Abstract
Introduction:
Enteral feeding tubes are commonly utilized in the neonatal intensive care unit (NICU) due to the underdevelopment of coordination between sucking, swallowing, and breathing, or due to various diseases affecting the respiratory, cardiovascular, gastrointestinal, or neurological systems. The aim of the study was to investigate the differences in bacterial colonization patterns in hospitalized infants who received enteral feeding through either nasogastric (NG) or orogastric (OG) feeding tubes.
Introduction
Enteral feeding tubes are commonly utilized in the neonatal intensive care unit (NICU) due to the underdevelopment of coordination between sucking, swallowing, and breathing, or due to various diseases affecting the respiratory, cardiovascular, gastrointestinal, or neurological systems.[1-3] Nonetheless, these feeding systems quickly develop biofilms composed of microorganisms. This can turn the feeding tubes into potential reservoirs for infection-causing pathogens in infants or for transmission between patients by health-care staff.[4-6] This concern is substantiated by the presence of shared bacterial strains among NICU infants.[3] This is especially worrisome as many of these strains exhibit resistance to multiple antibiotics. A significant portion of the bacteria identified in feeding tubes are hospital-acquired pathogens, including members of the Enterobacteriaceae family, Enterococcus spp., and coagulase-negative Staphylococcus spp.[5]
Either the nasogastric (NG) or the orogastric (OG) route can be utilized to insert feeding tubes successfully.[6] The nasopharynx is situated at the back of the nose in the upper part of the throat, and it is lined with ciliated and columnar epithelium. On the other hand, the oropharynx is located directly behind the mouth, and it is lined with nonkeratinized stratified squamous epithelium.[7,8] After birth, the nasopharynx and oropharynx quickly acquire a diverse initial microbiome, and within the first few days of a person’s life, there are already discernible differences between the microbiomes of neighboring anatomical regions.[9,10] The nostrils are home to bacteria belonging to the genera Corynebacterium and Staphylococcus, whereas the oropharynx contains species belonging to the genera Streptococcus, Haemophilus, and Neisseria, in addition to various anaerobic bacteria and Staphylococcus to a lesser extent.[11,12] There is a connection between the evolution of the infant’s upper respiratory tract microbes and external factors such as the method of delivery, breastfeeding versus formula feeding, and exposure to antibiotics. The maternal microbiota plays a role in the process of shaping the microbiome of the infant. Even though research into the clinical implications of neonatal intestinal tract colonization is still in its infancy, it is essential to have a solid understanding of both the composition and function of the microorganisms that colonize the neonatal intestinal tract to evaluate the effect these microorganisms have on the developing immune system.[13,14]
Infants in the NICU often require enteral feeding due to various medical conditions, and the choice of feeding tube type (NG vs. OG) can impact the colonization of potentially harmful bacteria. Understanding how bacterial colonization varies between these two methods is crucial, as it can influence infection rates, treatment outcomes, and overall neonatal health. By conducting this comparative analysis, the study aims to provide health-care practitioners with valuable insights into optimizing feeding tube strategies, enhancing infection control protocols, and ultimately improving the care and outcomes of vulnerable infants in the NICU setting. The purpose of this research was to gain a better understanding of the differences in bacterial colonization that occured in hospitalized infants who received enteral feeding through either NG or OG feeding tubes. In addition, we investigated clinical factors that affect the formation of biofilms, such as the mode of delivery, gestational age, the type of feeding, and the use of antibiotics.
Materials and Methods
This study was a hospital-based cross-sectional investigation that was carried out on infants who were admitted to the NICU. The NICU gathered feeding tubes throughout 4 months, beginning in August 2022 and continuing through December 2022. The research was carried out in a manner that was consistent with the principles outlined in the Declaration of Helsinki, and the Institutional Review Board gave its approval to each of the protocols. In addition, informed consent was collected from the participant’s parents or legal guardians for the entire group of people who took part in the research. The fact that the patient participated in this study had no bearing on the level of care that they were given. The research team gave serious consideration to recruiting any of the newborns who were receiving medical attention in the NICU and who had a feeding tube inserted during the time that sample collection was taking place. The infants did not receive any probiotics as a form of treatment in any of the cases.
