Abstract
Background:
Ameloblastomas are a rare type of benign neoplastic tumors that are locally invasive and make up around 1% of all head-and-neck tumors. Mostly diagnosed in people between the ages of 30 and 60 years, it has a global incidence of about 0.5% of cases per million person years. Ameloblastomas are classified as multicystic, unicystic, peripheral, and desmoplastic variants. Unicystic ameloblastomas (UAs), accounting for about 6% of the cases, have a high recurrence rate. It is histologically divided into luminal, intraluminal, and mural types. As the mural variant is aggressive and has the highest chance of recurrence, radical resection of tumor is required.
Introduction
Ameloblastomas are considered benign neoplastic tumors that are locally invasive and the second-most common odontogenic tumors after odontoma.[1] They make up around 1% of all head-and-neck tumors and are believed to originate from the dental lamina and odontogenic epithelium.[2] They can be clinically, radiologically, and histologically diverse which is why, they are also considered impactful neoplastic entities.[1] Malassez first reported it in 1890 under the name adamantinoma. Ameloblastomas often develop in the jaw, primarily in the third-molar region, accounting for around 80% of all cases, whereas the maxilla, particularly in the posterior area, accounts for 20%.[3] Ameloblastomas can grow to enormous size and can cause significant damage to the mandible or maxilla, as well as invade adjacent structures. There are four types of ameloblastomas according to the World Health Organization classification of head-and-neck tumors which consist of multicystic, peripheral, desmoplastic, and unicystic ameloblastomas (UAs).[4] Most instances are diagnosed in people between the ages of 30 and 60 years, with the global incidence estimated to be 0.5 cases per million person years.[5] In addition, the male-to-female ratio has been reported to be 1.2:1.[6,2]
UA was first reported in 1977. It refers to cystic lesions that are often diagnosed as jaw cysts but show ameloblastomatous epithelium upon histological examination.[7] About 6% of ameloblastomas are unicystic, making it the second frequent kind of ameloblastoma after the multicystic variant, which accounts for 92% of cases. UA is more prevalent in the mandible than in the maxilla and typically affects patients in their second decade of life.[8]
