Abstract
Background and Aims:
A major economic and social burden is associated with chronic obstructive pulmonary disease (COPD), an incurable lung illness that affects people all over the world. With a rising incidence rate, COPD is predicted to overtake all other causes of mortality by 2030. This study employs the BMI, Obstruction, Dyspnea scale, exercise tolerance (BODE) index as a multidimensional staging approach to evaluate the prescription pattern, safety, and efficacy of therapy in patients with COPD. The body mass index (BMI), forced expiratory volume in one second (FEV1), modified british medical research council questionnaire (MMRC) Dyspnoea scale, and the 6-minute walk test are among the factors that the BODE index uses to classify the severity of an illness.
Methods:
An observational and prospective study was conducted at a government teaching hospital’s outpatient department for one and a half years. Demographic, clinical, and treatment data were collected from patients with COPD, and severity was assessed using the BODE index. The study aimed to evaluate prescription trends, treatment effectiveness, and safety, with primary and secondary objectives.
Results:
Out of 100 enrolled participants, 79 completed follow-ups. Sociodemographic analysis revealed a male predominance (83.54%), with the highest prevalence in the 51–70 age group. Most patients exhibited moderate COPD severity (51.9%), based on the BODE index. Prescriptions included inhalation therapy (94.94%), predominantly combining corticosteroids and bronchodilators. Assessing treatment effectiveness, the BODE index significantly improved over 6 months (P = 0.0002), demonstrating reduced disease severity. Adverse drug reactions occurred in 13.92% of participants, primarily mild in nature.
Conclusion:
The study highlights prevalent prescription patterns, emphasising inhalation therapy, and demonstrates the effectiveness and safety of the selected treatments using the BODE index. The findings underscore the importance of tailored therapeutic approaches in COPD management.
Keywords
Introduction
The lung condition known as chronic obstructive pulmonary disease (COPD) is not entirely reversible and is characterised by a chronic obstruction of lung airflow that makes it difficult to breathe normally.[1] It is a major global source of illness and mortality, resulting in a significant and growing economic and social cost.[2] The most recent WHO figures state that 3 million individuals have died from COPD and that 64 million people now have the disease. According to World Health Organization (WHO) projections, COPD will overtake all other causes of mortality globally by 2030.[3] Any patient experiencing dyspnoea, a persistent cough or sputum production, and a history of exposure to disease-related risk factors should be evaluated for COPD. There are other ways to evaluate COPD; in this case, we used the BMI, Obstruction, Dyspnea scale, exercise tolerance (BODE) index, a multidimensional staging approach that takes into account both systemic and pulmonary disease manifestations. The four dimensions of this index are the following: the body mass index (BMI), which measures airway obstruction and cachexia; the forced expiratory volume in one second (FEV1), which measures dyspnoea severity; the modified british medical research council questionnaire (MMRC) Dyspnoea scale, which measures dyspnoea severity; and the 6-minute walk test, which measures exercise tolerance. The BODE index was built on a scale of 0 to 10, where 10 represented the most severe disease and the highest mortality.[4-6]
Pharmacological and non-pharmacological approaches are used to treat COPD symptoms. The primary categories of drugs given to COPD subjects are Beta 2 agonists, anticholinergics, methylxanthines and corticosteroids. Inhalation therapy plays an important role in management of COPD and is being extensively used. 6
Aims and Objectives
Aim
The aim of this study was to evaluate the prescription pattern, treatment effectiveness and safety in COPD outdoor patients.
Primary Objective
The primary objectives of the study were to evaluate the current trend of prescribing the patterns of the drugs used in COPD and to evaluate the effectiveness of therapy in patients of COPD using the BODE index.
Secondary Objectives
The secondary objectives of the study were to evaluate the association of sociodemographic parameters with COPD, to evaluate the severity of disease in patients with COPD using the BODE index, and to evaluate the adverse drug reactions observed during the study period.
Materials and Methods
Study Design
This was a prospective observational study.
Study Site and Duration
The study was conducted at the outpatient department of Tuberculosis (TB) and chest department in a government teaching hospital for one and half years.
Study Criteria
Inclusion Criteria
The inclusion criteria of the study were patients of all ages and from both genders, who had visited the outpatient department of TB-CD and were diagnosed with COPD alone or along with comorbidities (non-respiratory).
Exclusion Criteria
The exclusion criteria of the study were patients diagnosed with other respiratory disorders such as bronchial asthma, pneumonia and tuberculosis.
Patients not willing to participate and pregnant/lactating mothers were also excluded from the study.
This study was approved by the Institutional Ethics Committee of Govt. Medical College.
Patients’ demographic data, clinical data and treatment data were collected from patients’ case file and by interviewing patients. For assessment of severity, the BODE index was used.