Infants eligible for inclusion in this study were those who had been admitted to the NICU and required enteral feeding, either through NG or OG tubes. Their age at the time of tube insertion should be ≤28 days. In addition, infants included in the study had documented microbial analysis data from their enteral feeding tube samples. Cases with significant congenital anomalies affecting gastrointestinal function were excluded. For accurate bacterial colonization assessment, infants with feeding tubes in place for a minimum of 48 h were considered. Cases with a history of chronic medical conditions influencing the gastrointestinal tract or immune system were not considered. Incomplete or insufficient microbial analysis data for enteral feeding tubes were excluded. Finally, infants concurrently undergoing treatment with probiotics or other interventions that may impact bacterial colonization were excluded from the study.
After being removed by staff members of the NICU as per protocol, feeding tubes were subsequently placed in sterile sleeves before being chilled to a temperature of −40°C. Throughout the study, weekly logs were kept documenting the kinds of foods that were provided to each participant as part of their nutrition. In addition, the participant’s electronic medical records were reviewed to document the length of time that the feeding tube was in place, the route that it was inserted, the gestational age of the patient, the use of acid suppressors or antibiotics, and the diagnoses of necrotizing enterocolitis (NEC), sepsis, and spontaneous intestinal perforation. In the current study, we restricted our analysis to data collected from the pharyngeal segments because it was hypothesized that these would be most affected by the implant site (oral vs. nasal).
For continuous variables, descriptive statistics were reported as the mean accompanied by the standard deviation, and for categorical variables, frequencies were reported alongside percentages. To determine the degree of association between categorical variables, the Chi-square test was utilized. To determine the degree of association between the continuous variables of the two groups, an independent t-test was carried out. IBM SPSS Statistics for Windows, version 26.0, developed by IBM Corp. and based in Chicago, Illinois, USA, was used to perform statistical analysis on the collected data.
Results
