Abstract
Introduction:
Pial arteriovenous fistula (PAVF) is a rare intracranial vascular malformation with diverse clinical presentations. Early diagnosis and intervention are crucial due to its poor natural history. Endovascular treatment has emerged as a preferred option with promising results.
Introduction
Pial arteriovenous fistulas (PAVFs) are rare intracranial vascular malformations that could be either congenital or acquired. Although it can manifest in various forms, the most common presentations include congestive cardiac failure, seizures, headache, hemorrhage, macrocephaly, and focal neurological deficit.[1] A poor natural history with high mortality rates necessitates early diagnosis and treatment in these patients. Computed tomography (CT) angiography and magnetic resonance angiography (MRA) play a major role in diagnosis with digital subtraction angiography (DSA) being the gold standard.[2] In recent years, endovascular treatment with relatively high occlusion rates has become the preferred treatment for PAVFs.[1,3]
Case Report
An 8-month-old male child presented to the emergency department with left upper and lower limb seizures lasting for a few minutes without secondary generalization and was aborted by medication. The child had a high-grade fever for 1 day and 2–3 episodes of similar seizures before admission with no focal neurological deficits on examination.
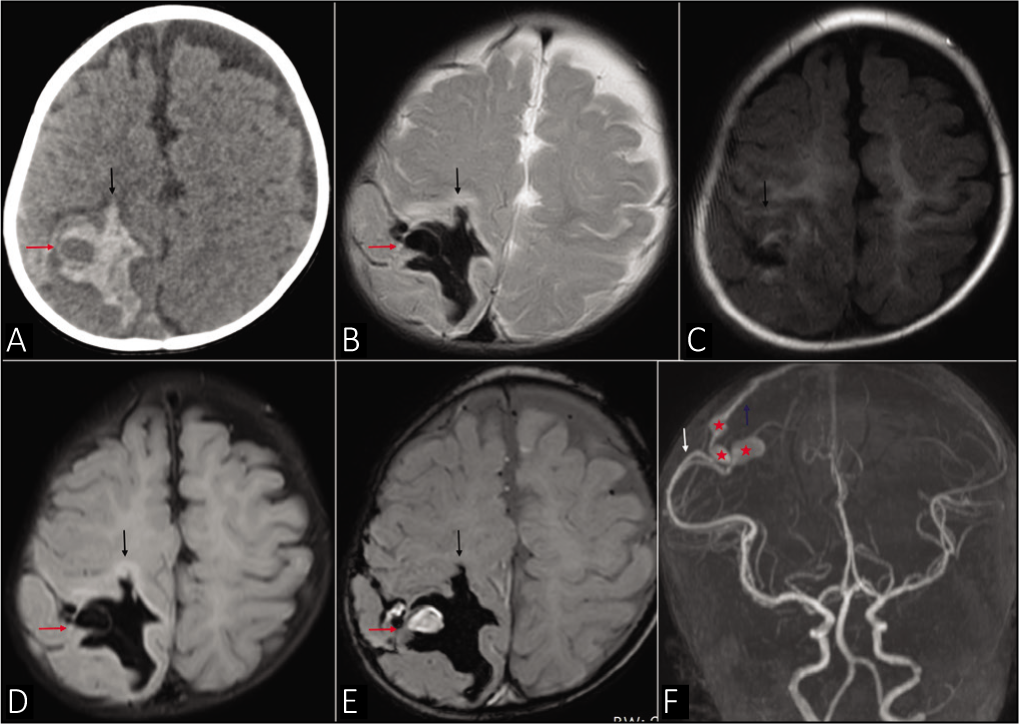
CT brain was done which showed acute intraparenchymal hematoma with mixed density in the right parietal lobe and subarachnoid extension [Figure 1a]. Magnetic resonance imaging (MRI) brain was performed to look for the underlying cause of the hemorrhage. A PAVF was noted along the right parietal convexity supplied by the ectatic parieto-occipital branch of the right middle cerebral artery (MCA) with two fistulous points and venous aneurysms at the site of the fistulae. The venous drainage was noted through the parietal cortical vein into the superior sagittal sinus. The PAVF was located along the lateral aspect of the hematoma with the venous aneurysm within the hematoma [Figure 1 b–f]. Thus, the hemorrhage was secondary to the rupture of the venous aneurysm. A blood workup showed iron deficiency anemia with hemoglobin of 5.8 g/dL for which packed red blood cell was transfused. Other workups including echocardiography, coagulation profile, and platelet counts were normal. Once neurologically stable, the patient was discharged on antiepileptics with a plan for definitive treatment once the anemia was corrected.
