Abstract
Background:
Neutrophil extracellular traps (NETs) have emerged as potential biomarkers in autoimmune diseases. This study aimed to evaluate NETs as markers of disease severity in rheumatoid arthritis (RA).
Methods:
A prospective cross-sectional study was conducted from May 2022 to December 2023 at a tertiary care hospital in Western India. Patients with RA were categorised into high disease activity (HDA-RA) and low disease activity (LDA-RA) groups based on the DAS-28-CRP score (≥3.2 vs <3.2). Age and sex-matched healthy individuals served as the control group. Levels of NETs markers, cell-free DNA (cfDNA), myeloperoxidase-DNA (MPO-DNA) complexes and citrullinated histone-3 (citH3), along with conventional markers (C-reactive protein [CRP] and erythrocyte sedimentation rate [ESR]) were measured and compared across groups.
Results:
Patients with HDA-RA demonstrated significantly elevated levels of NET markers—cfDNA (197.7 ± 18.7 ng/mL), MPO-DNA (67.3 ± 12.0 mAU/mL) and citH3 (4.1 ± 1.4 ng/mL)—compared to low disease activity (LDA-RA) and controls (P < .001). NET markers correlated strongly with CRP, ESR and DAS-28-CRP (r = 0.436–0.647, P < .001). Logistic regression confirmed cfDNA, MPO-DNA and citH3 as the independent predictors of RA disease severity, with citH3 showing the strongest association (odds ratio = 2.871, 95% confidence interval: 1.786–4.616, P < .001). Receiver operating characteristic analysis identified cfDNA as the most accurate marker for distinguishing HDA-RA from LDA-RA (area under the curve = 0.87, sensitivity 81%, specificity 79%), outperforming traditional markers.
Conclusions:
NET markers, especially cfDNA, may complement traditional inflammatory markers in assessing RA disease severity. While their diagnostic advantage appears modest, further research is warranted to explore their broader utility in RA management.
Keywords
Key Messages of the Study
NETs as RA biomarkers: NETs markers (cfDNA, MPO-DNA and citH3) are significantly elevated in RA patients, correlating moderately with disease activity.
Improved diagnostic accuracy: Future research should investigate the relationship between NET biomarkers and advanced imaging modalities to assess tissue-level inflammation and structural progression in early RA, thereby validating the clinical utility of NET markers.
Potential clinical utility: A novel NET-based scoring system could aid in early RA diagnosis and better disease monitoring.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease marked by chronic and progressive inflammation, responsible for increased morbidity and mortality. Therefore, monitoring inflammation in RA is critical. Commonly used markers in clinical practice to monitor RA disease activity, such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), along with the Disease Activity Score 28-CRP (DAS-28-CRP), have notable limitations in accurately assessing disease activity. 1 Although DAS-28-CRP is widely used, its clinical components are subjective, and ESR/CRP are non-specific acute phase reactants. Notably, CRP and ESR may fail to detect ongoing synovial inflammation, leading to under-treatment or delayed therapeutic escalation. Imaging modalities, such as MRI and ultrasound, provide a more sensitive assessment of synovial inflammation but are expensive, time-consuming and not feasible for frequent monitoring in routine practice. 2
DAS-28-CRP is calculated based on four components: tender joint count (TJC), swollen joint count (SJC) (both evaluated by the physician), the patient’s global health score on a visual analogue scale (VAS) and the inflammatory marker CRP. 3 This scoring system is well established, with cut-off values of <3.2 and ≥3.2 indicating low disease activity (LDA) and high disease activity (HDA), respectively. 4 However, joint counts and global assessments are subjective and can vary between physicians, reducing reproducibility and sensitivity to change. 5 Additionally, ESR and CRP levels are non-specific and can be influenced by factors unrelated to RA disease activity, such as infections, obesity and other inflammatory conditions. 6 In the inflamed synovial tissue of RA, a significant infiltration of activated leukocytes, including polymorphonuclear neutrophils (PMNs), T cells, B cells, macrophages (Mφ) and dendritic cells, has been observed. 7 PMNs play a crucial role in the immune response and the development of RA. 8 They are among the first immune cells to migrate to the inflammatory site during joint inflammation, actively participating in the local immune response within the synovial tissue.9–10
In the early stages of RA, neutrophils accumulate in the inflamed synovium and play an essential role in its pathogenesis. 11 Recent studies have demonstrated that neutrophils can form extracellular reticular structures known as neutrophil extracellular traps (NETs) in response to specific stimulatory factors.12,13 NETs are composed of a DNA scaffold embedded with proteins such as citrullinated histone-3 (citH3), myeloperoxidase (MPO), neutrophil elastase (NE) and proteinase 3, which contribute to antimicrobial activity and increased tissue permeability.14,15 NETs may exacerbate RA inflammation by binding proteolytic enzymes and granular proteins to extracellular free DNA (cfDNA), thereby enhancing the macrophage-driven production of proinflammatory cytokines.¹² Studies suggest that NETs contribute to tissue damage in RA by harming vascular endothelial cells, promoting synovial inflammation and inducing a procoagulant phenotype in damaged vasculature, leading to thrombus formation.16,17 Preliminary studies have shown that NET-related biomarkers, such as cfDNA and citH3, are elevated in patients with RA compared to healthy controls and correlate with disease activity scores, synovial inflammation on imaging and progression of joint damage.17–20 These findings highlight NETs as a mechanistic link between innate immunity and persistent inflammation in RA. However, despite their promise, NET biomarker assays are more expensive, require specialised laboratory facilities and are not yet standardised for routine clinical use.17,21,22 Therefore, it is important to establish whether their added diagnostic or prognostic value justifies their use over simple, established measures.