The demographic and clinical data of the infants in each group are summarized in Table 1. Infants with NG tubes had a significantly higher number of C-sections (P = 0.04), an infant with NG tube was exclusively breastfed (P = 0.02), and one or more dose of antenatal steroids was administered more in infants with OG tube (P = 0.04). There was no statistically significant difference between the infants who had NG tubes and those who had OG tubes in terms of the antibiotics they received on the day of tube collection or any of the other clinical factors.
Distribution of demographics and clinical outcomes of bacterial colonization between nasogastric and orogastric enteral feeding tubes in infants (n=50)
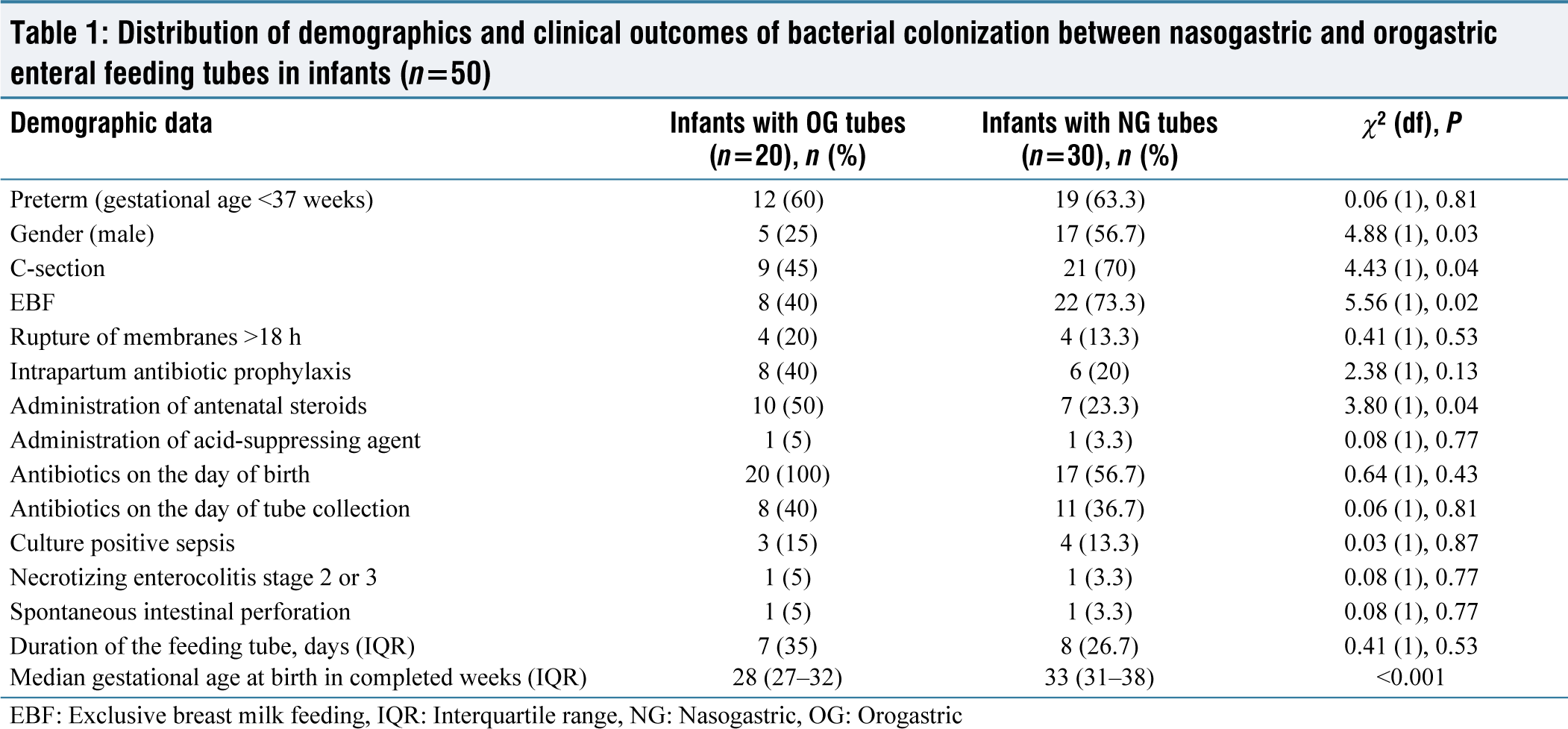
EBF: Exclusive breast milk feeding, IQR: Interquartile range, NG: Nasogastric, OG: Orogastric
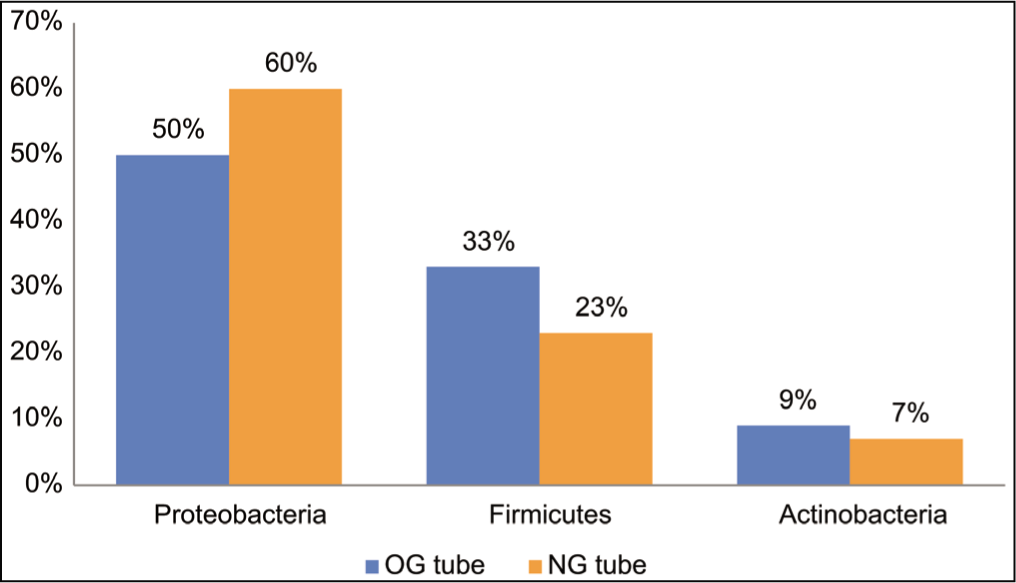
In the samples that were analyzed, it was discovered that the phyla known as Proteobacteria, Firmicutes, and Actinobacteria had the highest number of members. Researchers discovered Fusobacteria, Tenericutes, and Thermi in addition to Bacteroidetes and Cyanobacteria in their investigation. Figure 1 presents a visual representation of the various bacterial phyla that can be found in OG and NG tubes. Actinobacteria, Proteobacteria, and Firmicutes were the most common types of bacteria found in NG and OG tubes Proteobacteria, with a mean relative abundance of 50% and 60%, respectively Firmicutes, with a mean relative abundance of 33% and 23%, respectively Actinobacteria were the most common type of bacteria found (9% and 7%, respectively). It was found that there was a significant inverse correlation between the relative abundance of Proteobacteria and Firmicutes in all feeding tubes (Spearman correlation coefficient 0.62, P ≤ 0.001), and it was also found that there was a significant inverse correlation between Proteobacteria and Actinobacteria (Spearman correlation coefficient 0.37, P ≤ 0.001).
Distribution of bacterial phyla in orogastric and nasogastric tubes (n = 50). OG: Orogastric, NG: Nasogastric
Discussion
The microbiota of a newborn infant is highly dynamic and goes through rapid changes in composition throughout the first years of life. This process continues throughout the first decades of life. At various locations throughout the body, there are distinct microbial communities, in terms of both their taxonomy and their functions. A newborn baby’s microbiota is highly dynamic and subject to rapid changes in the composition of its components.