Abstract
Introduction:
The earlier the hearing loss in a child, the more difficult will it be for the child to achieve optimal development and lead a normal life. This study is directed toward early identification of deafness in neonates born to gestational diabetic mothers that help in the early rehabilitation of the child.
Materials and Methods:
Around 50 infants born to mothers with gestational diabetes were taken and screened after obtaining consent from parents and clearance from the ethics committee. Neonates born to gestational diabetic mothers in Father Muller Medical College were included. Critically ill neonates, neonates with hyperbilirubinemia, family history of deafness, use of ototoxic drugs, and low APGAR score babies (<6) were excluded. Detailed family history and information of the patients were obtained. The occupation of both parents, family income, and consanguinity was noted. It was also observed if the antenatal mother has attended ANC clinic regularly. Results: A longitudinal descriptive study was conducted on 50 infants born to gestational diabetic mothers. Hearing loss was assessed using distortion product otoacoustic emission. The majority of gestational diabetic mothers were homemakers followed by salesgirls. The other occupations also included Anganwadi workers, beedi workers, manual laborers, tailors, and farmers. The list also included cooks, hotel receptionist, office assistant, and floor manager. Most of the fathers were drivers (n = 10) followed by farmers. The list also included fishermen, manual laborers, painters, salesmen, security personnel, waiters, tailors, teachers, office assistants as well as storekeepers.
Conclusion:
Our study assessed if there was a correlation between hearing loss in infants and gestational diabetes. Hence, in our study, we found that there may not be any relationship between hearing loss and diabetes in pregnancy.
Introduction
It has been well documented that hearing plays a great role in the speech and language development of a child. Hearing loss not only delays communication but can also have a profound effect on the self-esteem and social skill development of the child. The earlier the hearing loss in a child, the more difficult will it be for the child to achieve optimal development and lead a normal life.[1] Therefore, it is of great importance to assess the hearing loss at the earliest and provide necessary care for the child. Diabetes is one of the most common diseases in the world. It has been known to affect approximately 62 million[2] people in India and the number is known to rise fast. The etiology of diabetes is multifactorial with genetics and obesity playing an important role in its onset.
Most organs of the human body are affected by diabetes.[3,4] Blood levels of glucose, lipids, and amino acids of the mother in case of gestational diabetes mellitus (GDM) make the intrauterine environment more challenging to the development of the fetus. It would therefore be expected that these maternal metabolic disturbances would have the potential to adversely affect the overall health of the newborn, and, specifically for this study the development of the auditory system.
GDM is defined as any degree of glucose intolerance with the onset or first recognition during pregnancy with or without remission after the end of pregnancy.[5] Criteria for diagnosis of GDM with 100 g oral glucose (O’Sullivan and Mahan modified by Carpenter and Coustan). Babies born to diabetic mother have an increased risk of macrosomia[5,6] that is much higher when compared to normoglycemic controls.
GDM has also been linked with cardiometabolic risk factors such as lipid abnormalities, hypertensive disorders, and hyperinsulinemia.[5] These might later result in the development of cardiovascular disease and metabolic syndrome. To provide adequate management of the disease in potential mothers who are at the risk of developing GDM, it is vital to understand the risk factors, the genetic factors, as well as the pathophysiological mechanism. Studies have shown that women with prior GDM can prevent type-2 diabetes mellitus by lifestyle modification.[7] The risk of birth defects in babies born to women with poor diabetic control is high compared with that of pregnant diabetic mother whose diabetes is well controlled. Birth defects include spinal cord defects (spina bifida), heart defects, skeletal defects, and defects in the urinary, reproductive, and digestive systems.[8] It has also been noticed that infants of women having diabetes have a greater chance of hypoglycemia, jaundice, and respiratory distress at birth. Various types of adverse outcomes for infants and mothers can be seen with abnormal glucose metabolism.[7]
Therefore, pregnant or expecting mothers are often screened for diabetes mellitus. The approach to GDM has altered markedly in the last decade. Universal screening recommends women with elevated blood glucose level monitor blood glucose levels at home to attain normoglycemia at the earliest. Majority of the women with gestational diabetes respond to diet control alone. However, if this fails, insulin therapy is recommended.[9]
