Abstract
Background and Objectives:
Pain perception in rheumatoid arthritis (RA) may be affected by Catechol-O-methyltransferase (COMT) polymorphism, which alters catecholamines-mediated central pain processing. The COMT Val158Met single nucleotide polymorphism (SNP) causes important functional alterations of the COMT enzyme. In this, the Val allele gives rise to an effective enzyme, whereas the Met allele produces a defective enzyme, which cannot clear catecholamines effectively from the system. The objective of this study was to determine the correlation of pain and disability with disease activity in RA patients with different COMT genotypes.
Methods:
This was a cross-sectional pilot study involving 60 female patients with RA who met the 2010 ACR/EULAR classification criteria. The Numerical Pain Rating Scale (NPRS) and Cochin Hand Function Scale (CHFS) were administered and 3 mL venous blood samples were taken for high-sensitivity C-reactive protein (hs-CRP) estimation and genotyping. Genotyping for the COMT Val158Met allele was performed via the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) method. The erythrocyte sedimentation rate (ESR) was determined as a routine hospital procedure.
Results:
The odds of developing moderate to severe pain in the Met/Met genotype group were 10.4 times greater than those in the Val/Val genotype group. There was a positive correlation between NPRS and ESR, Disease Activity Score 28 (DAS28)-ESR and CHFS, DAS28-CRP and CHFS, DAS28-ESR and NPRS, DAS28-CRP and NPRS in the Val/Val genotype. In the Val/Met genotype, DAS28-ESR and DAS28-CRP were positively correlated with NPRS. Thus, the COMT Val158Met polymorphism may be a factor which may moderate the relationship of pain and disability with disease activity in RA patients.
Keywords
Introduction
Catechol-O-methyltransferase (COMT) is a ubiquitous enzyme found in almost all human tissues and its main physiological function is the metabolic inactivation of endogenous catechol neurotransmitters. 1 The COMT gene is located in the 22q11.1-22q11.2 region of chromosome 22. There are different single nucleotide polymorphisms (SNPs) in the COMT gene that cause important functional alterations in the enzyme. One of the best-studied SNPs, rs4680, is located at codon 158 and involves a valine-to-methionine transition (Val158Met). The Val allele gives rise to an effective enzyme, whereas the Met allele produces a defective enzyme, which cannot clear catecholamines effectively from the system.2–4 Since dopamine, noradrenaline and adrenaline are metabolised by the COMT enzyme, they may act as key modulators of dopaminergic and noradrenergic/adrenergic neurotransmission. Considering the role of these neurotransmitters in pain modulation, the COMT polymorphism may influence the human experience of pain and may underlie inter-individual differences in the adaptation and response to pain. 5 The Val158Met COMT polymorphism was related to pain perception in chronic pain conditions such as fibromyalgia, where altered central nervous system pain processing is the main contributing factor.6-10 However, studies on its role in inflammatory diseases such as rheumatoid arthritis (RA) are limited.
RA is an autoimmune inflammatory rheumatic disease that affects approximately 0.34% of the Indian population and causes chronic synovial inflammation, eventually leading to joint destruction and disability. 11 Although RA pain is incited by inflammatory synovitis and structural knee damage, the response to pain is modified centrally in the brain and spinal cord. 12 Therefore, pain perception in individuals with RA may be influenced by the COMT polymorphism, which can alter central pain processing. The current study was a preliminary attempt to understand the relationship between pain and disability with disease activity in RA patients.
Materials and Methods
Patient Recruitment
This was a single-centre cross-sectional pilot study involving 60 RA patients of Indian origin recruited from the rheumatology outpatient department (OPD) of a tertiary care hospital. Since there was no previous study on the COMT polymorphism in RA the sample size was determined by convenience. The inclusion criteria were (a) diagnosed with RA fulfilling the 2010 ACR/EULAR classification criteria and (b) females between 18 and 49 years of age with a regular menstrual cycle. Patients with a history of other chronic pain conditions and neuropsychiatric illness were excluded. Pregnant and lactating females were also excluded. Informed written consent was taken from patients. The duration of the study was from December 2021 to May 2023.
Pain and Hand Disability Assessment
Pain intensity was assessed by the Numerical Pain Rating Scale (NPRS), which is an 11-point numerical scale ranging from ‘0’ meaning ‘no pain’ to ‘10’ meaning ‘pain as bad as you can imagine’ or ‘worst pain imaginable’. 13 Hand disability was assessed by the Cochin Hand Function Scale (CHFS). 14
Erythrocyte sedimentation rate (ESR) was measured as a routine hospital procedure. By venipuncture, 3 mL whole blood samples was collected in an EDTA vial and within 2 hours of collection samples were centrifuged at 2700 rpm for 15 minutes. The supernatant plasma was stored at −80°C. Plasma hs-CRP was estimated using a sandwich ELISA kit obtained from Elabscience according to the manufacturer’s instructions. Disease activity in patients was measured using the Disease Activity Score 28 (DAS28), which includes the 28 tender and swollen joint counts, the ESR or C-reactive protein and the patients’ global health. 15
Genotyping
For genotyping DNA was extracted from the whole blood sample using the QIAamp DNA Mini Kit according to the manufacturer’s instructions. DNA samples were quantified using the NanoDrop 1000 (Thermo Fisher Scientific). Afterwards, samples of satisfactory quantity and quality were stored at −80°C.