[15] It is highly likely that the formation of biofilms on feeding tubes is affected not only by the bacteria that are present at the site of placement (oral or nasal) but also by the bacteria that are present in the stomach and intestines. The symptoms of abnormal motility in infants frequently include abdominal distention, an overgrowth of bacteria, and gastroesophageal reflux, the latter of which can frequently be seen coming out of both the nose and the mouth. These biofilms will likely be affected in some way by the secondary effects of bacterial metabolic products or by the host mucosal immune responses.[16] These side effects are experienced by the digestive tract in addition to the respiratory tract. Columnar epithelial cells line the mucosal surfaces of the digestive and respiratory tracts. These cells can identify commensal bacteria and, as a result, shape both local and systemic immunity as infants develop and as a function of postmenstrual age. Mucosal surfaces of the respiratory and digestive tracts are lined with columnar epithelial cells. These embryonic beginnings are shared by the digestive and respiratory tracts of the developing body.[17] There is a good chance that the formation of feeding tube biofilms is influenced by a combination of factors, including dietary decisions, antibiotics, therapeutics, and environmental exposures in the NICU.[18] One example of the effect that these shifts have during the early postnatal period, which is a crucial time for the development of immune competence, is antibiotic-induced alterations of the neonatal gut microbiota.
In our research, we found that infants born with NG tubes had a significantly higher number of cesarean sections (P = 0.04), that infants born with NG tubes were breastfed exclusively (P = 0.02), and that infants born with OG tubes were given one or more doses of antenatal steroids more frequently (P = 0.04). In terms of the antibiotics they were given on the day of tube collection as well as any of the other clinical factors, there was no statistically significant difference between the infants who had NG tubes and those who had OG tubes. According to the findings of a study carried out by Vongbhavit et al.,[19] the OG group had a higher percentage of premature infants compared to the NG group (93% vs. 67%). There was no statistically significant difference between the infants who had NG tubes and those who had OG tubes in terms of the antibiotics they received on the day of tube collection or any of the other clinical factors.
