Abstract
Chyluria is caused by the development of the reno-lymphatic fistula leading to reflux of chyle in urine. The most common cause of chyluria is a parasitic infection, especially lymphatic filariasis; however, rarely chronic renal infections like tuberculosis can also cause chyluria. The presence of fat fluid level in the pelvicalyceal system, perinephric, retroperitoneal collections and in the urinary bladder on CT scan in a case of chronic pyelonephritis is an important indicator of the presence of the reno-lymphatic fistula. We describe clinical picture and CT images of a patient with chyluria showing fat fluid in abdominal collections.
Introduction
Dark fluid levels within the urinary system are commonly encountered in radiology practice, and its most common cause is an air–fluid level due to emphysematous infections. Rarely, a dark-fluid level may be caused by fat–fluid level occurring in the setting of chyluria. Chyluria is caused by the development of the reno-lymphatic fistula, leading to reflux of chyle in urine. The most common cause of chyluria is a parasitic infection, especially lymphatic filariasis, but rarely chronic renal infections such as tuberculosis can also cause chyluria. The presence of fat–fluid level in the pelvicalyceal system, perinephric, retroperitoneal collections, and in the urinary bladder on computed tomography (CT) scan in a case of chronic pyelonephritis is an important indicator of the presence of the reno-lymphatic fistula. Due to its rarity, it may be mistaken for the more common air–fluid level within the urinary system seen in emphysematous infections. Due to its rarity, imaging diagnosis of chyluria is often overlooked and confused with the more common air–fluid level in the pelvicalyceal system and urinary bladder. A measurement of the Hounsfield unit (HU) value of the hypodense component in any dark-fluid level with the urinary bladder is needed for a prompt radiological diagnosis. We describe clinical pictures and CT images of a patient with chyluria showing fat–fluid in abdominal collections.
Case Report
A 35-year-old female presented with fever, abdominal distension, white-colored urine, and left lumbar pain for 15 days. She was previously diagnosed with Staghorn calculus with moderate hydronephrosis in the left kidney and treated with percutaneous nephrolithotomy about 1.5 years back. About 6 months later, she again presented with pyonephrosis and multiple renal calculi, for which she underwent D-J stenting and had gradually improved.
Investigations
The present routine blood investigations revealed raised total leukocyte count (18,000 cells/mm3) with increased neutrophil counts. The rest of the blood parameters were unremarkable including the blood lipid profile. Urine examination revealed the presence of chyle in urine with raised triglyceride levels and multiple pus cells. Urine culture was negative.
Ultrasonography revealed a small left kidney with mild hydronephrosis, mildly thinned renal parenchyma, few renal calculi, and perinephric and capsular calcifications. Associated multiple loculated retroperitoneal collections were seen.
Contrast-enhanced CT with CT urography was done, and it revealed [Figure 1] a small left kidney (7.5 cm × 3.8 cm) with a distorted outline, focal areas of cortical thinning associated with mild hydronephrosis, 2 calculi, and DJ stent in situ. Compensatory hypertrophy was seen along the lateral cortex. Extensive calcification was seen in the perinephric space around the upper pole of the left kidney extending along with the renal capsule with minimal perinephric fluid and perinephric fat stranding. Fat–fluid level was noted in the dilated upper polar and interpolar calyces of the left kidney. Mild heterogeneous hypoenhancement and minimal contrast excretion were seen through the left kidney. An ill-defined retroperitoneal fluid collection was seen on the left side, measuring approximately 24.2 cm × 7.7 cm × 6.9 cm and extending into the left perinephric space. There was evidence of significant cortical thinning and a suspicious breach at the lower pole of the left kidney through which the collection was communicating with the pelvicalyceal system of the left kidney. Fatty attenuation was noted within the collection with evidence of fat–fluid levels (HU value ~ −98 to −115). The collection was seen to extend into the anterior abdominal wall and left psoas and quadratus lumborum muscles. Multiple calculi were noted within the collection in the psoas and quadratus lumborum muscles. Fat–fluid level was noted within the urinary bladder. Multiple subcentimetric-sized paraaortic lymph nodes were seen. Small nonobstructive calculus was seen in the right kidney also.