This index uses four dimensions to classify the severity of COPD, BMI (as a measure of cachexia and muscle wasting), forced expiratory volume in one second-FEV1 (to measure airway obstruction), MMRC Dyspnoea scale (to measure severity of dyspnoea) and 6-minute walk test (to measure exercise tolerance A scale of 0 to 10 was used for constructing the BODE index, where 10 represented the most severe illness with the highest probability of mortality. Assessment was done at first visit and then at successive 3 and 6 months of follow-up by interviewing patients.
Results
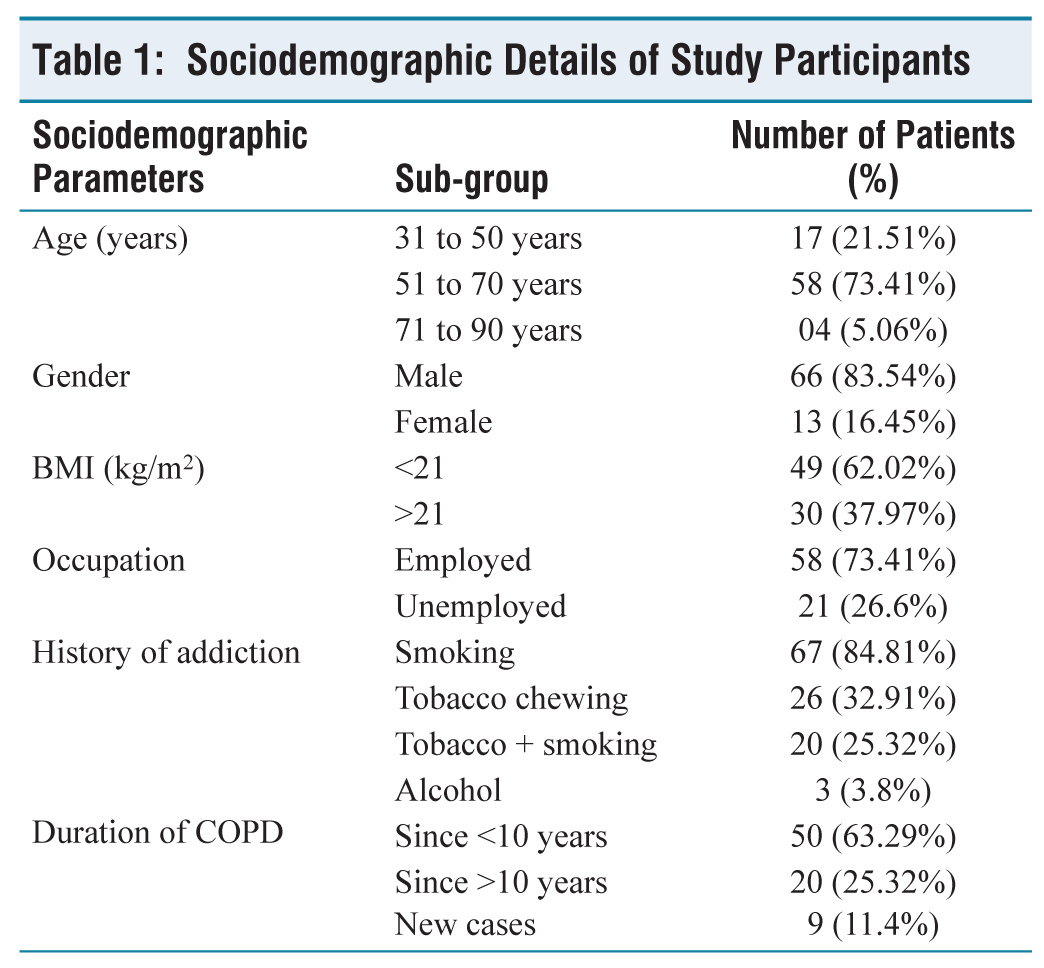
Out of the total 100 enrolled participants, 79 were evaluated as they completed two subsequent follow-up on the third and sixth month after their first enrolment. Twenty-one study participants were excluded from final analysis, 16 were unable to perform pulmonary function test and five participants were lost to follow up. Sociodemographic details and comorbidities of participants are mentioned in Table 1 and Figure 1, respectively.