The rationale for studying is rooted in the potential impact of maternal health conditions on fetal development and long-term health outcomes. GDM is a common pregnancy-related condition characterized by high blood glucose levels during pregnancy. It has been associated with various adverse maternal and fetal outcomes. Given that hearing is a crucial sensory function for communication, learning, and overall well-being, investigating the potential link between GDM and hearing impairment is both clinically relevant and scientifically intriguing. The prenatal environment significantly influences fetal development, and maternal health plays a pivotal role. Maternal hyperglycemia in GDM can lead to altered fetal growth and development, potentially affecting the auditory system’s formation. Understanding the impact of GDM on hearing can contribute to a broader understanding of how maternal metabolic conditions can influence fetal organogenesis. Hearing impairment in infants can have profound consequences on language development, cognitive skills, and social interactions. Identifying potential risk factors, such as GDM, enables early monitoring and intervention, which can significantly improve the long-term outcomes for affected infants. Exploring the potential association between GDM and hearing impairment could contribute to the development of targeted screening strategies and interventions. GDM is a prevalent condition affecting a significant number of pregnant women. As the incidence of GDM continues to rise due to lifestyle and dietary changes, understanding its potential impacts on various aspects of infant health becomes imperative. Addressing potential hearing impairment in GDM-affected infants can have substantial public health implications in terms of early diagnosis, treatment, and quality of life. While extensive research has been conducted on the maternal and fetal effects of GDM, the potential association between GDM and hearing impairment remains relatively underexplored. This research area offers an opportunity to contribute novel insights into the intricate relationship between maternal metabolic health and offspring outcomes.
Newborn hearing screening testing has long been technically safe and cost-effective and was introduced relatively early in many countries. This study could be used to assess the hearing loss in children born to gestational diabetic mothers. Hearing test is the right of every newborn child. Parents must be informed accordingly (parent information sheets are available) before carrying out the test, and it is their responsibility to decide on participation. If parents refuse the screening test, documentation of the same along with the signature of at least one parent is necessary. A binaural hearing screening test should be performed by the 3rd day of life by an automated Transient Evoked Otoacoustic Emissions[10] or Automated Auditory Brainstem Response (AABR) measurement. In children at risk, the AABR measurement is mandatory.[7] This study is directed toward the early identification of deafness in neonates born to gestational diabetic mothers that help in the early rehabilitation of the child.
Materials and Methods
After obtaining Institutional Ethical Committee clearance (FMMC/FMIEC/4474/2017, dated November 18, 2017) 50 infants born to mothers with gestational diabetes were taken and screened after obtaining consent from parents and clearance from the ethics committee. Neonates born to gestational diabetic mothers in Father Muller Medical College were included. Critically ill neonates, neonates with hyperbilirubinemia, family history of deafness, use of ototoxic drugs, and low APGAR score babies (<6) were excluded from the study.
A longitudinal descriptive study of the neonates born to gestational diabetic mothers at Father Muller Medical College Hospital, Mangalore. The study was conducted in November 2017–February 2018 (18 months). The sample size was calculated based on the article “Prevalence of gestational diabetic mellitus and its outcomes in Western Rajasthan.”[11] Sample size was calculated using the formula, n = Zα 2 P (1 − p)/e 2 where Zα =1.96 at 95% confidence value, P = 6.6, e = Allowable error (7%) and the estimated sample size was 50.
Detailed family history and information of the patients were obtained. The occupation of both parents, family income, and consanguinity were noted. It was also observed if the antenatal mother has attended ANC clinic regularly. Maternal and paternal family history of deafness, maternal ototoxic drug intake, and eruptive fever were noted. Infants born to mothers with preeclampsia, any radiation exposure, or TORCH infections were excluded from the study. It was also noted if the mother had malnutrition or history of repeated abortions. The parity of the mother was also noted. In addition to that incidence of prolonged labor, premature birth, low birth weight, and craniofacial anomaly, if any, were noted. Moreover, any history of NICU admission for more than 3 days was noted. Children born with craniofacial anomalies and hyperbilirubinemia were excluded from the study. APGAR score at 1 min and 5 min after birth was also noted.
