Abstract
Cyclophosphamide (CYC), a derivative of mustard gas, has been a cornerstone in treating life-threatening complications of various inflammatory rheumatic and musculoskeletal diseases (I-RMDs) since the mid-twentieth century, despite its well-documented adverse effects. The transformation of CYC from a chemical warfare agent in World War I to a life-saving drug is remarkable and crucial for rheumatologists to understand. This article offers a historical overview of the events and the scientists who were instrumental in this transformation, along with its key role in I-RMDs. Over the ensuing decades, numerous drugs have been developed to address conditions for which CYC was once the sole treatment option. Nevertheless, even in the third decade of the twenty-first century, CYC continues to serve as a dependable option for managing certain complex and challenging complications of I-RMDs. Rheumatologists, however, face a significant challenge when considering the initiation of CYC therapy. They must carefully weigh its proven efficacy against its potential toxicities. Furthermore, practical considerations such as the route of administration, dosage schedule and treatment duration must be guided by the best available evidence. Unfortunately, such information is often dispersed across decades of publications, with recommendations that are sometimes inconsistent and perplexing.
This review delves into two primary aspects of CYC: its historical evolution and the practical considerations for its use in contemporary rheumatology practice.
Keywords
Introduction
The devastation of World Wars (WW) I and II showcased both humanity’s capacity for destruction and its resilience in transforming calamity into progress. From the horrors of chemical warfare, particularly mustard gas (whose derivative is cycloph«osphamide (CYC)), emerged groundbreaking medical advancements. The accidental release of mustard gas in WWII’s ‘Little Pearl Harbor’ at Bari (Italy) led to investigations into nitrogen mustard, revealing its potential for cancer treatment.1,2 Pioneering researchers, including Goodman and Gilman, laid the foundation for using nitrogen mustards in cancer chemotherapy and recognised its ability to suppress white blood cells and shrink lymph nodes, laying the foundation for modern chemotherapy.3,4 This pivotal discovery eventually led to the development of CYC, a crucial drug in treating cancers and certain inflammatory rheumatic and musculoskeletal diseases: I-RMDs.
Nitrogen Mustard and Its Derivative Cyclophosphamide in the Management of I-RMDs
The use of nitrogen mustard for treating rheumatoid arthritis (RA) was first reported in the 1950s by Spanish physicians Jimenez-Diaz et al. and Robecchi et al.5,6 However, the credit for integrating chemical immunosuppression into routine rheumatology practice belongs to the rheumatologists at the Cleveland Clinic, Cleveland, Ohio, United States. In a series of landmark articles published in 1957, Scherbel, Schuchter, and Harrison from the Department of Rheumatic Diseases at the Cleveland Clinic successfully transitioned the use of nitrogen mustard (HN2) from a research concept to clinical application in managing I-RMDs.7–10 The concept proposed by Scherbel et al. at the Cleveland Clinic—using cytotoxic drugs to treat I-RMDs—quickly gained traction among physicians at several centres across the United States, with five significant reports emerging between 1960 and 1976.11–15 The rheumatologists at the Cleveland Clinic also acknowledged the contributions of several other prominent rheumatologists of that era in the United States, including Daniel McCarthy, Rollins and Shaw. These pioneers reported similar experiences with the use of nitrogen mustard in treating RA and other I-RMDs, such as systemic lupus erythematosus (SLE), polyarteritis nodosa, dermatomyositis (DM) and glomerulonephritis. 9
The first use of CYC in I-RMDs was in RA and was carried out by Fosdick et al. on 38 patients who had failed other therapies. They concluded that CYC showed promise as a therapeutic option for patients of RA who had failed other therapies. 16 Suarez-Almazor et al. shed more light upon the use of CYC in RA in their systematic review which concluded that CYC is effective for the treatment of RA in patients who have failed other therapies; however, its use is limited by its toxicity. 17 However, with the introduction of highly effective and less toxic therapies, such as low-dose methotrexate, the use of CYC in RA was eventually discontinued.
John Fahey et al. from Presbyterian Hospital, New York, the United States in 1954 did pioneering work in the description of granulomatosis with polyangiitis (GPA, earlier called Wegener’s granulomatosis). 18 The first use of CYC as single-drug therapy for GPA in four patients was reported by Carl Pearson’s group from Los Angeles on 29 April 1971. 19 Within eight months of the first publication (December 1971) Workers at NIH (National Institutes of Health), Bethesda, United States led by Anthoney Fauci, published their landmark article describing high efficacy of CYC in GPA in 9 cases. 20 Both the groups showed complete-to-partial remission in most of the patients treated with CYC in GPA. The NIH team published their collective experience with 180 patients with GPA with multiorgan anti-neutrophil cytoplasmic antibody-associated vasculitis (AAV) between 1967 and 1991. 21
The Mode of Action of Nitrogen Mustard and Cyc for the Treatment of Cancer and I-RMDs
The mechanism of action of nitrogen mustard which enables it to eliminate rapidly dividing cells, such as cancer cells and immune system cells, made it popular in both these fields.
Nitrogen mustards are polyfunctional alkylating agents. 22 In the body, nitrogen mustards (available in three forms: HN1, HN2, and HN3) react with two guanine bases on opposite strands of a DNA helix, forming interstrand cross-links. This process, known as DNA alkylation, prevents the unwinding of the DNA helix, thereby blocking essential processes such as replication and transcription. The resulting disruption leads to the death of rapidly dividing cells.22–24
Administering nitrogen mustard in its gaseous form posed significant challenges due to its instability and the need for specialised handling. This made the development of CYC, a more stable and practical derivative of nitrogen mustard, a pivotal advancement. Synthesised by Friedman and Seligman in 1954 25 and further developed by Norbert Brock et al. 26 CYC became available both in oral and intravenous (IV) form, thus making it as a reliable treatment option.