Genotyping was performed via the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) method. The PCRs were carried out in a total volume of 25 µL containing 100 ng of genomic DNA as the template (2 µL of 50 ng/µL DNA), 0.3 µL of each primer (10 µM; synthesised by Barcode Biosciences Pvt Ltd, Bangalore), 1.5 µL of 25 mM MgCl2, 0.3 µL of 10 mM each dNTP, 2.5 µL of 10X Taq buffer (100 mM Tris-HCl pH 8.8, 500 mM KCl) and 0.3 µL of 5 U/µL Taq DNA polymerase (Thermo Scientific™, USA). PCR amplification was carried out in a Bio-Rad S1000 thermal cycler with an initial denaturation step at 94°C for 5 minutes, followed by 30 cycles at 94°C for 30 seconds, 56°C for 30 seconds and 72°C for 10 seconds and a final extension step at 72°C for 5 minutes. The sequences for the forward and reverse primers were F5′-CTGTGGCTACTCAGCTGTG-3′ and R5′- CCTTTTTCCAGGTCTGACAA-3′, respectively. 16 The amplified product was digested by adding a PCR mixture of 10 µL to 18 µL of nuclease-free water, 2 µL of 10X Buffer G and 2 µL of NlaIII restriction enzyme (Thermo Scientific, Hin1II). The mixture was mixed gently, spun down for a few seconds and incubated at 37°C for 12 hours.
Amplified DNA samples and restriction enzyme digestion products were separated via gel electrophoresis in 2.5% and 4% agarose gels with 1× Tris-borate EDTA buffer. The DNA bands were visualised via ethidium bromide staining (5 µL in 100 ml gel). A 2 µL DNA sample was mixed with 5 µL of gel loading dye orange and loaded into the wells. The amplified fragment of the COMT gene was 168 base pairs (bps). After NlaIII restriction enzyme digestion of the amplicon, the Val/Val genotype produced three bands of 114, 29 and 25 bp long fragments. The Val/Met heterozygote produced five bands of 114-, 96-, 29-, 25- and 18-bp long fragments. The Met/Met homozygote produced four fragments of 96, 29, 25 and 18 bp. Fragments of 29, 25 and 18 bp size were not visualised in the 4% agarose gel. Hence, the presence of a single 114 bp long band indicated a homozygous wild-type genotype (Val/Val), the presence of a single 96 bp long band indicated a homozygous genotype (Met/Met) and the presence of both 114 and 96 bp long fragments indicated a heterozygous genotype (Val/Met).
Statistical Analysis