(a) Plain computed tomography brain showing right parietal lobe acute intraparenchymal hematoma (black arrow) with hypodense area (red arrow) within, suspicious for an underlying lesion. (b-e) magnetic resonance imaging brain images: (b) Axial T2, (c) Axial T1, (d) Axial FLAIR, (e) Axial susceptibility-weighted imaging (SWI): Showing right parietal lobe acute intraparenchymal hematoma (black arrow) with extension into the subarachnoid space. A well-defined SWI hyperintense area was seen within the hematoma (red arrow) appearing as signal void on T2 and FLAIR (red arrow) – suggesting an underlying vascular malformation. (f) Magnetic resonance angiography shows the pial arteriovenous fistula with arterial supply from the right middle cerebral artery (white arrow), venous drainage into the parietal cortical vein (blue arrow), and the venous aneurysms (red asterisk)
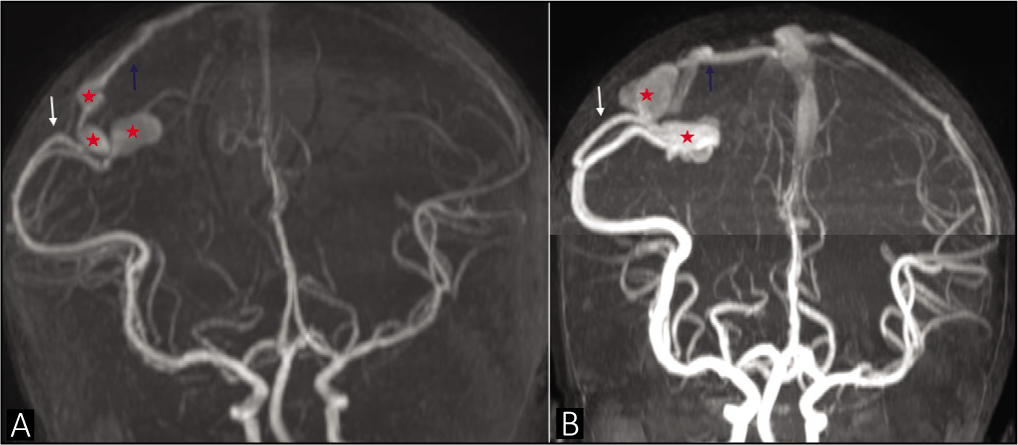
The patient was lost to follow-up and presented later at the age of 3 years and 7 months for planned definitive management. MRI now showed encephalomalacia with hemosiderin staining in the right parietal lobe. The PAVF showed an interval increase in the size of the venous aneurysm [Figure 2]. However, there was no evidence of new or acute hemorrhage and the child was neurologically stable.