Route of Administration
CYC is primarily administered through two routes: (a) oral and (b) IV route, each with distinct advantages and limitations. The oral route is convenient to administer and relatively cost-effective. However, it results in a higher cumulative dose compared to the IV route, which, while effective, is associated with increased toxicity. Improper adherence to oral CYC regimens, such as failing to take the medication in the morning or neglecting adequate hydration throughout the day, can lead to serious complications, including haemorrhagic cystitis and other urinary tract side effects. 27 Prolonged use of oral CYC beyond the prescribed treatment period, often due to loss to follow-up, can result in catastrophic outcomes, such as the development of secondary malignancies. On the other hand, IV-CYC offers better tolerability, facilitates easier monitoring and typically involves a lower cumulative dose compared to the oral formulation, making it a preferred option in many clinical scenarios.
Following the development of a stable compound-CYC, its use expanded and researchers began exploring CYC in various rheumatologic diseases.
CYC in Lupus Nephritis
Historically, the first formal trial of CYC for lupus nephritis was conducted in 1970 by Cameron, Boulton-Jones, Robinson and Ogg at Guy’s Hospital, London. 28 This open-label trial involved six patients with biopsy-confirmed lupus nephritis. The outcome was mixed: Three patients showed a significant reduction in proteinuria; the dose of corticosteroids in two patients could be reduced significantly while maintaining disease control; one patient who had active necrotising renal lesion succumbed. 28
Almost simultaneously, in 1971, a controlled trial led by A.D. Steinberg and his team at the National Institutes of Health (NIH), Bethesda, United States, examined the use of CYC in lupus nephritis. 29 This trial yielded positive results and was followed by eight additional NIH-led trials, the last of which was published in 1992. A summary of these trials is provided in Table 1.
Summary of National Institutes of Health (NIH) Studies on Cyclophosphamide Use in Lupus Nephritis.
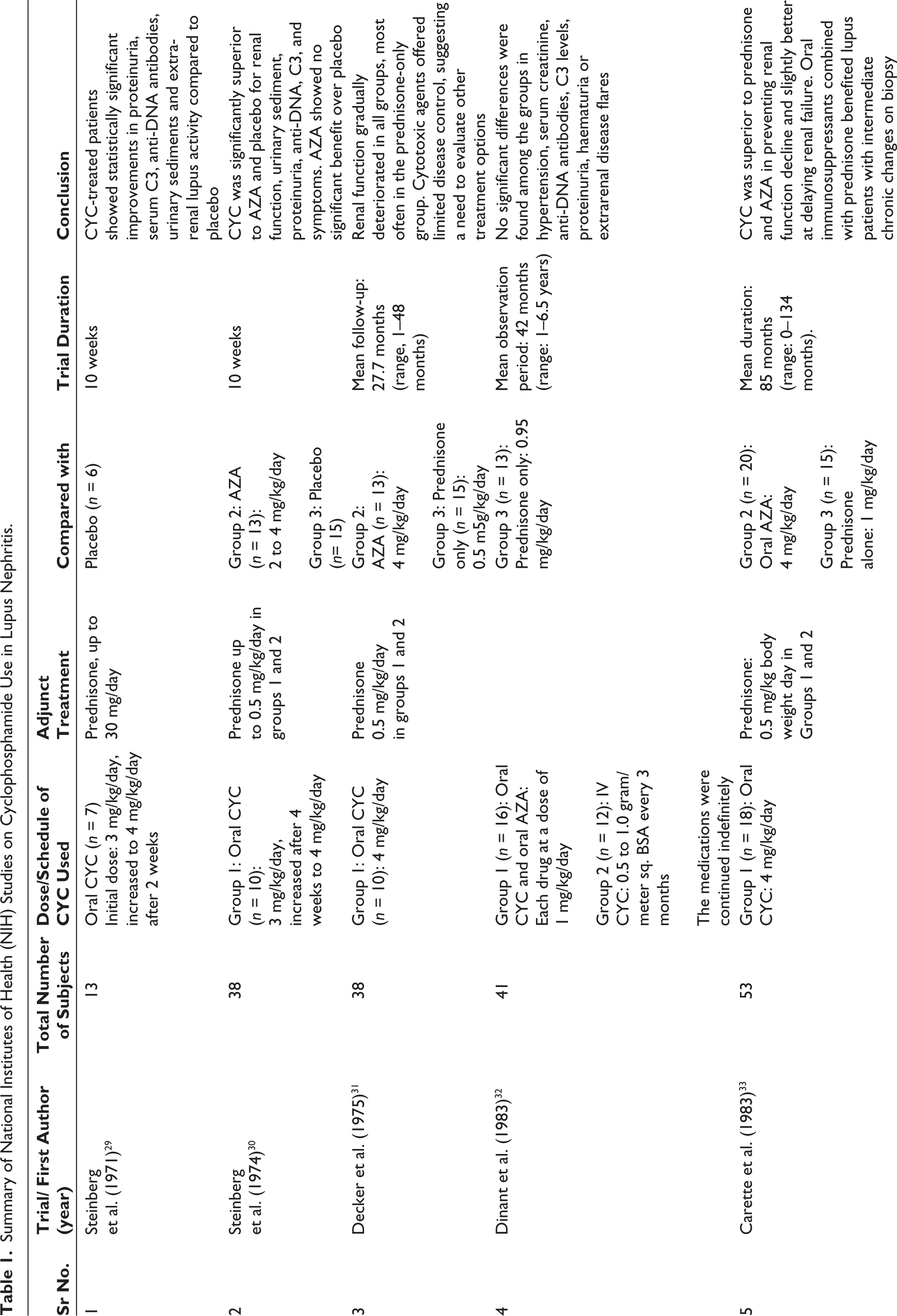
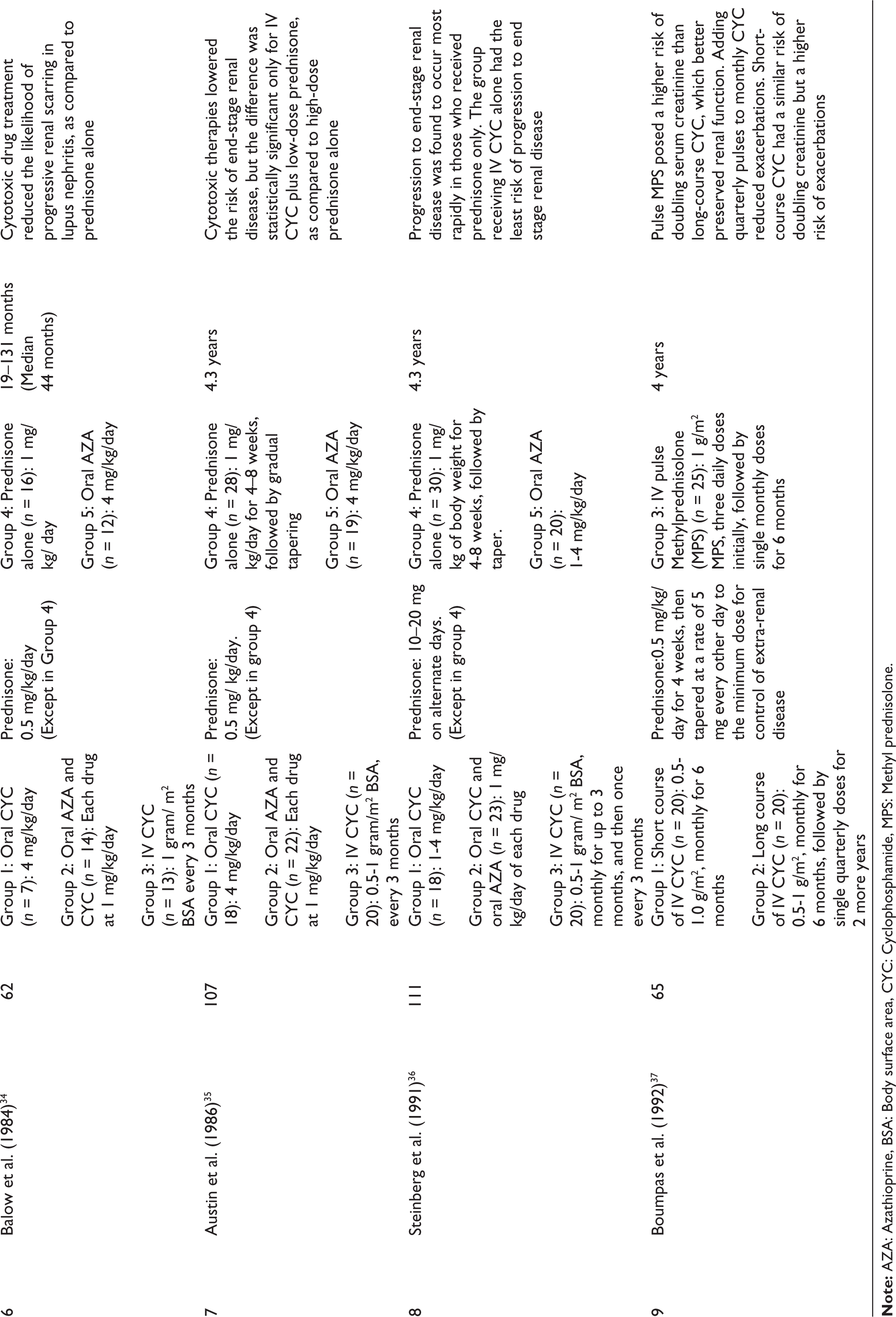
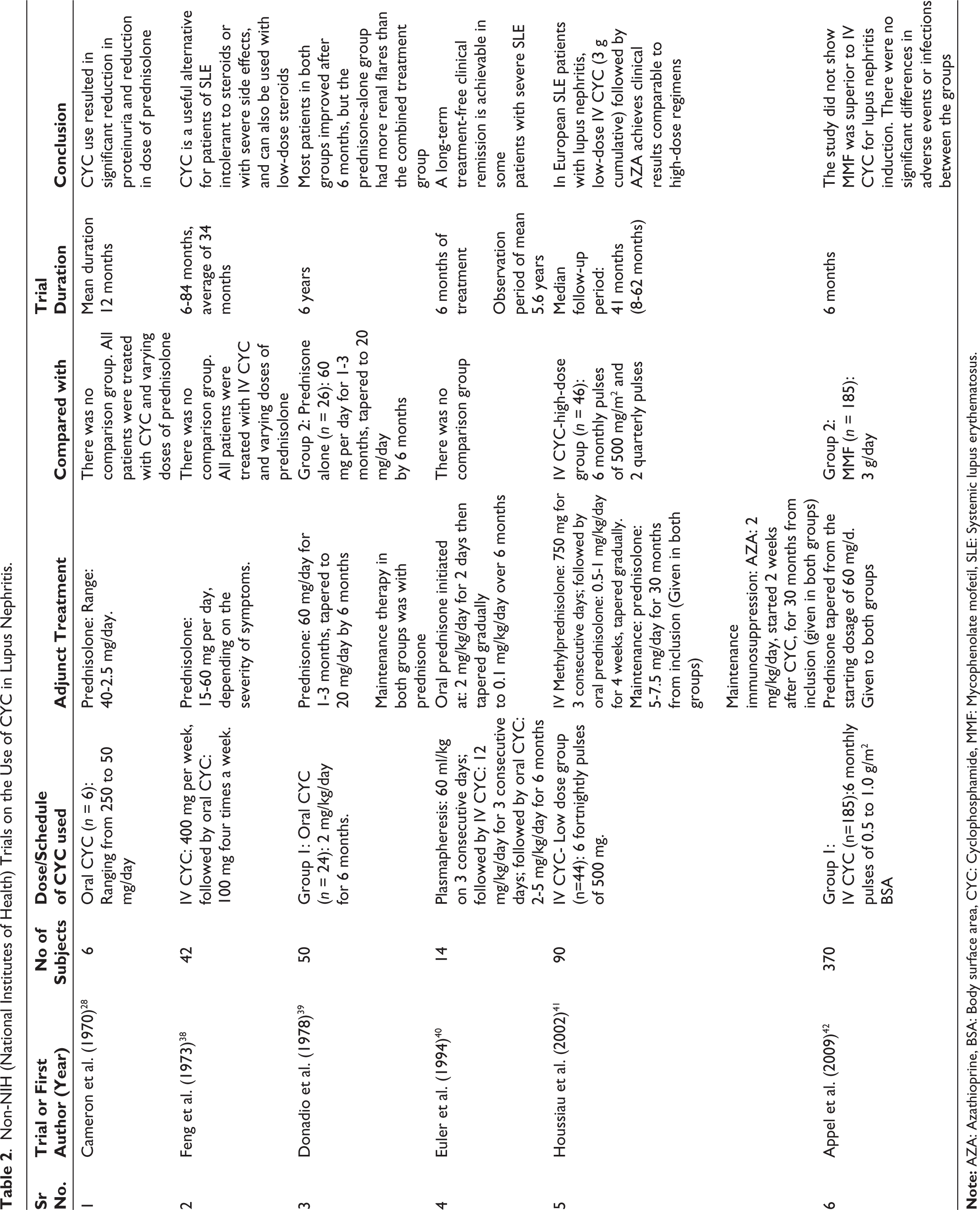
Furthermore, six independent trials were conducted at centres outside the NIH, as summarised in Table 2. Buoyed by the success of CYC in lupus nephritis, its use was expanded to other I-RMDs.
CYC in Neuro-psychiatric SLE (NPSLE)
