPRS score. At baseline, Group A had a mean ± SD score of 8.5 ± 1.25, when compared to Group B with 8.7 ± 1.20. After 2 months, Group A had a mean ± SD score of 2.5 ± 1.50, when compared to Group B with 4.5 ± 1.80, respectively, which was statistically significant with P < .001.
Conclusion:
This study suggests that gabapentin and pregabalin are equally effective at reducing pain in persons with neuropathic pain. In terms of NPRS score, pregabalin performs better than gabapentin.
Introduction
Neuropathic pain refers to the illness that affects the somatosensory nerve system which is characterised by spontaneous pain and pathologically increased sensitivity to both noxious and benign stimuli.[1] Changes in the way that sensory impulses are transmitted to the brain and spinal cord can result from lesions in the somatosensory nerve system. Every year, 7%–10% of the general population experiences neuropathic pain. Numerous reasons for neuropathic pain have been identified, and as the world’s population ages, the prevalence of diabetes mellitus will likely rise, and chemotherapy-induced cancer survival rates will likely rise.[2] Crucially, inflammatory pain – which arises, for instance, in rheumatoid arthritis, where inflammation is the primary cause with altered chemical events at the site of inflammation – is mechanistically distinct from neuropathic pain and is treated and diagnosed differently.[3] Neuropathic pain is linked to more prescription drug usage and medical visits.[4]
A unique collection of symptoms is usually experienced by patients, including burning and electrical-like sensations as well as pain from non-painful stimulations (such as light touching); these symptoms tend to linger, become chronic, and be less responsive to painkillers. Patients with neuropathic pain frequently experience significant anxiety, sadness, and sleep disruptions. Their quality of life is also more compromised in neuropathic pain patients than in non-neuropathic pain patients, who do not experience pain stemming from damaged or irritated nerves.[5] Neuropathic pain is characterised by a number of signs and symptoms, including allodynia (pain brought on by a stimulation that typically does not cause pain), hyperalgesia (an increase in the perception of pain generated by a stimulus that causes pain), and paresthesia (a condition that determines the perception of anomalous sensations comparable to needle bites, tingling, itching, reduced, or even loss of sensitivity). Neuropathic pain sufferers typically experience pain that is spontaneous and arises in response to no external stimuli. Patients’ psychological well-being is compromised by this pathological condition, which has a prominent negative effect on their quality of life.[6] Gabapentin, Pregabalin, Serotonin and norepinephrine reuptake inhibitors (SNRIs), and tricyclic antidepressants (TCA) are recommended for use as first-line treatment and were among the recently revised worldwide applied NeP pharmacotherapy recommendations from the special interest group on neuropathic pain.[7]
Tricyclic antidepressants (TCAs), selective SNRIs, nonsteroidal anti-inflammatory drugs (NSAIDs), opioids, and antiepileptic medications (AEDs) are among the many pharmacological classes that have been used to treat neuropathic pain. While each of these seminars has its limits, they have all shown some degree of efficacy. An anticonvulsant with analgesic and anxiolytic qualities is pregabalin. It functions as a structural analogue of the neurotransmitter gamma-aminobutyric acid (GABA), mediating its effects through binding to CNS voltage-gated calcium channels. Pregabalin functions as a high-affinity, selective ligand for the voltage-gated calcium channel’s α2-δ subunit, which is believed to be crucial in regulating neuropathic pain.[8-10]
An effective anticonvulsant and analgesic is pregabalin, which was approved by The Food and Drug Administration (FDA) as the first medication for the treatment of postherpetic neuralgia and neuropathic pain.[11] Studies in both preclinical and clinical settings have demonstrated how well pregabalin works to treat neuropathic pain. Research using animals has aided in elucidating the processes behind its anti-hyperalgesic and antiallodynic effects. Clinical research has also demonstrated the effectiveness of pregabalin in treating pain and associated symptoms, either alone or in conjunction with analgesics, as well as its dose-dependent effects. Pregabalin’s main benefits for neuropathic pain patients include its high tolerance, ease of use, and relative reliability.[12]
A frequent treatment for post-herpetic neuralgia (PHN) is gabapentin (GBP). GBP acts through altering the neurotransmitter release and lowering excitability of nerve cell by binding with higher affinity towards alpha-2-delta subunit of the calcium channels (voltage-gated) found in both peripheral and central nervous systems. It is this mechanism of action that may produce the analgesic effect in patients experiencing neuropathic pain.[13,14] The purpose of this study was to compare the short-term therapy of young adults with chronic neuropathic pain with gabapentin in order to assess the safety and effectiveness of pregabalin.