Sociodemographic Details of Study Participants
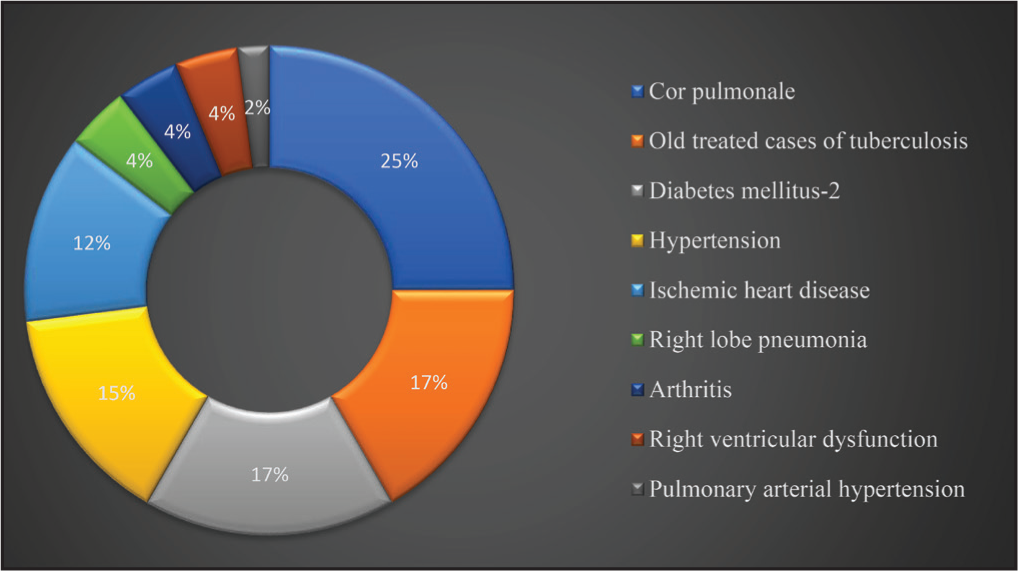
Distribution of Comorbidities Associated With COPD
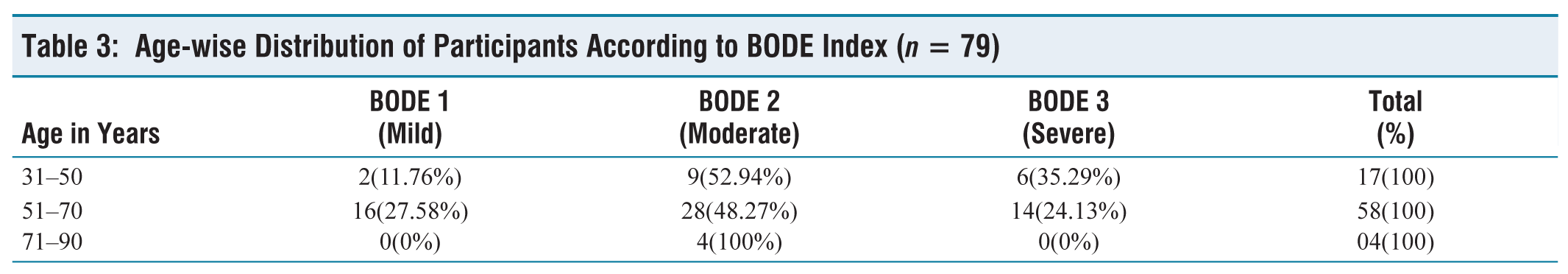
Severity of disease, age distribution and mMRC grading according to the BODE index are given in Tables 2 and 3. According to BODE index, we found maximum number of patients 41(51.9%) in moderate category followed by severe (25.31%) and mild category (22.8%).
Severity of COPD According to BODE Index

Age distribution of participants according to BODE index (Table 3) shows that maximum number of participants in 51–70 years age group, that too in moderate category followed by severe and mild. But we did not find statistically significant association between age and BODE index.
Age-wise Distribution of Participants According to BODE Index (n = 79)