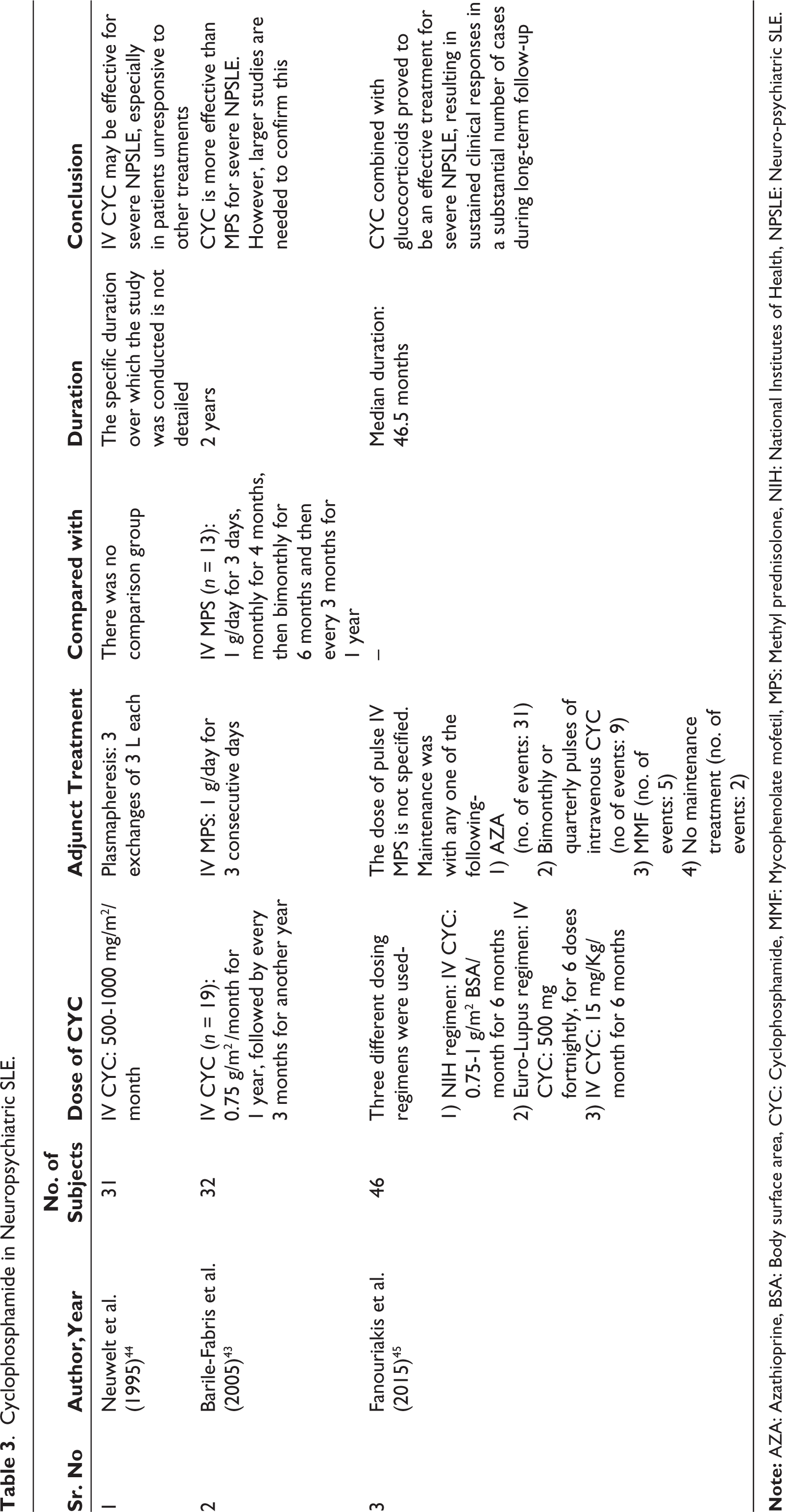
Central nervous system (CNS) involvement is a significant contributor to illness severity and death in patients with SLE. Managing NPSLE is a substantial challenge for clinicians in everyday practice. 43 In a randomised trial of 32 patients that compared IV CYC with IV methylprednisolone, IV CYC was found to be significantly better than IV methylprednisolone in treating NPSLE. This study also suggested that to achieve optimal response in NPSLE with no flares, treatment with CYC should be sustained for two years. 43 Table 3 summarises selected studies of CYC in NPSLE.
CYC in ANCA-Associated Vasculitis (AAV)
Vasculitis is among the most aggressive and difficult to treat conditions faced by rheumatologists. Before the advent of CYC treatment, mortality of severe AAV was ~80% within one year of diagnosis. 46 Table 4 gives the list of CYC trials in AAV along with the dose schedule.
The randomised controlled trials of CYC in AAV can be classified into:
Induction trials The CYCLOPS trial conducted in 2009 was a randomised trial comparing daily oral and monthly IV CYC regimens.
47