The present study aimed to investigate circulating NET biomarkers as potential novel indicators of disease severity in RA and compare them with conventional markers (CRP, ESR and DAS-28-CRP). Understanding their role could enable earlier assessment of disease severity, optimisation of treatment strategies and improved long-term disease management, ultimately enhancing patient outcomes and quality of life.
Materials and Methods
Study Design
This prospective analytic cross-sectional study was conducted at a tertiary care medical college-attached hospital in western India from May 2022 to December 2023.
Participant Selection
Patients diagnosed with RA based on the American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 classification criteria 19 were enrolled as cases. Age- and sex-matched controls were selected from healthy volunteers attending the hospital for routine health check-ups or blood donations. RA cases were categorised into HDA and LDA subgroups based on the DAS-28-CRP, with scores <3.2 indicating LDA and ≥3.2 indicating HDA.
Patients with autoimmune diseases other than RA, systemic diseases, including cancer, acute or chronic infections, hematologic abnormalities, liver, heart and renal failure, chronic pulmonary disease, coronary artery disease, type 1 diabetes mellitus, and pregnant patients were excluded from the study.
Data Collection
General data collected included demographic details, personal history medical history, smoking and alcohol consumption history, and current pharmacotherapy. Clinical parameters apart from general and systemic examination, musculoskeletal examination, including SJC and TJC were noted. Additionally, patient global assessment using a VAS was recorded. All participants underwent laboratory testing, including hemogram, ESR, CRP, rheumatoid factor (RF), anti-cyclic citrullinated peptide (anti-CCP) antibodies, anti-nuclear antibody (ANA), NET markers and a basic metabolic profile. The disease activity score for each patient with RA was calculated using the DAS-28-CRP calculator, which incorporates TJC, SJC, CRP and global health scores.
Laboratory Parameters
According to the manufacturer’s protocol, plasma cfDNA levels were measured using the PicoGreen™ dsDNA Quantification Kit (Invitrogen, USA). The myeloperoxidase-DNA (MPO-DNA) complexes were quantified using solid-phase-modified specific antibodies targeting MPO and dsDNA (ABTS ELISA Buffer Kit, ThermoFisher Scientific, MA, USA) (Pepro Tech, Rocky Hill, NJ, USA). Plasma citH3 levels were determined using anti-histone antibodies from the Cell Death Detection ELISA Kit (Roche Diagnostics, Switzerland).
Haematological parameters were measured using a Cell-Dyn 3700 SL haematology analyser (Abbott, Chicago, IL, USA). ESR was determined using the Westergren method. RF levels were measured via enzyme-linked immunosorbent assay (ELISA), with values above 20 arbitrary units considered positive. Anti-CCP antibodies were measured by ELISA, with levels above 5 arbitrary units considered positive. ANA positivity was determined using the immunofluorescent method. CRP was measured by ELISA, with results reported as mean ± standard deviation (SD). Laboratory testing was conducted by trained technicians following standardised protocols under the supervision of a pathologist.
Sample Size Calculation and Statistical Analyses
The sample size was determined based on previous studies to ensure adequate power (80%) to detect significant associations between inflammatory markers and disease severity in patients with RA. P value > .05 was considered statistically significant. All data analyses were performed using IBM SPSS Statistics version 22.0. Normally distributed data were expressed as mean ± standard deviation (SD). Comparisons between two independent groups (LDA-RA vs HAD-RA) were conducted using student t-tests. For comparisons involving more than two groups (e.g., LDA-RA vs HAD-RA vs healthy controls), one-way analysis of variance (ANOVA) was applied, followed by post hoc Bonferroni correction to identify between-group differences.
Categorical variables (such as sex distribution and positivity rates) were compared using the χ2 test or Fisher’s exact test, where appropriate. The correlation between plasma NETs’ levels and inflammatory markers (e.g., CRP and ESR) was assessed using Pearson correlation coefficients to determine the strength and direction of linear relationships. To identify independent disease severity markers of RA, binary logistic regression analysis was conducted. The dependent variable was RA diagnosis (yes/no), and the independent variables included plasma NET levels along with other clinical and inflammatory markers. This analysis was performed to assess the contribution of NETs beyond other known risk factors.
For the diagnostic evaluation, the receiver operating characteristic (ROC) curve analysis was performed to assess the discriminative ability of plasma NETs in distinguishing patients with RA from healthy controls. The dependent covariate (test variable) in the ROC analysis was the plasma NET level. The area under the curve (AUC) was calculated to summarise diagnostic accuracy. The optimal cut-off value was determined using the Youden index (sensitivity + specificity − 1), from which sensitivity, specificity, positive predictive value and negative predictive value were derived.