Materials and Methods
This is a prospective, randomised, comparative, single centre and open-label cross-sectional study conducted at neurology, orthopaedics, general medicine, and psychiatry outpatient departments in collaboration with the Department of Pharmacology at a tertiary care institute of India. Following Institutional Ethics Committee (IEC) permission, 270 patients were randomly assigned to 2 groups. Before any patients were included in the trial, their informed consent was obtained. Group A included 135 patients who received Pregabalin 75mg twice daily orally for 7 days initially and the dose of pregabalin increased to 150mg orally twice daily from the 8th day. Group B included 135 patients who received Gabapentin of 150 mg twice daily on the first day and 300mg orally twice daily from 2nd day onwards. The Numeric Pain Rating Scale (NPRS) was used to quantify pain intensity at baseline, 1 month, and 2 months later in order to compare the negative effects of pregabalin to those of gabapentin. Participants in the study had to be willing to participate and give written and informed consent. Patients of either gender and age group (20–45 years) who were diagnosed with chronic lumbar radiculopathy depending on the symptoms, clinical assessment, MRI scan and X-ray of the lumbosacral spine were included in the study. individuals with a background of diabetes, liver and kidney disorders, heart illness, TB, and renal disease. Exclusions from the trial were women who were pregnant or nursing, patients with immunocompromised conditions, patients with radiculopathy resulting from tumours, and patients who had a history of medication hypersensitivity. By convenient sampling and after obtaining informed consent, a total of 270 patients were included from outpatient departments of medicine, orthopaedics, neurology, and psychiatry. Following enrolment and before treatment started, the following tests were recorded: clinical assessment for chronic lumbar radiculopathy, digital X-ray of the lumbosacral spine in anteroposterior and lateral views, physical examination, systemic examination including vitals, MRI scan of the lumbosacral spine, and past medical history. NPRS were used to assess pain at the beginning of the trial (0 day), 1 month, and 2 months). All the patients between 20 years to 45 years who are attending the outpatient department and are on treatment with pregabalin and gabapentin will be examined and history taken after obtaining informed consent. Age, gender, additional drug use, history of falls within the past year, dizziness, and peripheral oedema will be evaluated for all cases, and physical examinations will be performed. The ADR reporting form was used to report any adverse drug reactions that were noticed by the clinician or reported by the patient during the trial.
Gabapentin 300 mg once daily as the starting dose, then 300 mg twice daily starting on day two. High dosages of gabapentin (900–180,000 mg/day) have been utilised in some studies; however, some patients may not be able to tolerate these levels of side effects. It’s possible that low dosages of gabapentin produce satisfying effects.
Statistical Analysis
All the data was represented as Mean ± SD with Microsoft Excel 2019 and SPSS v 24.0 (“IBM Corp. Released 2016. IBM SPSS Statistics for Windows, Version 24.0. Armonk, NY: IBM Corp.”). Students unpaired t-test was applied for obtaining the P value significance between cases and controls. A P value of .05 or less was regarded as statistically significant.