GraphPad Prism, version 9.5.1 for Windows (GraphPad Software, San Diego, California USA) was used for all the statistical analyses. The normality of the data was assessed using the Shapiro-Wilk test. Compatibility of the Val allele and Met allele distributions with expected frequencies was assessed using the chi-square test. Baseline scores of the parameters among the 3 groups of COMT genotypes were compared using one-way ANOVA or the Kruskal-Wallis test. The correlations between parameters were tested using the Spearman correlation test. P < .05 was considered to indicate statistical significance.
Results
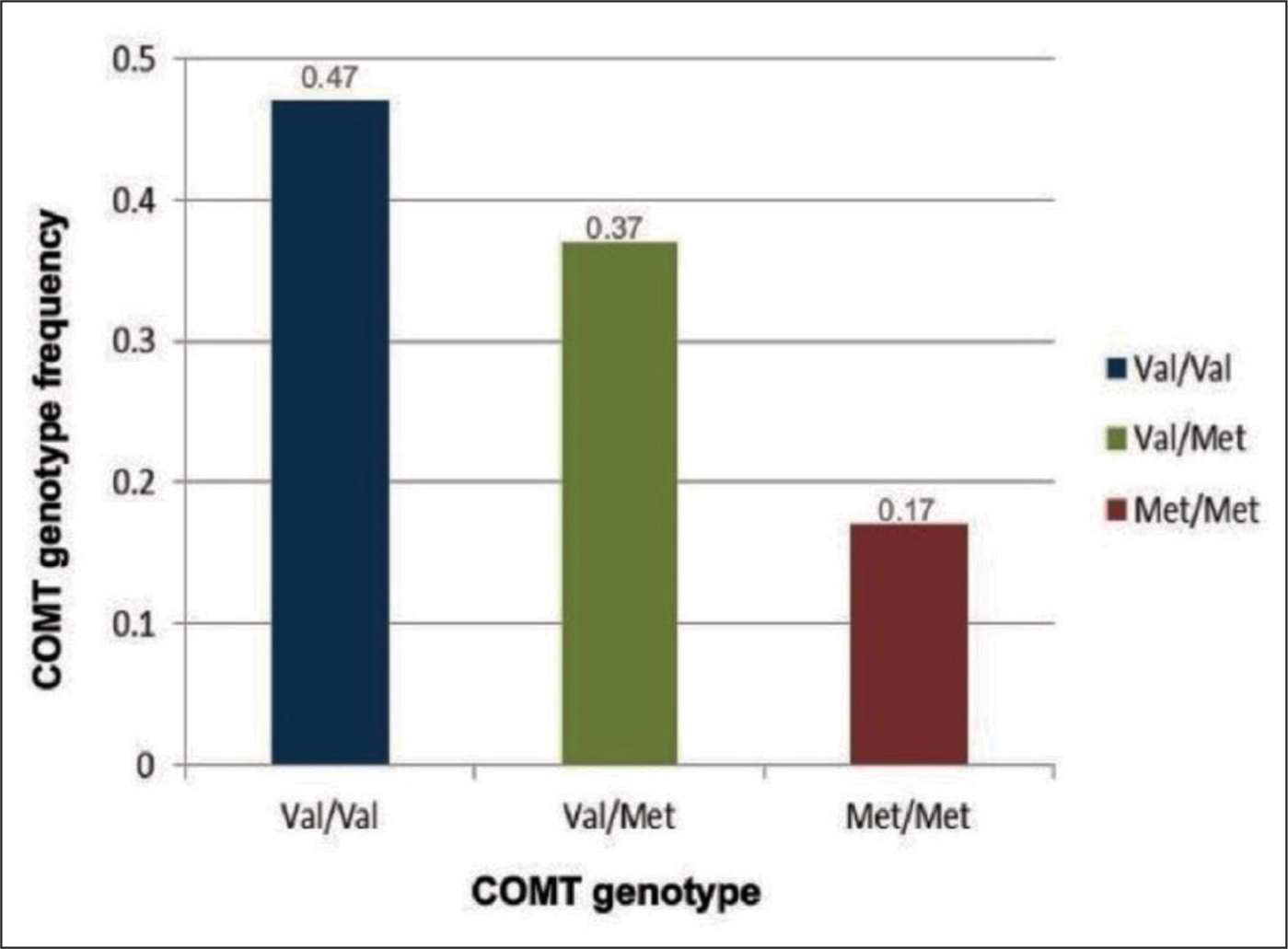
The patients were divided into 3 groups on the basis of their COMT Val158Met gene SNPs. The three different genotypes were homozygous Val/Val, Met/Met and heterozygous Val/Met. The prevalence rates of Val/Val, Val/Met and Met/Met were 46.7% (n = 28), 36.7% (n = 22) and 16.7% (n = 10), respectively, as shown in Figure 1. The respective genotype frequencies were 0.47, 0.37 and 0.17. This polymorphism was compatible with Hardy-Weinberg equilibrium (χ² = 1.20; df = 2; P = .55).
Bar Diagram Showing the Frequency of the Three COMT Genotypes in Rheumatoid Arthritis Patients.
The measured parameters of the RA patients of each genotype are listed in Table 1. The mean ages of patients with the Val/Val, Val/Met and Met/Met genotypes were 38.8 ± 6.3 years, 37.8 ± 7.7 years and 38.2 ± 6.3 years, with median body mass indices (BMIs) of 24.8 (22.–28.3), 24.2 (21.9–27.9) and 26.7 (23.6–30.2) kg/m 2 and respectively. The median symptom duration was 8 (4.0–12.0) years, 9 (5.8–13.0) years and 5.5 (3.5–10.5) years in the respective groups. The median high-sensitivity C-reactive protein (hs-CRP) levels were 7.2 (3.3–18.0) mg/L, 8.8 (3.178–26.50) mg/L and 8.6 (1.933–26.17) mg/L and the mean ESRs were 31.9 ± 19.7 mm/h, 31.4 ± 14.2 mm/h and 46.3 ± 29.3 mm/h for the Val/Val, Val/Met and Met/Met genotypes, respectively. The median NPRS and CHFS scores were 3.5 (2.0–5.0), 5.0 (2.0–7.0) and 4.5 (4.0–7.3) and 6.0 (1.0–14.5), 7.5 (1.8–26.8) and 6.5 (2.3–25.3), respectively.
Demographic Details, Inflammatory Marker Levels, Disease Activity, Pain Scores and Disability Scores of Rheumatoid Arthritis Patients.