Results
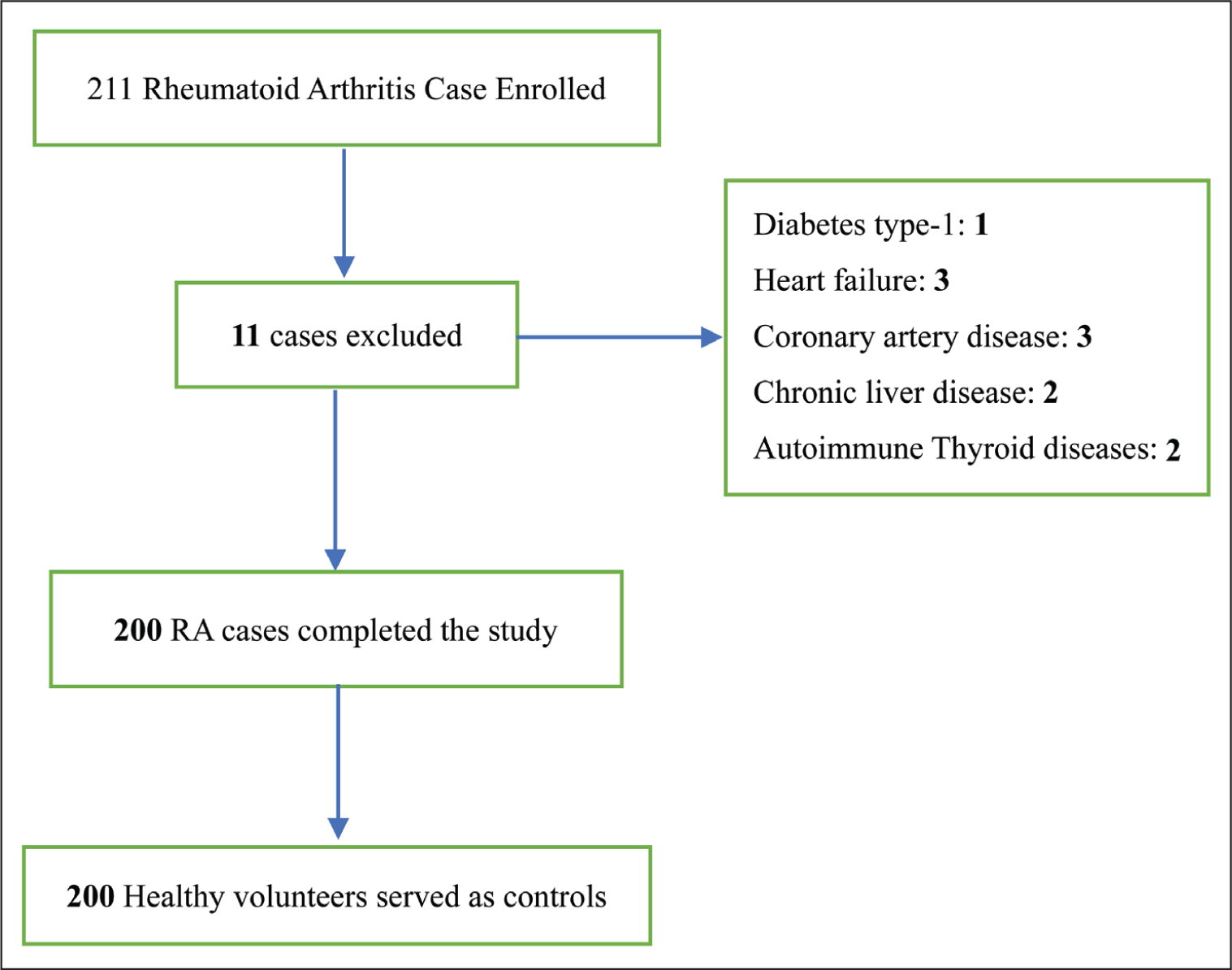
Initially, 211 patients with RA were enrolled. After excluding 11 cases, the study was completed by 200 patients with RA along with an equal number of matched controls (Figure 1).
Flow Chart Showing Inclusion and Exclusion of Participants.
