Abstract
Background and Aims:
Oesophageal malignancies tend to have poor prognosis and thus there is a need to identify newer therapeutic targets for their treatment. This study aimed at studying the incidence and histopathological spectrum of oesophageal malignancies with special emphasis on immunohistochemical expression of HER2/Neu in adenocarcinomas.
Material and Methods:
This study was a retrospective descriptive study conducted in a tertiary care centre in South India. Endoscopic biopsies and resection specimens of oesophageal malignancies were included in the study. Immunohistochemical analysis for HER2/Neu was performed on all oesophageal adenocarcinomas.
Results:
Squamous cell carcinoma constituted the most common malignancy in this study. Approximately 20% of oesophageal adenocarcinomas showed positivity for HER2/Neu. A prominent association of Barrett’s metaplasia with oesophageal adenocarcinoma was noted.
Introduction
Oesophageal carcinoma contributes a huge deal to the global cancer burden and constitutes the world’s eighth[1] most common cancer[2] and represents the world’s sixth most common cause of cancer death.[1,2] Oesophageal malignancies tend to have a very poor prognosis, with 5-year survival ranging between 5% and 20%,[3] even after several recent advances in surgical and chemotherapeutic agents. Thus, it is essential to discover newer therapeutic targets to treat these tumours with more aggressive targeted regimens.
HER2/Neu, also known as ERBB-2, is a protooncogene and a member of the human epithelial growth factor receptor family. Its role in oncogenesis has been proven beyond doubt in many tumours. This growth factor receptor has been most comprehensively studied from the perspective of breast cancer. Chemotherapy with trastuzumab, a monoclonal antibody targeting HER2, has become a standard of care for breast cancer positive for HER2/Neu.[4,5] The trastuzumab for gastric cancer (ToGA) trial has demonstrated that addition of trastuzumab in a subset of esophageal carcinomas that express HER2/Neu has a definite survival advantage.
This study aimed at studying the incidence and histopathological spectrum of esophageal malignancies with special emphasis on immunohistochemical expression of HER2/Neu.
Material and Methods
This study was a retrospective 5-year study conducted in a tertiary care hospital in South India. Endoscopic biopsies and resection specimens of esophageal tumours that were received during this period were included in the study. Institutional ethical clearance was taken as per departmental protocol. All specimens were routinely fixed in 10% buffered formalin and processed. Paraffin-embedded tissue blocks that were obtained were sectioned and stained with hematoxylin and eosin. Immunohistochemical analysis was performed using a combination of microwave oven heating and standard streptavidin–biotin–peroxidase complex, using Dako kit for HER2/Neu. Modified Ruschoff’s criteria were used for assessing HER2/Neu immunohistochemical expression.[6,7]
Results
A total of 84 oesophageal malignancies were included in the study. Table 1 depicts the detailed demographic distribution, histopathological diagnosis, differentiation and immunohistochemical expression of HER2/Neu. The most common age group was the seventh decade of life, with a male preponderance (male-to-female ratio: 1.62:1). Most patients presented with dysphagia (83.3%), followed by hematemesis (12%) and epigastric pain (4.7%). The most common endoscopic appearance was ulcero-proliferative growth, which constituted 64 of the total 84 cases. The most common site for oesophageal tumours was the lower third, which comprised 69% of all the cases, followed by the middle third. The most common tumour in the oesophagus was squamous cell carcinoma, constituting 50 of 84 cases (59.5%). Moderately differentiated squamous cell carcinoma was the most predominant sub-type. Oesophageal adenocarcinomas (EAC) constituted 30 out of the total 84 oesophageal malignancies that were reported. The most common site for EAC was the lower third (24 of 30 cases). There were no cases that occurred in the upper third of the oesophagus.