Magnetic resonance angiography (a) on initial presentation at 8 months of age and (b) later at 3 years 7 months of age showing interval increase in the size of the venous aneurysm (red asterisk). The pial arteriovenous fistula has the arterial supply from the right middle cerebral artery (white arrow) and venous drainage into parietal cortical vein (blue arrow)
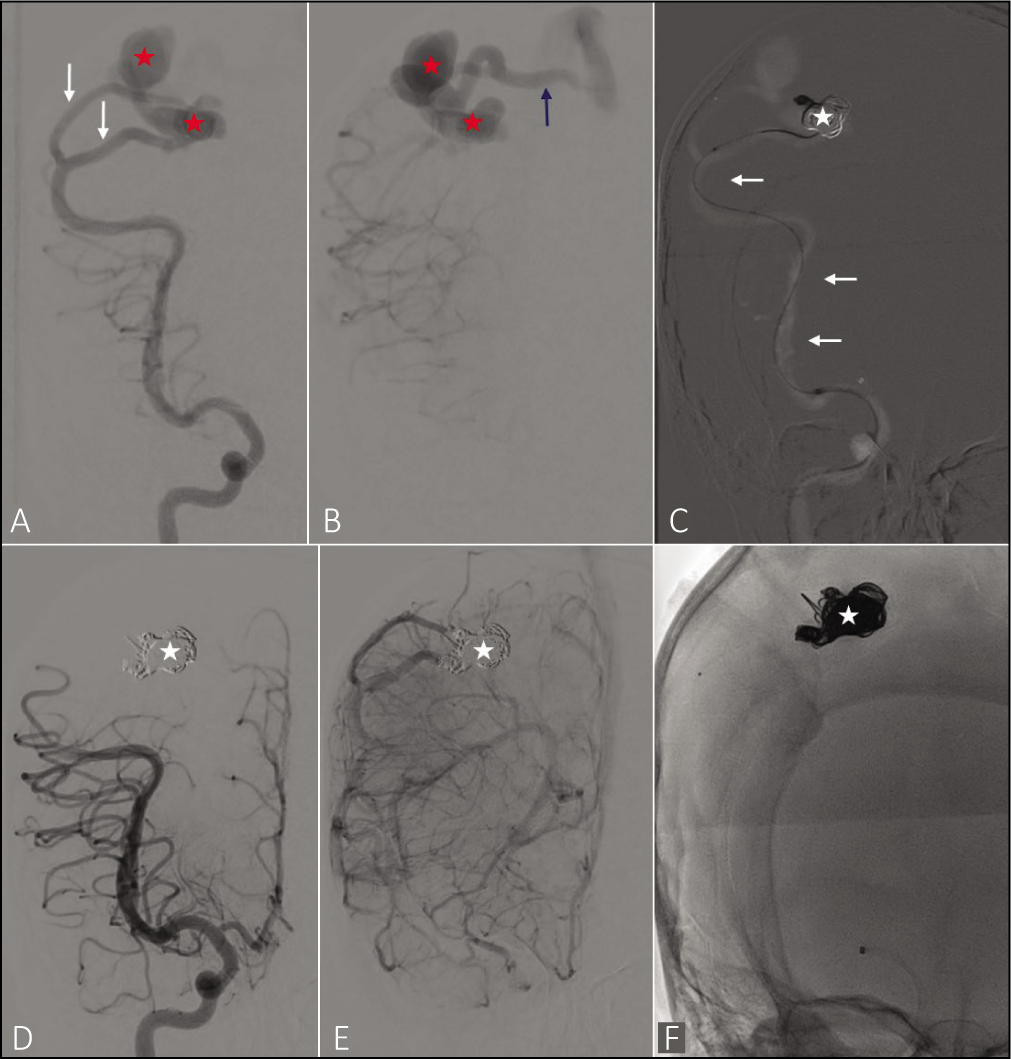
The child was taken up for a DSA which confirmed PAVF and was subsequently taken up for endovascular embolization. The right femoral access was obtained and a 5-French sheath was secured. The distal access catheter was placed in the right proximal M1 MCA. Selective cannulation of both the fistulous points was done using a microcatheter and the venous aneurysm, fistulous site, and distal aspect of the feeding artery were embolized with coils. The final angiogram showed complete exclusion of the fistula with good flow in the right MCA and ACA branches [Figure 3]. The postprocedure period was uneventful with no neurological deficits. The child was healthy at 6 months of clinical follow-up, with no new symptoms and normal growth and development.