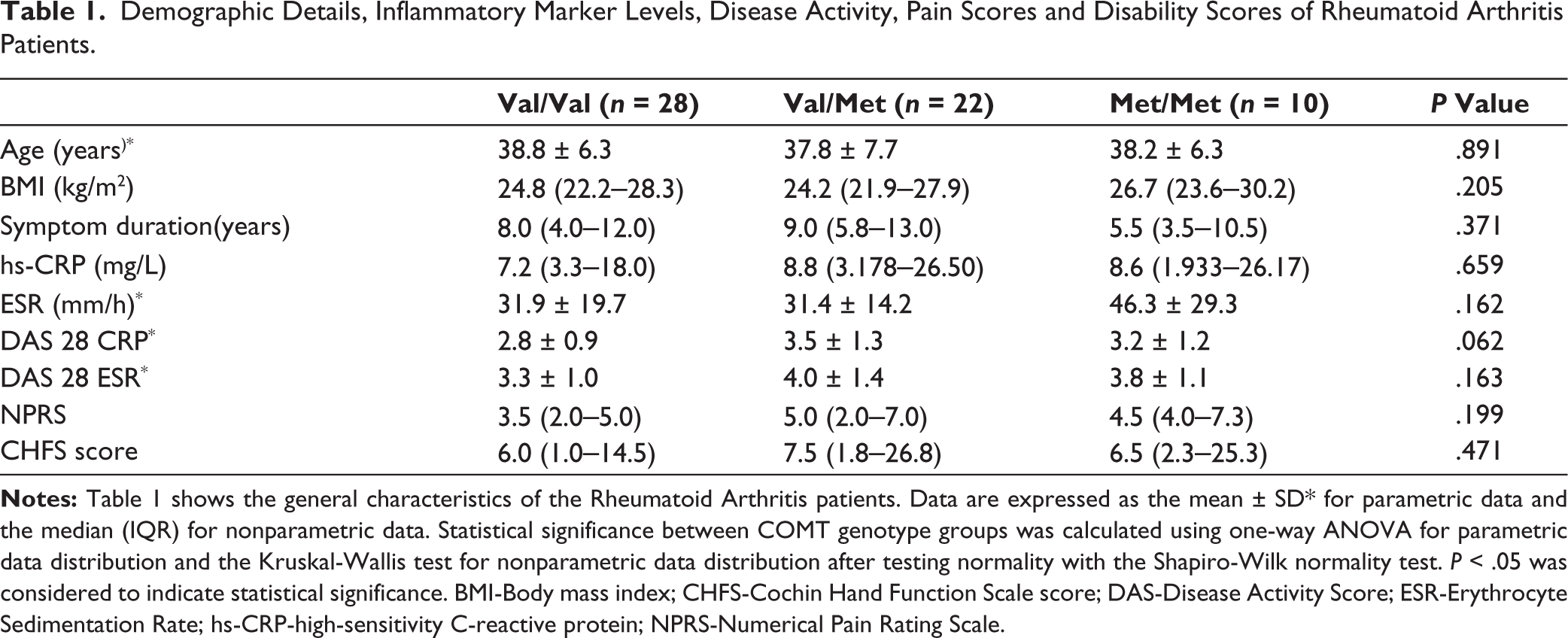
All the above parameters were comparable between the groups. All the patients were receiving nonbiological disease-modifying antirheumatic drug (DMARD) treatment and were taking nonsteroidal anti-inflammatory drugs (NSAIDs) for pain relief. There were three patients with the Val/Val genotype with a family history of RA who were first-degree relatives. Seventeen patients with the Val/Val genotype, 13 patients with the Val/Met genotype and two patients with the Met/Met genotype were rheumatoid factor positive.
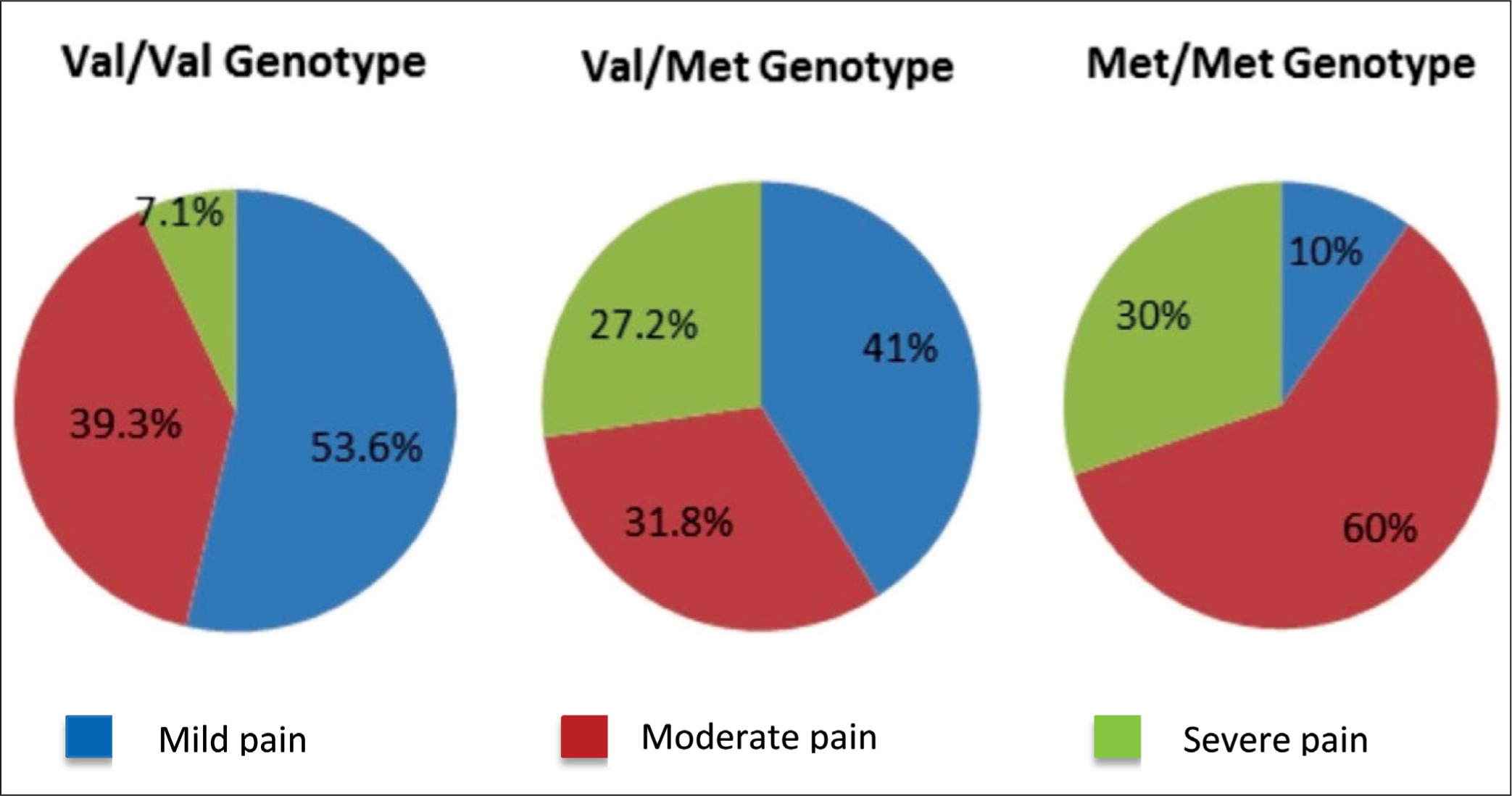
The severity of pain was classified as NPRS score from 1 to 3 as mild pain, score from 4 to 6 as moderate pain and score from 7 to 10 as severe pain. The proportions of RA patients with mild, moderate and severe pain with three catechol-O-methyl transferase genotypes are shown in Figure 2. A total of 53.6% of the Val/Val genotype patients, 41% of the Val/Met genotype patients and 10% of the Met/Met genotype patients experienced mild pain. Moderate pain was experienced by 39.3%, 31.8% and 60% of patients with the Val/Val, Val/Met and Met/Met genotypes, respectively. The percentages of patients who experienced severe pain were 7.1%, 27.2% and 30% for Val/Val, Val/Met and Met/Met, respectively.
Pie Chart Showing the Proportions of Rheumatoid Arthritis Patients with Mild, Moderate and Severe Pain in the Val/Val, Val/Met and Met/Met Genotypes.
The odds of developing moderate to severe pain in the Met/Met genotype group were 10.4 times greater than those in the Val/Val genotype group (P = .02).