It showed that the rate of induction of remission and the number of patients who achieved remission is almost equivalent in both the regimens. Further details of the CYCLOPS and several other trials are mentioned in Table 4. Maintenance trials The CYCAZAREM trial was a randomised control trial evaluating the role of oral CYC as a maintenance therapy for patients with AAV and renal involvement and compared it to AZA. The trial advocated that AZA can be safely used as a substitute to CYC during the remission phase.
48
Table 4 provides further details on CYC In AAV.
Non-NIH (National Institutes of Health) Trials on the Use of CYC in Lupus Nephritis.
Cyclophosphamide in Neuropsychiatric SLE.
Cyclophosphamide in ANCA-Associated Vasculitis.


Since these important early reports, major advances in the treatment of AAV have improved five-year survival to 74%–91% and 45%–76% for GPA and MPA respectively. 49
CYC in Systemic Sclerosis-related Interstitial Lung Disease (SSc-ILD)
As such, immunosuppressive drugs have been used mainly for SSc-ILD. There have been several uncontrolled studies performed with CYC that revealed a positive effect on pulmonary functions in patients with SSc-ILD. 55 These led to the first randomised controlled trial with CYC for SSc-ILD, the Scleroderma Lung Study I (SLS I) 56 which was followed by the ‘Scleroderma Lung Study II’ (SLS II), 57 detailed in Table 5.
Cyclophosphamide in Other I-RMDs.