The general physical examination of the neonates was done. The length, weight, the chest circumference, abdominal girth, and head circumference of the child were noted. The preaural area, pinna, and postaural area of the newborn were examined for any congenital anomaly. The external auditory canal was examined. If there was any atresia or wax, the test would be done at a later date. The external appearance of the nose and oral cavity was examined. The infant was also screened for any neurological, cardiovascular, abdominal and respiratory problems. The test was done, and if an infant failed to pass the test, the test was repeated after 6 weeks or whenever the child came for immunization. If the child failed the second time as well, brainstem evoked response audiometry (BERA) was done. The 1st recording of DPOAE was done within the first 3 days of birth. If the result is passed, hearing is normal (phase 1). If the result was negative, DPOAE was repeated in the next visit (phase 2). This was done after 6 weeks of birth or when the child came for immunization. If the result is passed, the hearing is considered to be normal. If the result was negative, DPOAE was repeated again at 3 months (phase 3). If the result was still fail, neonates were to be assessed with further intervention (BERA). At the end of the study, all the data were combined and assessed using mean, standard deviation, frequency percentage, and Chi-square test. Statistical software, namely SPSS version 26.0, (IBM Corp, Chicago, Illinois, USA) was used for the analysis of data, and Microsoft Word and Excel have been used to generate graphs, tables, etc.
Results
Fifty infants born to gestational diabetic mothers were included in the study. The assessment of hearing was done using otoacoustic emission (OAE).
Table 1 compares the occupation of both father and mother of infants born to gestational diabetic mothers. The majority of gestational diabetic mothers were homemakers followed by sales girls. The other occupations were also included Anganwadi workers, beedi workers, manual laborers, tailors, and farmers. The list also included cooks, hotel receptionist, office assistant, and floor manager. The father’s occupation was also taken into consideration. Most of the fathers were drivers (n = 10), followed by farmers. The list also included fishermen, manual laborers, painters, salesmen, security personnel, waiters, tailors, teachers, office assistants as well as storekeepers [Table 1].
Distribution of demographic details among the study participants (n = 50)

All mothers attended ANC regularly. The mothers were screened for gestational diabetes. 100 g of oral glucose was given to the mother and their blood sugar levels were monitored. The acceptable fasting glucose level was 95 mg/dL. The blood glucose level was monitored after 1 h. The normal blood glucose level should be <180 mg/dL and at 2 h after the test, the blood glucose limit should be within 150 mg/dL. At 3 h post the test the blood glucose should be within 140 mg/dL. GDM is diagnosed when any two values are met or high.
None of the infants born to gestational diabetic mothers had family history of deafness, birth asphyxia, icterus, or any other illness. The mothers had no history of repeated abortions, radiation exposure, or ototoxic drugs. The APGAR score was also calculated. It calculated the heart rate of the child, respiratory effort, muscle tone, skin color, and reflects irritability. Each of these parameters was scored from 0 to 2. The score between 0 and 3 is low score, 4–6 is moderately abnormal, and between 7 and 10 is reassuring (normal). The APGAR score was routinely done 60 s after birth of the infants and then repeated again 5 min after birth. In case there is difficulty in resuscitating the child, PGAR score may be done again at 10, 15, and 20 min. A perfect APGAR score of 10 means that the infant is in the best possible condition. Immediate resuscitation is needed when an infant is born with APGAR score of 0–3.
Persistently, low APGAR score may be the result of fetal distress, prematurity, drugs given to the mother during labor. The persistence of low APGAR score (0–3) at 20 min of age is predictive of high rates of morbidity (disease) and mortality [Table 2]. The parity of the mothers was also noted. 58% of GDM mothers were P2 where as 42% of mothers were P3. The mean age of doing OAE was also calculated. Majority of the infants, OAE was done on the 2nd day of life. It was also done at 1st day of life up to the 5th day of life. The DPOAE was recorded and 88% of the newborns passed the test. Twelve percent failed the test and the child was asked to repeat the investigation after 6 weeks or when the child is bought for his next immunization. Out of the 12% whose DPOAE was repeated all the infants passed the test in the second visit.