The correlations between the different parameters are listed in Table 2. We found a significant positive correlation between the ESR and NPRS score in the Val/Val genotype (r = 0.52, P < .01). No significant correlation was found between hs-CRP and the NPRS score or ESR and hs-CRP with the CHFS score in any genotype.
Correlations Among Inflammatory Markers, Disease Activity Scores, Numerical Pain Rating Scale Scores and Cochin Hand Function Scale Scores in Catechol-O-Methyltransferase Genotypes.
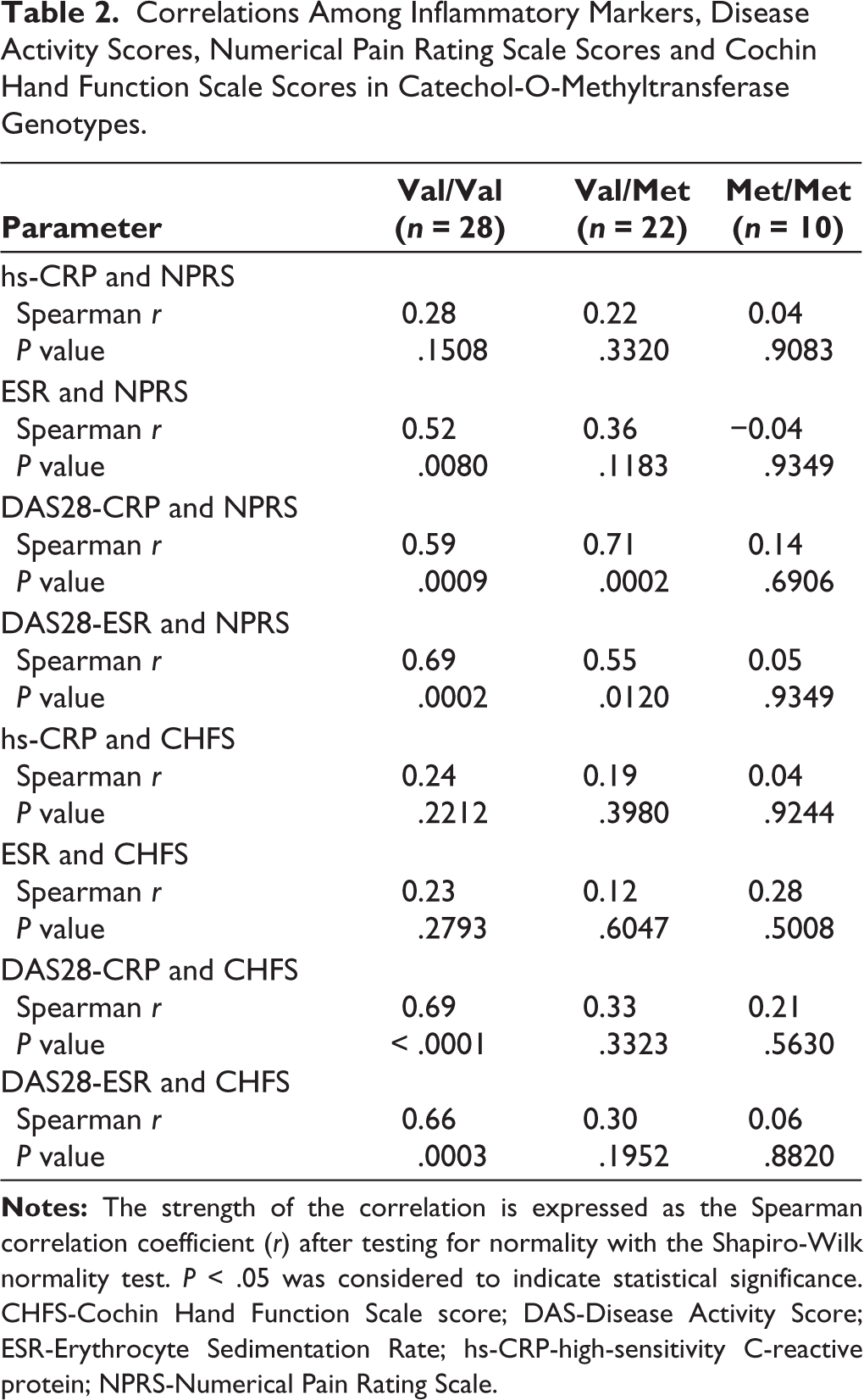
There was a significant positive correlation between the DAS28-ESR and NPRS scores in the Val/Val (r = 0.69, P < .001) and Val/Met (r = 0.55, P < .05) genotypes. Similarly, a positive correlation was found between the DAS28-CRP and NPRS scores in the Val/Val (r = 0.59, P < .001) and Val/Met (r = 0.71, P < .001) genotypes. No significant correlation was found between DAS and NPRS in the Met/Met genotype.
In terms of the disability score, there was a significant positive correlation between the DAS28-ESR and CHFS score (r = 0.66, P < .001) and between the DAS28-CRP score and CHFS score (r = 0.69, P < .0001) in the Val/Val genotype. No correlation was found between the DAS and CHFS score in the Met allele carriers.
Discussion
Chronic pain in RA patients is attributed to multiple mechanisms, including peripheral sensitisation, central sensitisation, diffuse noxious inhibitory control dysfunction and neuropathic pain. This pain sensitisation is strongly influenced by other factors, such as psychological baggage and genetics. In our study of 60 RA patients, there was an increased risk of developing moderate to severe pain in the Met/Met COMT genotype group compared with the Val/Val COMT genotype group. This is a pilot study investigating the associations of pain and COMT polymorphisms