mMRC grading is one of the components of BODE index. Table 4 shows the distribution of participants according to mMRC grading and BODE index. Maximum number of participants was in the moderate category according to mMRC grading.
Distribution of mMRC Grade According to BODE Index (n = 79)

mMRC grade, modified British Medical Research Council (mMRC) grade.
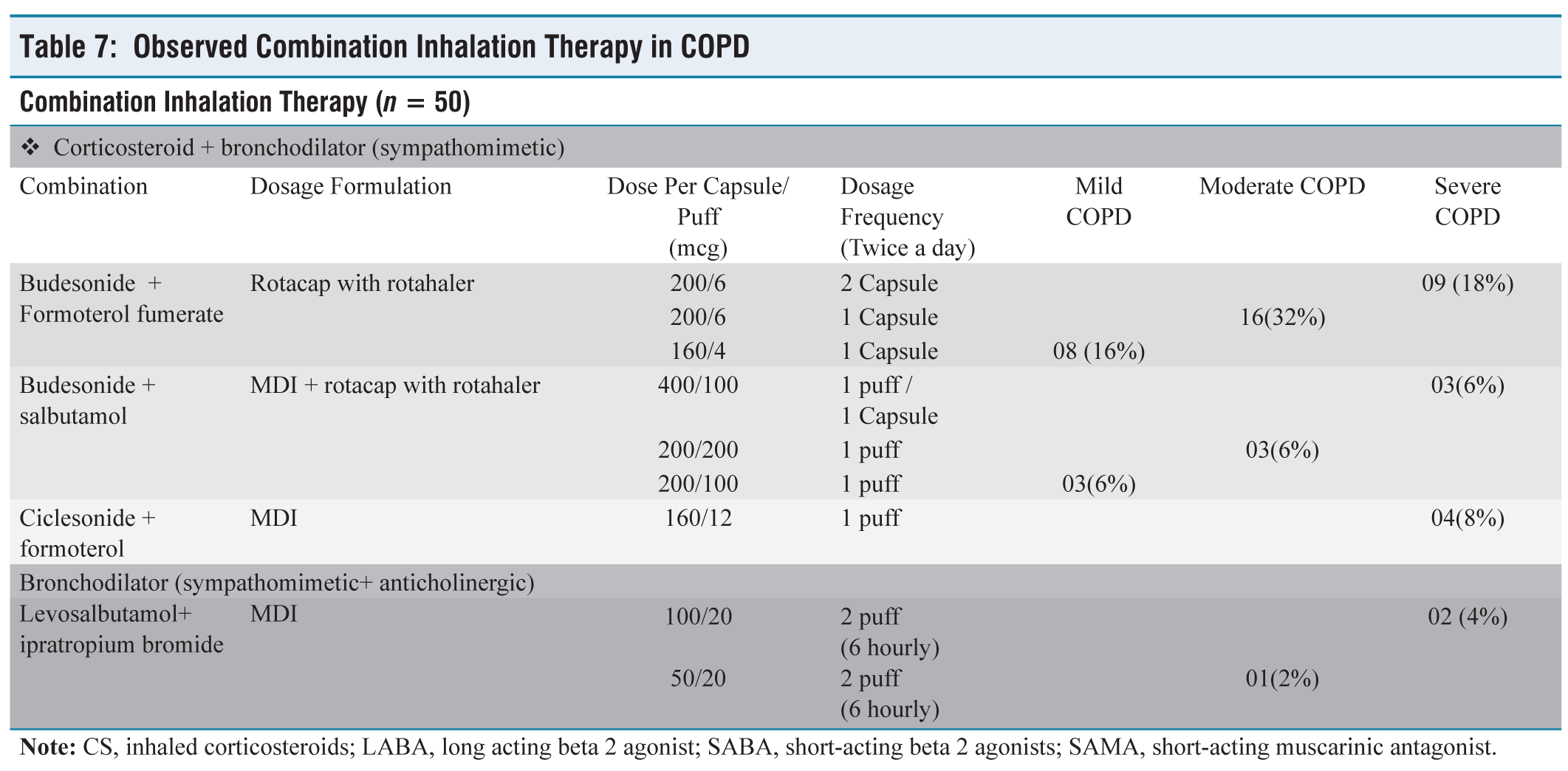
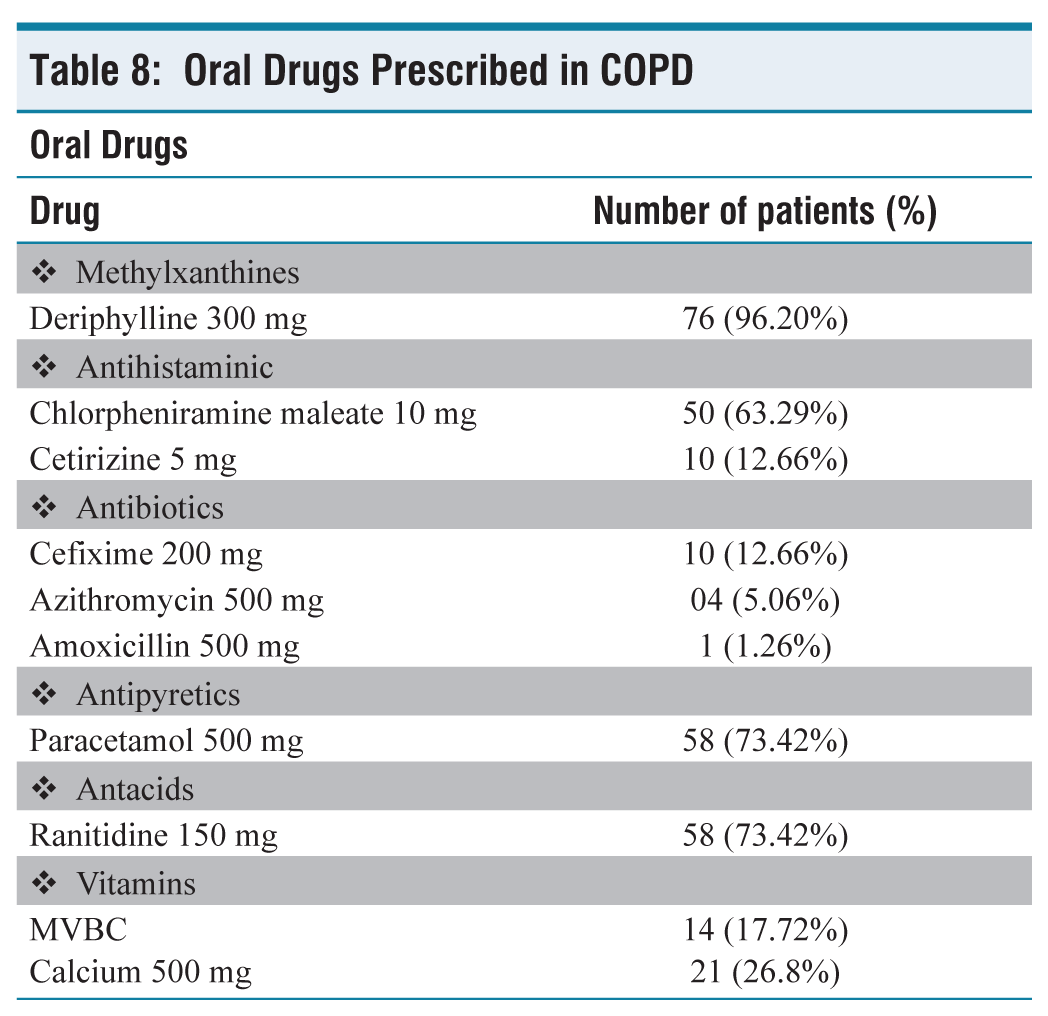
Prescriptions were evaluated according to prescribed drugs, dosage and routes of administration. Injectable, inhaled and oral drugs were prescribed and evaluation is given in Tables 5–8. Total 74 patients received inhalation therapy out of 79, among them 50(67.6%) received combined inhalation therapy and 24(32.43%) received single-drug inhalation therapy (Tables 6 and 7).