Non contrast Computed Tomography (NCCT) coronal (a) and axial (b) images show a small left kidney, dense calcification in the perinephric space, and renal capsule at the upper pole (blue arrows) and two calculi in lower pole calyx (white arrow). Contrast-enhanced computed tomography images in axial (c, e and f) and coronal planes (d)mild hydronephrosis with the fat–fluid level in pelvi-calyceal system (PCS) (white arrow with black outline), surrounding collections with fat–fluid level (yellow arrow) and calculi (white arrow in e), and fat–fluid level in the urinary bladder (red arrow)
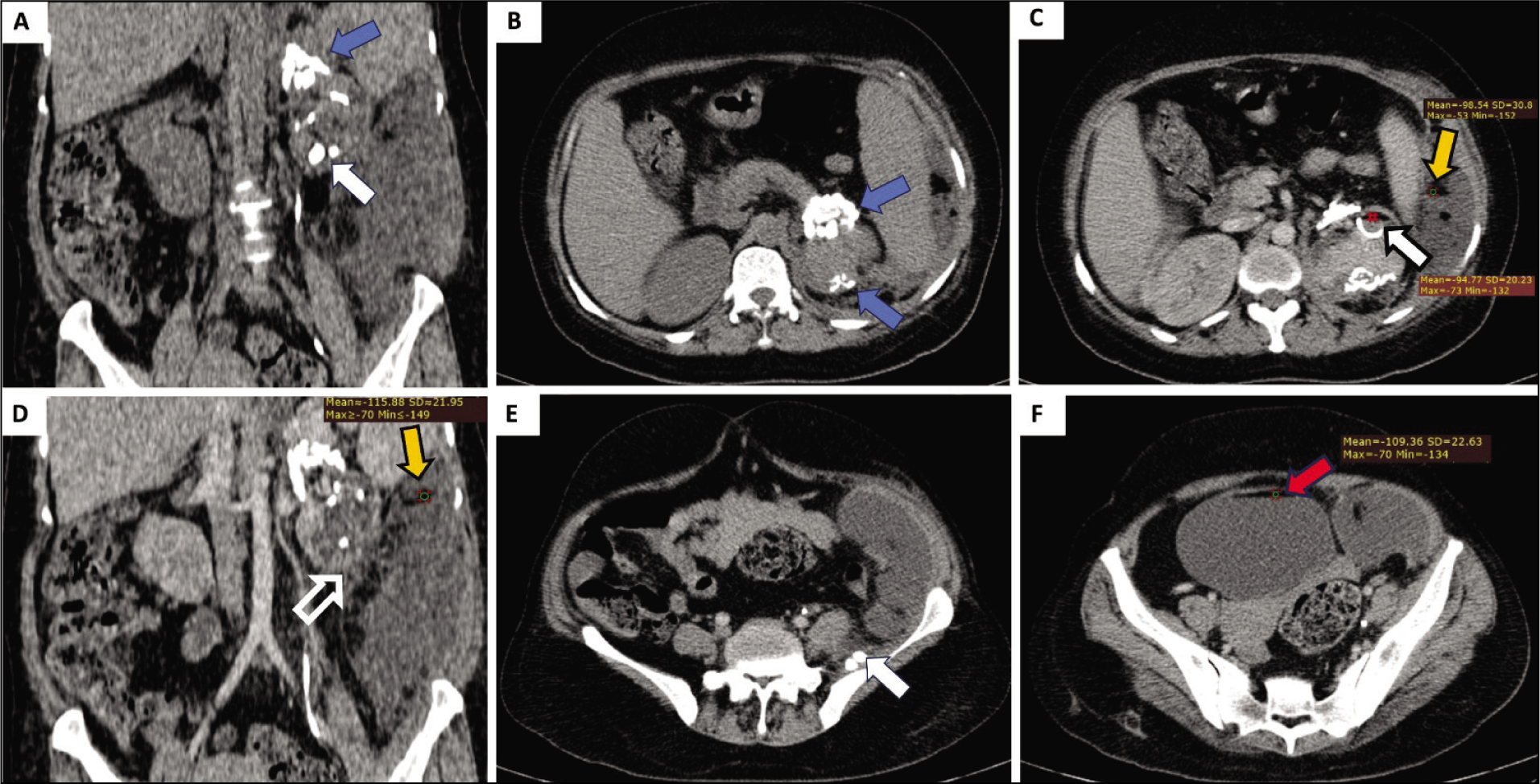
All these CT findings of small and poorly excreting kidney with capsular and perinephric renal calcifications, renal calculi, mild hydronephrosis, and perinephric collections suggest the presence of chronic left pyelonephritis. The presence of calculi in retroperitoneal and abdominal wall collections suggests communication of calyces with collections. Moreover, chyluria, fat–fluid levels in the left pelvicalyceal system and retroperitoneal collections suggest the development of renolymphatic fistula which can either be due to chronic infection or injury to renal parenchyma during prior surgery. The patient was initially treated with percutaneous drainage of retroperitoneal collections and broad-spectrum antibiotics. Her pus culture, later on, came positive for the tubercular organism, following which she was kept on antitubercular treatment.