in RA in the Indian population. Earlier studies reported an association between pain and the COMT Val158Met polymorphism in other chronic pain conditions. There was an increased risk of hip pain in confirmed radiographic osteoarthritis patients who were Met allele carriers. 17 Additionally, in fibromyalgia patients, those with the Met/Met genotype presented increased pain scores.6–10 Fibromyalgia patients with Met/Met genotype showed higher sensitivity to thermal and pressure pain stimuli and higher fibromyalgia impact questionnaire score.6,7 The possible mechanism that can explain the increased risk of high-intensity pain in the Met/Met genotype is the difference in the enzymatic activity of the COMT gene product in Met allele carriers. The Val allele gives rise to an effective enzyme that is stable at 37°C, whereas the Met allele produces a defective enzyme with low stability at 37°C that is incapable of effectively clearing catecholamines from the system.2–4 Low COMT enzyme activity can lead to a reduction in the phasic release of dopamine from the nucleus accumbens and thus can decrease the descending inhibition of pain at the ventral pallidum, thalamus and dorsal horn. 18 Additionally, only non-pregnant, non-lactating female patients of reproductive age were included in the present study to minimise the influence of oestrogen levels, since oestrogen is known to regulate COMT expression. 19 The previous study on the prevalence of the COMT Val158Met polymorphism in 10 Sudanese RA patients has shown a 50% prevalence of Val/Val and Val/Met genotype without any Met/Met genotype. 20 To the best of our knowledge, this is the first study to assess pain and disability status in RA COMT genotype.