Injectable Drugs in Treatment of COPD
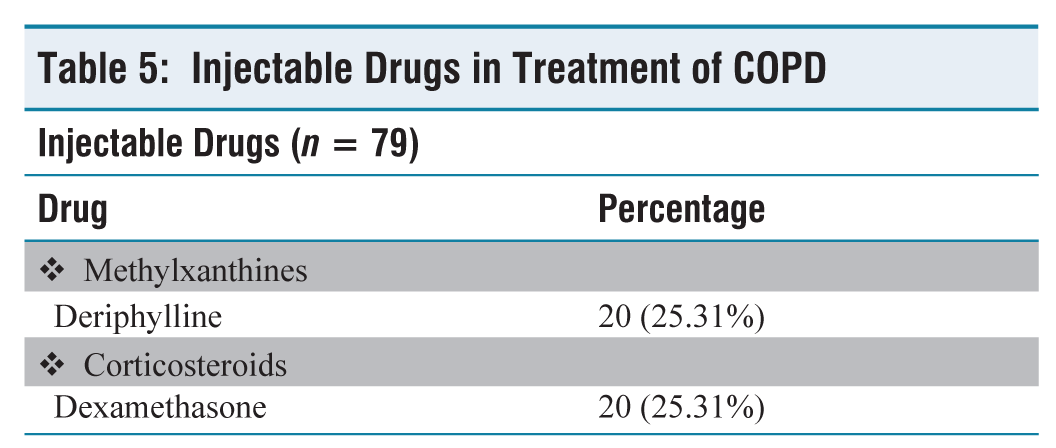
Observed Single Inhalation Therapy in COPD
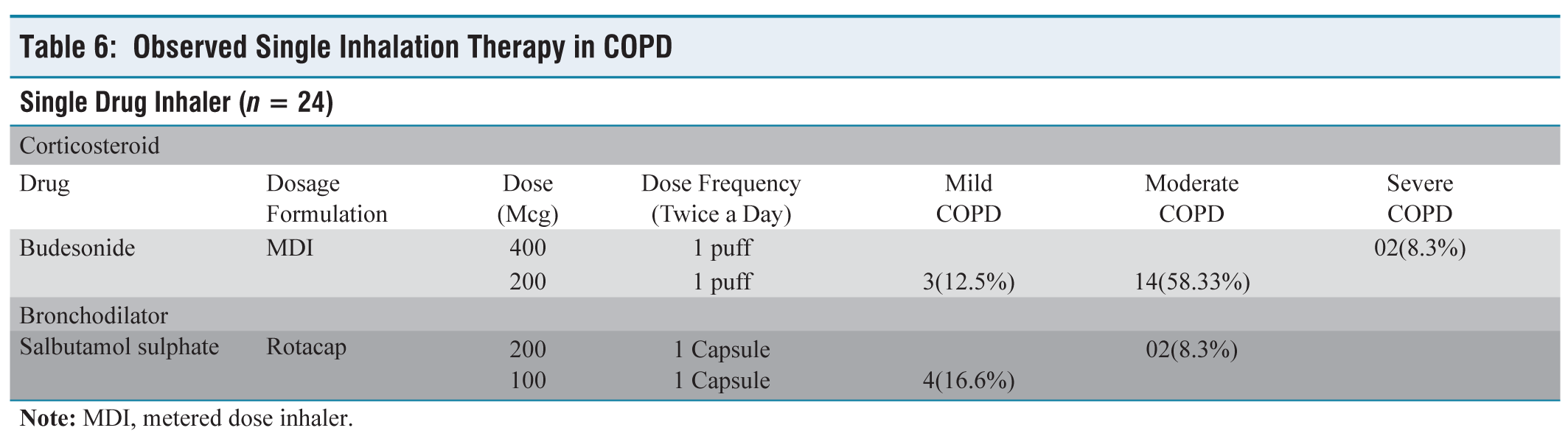
Observed Combination Inhalation Therapy in COPD
Oral Drugs Prescribed in COPD
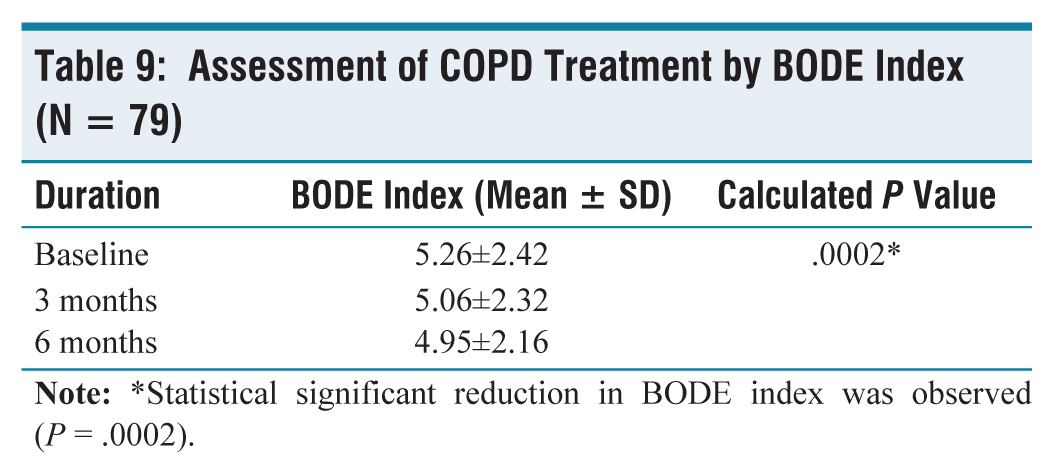
Analysis of effectiveness of treatment (Table 9): At baseline, average BODE index was improved significantly at the end of 6 months with a mean difference of –0.31 and P = .0002 (Friedmen test) and post hoc analysis (Tukey’s pairwise test) showed the statistically significant improvement from baseline to 3 months and 6 months.
Assessment of COPD Treatment by BODE Index (N = 79)
Cumulative prescription analysis was done using WHO prescribing indicators (Table 10).