Comparison of Clinical Characteristics
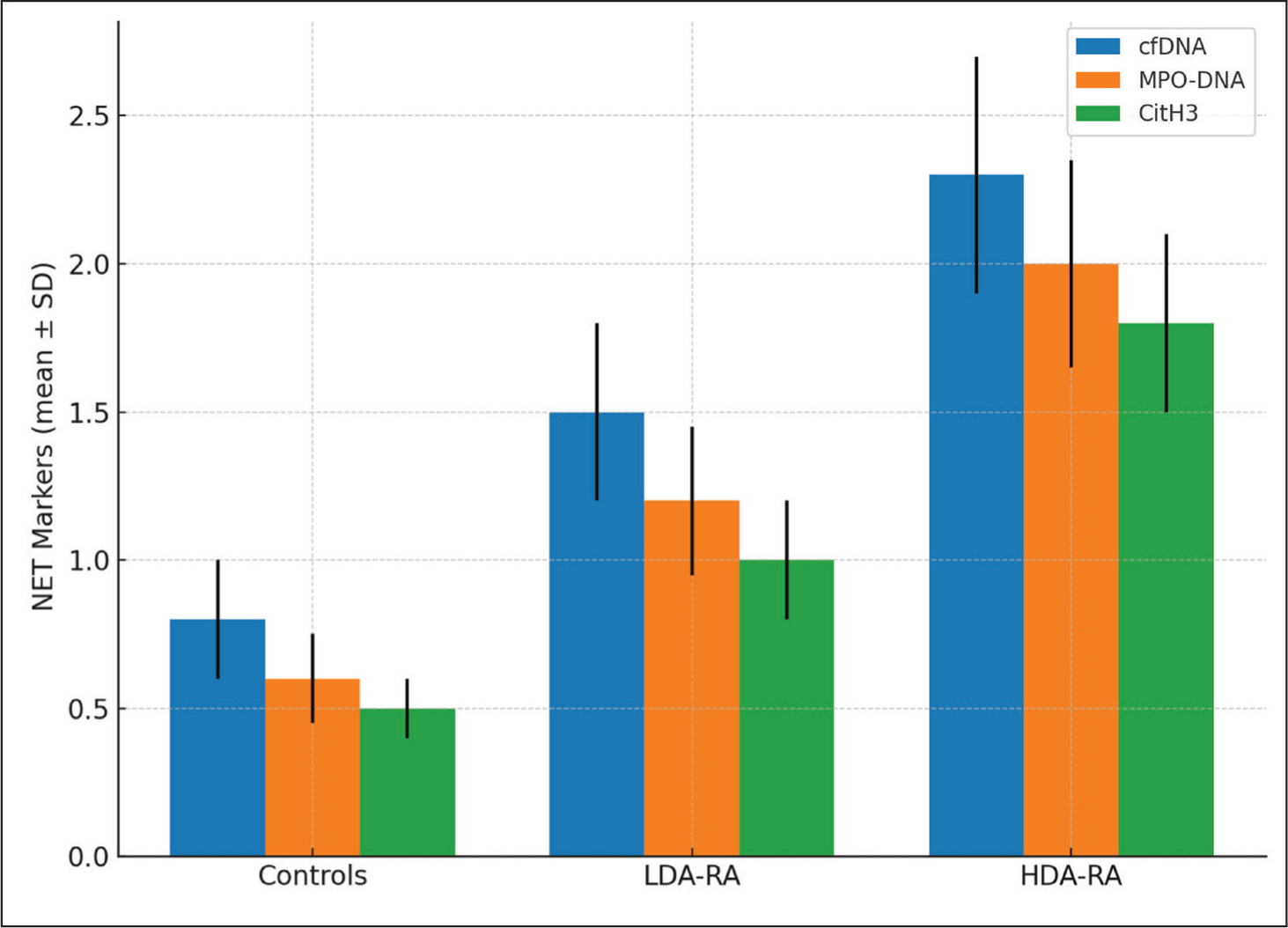
Table 1 summarises the demographic and clinical profiles of 160 patients with HDA-RA, 40 patients with LDA-RA and 200 healthy controls. No significant differences were observed between groups regarding age, sex distribution or smoking/tobacco use. However, markers of inflammation differed markedly. The ESR was significantly elevated in HDA-RA (37.2 ± 3.4 mm/h) compared to LDA-RA (29.9 ± 2.9 mm/h) and controls (8.2 ± 0.3 mm/h; P < .001). Likewise, CRP levels were highest in HDA-RA (16.1 ± 2.5 mg/dL), followed by LDA-RA (11.4 ± 2.1 mg/dL) and controls (1.8 ± 0.2 mg/dL; P < .001). NET markers were significantly elevated in HDA-RA versus LDA-RA and controls: cfDNA (197.7 ± 18.7 ng/mL vs 165.9 ± 18.7 vs 132.9 ± 32.8), MPO-DNA (67.3 ± 12.0 vs 55.2 ± 12.6 vs 36.0 ± 16.3) and citH3 (4.1 ± 1.4 vs 3.2 ± 1.2 vs 1.9 ± 1.0; all P < .001).
Demographic Details, Clinical Characteristics and Laboratory Parameters of Rheumatoid Arthritis Patients.