Our study did not reveal a correlation between hs-CRP and pain intensity in RA patients. This can be attributed to small sample size of the current study. Also, the absolute values of acute-phase reactants may not be the best indicator of inflammation, as these markers are influenced by lifestyle, genetics and diet.21,22 Additionally, the correlation between the ESR and pain intensity was found in the Val/Val genotype only, but not with the Val/Met or Met/Met genotypes. This might be because in Met allele carriers, low COMT enzyme activity is becoming a modulating factor in pain perception. 18 The influence of the COMT polymorphism in distorting the correlation between pain and inflammation in Met allele carriers needs to be confirmed in a large sample size study.
In our study, there was a positive correlation between DAS and pain intensity in the Val/Val and Val/Met genotypes, but not in the Met/Met genotype. The DAS is calculated from the tender joint count, swollen joint count, patient global health and ESR or CRP levels. This score is a composite of a subjective assessment and an objective assessment of disease activity and it is correlated with pain intensity, as pain is a manifestation of disease activity. Studies have shown a strong temporal relationship between changes in disease activity and pain intensity in the course of disease. 23 The discordant relationship between disease activity and pain intensity in the Met/Met genotype might be due to the decrease in descending pain inhibition mediated by the Met allele. 18
Functional disability in RA patients is an important outcome measure, as it contributes to low work productivity and impaired quality of life. Studies have shown that there is a positive correlation between the hand function score and occupational performance.24,25 Therefore, we have taken the CHFS score as a scale of functional disability. In our study, hand function disability did not correlate with inflammatory markers in RA patients. However, the study showed a positive correlation between the DAS and functional disability in the Val/Val genotype. The DAS, being a composite score that includes the tender joint count, swollen joint count, patient global health and inflammatory markers, is likely to influence functional ability more than inflammatory markers alone. Also, the hand function performance depends on the number and type of joints affected with pain and swelling. Functional disability is also influenced by the emotional processing and behaviour of individuals. Compared with Val homozygotes, Met homozygotes showed a more internal negative affective state; the Met allele has been suggested to underlie inter-individual differences and less efficient adaptation to painful and stressful stimuli. 5 Increased cortico-limbic reactivity in Met carriers is suggested to underlie emotional dysregulation. 26 Thus, the discordant relationship between disease activity and functional disability might be due to altered emotional processing in Met allele carriers.
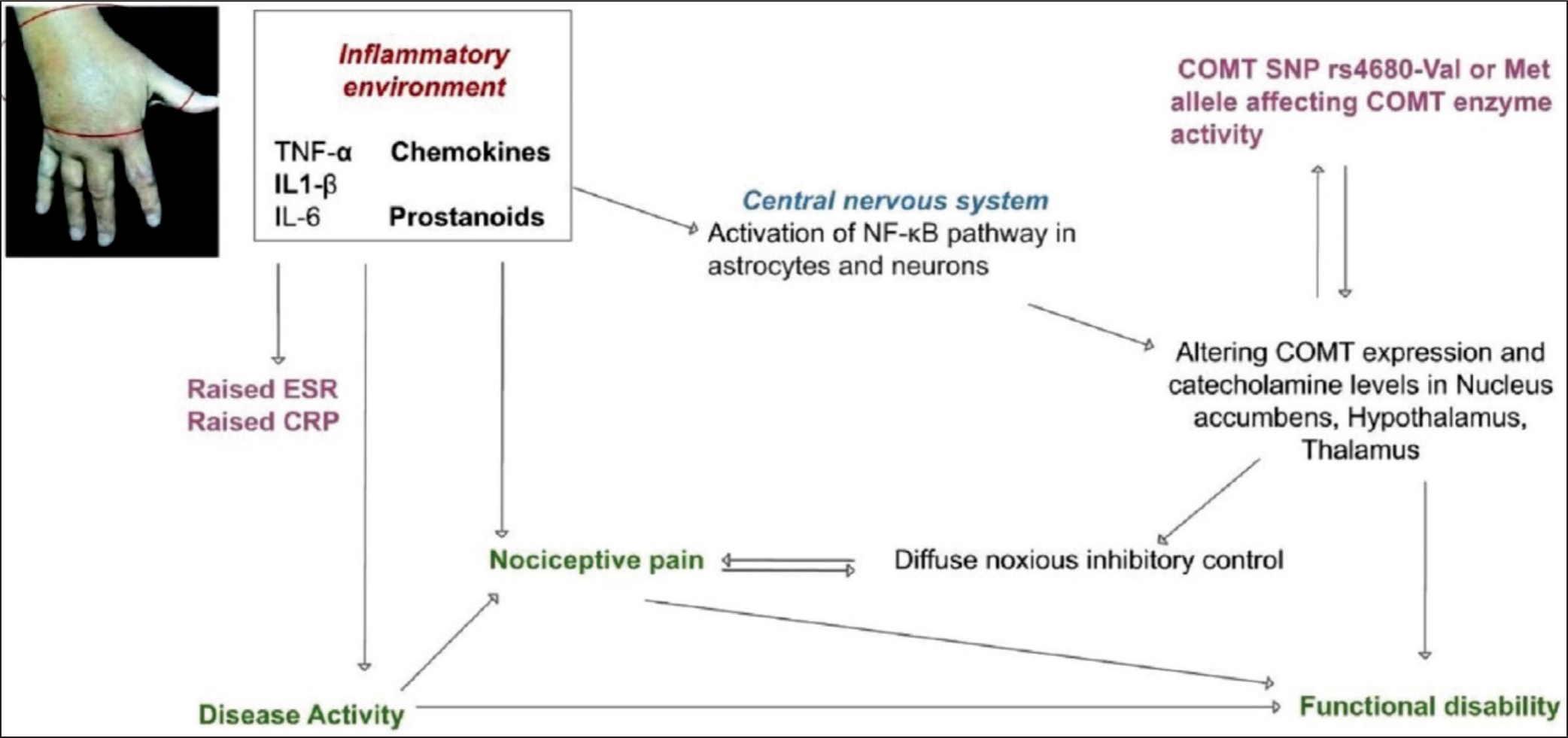
The proposed mechanism by which inflammation and the COMT allele expression influence pain intensity and functional disability is shown in Figure 3. Synovitis of the affected joint in RA can induce the production of molecules such as TNF-alpha, IL-1β, IL-6, chemokines and many more, which are responsible for nociceptive pain at affected joints. Elevated levels of interleukins and chemokines in affected joints and the circulation result in increased ESR and the release of CRP from the liver. Molecules such as TNF-alpha may induce activation of the nuclear factor kappa B (NF-kB) pathway in astrocytes and neurons, which has been shown to reduce COMT gene expression in the forebrain and midbrain areas and may inhibit descending diffuse noxious inhibitory control, resulting in heightened pain perception.18,27,28 In the Met/Met genotype, we hypothesise