WHO Core Prescribing Indicators

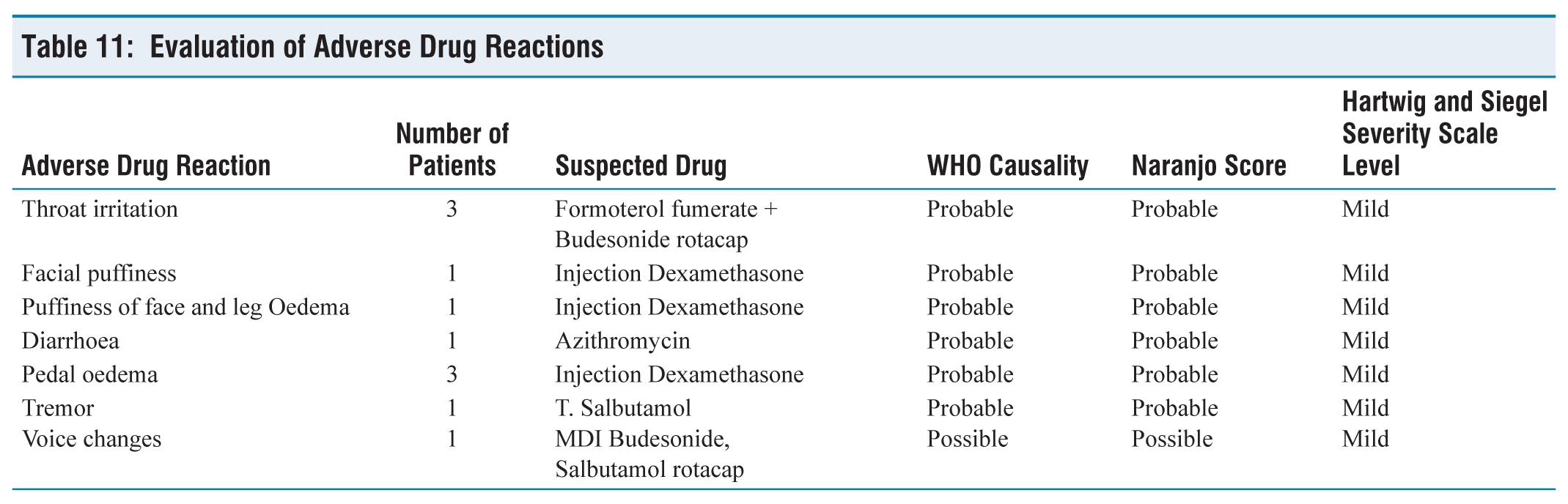
Evaluation of Adverse Drug Reactions
Discussion
Pre-marketing clinical trial data are anticipated to form the foundation of therapeutic practice, but post-marketing data are equally crucial for enhancing medication therapy. Drug utilisation studies (DUS) are carried out to assess the type, amount, and drivers of usage as well as to learn about current prescribing patterns. This aids in identifying and implementing drug usage among individuals that is appropriate. In this study, we have analysed the prescription pattern and evaluated the effectiveness and safety of therapy in patients with COPD in tertiary care teaching hospitals.