Demographic Distribution, Clinical Features, Histopathological Diagnosis, and Differentiation of the Tumours Included in the Study
Discussion
In this study, males constituted 52 out of 84 cases of oesophageal carcinoma, outnumbering females by a ratio of 1.62:1. The present findings correlate well with the observations of Sons et al.[8] The most common site for oesophageal carcinomas was distal one-third, constituting 66.7% of cases. Previously, the most common site for oesophageal cancer was the middle third; however, with the changing trends in the incidence of upper gastrointestinal tract malignancies, this site has also changed to the lower third.[9,10] as reflected by this study. In the current analysis, the most common site for SCC was the lower oesophagus followed by the middle and upper, and 24 adenocarcinomas and two adenosquamous carcinomas reported occurred in the lower third of the oesophagus. However, Li et al.[11] and Shimada et al.[12] reported that SCC occurred most commonly in the middle third followed by the lower third of the oesophagus.
Squamous cell carcinoma was the most common neoplasm of the oesophagus comprising 59.5% of all the oesophageal tumours in this study. This finding is in parallel to that of Pun CB et al.[13] and Wang et al.[14] However, in the studies by Sons et al.,[8] Vidhyavathi et al.[15] and Derakhshan et al.[16] the incidence ranged between 84% and 89%.
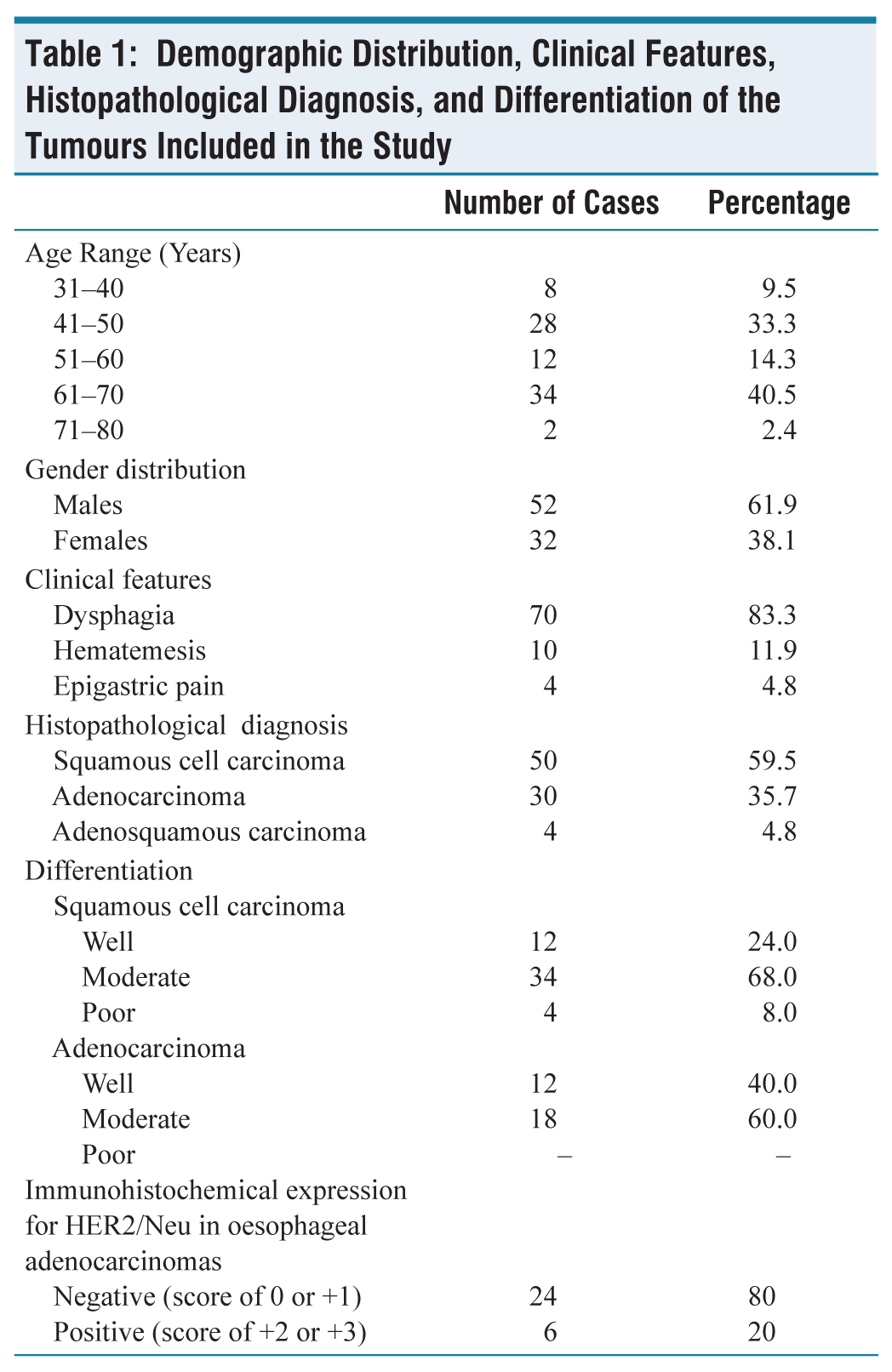
Squamous cell carcinomas were categorised as well, moderately and poorly differentiated based on the cell morphology. Tumours were categorised as well differentiated when they had cytological and histological features similar to the normal oesophageal squamous epithelium, when large differentiated keratinocyte-like squamous cells constituted a large proportion of the tumour and were arranged in sheets and when keratinisation and pearl formation akin to the appearance of non-neoplastic squamous epithelium was seen. These tumours typically had lower mitotic activity (Figure 1A). Tumours were categorised as poorly differentiated when they contained predominantly basal type of cells arranged in sheets with high mitotic activity, and with occasional parakeratotic cells (Figure 1B). Tumours with intermediate features were grouped under moderately differentiated squamous cell carcinomas (Figure 1C).