We report a rare case report of UA mural variant in the right mandible, which caused thinning of the cortex and erosion of the roots of the right lower molar teeth. The case underscores the importance of prompt diagnosis and treatment of this type of ameloblastoma, particularly in a location where they can cause significant damage.
Case Report
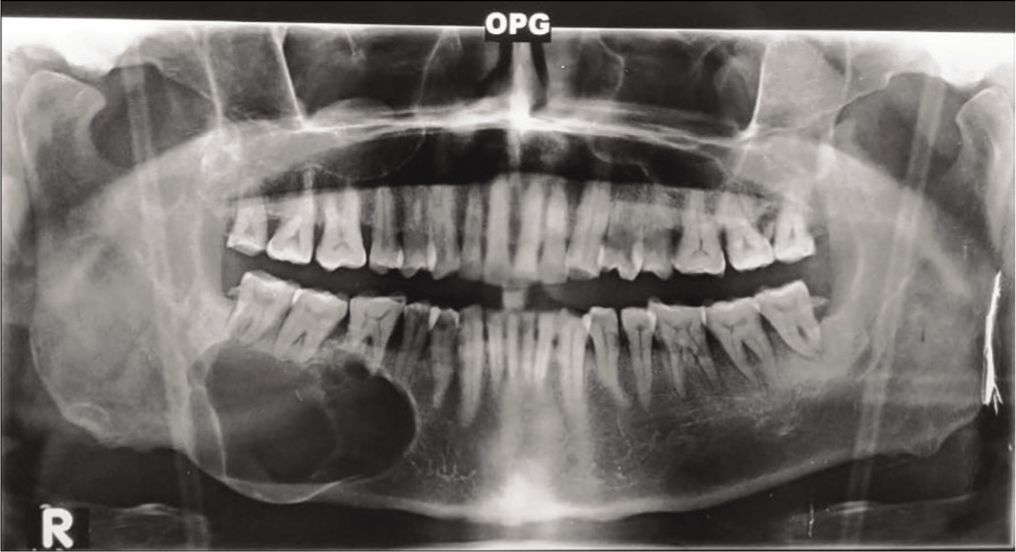
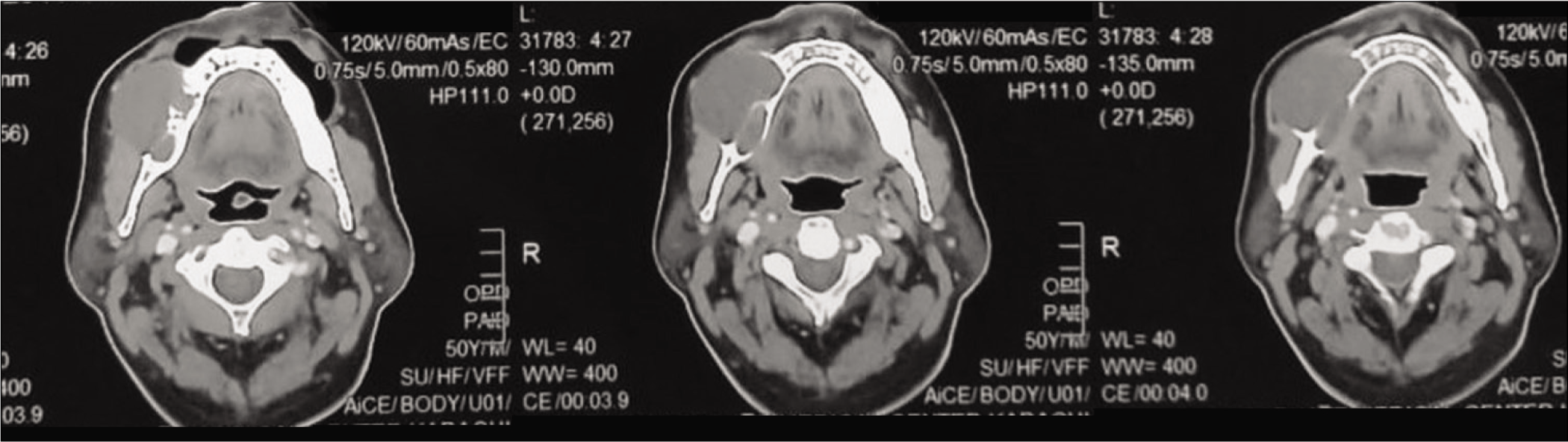
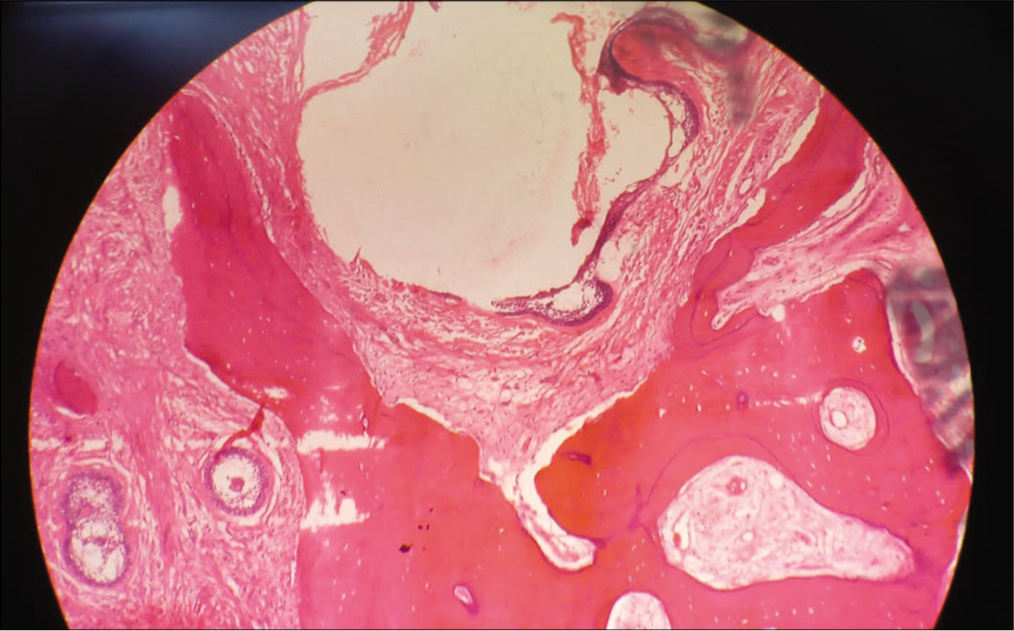
A 47-year-old male patient, hypertensive, presented to the outpatient department of head and neck surgery, on March 2023 with a complaint of painless swelling over the right cheek for 3 years, progressively increasing in size for 2 months. On physical examination, a firm nontender mass was palpated over the right mandible, measuring about 4 cm × 3 cm in size, showing facial asymmetry. The swelling was arising from the right lower first premolar tooth, extending up to ramus of the mandible. Intraorally, buccal mucosa was normal. No cervical nodes were palpable. An orthopantomogram was done, which showed osteolytic lesion involving body of the right mandible, causing thinning of overlying cortex and erosion of the roots of all molar teeth [Figure 1]. Contrast computed tomography scan head and neck showed heterogeneously enhancing soft-tissue density in the right buccal mucosa causing obliteration of buccogingival recess and ipsilateral buccolingual recess with extension up to retromolar trigone. It was causing erosion and destruction of adjacent hemimandible. Multiple subcentimeter lymph nodes were seen at cervical levels Ib and II [Figure 2]. The patient was then planned for surgery under general anesthesia and underwent a right hemimandibulectomy through lip-split incision along with ipsilateral neck dissection. The defect was covered with a free fibular graft taken from the left leg. The resected specimen was sent for histopathology that reports a fibrocartilaginous cyst wall with an intramural component, features suggestive of ameloblastoma [Figure 3].