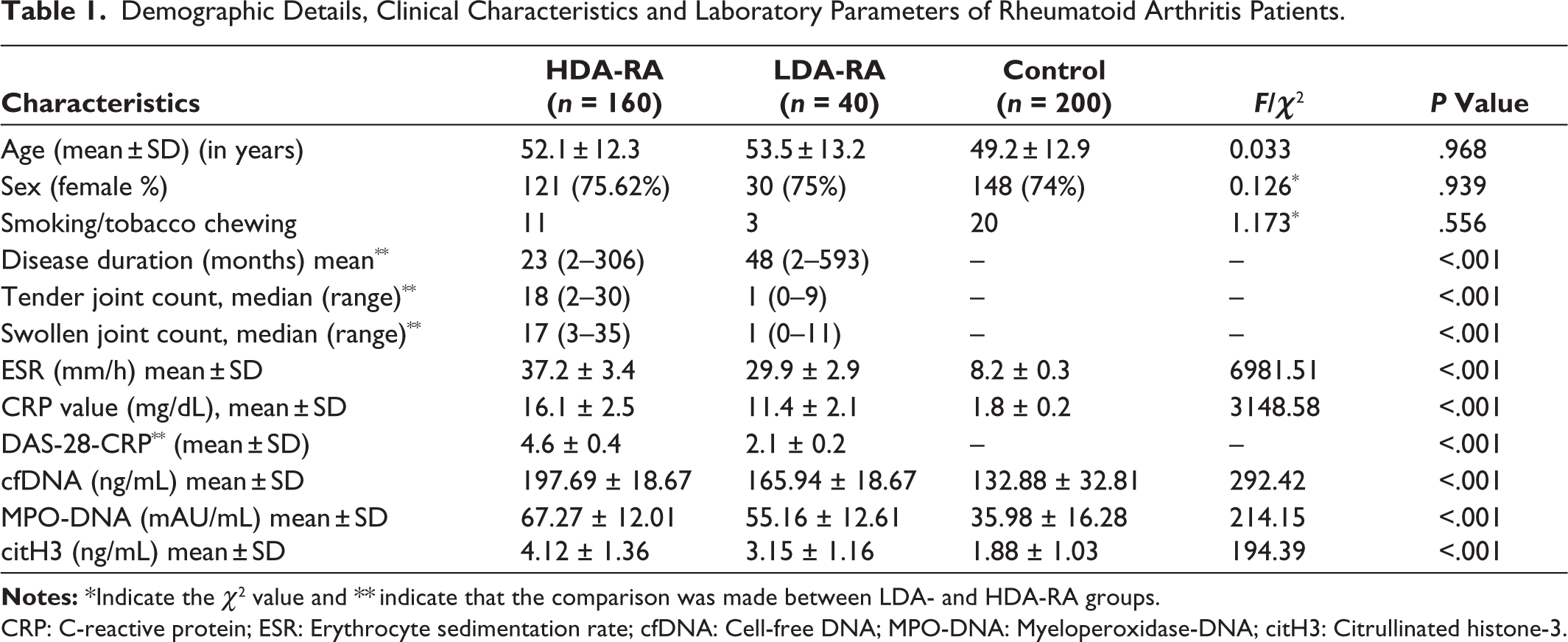
CRP: C-reactive protein; ESR: Erythrocyte sedimentation rate; cfDNA: Cell-free DNA; MPO-DNA: Myeloperoxidase-DNA; citH3: Citrullinated histone-3.
Correlation Analysis Between Circulating NETs and Inflammatory Markers
Table 2 shows strong positive correlations between NET markers and inflammatory parameters. For example, cfDNA correlated significantly with CRP (r = 0.531, P < .001), ESR (r = 0.621, P < .001) and DAS-28-CRP (r = 0.571, P < .001). Similar significant correlations were seen for MPO-DNA and citH3.
Correlation Between Circulating NET Markers and Inflammatory Factors.
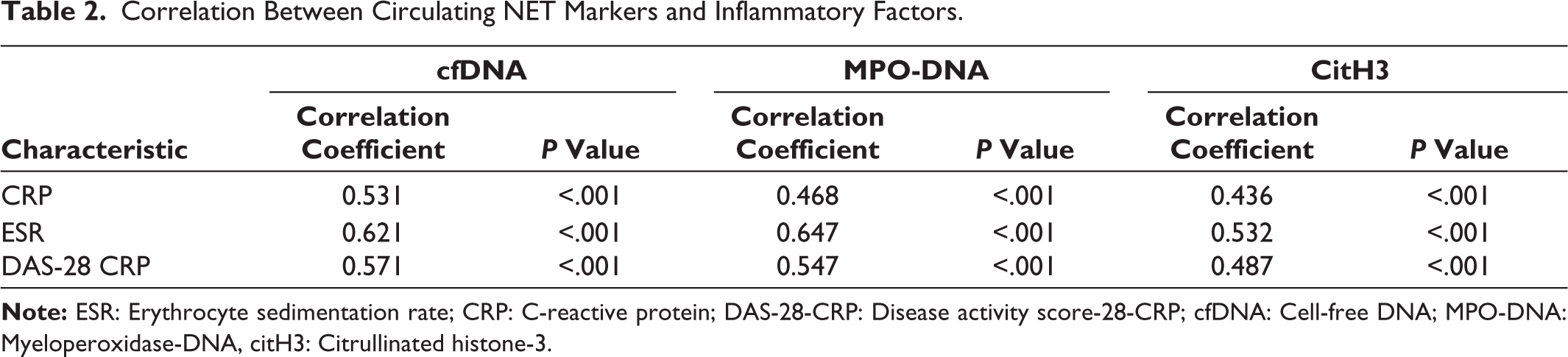
Figure 2 illustrates a bar diagram comparing NET markers (cfDNA, MPO-DNA and citH3) across LDA-RA, HDA-RA, and control groups, with error bars representing mean ± SD.
Logistic Regression Analysis of NET Markers as Disease Severity Markers of RA Using the ROC Curve
Table 3 presents logistic regression findings: cfDNA (OR = 1.045, 95% CI: 1.025–1.067), MPO-DNA (OR = 1.084, 95% CI: 1.048–1.122) and citH3 (OR = 2.871, 95% CI: 1.786–4.616) were identified as statistically significant independent markers in differentiating HDA-RA and LDA-RA.