More recent data from developed nations indicates that the prevalence is nearly similar for both genders currently, most likely owing to shifting smoking patterns.[7] In this study, COPD was reported more in males 66 (83.54%) as compared to females 13(16.45%) participants. Male to female ratio was 5.5:1. This was similar to the result of studies done by Mahmoodan et al. (male 62% vs. 38% female), and Vyas et al. (male 73.33% vs. 26.66% female).[8,9] Although exposure to other risk factors is frequently mentioned together with age as a risk factor for COPD, it is uncertain whether or not healthy ageing alone causes COPD. With a minimum age of 37 years and a maximum age of 85 years, the age group of 51–70 years had the highest percentage of patients (73.42%). According to Sharon et al., the majority of patients (63%), were in the 51–70 year age range.[10]
Muscle wasting and weight loss are common manifestations of COPD and it causes limitations in the activities and the exercise capacity of the patient with COPD and indirectly on the quality of life. We observed 62.02% of participants with <21 BMI and 37.97% with BMI >21. Jangid et al. found a similar result with 72% of participants with < 21 BMI and 28% with > 21 BMI.[11]
According to the Centers for Disease Control and Prevention, smoking accounts for up to 8 out 10 COPD-related deaths. As smoking cessation is the primary goal in COPD management, a personal history of addiction such as smoking, tobacco chewing and alcohol was also analysed. We found history of smoking in 67(84.81%) participants followed by 26(32.91%) tobacco chewing, 20(25.32%) tobacco plus smoking and 3(3.8%) with history of alcohol. Of patients having a history of smoking, 50(74.62%) smokers had continued the smoking while only 25.37% of patients discontinued the smoking. Vyas et al. found history of smoking in 56% of participants, of which 29% were current users and 27% were past users.[9]
Whether COPD and some of its comorbidities are related or not, the comorbidities of patients with COPD must be identified and treated as part of their care. In our study, 37 (46.8%) participants were found to have history of comorbidities which varies in other studies and one of the valid reason for polypharmacy.[8-10]
The severity of COPD was assessed by BODE index. According to BODE index, we found maximum number of patients 41(51.9%) in the moderate category followed by severe (25.31%) and mild category (22.8%). Jangid et al. found maximum in moderate category (45%) followed by severe category (32%) and mild category (6%).[11] mMRC grading is one of the components of BODE index, based on mMRC grading, maximum number of participants were also in moderate category(32% -Grade 2) followed by severe and mild.it was also observed that 48% of the study participants had Grade-2 dyspnoea in similar other study carried out by Mahmoodan et al. No statistically significant association between age and BODE index was observed.[8]
In our study, a statistically significant reduction in the BODE index was observed (P = .0002, Friedman test). Post hoc analysis (Tukey’s pairwise test) showed a statistically significant improvement from baseline to 3 months and 6 months. This is observed in patients who had discontinued smoking and were on regular treatment. These findings are consistent with those of a research by Linderman et al., which examined the impact of proactive integrated treatment for patients with COPD and discovered a 12-week drop in mean BODE index.[12] After 12 months, the baseline average BODE index, which was 1.5, had dramatically improved, with a mean difference of –0.40 and a P = .03, according to another study conducted at the University of Louisville.[13] There was also decline in BODE index at 24 months (mean difference 0.14) but it’s not statistically significant from baseline as there are fewer subjects at 24 months and thereafter.