Results
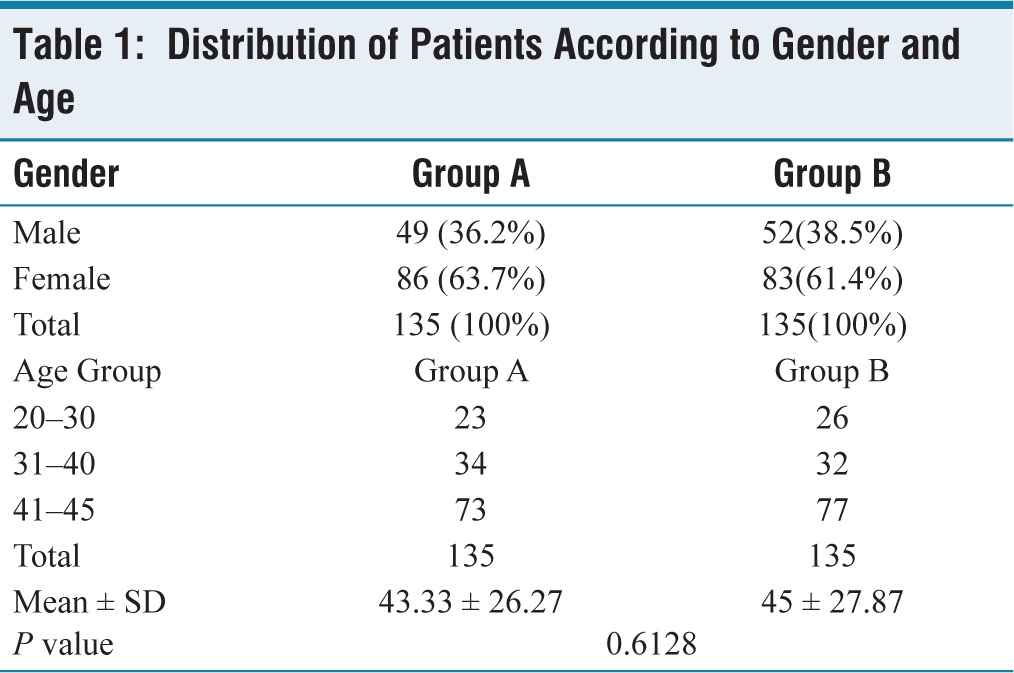
There were 135 patients in total in each group. Within Group A, there were 86 (63.7%) girls and 49 (36.2%) males. Fifty-two (38.5%) men and 83 (61.4%) women made up Group B. In Group A: the mean age of patients was 43.33 ± 26.27 years. In group B the mean age of patients was 45 ± 27.87 years (Table 1). Most number of patients suffering from Neuropathic pain fall in the age group of 41–45 years, 86 (63.7%) in Group A and 83(61.4%) in Group B.
Distribution of Patients According to Gender and Age
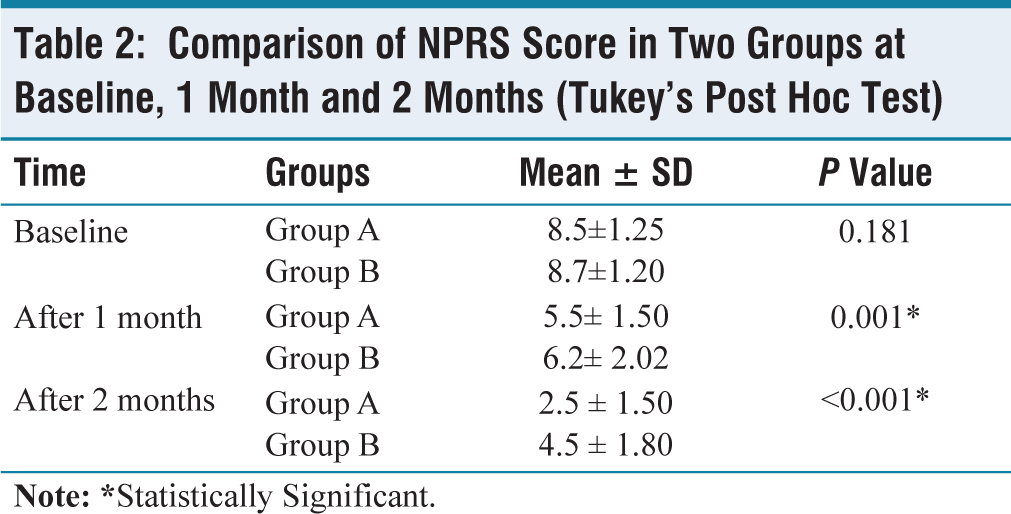
NPRS score when compared among the two groups at baseline, at 1 month and 2 months, the NPRS was relatively lower in Group A (Pregabalin). At baseline, Group A has a mean ± SD score of 8.5±1.25, when compared to Group B with 8.7±1.20. After 1 month, Group A had a mean ± SD score of 5.5±1.50, when compared to Group B with 6.2±2.02 respectively, which was statistically significant with a P = .001. After 2 months, Group A had a mean ± SD score of 2.5±1.50, when compared to Group B with 4.5±1.80 respectively, which was statistically significant with P < .001 (Table 2).
Comparison of NPRS Score in Two Groups at Baseline, 1 Month and 2 Months (Tukey’s Post Hoc Test)
In this study, the incidence of dizziness has been significantly higher among 34 patients of group B (25.1%) as compared to group A among 30 patients (22.2%). The sedation was observed in 42 patients of Group B (31.1%) and constipation was seen in nine patients of Group B (6.6%) and four patients of Group A (2.4%). Dryness of mouth was observed in both groups, the drowsiness is predominantly seen in 26 individuals in group B (19.2 %) as compared to that of 21 patients (15.6%) in Group A, with P = .732 (Table 3). Sedation and Drowsiness are not the same adverse effect. Sedation is the slower reaction time with impaired performance in activities that require diligent attention Drowsiness is a subjective feeling of being tired or being unable to keep your eyes open during the day.