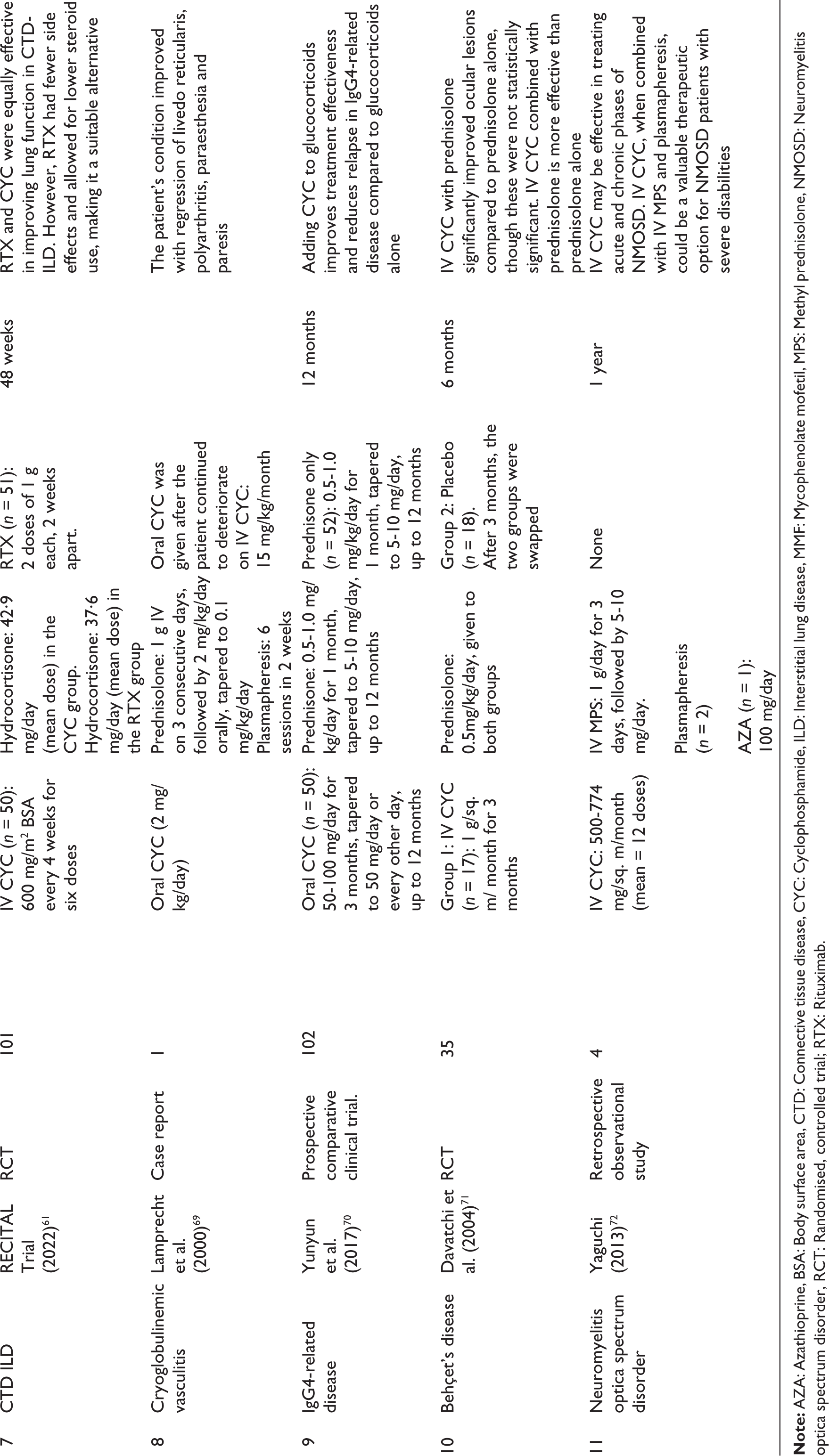
The 2023 guidelines by American College of Rheumatology (ACR)/American College of Chest Physicians (CHEST), for the treatment of CTD-ILD state that in SSc-ILD, CYC may be used as an additional drug, after the recommended first-line agents for SSc-ILD (MMF, tocilizumab and RTX) have shown inadequate response. 58 In such a scenario, CYC may be administered either IV or orally, but the use of intermittent IV pulses is preferred because it is less toxic. The recommended dose of IV CYC is 500–750 mg/m2 of BSA (maximum dose of 1200 mg), given monthly for 6 months to be followed by maintenance with a less toxic agent, for example, MMF (2–3 g/day). 58
CYC in Skin Involvement in Systemic Sclerosis (SSc)
Skin involvement in SSc has been a challenge to treat. Initiation of treatment is done with CYC at a dose of 500 mg/m2 for 6–12 months. Maintenance agents commonly used after 6 to 12 months of CYC include methotrexate, MMF or AZA (Table 5). 59
CYC in Idiopathic Inflammatory Myopathy (IIM)
In a systematic review on IIM patients with progressive ILD, IV-CYC was administered at the dose of 300–800 mg/m2 given once in four weeks for six cycles. This was combined with oral prednisolone at a dose between 30 and 60 mg/day for the first two weeks and gradually tapered in all patients. It was shown that IV-CYC improved symptoms, pulmonary function tests and high-resolution computerised tomography (HRCT). The same review also showed improvement both in muscle strength as also in creatine kinase levels. 60
The RECITAL trial compared RTX versus IV-CYC in patients with ILD in various CTDs, including IIM. It concluded that RTX was not superior to CYC for treating CTD-ILD. However, RTX was associated with fewer adverse events (Table 5). 61
CYC in Catastrophic Antiphospholipid Syndrome (CAPS)
In rare cases, CYC is a part of combination therapy to manage CAPS, especially if secondary to lupus or other autoimmune diseases. 62
In SLE-associated CAPS, CYC has been used at a dose of 500–750 mg/m2 BSA, as has been administered in other organ or life-threatening manifestations of SLE. 63
CYC in Mixed Connective Tissue Disease (MCTD)
For cases with severe systemic involvement, including ILD or renal crisis, CYC has been employed. 61
In a review of 495 participants (most with systemic sclerosis), two separate comparisons were done—CYC versus placebo and CYC versus mycophenolate. The data demonstrated significant improvement in lung function with CYC compared with placebo with regard to FVC%, but no significant impact on lung function when CYC was compared with mycophenolate, at 12 months. CYC had more adverse events as compared to mycophenolate, in particular, leukopenia and thrombocytopenia. 64
CYC in Sarcoidosis
For refractory cases with extensive pulmonary or extrapulmonary involvement, CYC has been explored as a treatment option. It has been found to be an effective treatment in corticosteroid-resistant disease in both neuro-sarcoidosis and cardiac sarcoidosis, and has been associated with a lower relapse rate for patients with neuro-sarcoidosis. 65
CYC in Relapsing Polychondritis
In patients with life- or organ-threatening relapsing polychondritis, such as involvement of laryngotracheal and bronchial cartilage, cardiovascular, renal, neurologic disease, ocular or inner ear involvement, a combination of prednisolone (usually 1 mg/kg daily) with oral CYC at a dose of 2mg/kg/day has been used. There are no fixed guidelines about maintenance therapy, but usually it is continued for at least 6 months of disease remission. Once the patient is stable, any of the agents among AZA, Methotrexate (MTX) or IV-pulsed-CYC every one to two months may be considered. 66
Use of CYC in various other I-RMDs has been described in Table 5.