Logistic Regression Analysis of Independent NET Markers for Rheumatoid Arthritis Disease Severity.
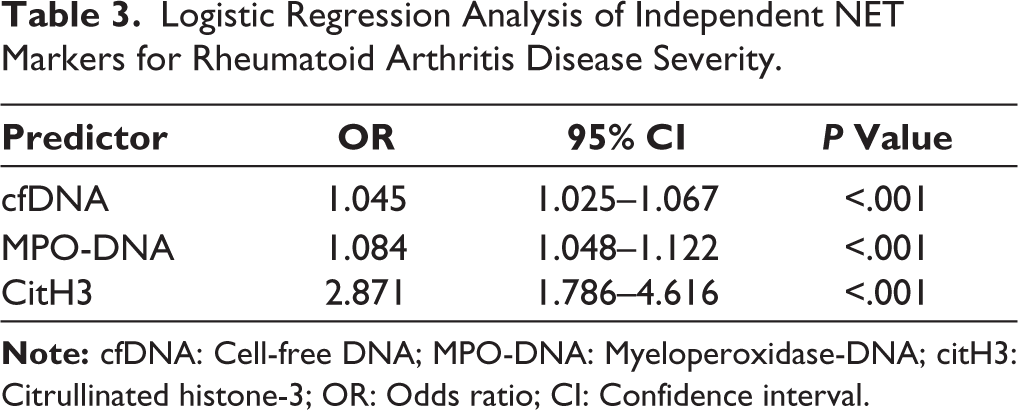
Table 4 describes the ROC curve analysis assessing the diagnostic performance of cfDNA, MPO-DNA, citH3, ESR and DAS-28-CRP in differentiating HDA-RA from LDA-RA. Among these, cfDNA demonstrated the highest diagnostic accuracy, with an AUC of 0.87 (95% CI: 0.82–0.92), achieving 81% sensitivity and 79% specificity at an optimal cut-off of 182 ng/mL. MPO-DNA and citH3 also showed strong performance, with AUCs of 0.86 and 0.84, respectively. The optimal cut-offs were 61 mAU/mL for MPO-DNA (80% sensitivity, 78% specificity) and 3.5 ng/mL for citH3 (77% sensitivity, 75% specificity). Traditional markers such as ESR and DAS-28-CRP demonstrated slightly lower diagnostic ability, with AUCs of 0.80 and 0.82, respectively. All markers showed statistically significant P values (P < .001), reinforcing their clinical utility. Additionally, for distinguishing controls from LDA-RA, the best cut-offs were 145 ng/mL for cfDNA, 42 mAU/mL for MPO-DNA and 2.3 ng/mL for citH3, each offering good sensitivity (72%-78%) and specificity (74%-80%).
ROC Curve Analysis of cfDNA, MPO-DNA, CitH3, ESR and CRP as Key Factors in Differentiating Active Rheumatoid Arthritis from Remission.