along with NF-kB-mediated alterations in COMT enzyme expression, the low enzyme activity of COMT may result in discordance of the relationship between disease activity and pain. Experimental evidence from further studies is needed to substantiate this hypothesis. Functional disability is influenced by disease activity, pain perception and disability perception. Disability perception is negatively affected by emotional dysregulation and less efficient adaptation to stressful stimuli, which might lead to a discordant relation between disease activity and functional disability in Met allele carriers.
The Proposed Mechanism by Which Inflammation and COMT Gene Expression Influence Pain Perception and Functional Disability in Rheumatoid Arthritis Patients. The Inflammatory Environment at Affected Joints Results in Nociceptive Pain. 12 The Chemokines Released from the Inflammatory Environment, such as TNF-alpha, Activate the NF-kB Pathway, which Reduces the Expression of the COMT Enzyme in Astrocytes and Neurons of the Nucleus Accumbens, Hypothalamus and Thalamus.27,28 This May Decrease Descending Diffuse Noxious Inhibitory Control and Increase Pain Perception. 18 The Continuous Nociceptive Input Itself Can Alter Diffuse Noxious Inhibitory Control and Make it Dysfunctional. 12 With Inflammation, the Blood CRP Concentration and ESR Increase. Inflammation is a Determinant of Disease Activity. In the Met/Met Genotype, NF-kB-mediated Alterations in COMT Enzyme Expression Might be Affected, Resulting in Discordant Relation between Disease Activity and Pain. Functional Disability is Influenced by Disease Activity, Pain Perception and Disability Perception. Disability Perception Depends on Emotional Processing. Emotional Dysregulation and Less Efficient Adaptation to Stressful Stimuli Might Result in Discordance between Disease Activity and Functional Disability in Met Allele Carriers.
In conclusion, the subjective experience of pain and disability in patients with RA is influenced not only by disease activity but also by the COMT Val158Met polymorphism. The interaction of inflammation with allele-dependent expression of the COMT gene may contribute to the variable correlations of pain and functional disability with inflammation and disease activity in three COMT genotypes of RA. Further validation of these findings is necessary and will be helpful in deciding personalised pain management options for RA patients.
The limitation of this study is that the dose-dependent effects of DMARDs and NSAIDs on pain, inflammatory markers and disability are not taken into consideration. Even though oestrogen influences COMT expression, the phase of the menstrual cycle or the serum oestrogen level was not taken into consideration during analysis. Another limitation is the small sample size and exclusion of men from the study population. Also, the pain assessment relied only on subjective and patient-dependent scores, which may not fully capture the multidimensional nature of pain in RA. Therefore, it is important to validate gene-inflammation interactions with further longitudinal studies of large sample size to understand the complex pathophysiology of pain in RA. The role of other gene polymorphisms and their interactions in RA also needs to be incorporated in further studies.
Footnotes
Acknowledgements
The authors acknowledge the research staff and PhDs of Pain and Transcranial Magnetic Stimulation Laboratory, Integral Health and Wellness Clinic, Department of Rheumatology and Department of Anatomy, All India Institute of Medical Sciences, New Delhi.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval obtained from Institute Ethics Committee for Postgraduate Research, All India Institute of Medical Sciences, New Delhi. Ref No. IECPG-649/ 28.10.2021.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
All the data are collected after written informed consent from patients.