(a and b): Right internal carotid artery angiogram showing a parietal pial arteriovenous fistula with arterial supply from the right middle cerebral artery (MCA) (white arrow), venous drainage into parietal cortical vein (blue arrow) and venous aneurysms (red asterisk). (c) Endovascular coiling of the venous aneurysm and the distal feeding artery via the MCA (white arrow). Coil mass was noted within the venous aneurysm (white asterisk). (d and e): Postcoiling angiogram showing complete obliteration of the fistula with coil mass in the venous aneurysm (white asterisk). (f): Fluoroscopy image showing the coil mass (white asterisk)
Discussion
PAVF is a rare entity accounting for only 1.6% of all intracranial vascular malformations and is most commonly seen in children.[1] They represent a direct arteriovenous shunt between pial or cortical arteries and a cortical draining vein with the characteristic absence of an intervening nidus, differentiating it from other classic arteriovenous malformations. Unlike dural AVF which occurs within a dural venous sinus, PAVFs occur in the subpial space or within the brain itself.[4]
PAVFs can be congenital or acquired. The etiology of congenital PAVF is not well understood but likely occurs due to the failure of regression of arteriovenous shunts that forms during the transient stages of vascular genesis. Acquired PAVF in adults can occur rarely following trauma, brain surgery, or cerebral vein thrombosis as a result of the reconstruction of blood flow.[5]
Slight male preponderance is established in previously published literature.[1,4] In neonates, the most common presentation is congestive heart failure resulting from fistula overload. Common presentations in infants and older children include seizures, headache, cerebral hemorrhage, macrocephaly, and focal neurologic deficits. In the majority, seizures are focal as observed in this case.[1]
Eighty percent of the PAVFs are supratentorial and are usually associated with venous varix.[3,4] The most common location of the fistula is the frontal lobe followed by temporal and parietal lobes.[1]
On imaging, they are difficult to detect on plain CT. However, on CT Angiography or MRA, the feeding arteries and draining veins can be identified. The absence of a nidus should raise suspicion of PAVF. DSA is the gold standard for imaging as it allows accurate assessment of the angio-architecture of the lesion.[2,5] In this patient, the presence of a large intracranial hematoma in CT raised the suspicion of an underlying lesion, and MRA established the diagnosis of PAVF. DSA further confirmed the diagnosis and allowed treatment in the same session.
If left untreated, these are associated with 63% mortality.[2,5] Hence, early treatment either by surgical or endovascular route to obliterate these AVFs should be done to prevent life‑threatening intracranial hemorrhage. Improvements in neurointerventional techniques have recently made endovascular embolization the first-line treatment. Endovascular embolization is safe and effective for pediatric PAVFs as a complete obliteration of the pial fistula is achieved in a single session in the majority of cases.[1,3]
Endovascular methods include n-butyl cyanoacrylate embolization, coiling, onyx embolization, or a combination of these. The choice of the embolic agent is determined by the angio-architecture of the AVF.[6] In this case, endovascular embolization with coiling alone was performed with no postprocedural complications and a good clinical outcome.
Challenges in endovascular technique in the treatment of pediatric PAVFs are the presence of high-flow fistulas, tortuous intracranial feeding arteries, small femoral artery access site, limitations in the volume of contrast, and risk associated with general anesthesia in a critically ill neonate.[2,3]
Conclusion
A PAVF is a rare intracranial vascular malformation seen in children. As it has an unfavorable natural history with high mortality and morbidity, high clinical suspicion, and early diagnosis followed by prompt treatment are necessary. Endovascular embolization has shown excellent outcomes in the treatment of pediatric PAVFs with low postprocedure complication rates in expert hands.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Patient Consent
The authors certify that he has obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that his name and initials will not be published, and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
CRediT Author Statement
Dr. Savith Kumar: concept and review of the article Dr. Gayathri S Menon: drafted the article Dr. Prashanth S Urs: reviewed the article Dr. Anup Itihas: reviewed the article.
The manuscript has been read and approved by all the authors, the requirements for authorship as stated earlier in this document have been met, and each author believes that the manuscript represents honest work.
Data Availability
No additional data available.
Use of Artificial Intelligence
The authors declare that they have not used any type of generative artificial intelligence for the writing of this manuscript, nor for the creation of images, graphics, tables, or their corresponding captions.