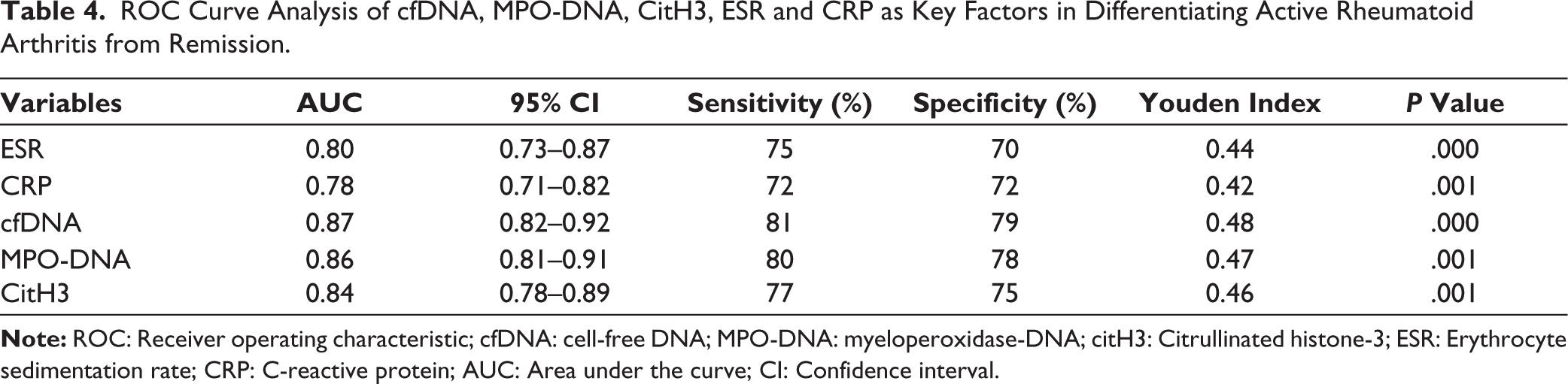
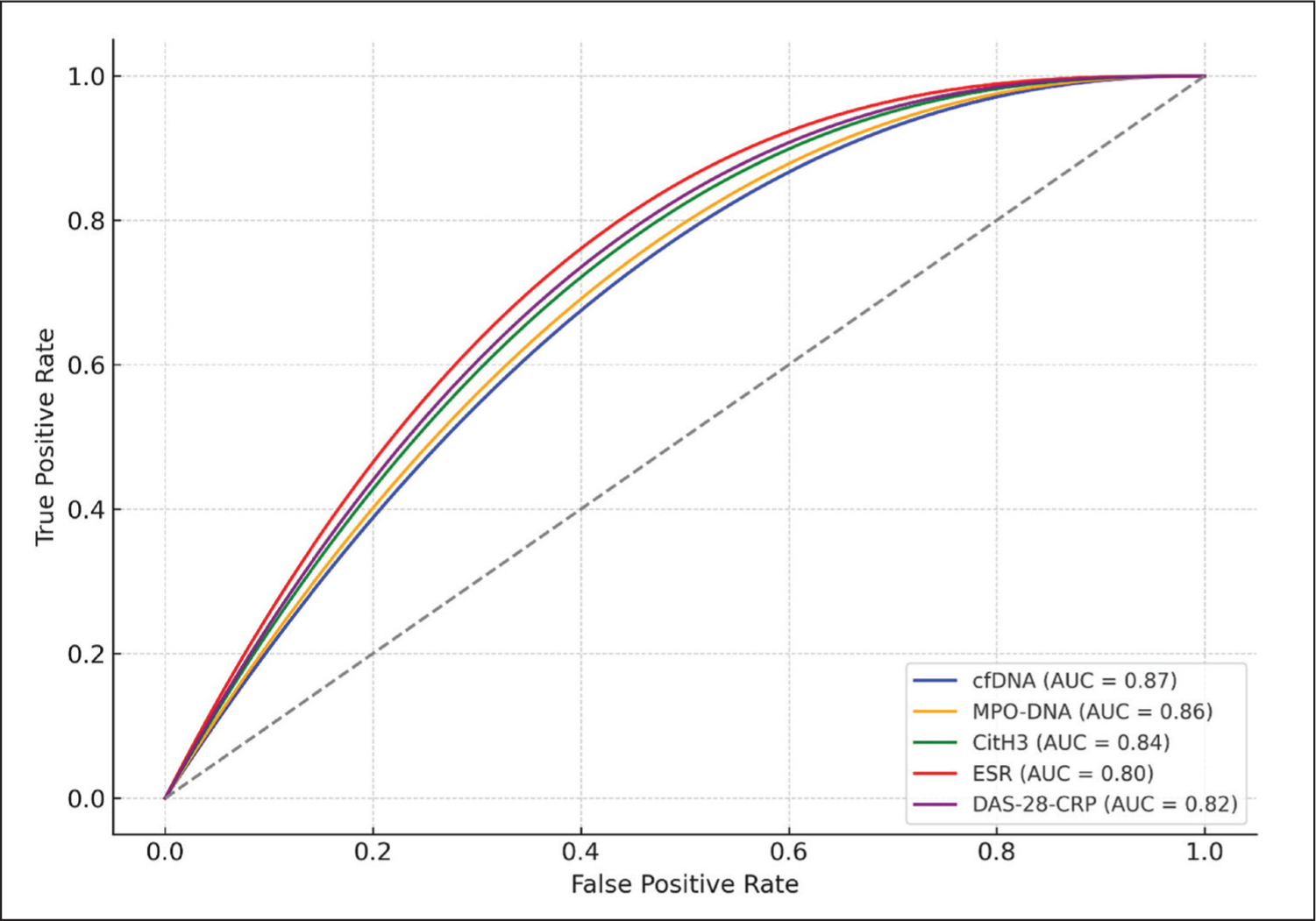
Figure 3 demonstrates the ROC curve comparing cfDNA, MPO-DNA, citH3, ESR and DAS-28-CRP. This visualises their diagnostic performance for distinguishing HDA-RA from LDA-RA.
Discussion
This study systematically evaluated NET markers, cfDNA, MPO-DNA and citH3 as potential biomarkers for assessing disease severity in RA. While prior research has established the involvement of NETs in RA pathogenesis,16–20 few studies have directly compared NET markers across well-defined disease activity categories (HDA-RA vs LDA-RA) alongside healthy controls, particularly in the Indian population. By integrating standard inflammatory markers (CRP and ESR) with NET markers and employing robust statistical tools, including logistic regression and ROC analysis, this study provides novel insights into the relative diagnostic utility of NETs for RA severity stratification. Our findings address a key knowledge gap regarding whether NET markers offer meaningful advantages over conventional markers for routine clinical use and help clarify their potential role in complementing existing RA disease activity assessments.