(A) Well-differentiated Squamous Cell Carcinoma Showing Pleomorphic Cells Arranged in Nests and Lobules. Keratin pearls are also seen. (B) Poorly differentiated squamous cell carcinoma showing basal-like pleomorphic cells arranged in diffuse sheets with occasional parakeratotic cells. (C) Moderately differentiated squamous cell carcinoma showing pleomorphic cells showing individual cell keratinisation
This study portrayed that most of the squamous cell carcinomas occurring in oesophagus were moderately differentiated. This finding correlates well with that of Vidhyavathi K et al.[15] However, Wang J et al.[14] and Suzuk l et al.[17] quoted a higher incidence of poorly differentiated carcinomas.
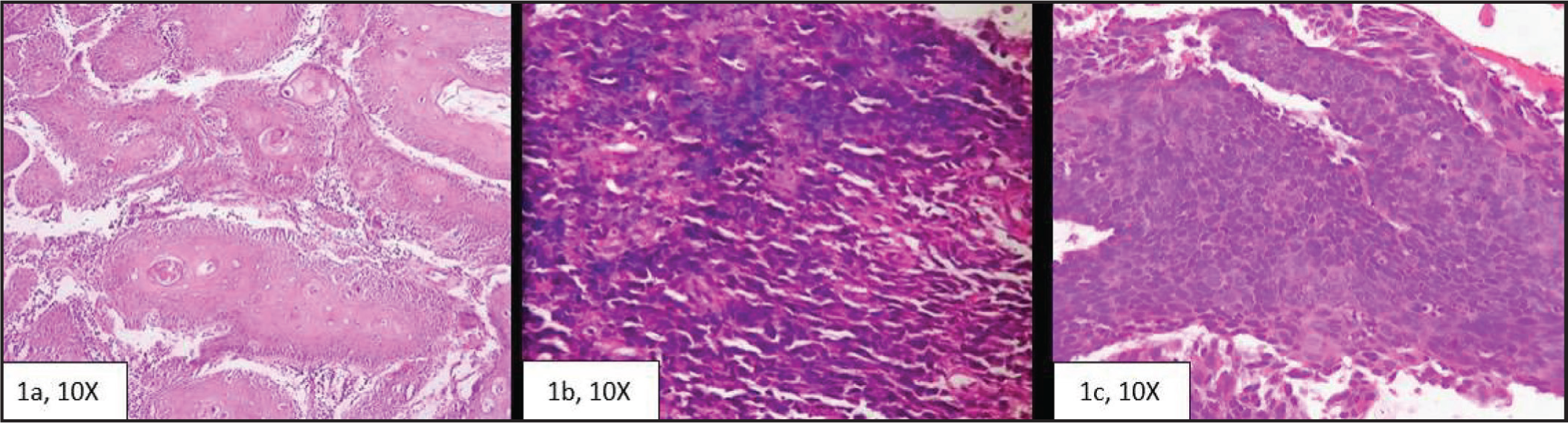
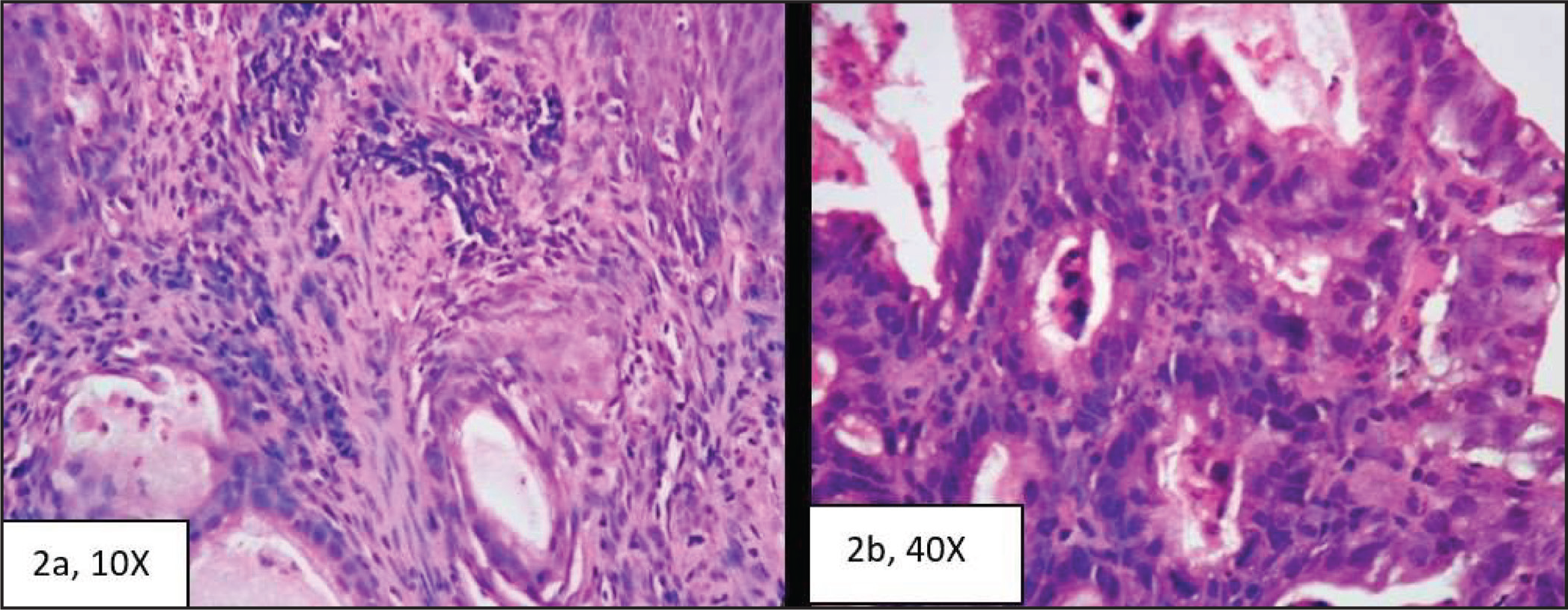
We reported four cases of oesophageal adenosquamous carcinoma all of which occurred in male patients above 50 years of age. The tumours were located in the lower oesophagus. Endoscopically, they presented as a fungating growth as shown in Figure 3A. Histologically, they were composed of mixed elements of both adenocarcinoma and squamous cell carcinoma which remained clearly distinguishable within the same tumour(Figure 2A).