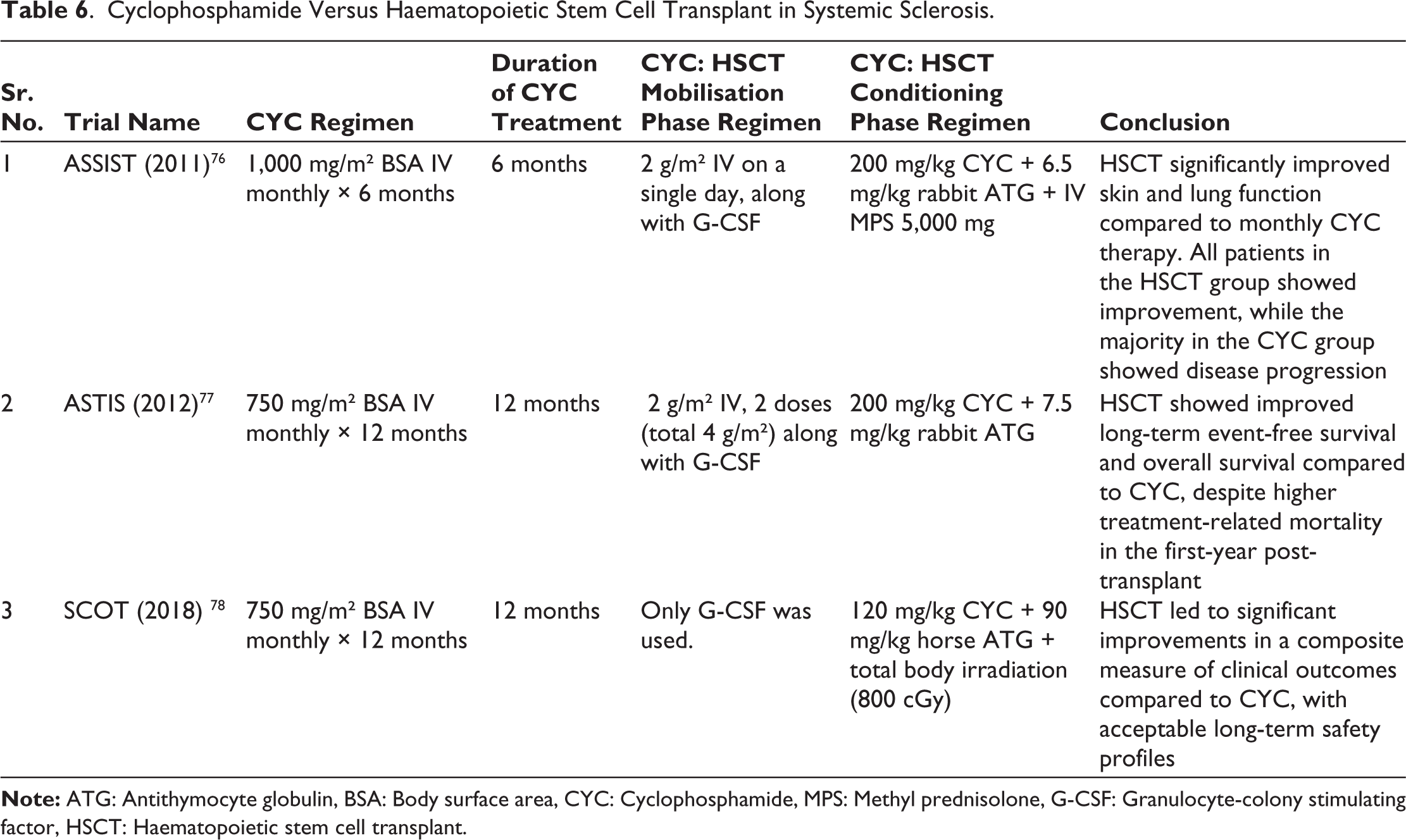
CYC in Autologous Haematopoietic Stem Cell Transplant
In patients with scleroderma, autologous haematopoietic stem cell therapy (HSCT) has emerged as a potential treatment option. In this context, CYC has a dual role to play:73–75
Stem cell Mobilisation (Mobilisation phase): CYC facilitates the release of haematopoietic stem cells into the peripheral bloodstream, making them more accessible for collection in autologous stem cell transplantation procedures. Immune System Reset (Conditioning phase): Prior to reinfusion of stem cells, CYC is administered for its immunoablative effects and this eliminates autoreactive immune cells, thus leading to long term disease remission.
The trials comparing HSCT and CYC in SSc are included in Table 6.
Cyclophosphamide Versus Haematopoietic Stem Cell Transplant in Systemic Sclerosis.
