Abstract
Introduction:
Ischemic stroke in younger adults has been increasing over the years. It is often challenging to identify the underlying etiology and pathogenesis of stroke in the young. Inherited thrombophilias contribute to about 1%–4% of ischemic strokes in the young. Of the inherited thrombophilias, isolated protein S deficiency is an important risk factor for the development of arterial ischemic strokes. It is commonly associated with venous thrombosis but it is rarely associated with arterial ischemic strokes, contributing to 4%–12% of young strokes.
Introduction
It has been reported that about 10% to 15% of all strokes occur in the age group of 18–50 years.[1] Primary thrombophilic disorders are well known to cause venous thrombosis, but they also contribute to about 1%–4% of ischemic strokes. Inherited thrombophilias include protein C, protein S, antithrombin III (AT III) deficiency, and factor V Leiden mutation.[2] Although protein S deficiency has been frequently associated with deep vein thrombosis and pulmonary embolism, it has been rarely reported in arterial strokes. Isolated protein S deficiency may be seen in up to 10% of patients with young ischemic strokes.[3] Detection of thrombophilia in young patients with stroke is very important as it can change the clinical management, thereby preventing recurrent episodes.
Case Report
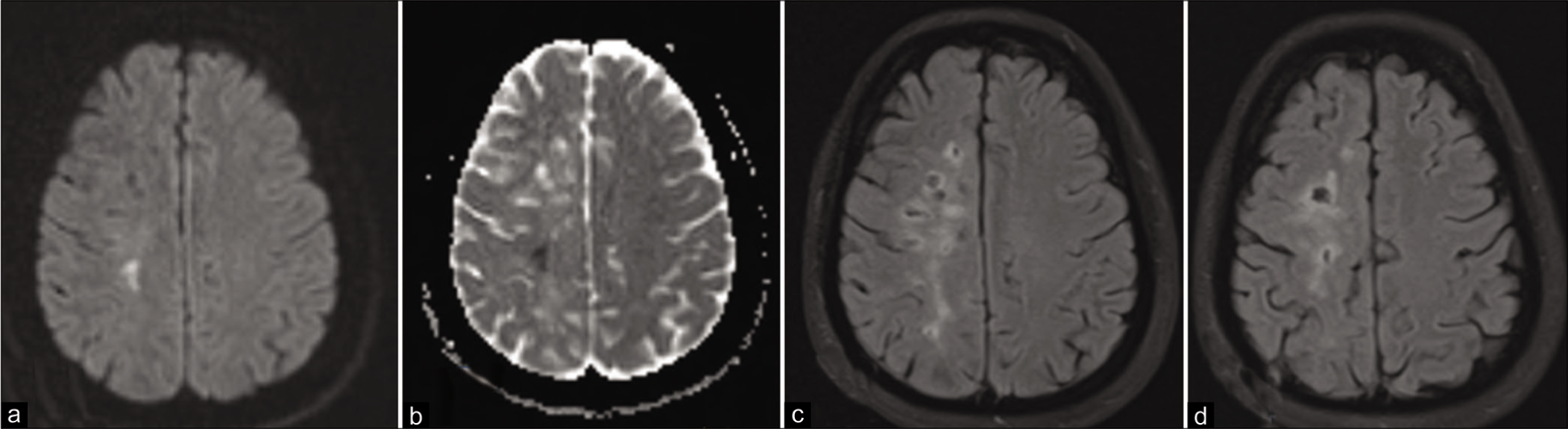
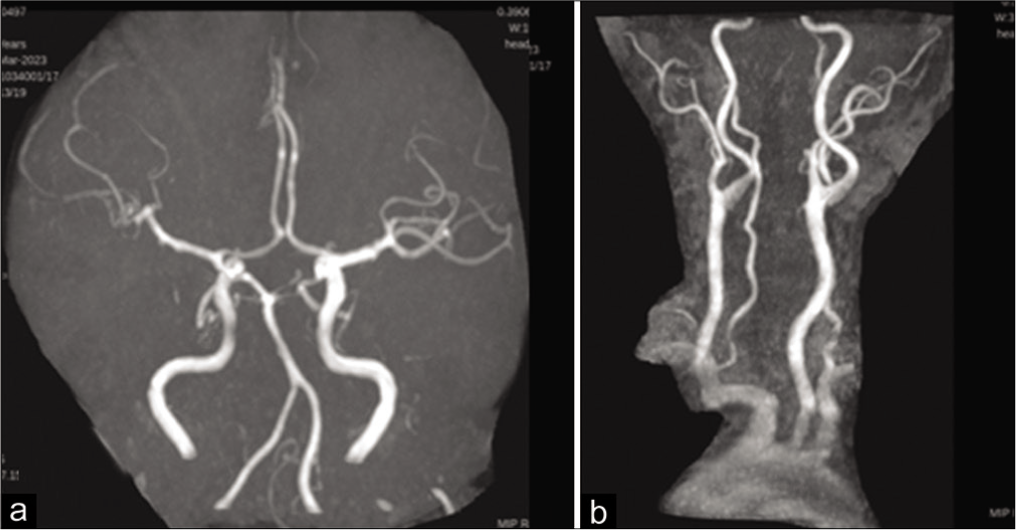
A 38-year-old female with no known comorbidities, presented to us with complaints of acute onset of unsteadiness on walking and weakness of the left upper and lower limbs for 2 days’ duration. Two days back, on waking up, she was unsteady and was swaying to the left side; however, she did not seek medical help on that day. On the next day, she developed difficulty in holding objects with her left hand and had dragging of the left lower limb while walking. On examination, she was conscious and oriented, had left upper motor neuron type of facial palsy, grade 4/5 power of left upper and lower limbs, with brisk deep tendon reflexes on the left side, left extensor plantar, and left-sided cerebellar signs. Magnetic resonance imaging (MRI) of the brain showed a small lesion showing diffusion restriction in the right corona radiate suggestive of acute infarct; multiple T2 fluid-attenuated inversion recovery hyperintensities were noted in the right centrum semiovale suggesting multiple chronic infarcts [Figure 1]. Magnetic resonance (MR) angiography was normal [Figure 2]. She was started on dual antiplatelets and high-intensity statin with supportive physiotherapy and she improved significantly over 1 week. She had no clinical history of stroke in the past. She has two children and she has no history of abortions. She has no history of joint pains, joint swelling, or skin rashes. Her mother had developed a stroke at the age of 54 years and expired. Her HbA1C and fasting lipid profile were normal. The cardiac evaluation was done, echocardiography was normal, and Holter showed no arrhythmias. Since her MRI showed evidence of recurrent infarctions, a workup for the same was done. A vasculitic workup was done, rheumatoid factor, antinuclear antibody (ANA)-indirect fluorescent, ANA profile, cytoplasmic antineutrophilic cytoplasmic antibody, perinuclear antineutrophil cytoplasmic antibody were negative. Serum homocysteine level was normal. After 2 weeks of the onset of symptoms, a hypercoagulopathy workup was done, protein C function was 97%, AT III level was normal and factor V Leiden mutation was not detected in exon 10 of the F5 gene. The antiphospholipid antibody panel (antibeta-2 microglobulin/lupus anticoagulant and anticardiolipin antibody) was also negative. Her protein S function was only <10%, indicating a protein S deficiency. She was initiated on apixaban 5 mg twice daily and has been under follow-up for the past 3 months. She had no further strokes during this period of follow-up.