(A) Tumour Composed of Both Adeno and Squamous Components. (B) Showing oesophageal adenocarcinoma composed of moderately pleomorphic epithelial cells arranged in tubular patterns with adjacent areas showing mononuclear infiltrate
Tumours were diagnosed as EAC, when they were located in the oesophagus and histologically showed typical tubular (Figure 2B) or papillary or a combination of the two patterns. There was one tumour that showed cells with vacuolated cytoplasm. Individual cells were cuboidal to columnar with round to oval hyperchromatic nucleus and scant to moderate amount of cytoplasm. The overlying squamous epithelium showed mild dysplastic changes in one case.
Of the 30 cases, 12 were well differentiated and 18 were moderately differentiated.
Sixteen out of 30 EAC were associated with Barrett metaplasia in the adjacent mucosa. Arriving at a diagnosis of Barrett metaplasia is a joint venture of endoscopist and pathologist. Endoscopically, Barrett metaplasia can be identified as tongues of salmon pink patches extending in between pearly white normal mucosa of oesophagus, as shown in Figure 3B. Barrett metaplasia can occur as different phenotypes including intestinal (with goblet cells), cardiac and fundic (cardio-oxyntic). Differentiating reactive changes from dysplastic changes in the Barrett oesophagus can be difficult, and hence reassessing in such doubtful cases may be required after a period of acid suppression. In the present case, Barrett metaplasia showed intestinal phenotype with goblet cells(Figure 4A and 4B).