OPG showing osteolytic lesion involving body of the right mandible, causing thinning of overlying cortex and erosion of roots of all molar teeth. OPG: orthopantomogram.
Contrast computed tomography scan of head and neck showed heterogeneously enhancing soft-tissue density in the right buccal mucosa causing obliteration of buccogingival recess and ipsilateral buccolingual recess with extension up to retromolar trigone. It was causing erosion and destruction of adjacent hemimandible.
Fibrocartilaginous cyst wall showing areas of ulceration replaced by granulation tissue; suggestive of intramural variant of unicystic ameloblastoma.
Discussion
UAs are a rare category of ameloblastomas, diagnosed histologically by the occurrence of a single cystic sac that is lined by odontogenic ameloblastomatous epithelium, only in focal areas.[4] It usually occurs in the second or third decades of life, associated with impacted tooth in 50%–80% of the cases, with third molar being most frequently involved.[4] More than 90% of lesions involve a posterior area of the mandible, followed by parasymphysis. In most of the cases, the lesion arises from dentigerous cyst as well as residual, primordial, root, and globulomaxillary cysts.[8] UAs can also develop from solid ameloblastomas that form multiple cysts after going through cystic degeneration which then fuses into unicystic entity.[7] Ackermann, after conducting a study on 57 cases of UA, categorized it into three different histologic groups. Group I includes luminal UA in which tumor is limited to the luminal surface of the cyst. Group II includes intraluminal or plexiform UA in which tumor cells show nodular proliferation into the lumen without infiltrating into surrounding connective tissue. Moreover, Group III includes mural UA with tumor infiltrating ameloblastomatous epithelium in the connective tissue but not the entire epithelium,[4] our case belongs to this group. Most commonly it presents asymptomatically in early stages, although large lesions cause painless swelling, facial asymmetry caused by adjacent soft tissue, and buccal or lingual cortical infiltration, rarely associated with mucosal ulceration in case of the continued growth of tumor.[9] However, UA is less aggressive than the other types, but it has a high recurrence rate (10%–25%).[9] Taking this into consideration, two treatment regimens have been recommended; radical and conservative treatment which includes marsupialization followed by enucleation. UAs of luminal and intraluminal types can be conservatively managed, however, those with intramural growth require radical resection.[6] It is also advised that curettage of bone, following enucleation, should not be done briskly, as it can lead to focal implantation of ameloblastomas into the bone more deeply. The radical management involves resecting the tumor in a block along with bony margins and then reconstructing the defect side with graft. This regimen is associated with multiple complications including problems in jaw movement, facial disfigurement, and mastication difficulties, but at the same time, it has shown a low recurrence rate in comparison with a conservative approach. According to a research conducted by Kim et al., the recurrence rate is 3.6% after radical resection and 30.5% after a conservative approach through marsupialization and enucleation.[8,9]
In this case, while we could go for conservative surgery, radical resection was opted for other pathological factors such ase size, extent, and location had to be kept in mind and the choice of treatment should be made accordingly. Regardless of the surgical approach taken by surgeon, long-term follow-up of the patient should be considered delayed recurrence has been observed in UA. The patient is kept in close 6-month follow-up with no aggressive outcome as of yet.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Patient Consent
The authors certify that he has obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that his name and initials will not be published, and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
CRediT Author Statement
Soubia Akhtar: Acquisition of data, drafting the article and revising it critically for important intellectual content Ashar Masood Khan: Acquisition of data and drafting the article Yumna Afzal (corresponding author): Acquisition of data and drafting the article Mubasher Ikram: Final approval of the version to be published.
Data Availability
Not applicable for this case report.
Use of Artificial Intelligence
Not applicable for this case report.