At the level of the phylum, the results of our preliminary research revealed that Proteobacteria and Firmicutes predominated in biofilms on feeding tubes, with Actinobacteria being present, but in much lower concentrations than the other two groups. In the past, it has been reported that these bacteria make up the majority of the bacterial populations that can be found in the feeding tubes of NICU patients, the extension tubing for enteral feeding systems, and the gastroesophageal microbiota of newborns.[20]
The predominance of Proteobacteria at the phylum level and Enterobacteriaceae at the family level in both OG and NG tubes has clinical relevance because these organisms have been described as a marker of intestinal dysbiosis[21] and are associated with increased risk of NEC in preterm infants.[22] It is noteworthy that there were no significant differences between NG and OG tubes for the Enterobacteriaceae that are most commonly associated with NEC and late-onset sepsis at the genus or species level. This finding is important because it indicates that NG and OG tubes contain the same bacteria (Escherichia and Klebsiella). When we compared OG and NG tubes for Pseudomonas, we found that there were differences at the genus and species levels, with different species being more common in each. These differences were discovered when we compared the two types of tubes for the bacterium. If this hypothesis is supported by further investigation, it would be extremely useful to acquire an understanding of the processes that are accountable for the observed differences in preference. This is because many species of Pseudomonas are opportunistic pathogens, and they form biofilms with other species that colonize hospitals and cause NICU outbreaks. These biofilms are resistant to penicillin and other beta-lactam antibiotics.[23] It is consistent with differences in the colonization of the nasopharynx and oropharynx, respectively, that we observed increased Moraxellaceae and Staphylococcaceae in NG tubes and increased Streptococcaceae and Neisseriaceae in OG tubes. In the future, it would be beneficial to conduct research on the impact that changing the nasal microbiota has on the biofilms that form on feeding tubes (e.g., by decolonizing methicillin-resistant strains of Staphylococcus aureus), as well as research that compares the biofilms that form on NG tubes in infants who are colonized with coagulase-positive and negative staphylococci.
Previous research found evidence of the presence of specific infant gut-associated strains in the environment of the NICU room,[24] as well as evidence of the exchange of those strains between the infant and the environment of the room. This room also contained evidence of the exchange of those strains between the infant and the environment of the room. There is a need for future research that investigates the effect that NICU environmental cleaning routines have on feeding tube biofilms as well as outbreaks of disease that are caused by common organisms found in biofilms. This line of inquiry is necessary because there is a need. However, it would be beneficial to investigate the influence that different probiotic strains have on feeding tube colonization because probiotic dietary supplements were not given to any of the infants who participated in this study. It may also be beneficial to investigate the clinical impact of feeding tubes coated with commensal organisms or antimicrobial molecules.
The fact that the analytical methods used in this study are unable to differentiate between commensal and pathogenic strains is one of the study’s limitations. This has important implications for clinical practice because diseases that threaten life have been linked both to a deficiency in bacteria that are beneficial to health and to an abundance of bacteria that could be pathogenic.[25] In addition to this, it has been discovered that many different strains of Enterobacteriaceae that have been isolated from NICU feeding tubes are resistant to antibiotics. Although numerous microbial communities found within individual body sites have been described, the associations between the microbiota found across multiple body sites or systems have received less attention from researchers.
Conclusion
Oral versus nasal feeding tube insertion was found to have a greater influence on bacterial colonization than any of the other clinical factors that were evaluated. The microbial analysis of the samples unveiled predominant phyla, including Proteobacteria, Firmicutes, and Actinobacteria, with varying relative abundances in both NG and OG tubes. Noteworthy findings encompassed additional phyla such as Bacteroidetes, Cyanobacteria, Fusobacteria, Tenericutes, and Thermi. A significant inverse correlation was established between the relative abundances of Proteobacteria and Firmicutes in all feeding tubes. Similarly, a significant inverse correlation was observed between Proteobacteria and Actinobacteria. These findings collectively provide valuable insights into the microbial composition associated with different feeding tube types and offer a basis for further investigation into potential implications for neonatal health and care.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional Ethical Committee Approval Number
Independent Ethics Committee – PR/2022/IEC/019.
CRediT Author Statement
Conceptualization: MS; Manuscript writing: MS and RSM; and Manuscript proofing: RSM. Both the authors have agreed to publish the manuscript.
Data Availability
All the data is confined within the manuscript.
Use of Artificial Intelligence
No AI tool is used to write the manuscript.