Endoscopic Photographs: (A) fungating growth in oesophagus and (B) Barrett oesophagus- Salmon pink tongue-like extension into the oesophagus
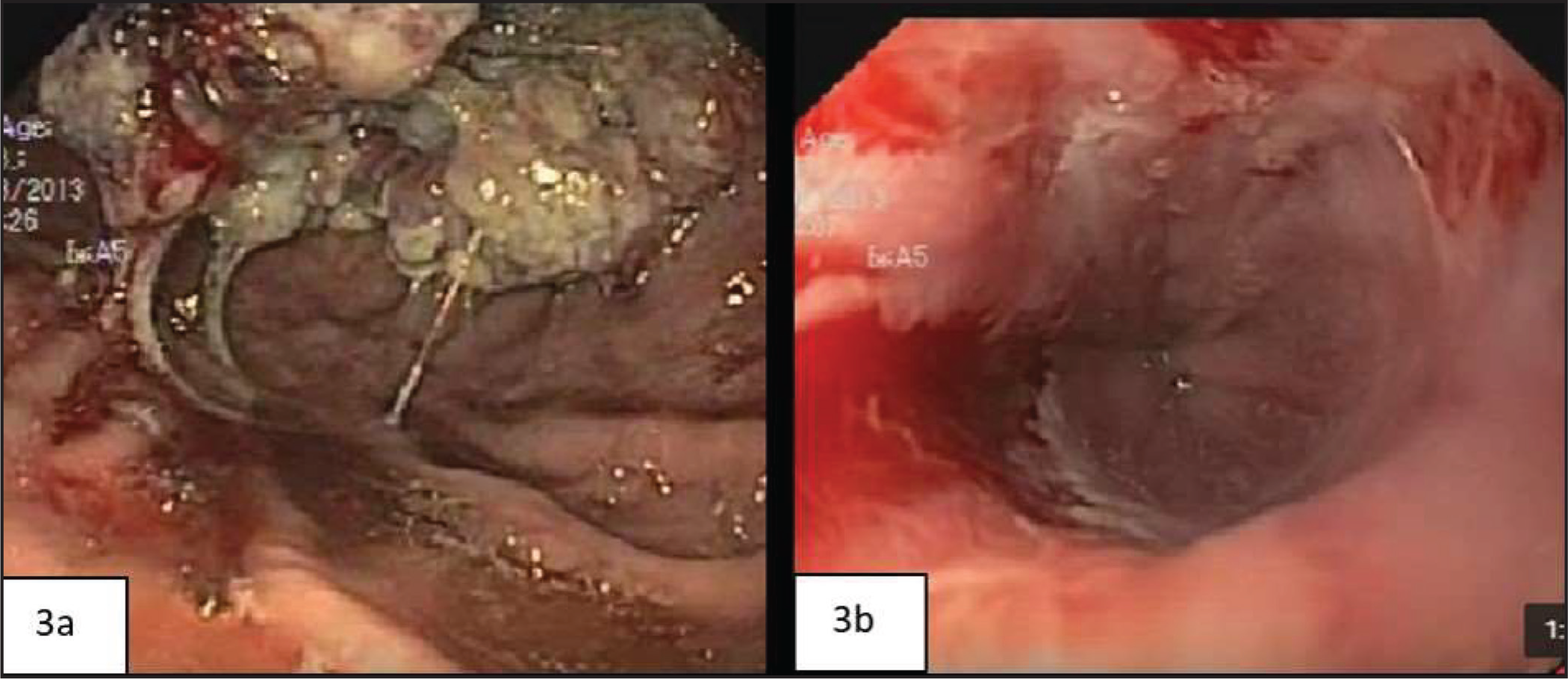
(A and B) H&E: 10X and 40X Intestinal Metaplasia of Esophageal Mucosa Along With Bits Showing Esophageal Submucosal Glands. Bits of stratified squamous epithelium were present elsewhere in the biopsy
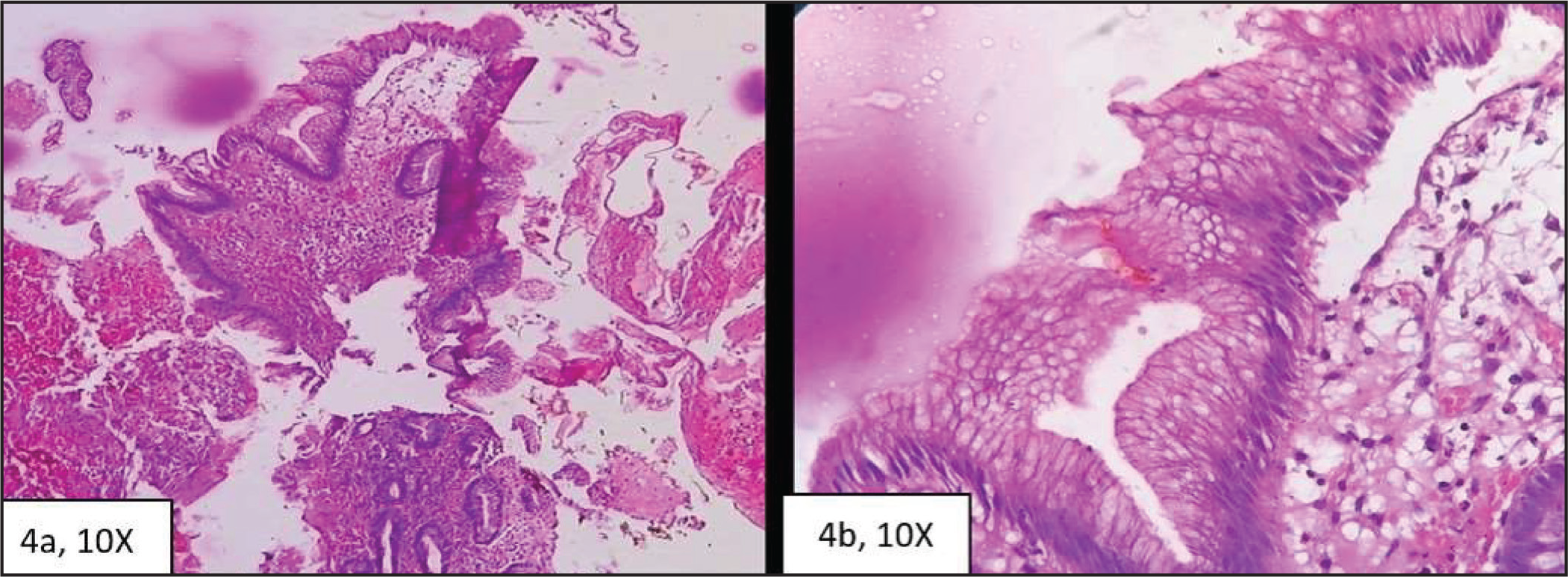
Presence of oesophageal sub-mucosal glands, bits of native squamous epithelium can help in correctly labelling these biopsies as those from oesophagus and as those showing Barrett metaplasia. Abrupt transition between morphologically different epithelia can help in confirming dysplasia in the Barrett’s oesophagus.[18]
Differentiating adenocarcinomas from high-grade dysplasia of Barrett oesophagus can be difficult, especially in small biopsies. Features such as associated necrosis, desmoplasia, presence of small clusters of dysplastic cells, back to back and cribriform arrangement of glands favour adenocarcinoma and help in distinction.[19]
(A and B) H&E and Corresponding IHC. Well-differentiated esophageal adenocarcinoma showing intense membranous staining – HER2/Neu score +3