Magnetic resonance imaging brain showing hyperintensity on diffusionweighted image, (a) with corresponding low apparent diffusion coefficient (ADC) values, (b) in the right corona radiata, suggestive of acute infarct, (c and d) show multiple hyperintensities with cystic changes involving the right centrum semiovale, on fluid-attenuated inversion recovery sequences suggestive of old infarct.
Magnetic resonance angiography showing normal intracranial arteries, (a) and normal neck vessels (b).
Discussion
Protein S is a Vitamin K-dependent glycoprotein, which, along with active protein C, inhibits the coagulation cascade. Protein S is reported to be a cofactor for activated protein C, which inhibits Factor Va and Factor VIIIa.[3] Protein S deficiency is commonly associated with venous thromboembolic episodes; however, arterial ischemic strokes have been reported rarely with protein S deficiency. One of the first published reports of the same was by Girolami et al. and Sié et al.[4,5] In another study, Wiesel et al. studied 105 patients with protein S deficiency, and noted 64 thromboembolic episodes, including 14 arterial thrombotic episodes involving the heart or brain; these occurred in young individuals with a mean age of 25 ± 13 years.[6] Hooda et al. have reported a case of acquired protein S deficiency-related recurrent ischemic stroke in a 16-year-old girl.[3] Our patient was a 38-year-old female with no vascular risk factors who presented with her first clinical stroke; however, her MRI brain showed evidence of recurrent old infarctions. It was interesting to note that all the infarcts were in the right middle cerebral artery (MCA territory); however, MR angiography showed no abnormality of the right MCA; the vasculitic workup was also negative. Her mother (who had no vascular risk factors) also had a stroke at 54 years of age; hence, our patient could probably have an inherited protein S deficiency. Congenital protein S deficiency is an autosomal dominant disease.[7] Fearon et al. reported an arterial thromboembolism in a patient with a family history of protein S deficiency.[8]
It is very important to identify the underlying etiology in a young patient with stroke. Detection of underlying thrombophilia warrants treatment with oral anticoagulation, ensuring prevention of further thrombotic episodes. In prior studies, Warfarin was used to treat and prevent thromboembolic events in patients with a protein S deficiency.[9] However, it has been reported that Vitamin K antagonists may transiently suppress the activity of protein C or S, which may promote thrombosis formation in thrombophilia. Recent studies have shown that direct oral anticoagulants (DOACs) such as apixaban and rivaroxaban have been used in patients with ischemic stroke caused by protein S deficiency.[9,10] Although controlled studies on administering DOACs for preventing thrombotic events in patients with protein C deficiency are lacking, observational studies have demonstrated that DOACs can be safe and effective in treating patients with thrombophilia.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Patient Consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
CRediT Author Statement
Dr. Sheetal S: Conception of the idea, drafting the article, critical revision of the article and Final approval of the version to be published;
Dr. Nikhil Gladson: Critical revision of the article and final approval of the version to be published;
Dr. Joseph George: Critical revision of the article and final approval of the version to be published;
Dr. Jennifer Daniel: Critical revision of the article and final approval of the version to be published.
Data Availability
Data is available upon reasonable request from the corresponding author.
Use of Artificial Intelligence
Artificial intelligence was not used for this study.