Discussion
Chyle is a mixture of emulsified fat, albumin, and fibrin, and chyluria is a passage of chyle in the urine. The patient mostly presents with milky urine, more pronounciated in the postprandial period, which may not be associated with other symptoms (mild) or may be associated with other symptoms such as colicky pain (moderate) or hematochyluria or weight loss (severe) which increases the severity and requires additional treatment. Urinalysis shows the presence of chylomicrons and triglycerides in the supernatant fluid.
Chyluria develops due to abnormal connections between the lymphatic and urinary systems. Milky urine is the most common presenting finding, and its biochemical demonstration is required for clinical diagnosis. There are two theories proposed for the formation of chyluria: (1) regurgitation of chyle into the renal excretory system and (2) obstruction by parasitic worms, leading to fistula formation.
Ultrasonography and CT can be used to diagnose chyluria which shows fat–fluid levels in renal, perirenal regions, or the urinary bladder. Magnetic resonance imaging is useful to anatomically delineate any fistulous communication between the lymphatic and renal excretory system. Although invasive and technically challenging, lymphangiography can be used as a preoperative tool to assess the size, site, and number of fistulous communications. Lymphangioscintigraphy is a noninvasive way to visualize the transport and drainage of lymphatic system which can be used to identify the site of the fistula. For grading, retrograde pyelography can be used to assess the severity of the disease depending on the number of calyces involved (one = mild/moderate depending on the clinical condition, >1 = severe) and to manage the case accordingly.
The etiology of chyluria includes parasitic, trauma, surgery, malignancy, radiation, lymphatic malformation, and rarely chronic infections such as tubercular infections. Most of the cases described in the literature are caused by parasitic infection, especially lymphatic filariasis due to Wuchereria bancrofti.[1] Chronic pyelonephritis and renal tuberculosis can rarely cause chyluria due to perirenal lymphatic blockage, chronic inflammation, and rupture of the fragile fornix.[2] In our case, the etiology may be either prior surgery (nephrolithotomy) or chronic tubercular infection, or a combination of both. On lymphangiography, fistulae between the renal collecting system at the fornix and the relatively abundant lymphatic drainage have been described.[3] These eventually communicate with the retroperitoneal lymphatic system through the peripelvic system, leading to chyluria.[4]
Dark fluid levels within the urinary system may be due to fat–fluid level or the more common air–fluid level. Subjectively, both of these look similar fat–fluid levels within the urinary system may be erroneously labeled as the air–fluid level as the latter is fairly common. Air–fluid levels within the renal pelvis are seen in emphysematous pyelonephritis. Within the urinary bladder, air–fluid levels are extremely common in catheterized patients. The presence of air-fluid levels in the abdominal collections represents either infection by gas-forming organisms, fistulous communication with the bowel, or postintervention changes. These potential pitfalls must be properly addressed before making a radiological diagnosis.
Patients with chronic chyluria may have hypoproteinemia, hypolipidemia, and mild variable immunodeficiency. Treatment of chyluria is conservative with the management of underlying etiology. However, in nonresponsive cases with persistent chyluria, sclerotherapy, or surgical treatment is required[3] which may include stripping and ligation of all the lymphatic vessels to the kidney and pelvis, development of a lymphovenous anastomosis, autotransplantation of the kidney, and nephrectomy as a last resort.
Conclusion
Imaging diagnosis of chyluria requires demonstration of fat–fluid levels in the urinary bladder and pelvicalyceal system of the kidney. Depending on severity, the fatty components may also be seen in the perinephric and retroperitoneal collections. The fat–fluid level in the urinary bladder and pelvicalyceal may be mistaken for the more commonly seen air–fluid level which is seen in emphysematous infections, and we advocate measurement of the HU value of the hypodense component in the dark fluid level for correct diagnosis.
Footnotes
Acknowledgements
None
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Patient Consent
The authors certify that he has obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that his name and initials will not be published, and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
CRediT Author Statement
All authors contributed equally to the final version of manuscript.
Data Availability
The data that support the findings of this study are available on request from the corresponding author, [MK]. The data are not publicly available as it can compromise the privacy and confidentiality of the patient.
Use of Artificial Intelligence
No use of AI.