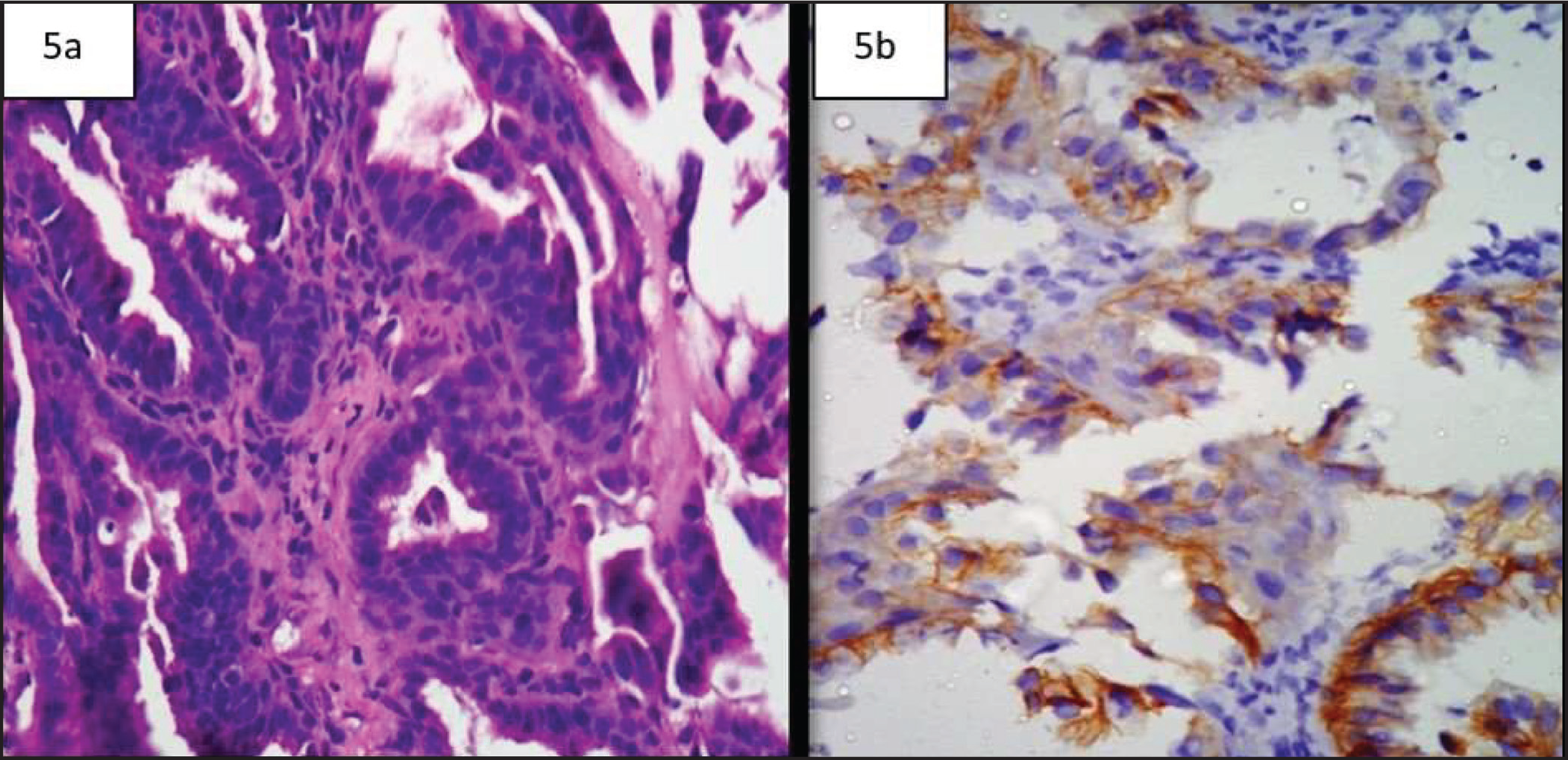
Differentiating lower EACs from gastric cardiac tumours based on histopathology is almost unfeasible. There are many studies which emphasise on these criteria.[20,21] However, in most of these classification systems, the anatomical location of the epicentre of the bulk of these tumours is taken to determine the origin of the tumour. There are some classification systems,[22] which include lower EACs, gastric cardiac tumours and subcardial tumours under tumours of esophagogastric junction tumours. Nevertheless, there are several studies which have used cyclooxygenase2,[23] P53 expression[24] and other genetic analyses,[24] in order to differentiate them. However, as for now none of them are rewarding.
In the current study, six cases of EAC showed HER2/Neu positivity, all the three tumours showed invasion into the serosa, and four cases presented with positive lymph nodal status. Due to the small sample size, statistical significance of the present findings could not be elicited. However, in the review article by Cathy B et al.[25] they stated that Her 2 overexpression correlates with tumour invasion and lymph nodal metastasis and thus indicates a poor prognosis and reduced overall survival rate. Out of the 30 EAC, 16 showed Barrett metaplasia. The non-dysplastic Barrett mucosa was negative for HER2/Neu expression in all these cases. This finding correlates well with that of Flejou et al.[18] The percentage of Her 2 overexpression in EACs ranged between 7% and 43% in various studies.[26]
Conclusion
To conclude, with the evolution of genetic and molecular testing and increasing understanding of cancer biology, the formerly considered single entity of oesophageal adenocarcinoma is now known to be conglomerate of histologically, biologically and molecularly heterogenous group of cancers. HER2-overexpression/amplification is now an established therapeutic target and use of trastuzumab has demonstrated improvement in overall survival in various studies.[27]
Footnotes
Acknowledgements
XX
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional Ethical Committee Approval Number
The study was approved by the institutional ethics committee vide approval number GMC/IEC/12/2020.
Informed Consent
Patients or their relatives provided written informed consent for use of their clinical material in the present study.
CRediT Author Statement
All authors have contributed to the study conception and methodology. RK: Data curation, writing- original draft preparation, PRK: reviewing and editing, BS: supervision.
Data Availability
Data supporting the findings of this study are available within the article text and tables.
Use of Artificial Intelligence
No artificial intelligence was used in the present study.