Among 79 prescriptions, it is observed that all the prescriptions contained more than three drugs. The potential that some individuals present with acute and chronic exacerbations of COPD with or without co-morbid disorders, requiring antibiotics, co-morbidity treatment, and corticosteroid treatment, may be the reason for prescribing more than three medicines to a single patient. We observed prescriptions with minimum 3 to maximum 11 drugs. In our study injectable drugs such as deriphylline and dexamethasone is used in 25.32% of prescriptions, whereas in other similar study parenteral steroids were used in 77.7 % of patients, in which hydrocortisone was prescribed followed by prednisolone and parenteral methylxanthines was given to 65.2% of participants.[10]
In our study total of 74 participants received inhalation therapy out of which 50 participants were receiving combination therapy and 24 were receiving single drug therapy. In our study, irrespective of severity, prescribed combinations were Budesonide plus formoterol fumerate rotacap in 33 (45.83%) participants followed by MDI budesonide plus rotacap salbutamol (11.11%), MDI levosalbutamol plus ipratropium bromide (6.94%) and MDI Ciclesonide plus formoterol (5.5%). Thus we can say that ICS plus LABA is used in 74% of participants followed by ICS plus SABA in (16%) and SAMA plus SABA in 10% of participants. A prospective study carried out at Bangalore showed different patterns of treatment where 45% percent of the patients were on combination therapy; the most popular combination was Salbutamol + Ipratropium bromide + Budesonide (40%) followed by Etophylline + Theophylline (22%) and Budesonide + Formoterol (14%).[10] Mahmoodan et al. found salbutamol+ ipratropium bromide (54.5%) followed by ipratropium bromide + fluticasone (3%) and Budesonide + formoterol(1%).[8] According to the severity of disease increased in dose/ number of capsule/puff per day is observed in our study.
In single-drug inhalation therapy, 25.67% of participants received MDI budesonide and 5.5% of participants received rotacap salbutamol sulphate, whereas in a similar study 15.5 % of participants received inhaled corticosteroids-budesonide.[10,14] ICSs are advised for individuals with moderate to severe COPD who have frequent exacerbations since they lower the frequency of exacerbations in those patients.
Among orally prescribed drugs, methylxanthines (96.20%) were mostly prescribed class of drug followed by antihistamines (75.95%), antipyretics and antacids (73.42%), Vitamins (44.52%) and antibiotics (18.98%). Theophylline tends to be added to inhaled bronchodilators in patients with more severe disease and has been shown to give additional clinical improvement when added to LABA. Theophylline enhances the anti-inflammatory effect of corticosteroids and its withdrawal in patients with COPD results in the worsening of disease. Use of low dose of methylxanthines largely avoids side effects and drug interactions, that’s why we can justify the 96.2% usage of methylxanthines. These findings were not in accordance with the previous studies done by Sharon et al. (42% use of methylxanthines) and Veettil et al. (86.7% use of methylxanthines).[10,14]
The prescription practice indicators assess how well medical professionals operate in a number of crucial domains pertaining to the responsible use of medications. Regardless of a particular condition, the basic prescription indicators assess overall prescribing behaviours within a particular scenario. The average number of medicines per prescription in our analysis was 6.35443, and comparable findings from previous studies also showed that prescriptions typically contained more than three substances.[7,8] Our research revealed that 62.7% of medications were administered under their generic names. This lowers the possibility of mistakes in prescription and administration. 55.74% of prescription medications were from the WHO’s essential medicine list. 19% of interactions resulted in an antibiotic prescription; this percentage did not match the results of earlier research by Sharon Sunil and Veettil SK which revealed 42% and 86.7% use of antibiotics. The percentage of encounters with an injection prescribed was 25.31%.[10,14]
We found adverse events in 11 (13.92%) participants, which included facial puffiness and oedema due to dexamethasone in five participants followed by throat irritation and voice changes due to inhalation therapy in four participants and tremor due to oral salbutamol and diarrhoea due to azithromycin found in one patient. A high average number of prescription prescriptions indicates a propensity among doctors to prescribe more medications to manage acute exacerbations of COPD; this trend is also reflected in the adverse event profile. We have evaluated the causation and severity of all adverse medication responses using the Naranjo, Hartwig, and Siegel severity scales, as well as the WHO causality evaluation scale.[15-17] We found all the adverse drug reaction (ADR) associated with drugs causing it probably only one ADR is associated possibly and all were with mild severity. We did not find any serious adverse drug reactions during the study period.
Considering the adverse outcomes associated with polypharmacy including adverse drug events, drug-drug interactions (often very complex), increased cost of treatment, medication errors, patient non-adherence with treatment (which increases with complex regimens), we need to take appropriate measures for minimising the extent of polypharmacy when possible.
Conclusion
Most of the patients were prescribed multiple drug therapy; out of all the inhalation route was most preferred. The prescribed patterns in COPD, in which mainly methylxanthines, followed by corticosteroids, β2 agonists, and anticholinergics were used for the management. Though anticholinergic drugs are proven to be as effective as or even better than beta agonists we found its usage in only 10% of COPD participants. Theophylline enhances the anti-inflammatory effect of corticosteroids and its withdrawal in patients with COPD results in the worsening of disease. In this study use of low doses of methylxanthines largely avoids side effects and drug interactions, that is why we can justify the 96.2% usage of methylxanthines.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
PDUMCR/IEC/19541/2018 dated 28/11/2011.
Credit author statement
All authors contributed to the study conception, design and data collection. Material preparation and analysis were performed by all authors. All authors read and approved the final manuscript.
Data availability
Data supporting the finding of this study are available within the article text and tables.
Use of artificial intelligence
NA.