Distribution of study variables among the study participants (n = 50)
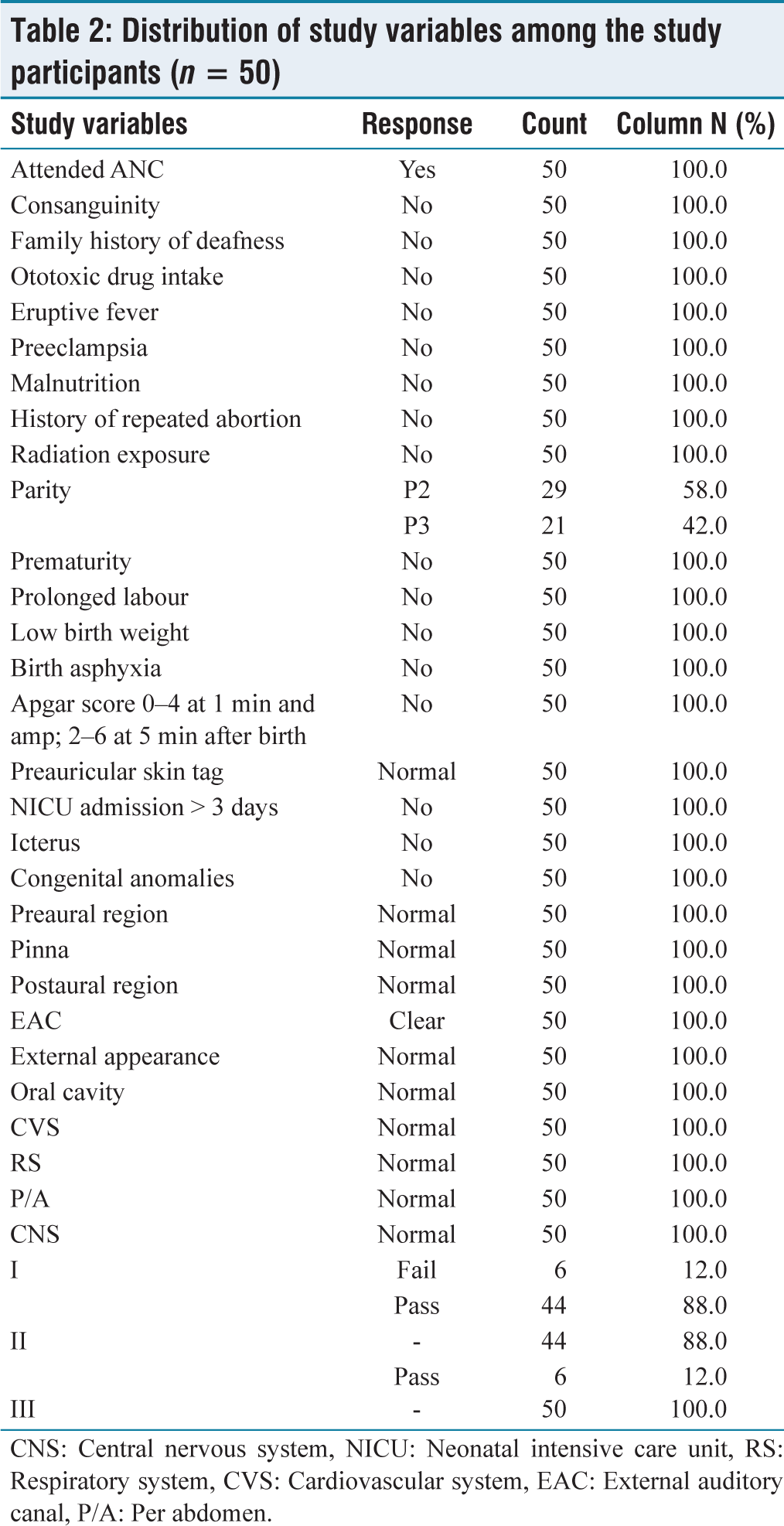
CNS: Central nervous system, NICU: Neonatal intensive care unit, RS: Respiratory system, CVS: Cardiovascular system, EAC: External auditory canal, P/A: Per abdomen
Discussion
Congenital disorders can cause severe hearing loss and are present at birth. Development anomalies of the bony and membranous labyrinth can occur. It is also important to assess if the hearing loss is unilateral or bilateral. It is estimated that there are 6.5 million Americans with some degree of unilateral hearing loss.[12] Binaural hearing is important due to “summation effect,” there is an improvement in auditory thresholds for pure tone and speech of approximately 2–3 dB. Although it may seem inconsequential, its favorable effect on speech understanding and case of listening is well documented.[12] Another important factor is the “head shadow effect.” The head attenuates the speech intensity as much as 6.4 dB as signal progresses from one ear to the other. Yet another factor is the “Squelch effect.” It is the ability of the two ears to suppress background noises to the advantage of the primary signal.[13] Bess and Tharpe found that children with unilateral hearing loss experience considerable difficulty in understanding speech in the presence of competing signals, and they performed poorer than those with normal hearing even when the primary signal was presented to the good ear.[12] Acquired hearing loss is common in children in children due to prenatal “TORCH” organisms (i.e., toxoplasmosis, rubella, cytomegalovirus, and herpes). Sensorineural hearing impairment is common in consanguineous marriage.