Adverse Drug Reaction in Patients in Two Groups

Discussion
At the end of the 2-month study, pregabalin demonstrated superior pain reduction over gabapentin., which is in line with the study by Dongre et al., that supported the better pain reduction scores of pregabalin in comparison to Gabapentin.[15] At the end of the 2-month study, pregabalin was more effective than gabapentin in lowering pain. This effect is explained by the agonistic activity of pregabalin on a certain subset of the GABA-B receptors, that inhibit hyper-excitability and abnormal activity of sensory neurons, thereby reducing pain. It also further causes negative regulation of the alpha-2-delta subunit of voltage-gated calcium channels, blocks calcium and Na± channels and activates inwardly rectifying K± channels.[16]
In contrast with the previous research that has revealed a male predominance, the results of this study show a female predominance in either treatment groups. This may be caused by differences in geography, a lower pain threshold, or the emotional susceptibility of women. Within five to six days of beginning medication, patients who got gabapentin reported significant pain alleviation and an expected reduction in pain score. Pregabalin-treated individuals also showed a good reduction in pain score, matching that of previous studies, and reported pain alleviation within four days after beginning medication. A study conducted in India by Bansal et al. also found that pregabalin relieves pain significantly more quickly and effectively than gabapentin.[17] There aren’t many other placebo-controlled trials that used pregabalin for 5 or 8 weeks that show a discernible decrease in pain when compared to placebo (P < 0.001).[18]
There were no uncommon or severe adverse events reported in the safety and tolerability evaluations for this study. For those suffering from neuropathic pain, the dose-limiting side effects continue to be a significant issue. This study’s tolerability profile was largely in line with that of the earlier research.[19,20] Even though gabapentin and Pregabalin share a relative mechanism of action as both suppress calcium influx and the release of excitatory neurotransmitters that follow, their pharmacokinetic and pharmacological properties are different. Following oral dosage, gabapentin is absorbed slowly, reaching maximum plasma concentrations in 3–4 hours. Because of its saturable absorption, which is a nonlinear, zero-order process, gabapentin taken orally has less predictable pharmacokinetics. Gabapentin plasma concentrations do not rise in direct proportion to dosage. On the other hand, pregabalin taken orally absorbs more quickly, reaching its maximal plasma concentrations in about an hour. First-order linear absorption results in plasma concentrations rising in direct proportion to dosage.[21]
The authors have analysed the differences between the two groups by applying a percentage reduction in the major efficacy metric. To the best of our knowledge, percentage reduction which is better sensitive among a smaller number of individuals than basic absolute parameters has not been employed in many investigations. A few restrictions apply to this study. There was no blinding in this open-label investigation. Because the patients were only followed for 2 months, it was not possible to evaluate the study medications’ long-term safety and efficacy.
Conclusion
This study suggests that gabapentin and pregabalin are equally effective at reducing pain in persons with neuropathic pain. In terms of NPRS score, pregabalin performs better than gabapentin. Additionally, patients are more likely to adhere to long-term treatment because less side effects have been reported with pregabalin. The cost-effectiveness of pregabalin compared to gabapentin is a significant factor for professionals caring for patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional Ethical Committee Approval Number
Ethical approval /clearance has been obtained from the Institutional Ethics Committee of Great Eastern Medical School and Hospital (GEMS) vide letter no. 127/IEC/GEMS&H/2022.
Informed Consent
Written Informed consent was taken from each patient before collecting the information from them.
CRediT Author Statement
Neeraja Rani Guruvu: Conceptualisation, data collection, manuscript writing, editing and submission. Bapugouda
Patil: Conceptualisation, data collection, and manuscript writing.
Dayakar Marri: Conceptualisation, data collection, manuscript writing.
L. V. Simhachalam Kutikuppala: Manuscript writing and editing.
Tirumalasetty Devika: Manuscript Writing, editing and review.
Katiboina Srinivasa Rao: Manuscript writing, editing and review.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Use of Artificial Intelligence
None of the Artificial Intelligence tools were used in preparation of this work.