NETs play a key role in autoimmunity, with markers such as cfDNA, MPO-DNA, and citH3 emerging as potential indicators of many inflammatory diseases such as systemic lupus erythematosus (SLE), inflammatory bowel diseases and RA.17,18,23 NETs are web-like chromatin structures released by neutrophils in response to inflammation, contributing to both defence and tissue damage. 24 In this study, NET markers were significantly elevated in patients with HDA-RA compared to those with LDA-RA and healthy controls, reinforcing prior findings that NETs may serve as sensitive biomarkers of RA activity. A moderate correlation was found between NET markers and inflammatory indices (ESR, CRP and DAS28-CRP), underscoring the link between NETs and systemic inflammation. NETs amplify immune responses and cause tissue damage via proteolytic enzymes and granular proteins attached to cfDNA.16–25 Beyond their role in disease activity, NETs are implicated in other pathogenic processes, including vascular inflammation, thrombosis and tissue remodelling, as seen in conditions such as SLE, cystic fibrosis and atherosclerosis.26–31 These broader roles suggest that future research should explore NETs not only as markers of RA disease activity but also as potential indicators of cumulative joint damage, cardiovascular risk and extra-articular complications in patients with RA patients. cfDNA, MPO-DNA and citH3 were identified as independent RA risk factors, with a combined diagnostic efficacy of AUC = 0.949. However, the precise molecular mechanisms of NETs in RA pathogenesis remain unclear.31,33 Proteomic analysis shows alterations in proteins involved in NET formation, further supporting their role in RA and other inflammatory diseases. 34 NETs may increase endothelial permeability, exacerbate tissue oedema and induce oxidative stress-related injury in synovial tissues. 35 Thus, NET markers hold promise as diagnostic and prognostic biomarkers, as well as therapeutic targets.
To leverage NETs clinically, a novel diagnostic system has been proposed that integrates measurements of cfDNA, MPO-DNA and citH3. By converting these NET marker values into binary categories and applying weights derived from ROC curve analysis, a composite NET score was developed. 10 This scoring system could facilitate the early identification of patients with RA with heightened NET activity, allowing timely therapeutic intervention. Further refinement of the system may enhance its predictive accuracy and clinical utility. Conventional inflammatory markers have limited diagnostic value in assessing RA disease activity at the subclinical or early stage, where sensitive imaging modalities often detect disease activity before overt clinical manifestations. 2 Future research could explore the relationship between NET biomarkers and imaging findings to better assess early RA disease activity.
Limitations
Despite these promising findings, several limitations must be acknowledged. First, the relatively small sample size restricts the statistical power of the study and may limit the robustness of the conclusions. Larger cohorts are essential to validate the reliability of the NET score across different clinical settings. Second, the cross-sectional design precludes any assessment of temporal or causal relationships between NET marker levels and RA disease progression. Longitudinal studies are needed to determine whether changes in NETs markers over time correlate with disease flares, remission or long-term outcomes. Third, as this was a single-centre study, there is potential for selection bias, and the findings may not be generalisable to broader populations with different genetic, environmental or clinical characteristics. Multi-centre studies involving diverse patient populations are necessary to confirm the external validity of the NET score.
Additionally, while the NET composite score adds diagnostic value, its marginal benefit over conventional inflammatory markers such as ESR and CRP, particularly given the higher costs and technical demands of NETs assays, may limit its routine adoption in standard clinical practice. The practicality and cost-effectiveness of implementing such a diagnostic tool in various healthcare settings should be carefully evaluated. Finally, this study did not assess the relationship between NET biomarkers and advanced imaging modalities, which are increasingly used to detect subclinical synovitis and early joint damage. Future research should integrate imaging findings, such as ultrasound or MRI data, to clarify how NET’s activity correlates with tissue-level inflammation and structural progression in early RA.
In summary, while the NET-based diagnostic system shows considerable promise as an adjunctive biomarker tool for RA disease activity, further refinement, validation in larger and more diverse cohorts and integration with imaging and clinical outcomes are essential steps before it can be translated into widespread clinical use. Moreover, expanding research to explore the roles of NETs in RA-related complications, such as cardiovascular disease and extra-articular manifestations, may reveal broader applications for risk stratification and therapeutic targeting.
Conclusion
This study highlights NETs markers as novel biomarkers for diagnosing & monitoring RA activity. Their moderate association with inflammatory indices supports their role as sensitive disease severity indicators. A NETs-based diagnostic system offers a promising approach for early detection & improved management. Further research is needed to clarify NETs’ role in RA pathogenesis & optimise their clinical application. Integrating NETs biomarkers into routine clinical practice could enhance disease monitoring, support more personalised treatment approaches, and ultimately lead to better patient outcomes and improved quality of life.
Footnotes
Authors’ Contributions
Data collection, patient recruitment and manuscript drafting: MMP.
Data analysis, interpretation and literature review: DKP.
Overall study supervision, conceptualisation and critical manuscript revision: LBP.
Study design, statistical analysis and manuscript editing: DMP.
Assisted in clinical data collection, patient follow-up and preliminary data analysis: KJP and VAP.
As the corresponding author coordinated the research process, ensured ethical compliance and provided final approval of the manuscript: MVP.
All authors have read and approved the final manuscript.
Data Availability
All relevant data are contained within the article. Additional data supporting the findings of this article are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This study was conducted in compliance with ethical standards, with approval from the Zydus Medical College and Hospital institutional ethics committee, Dahod Ref. No: ZMCH/IEC/2018-19/003 dated 26 April 2019.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Human and Animal Rights
Human participants’ rights were protected in accordance with the Helsinki Declaration guidelines, and no animals were used in this study.
Informed Consent
All participants were informed of the study design, and written informed consent was obtained from the participants.
Institute Details
The study was conducted at the Rheumatology and General Medicine departments of Zydus Medical College and Hospital, Dahod, Gujarat.