According to Vohr et al.[14] parental consanguinity is more common in hearing loss among infants. Hence, in our study, consanguinity was ruled out. In our study, there was no history of hearing loss in infants born in previous pregnancies. It was also in our study, there were no infants born with birth asphyxia and low APGAR score. Hypoxia has a strong association with hearing loss. Adequate oxygenation and perfusion are needed for normal cochlear function. In our study, infants born with neonatal jaundice and history of exchange transfusion were excluded as it is known to cause hearing loss. Babies whose mothers received ototoxic drugs were also excluded from our study as they are known to be a significant risk factor for hearing loss.
The investigation into the potential correlation between hearing loss in infants and gestational diabetes has illuminated a complex interplay between maternal health and fetal outcomes. The quest to understand the factors influencing the auditory development of newborns gains significance as hearing holds a paramount role in the cognitive, linguistic, and social development of children. This study has delved into the intricate web of congenital and acquired factors that contribute to hearing impairment, ultimately revealing a nuanced perspective on the role of gestational diabetes in this context.
Congenital disorders can cast a profound impact on hearing, exerting their influence right from birth. Developmental anomalies affecting the bony and membranous labyrinth can manifest as hearing loss, underscoring the importance of meticulous assessment and early intervention. Differentiating between unilateral and bilateral hearing loss holds pivotal importance, with the prevalence of unilateral hearing loss being substantial. This phenomenon prompts the exploration of binaural hearing advantages, as exemplified by the “summation effect.” Despite the seemingly marginal improvement of auditory thresholds, this effect has demonstrated a tangible enhancement in speech comprehension and overall auditory experiences.
Furthermore, the intriguing dynamics of the “head shadow effect” and the “Squelch effect” underscore the remarkable intricacy of the auditory system. The former elucidates the ability of the head to attenuate speech intensity during its transition between ears, while the latter highlights the ear’s remarkable capacity to suppress background noise, favoring the perception of the primary auditory signal.
Our longitudinal descriptive study, encompassing 50 infants born to mothers with gestational diabetes, applied distortion product otoacoustic emission as a comprehensive assessment tool. Encouragingly, all infants born to mothers with gestational diabetes showcased successful test results. This finding augments the current understanding of gestational diabetes’s potential impact on auditory development, suggesting that, in the context of this study, gestational diabetes may not be significantly correlated with hearing loss.
Conclusion
This study has provided a nuanced perspective on the potential correlation between hearing loss in infants and gestational diabetes. It navigated the labyrinthine pathways of congenital and acquired factors that shape auditory development, elucidating the intricate interplay between maternal health and infant outcomes. While the study’s findings suggest a lack of significant relationship in the context studied, the journey through these complexities underscores the need for continued research and comprehensive assessments to fully comprehend the multifaceted world of infant hearing impairment and its potential connections to maternal health conditions. The study contributes to the broader understanding of maternal-fetal health, sparking future explorations that stand to benefit both medical understanding and the lives of newborns.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional Ethical Committee Approval Number
FMMC/FMIEC/4474/2017, dated November 18, 2017.
CRediT Author Statement
Conceptualization: JKP and MBT; Manuscript writing: JKP and MBT; Manuscript revision: JKP, MBT, and JJ; Proof reading: JKP, MBT, and JJ. All authors accepted to publish the manuscript.
Data Availability
No.
Use of Artificial Intelligence
In the publication, there is no use of Artificial Intelligence.