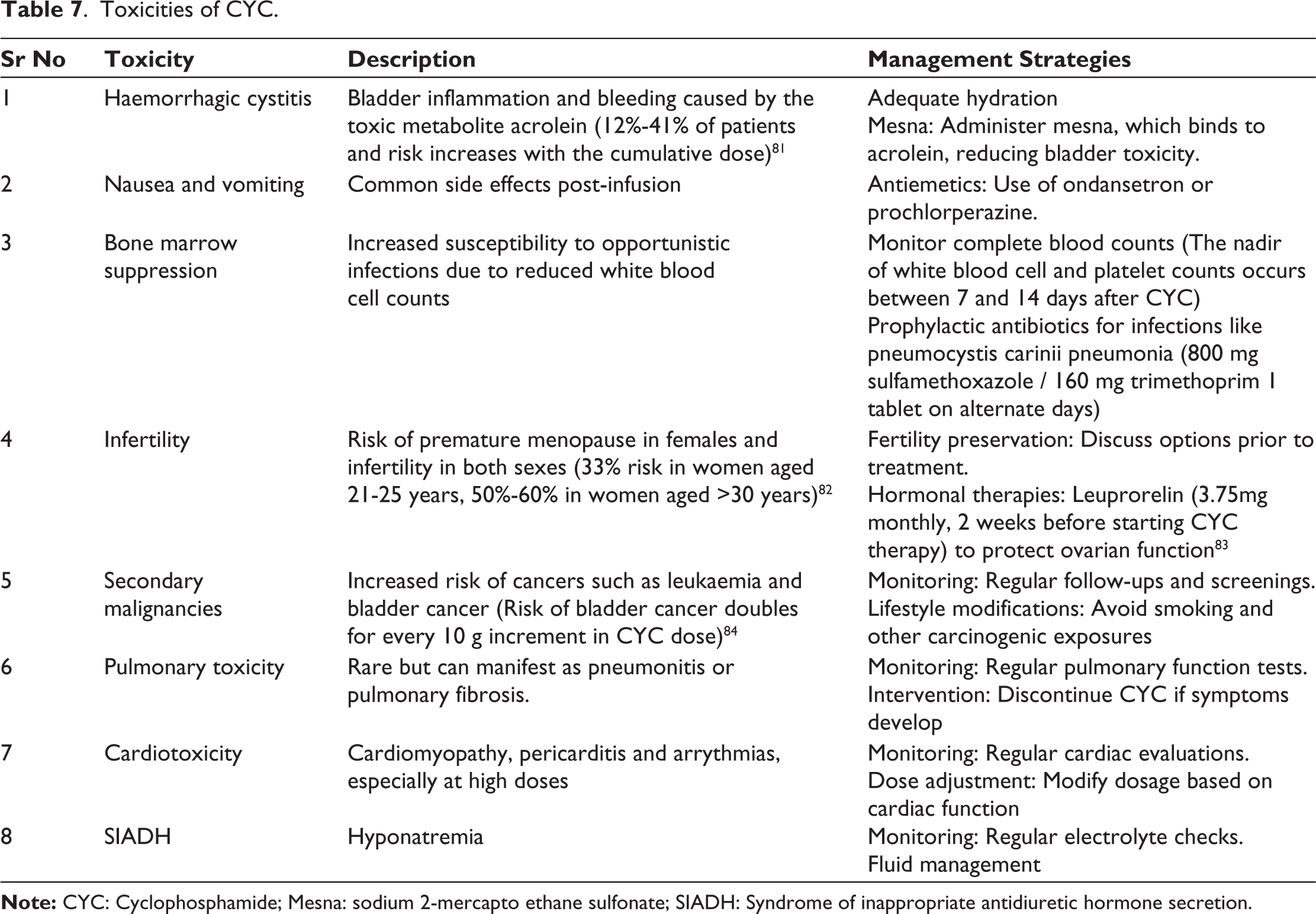
Toxicities of CYC
CYC is associated with a considerable number of toxicities,79,80 some of which are enlisted in Table 7. Due to its toxicity profile, CYC is increasingly being replaced by relatively safer alternatives such as MMF and RTX. This shift likely explains the decline in its use in recent years.
Toxicities of CYC.
Conclusion
In conclusion, the evolution of CYC from a lethal nitrogen mustard gas to a life-saving therapeutic agent highlights the remarkable potential of scientific innovation in transforming harmful substances into invaluable medical tools. In current practice,
While newer therapies have emerged for the treatment of I-RMDs, CYC remains a cost effective and time-tested drug in life-saving situations, despite its toxicities. CYC is one of the first-line drugs in the management of proliferative classes of lupus nephritis and AAV. CYC is highly efficacious in the management of severe forms of SSc, NPSLE and IIM. The emerging role of CYC in HSCT for SSc in both the mobilisation and conditioning phases is noteworthy. The toxicities of CYC, particularly infertility, bone marrow suppression and bladder cancer should be borne in mind when using CYC. Monitoring the total white cell count, specifically between day 7 to 14 after administering CYC, and monitoring the renal function of a patient on CYC is crucial to prevent its life-threatening complications. A constant vigilance for development of infections and malignancies is a must.
Continued research into optimising its use and minimising its adverse effects will further solidify its legacy as a cornerstone in the history of modern medicine.
Footnotes
Authors Contribution
NYK and ANM contributed to literature search and writing of the manuscript.
Data Availability
All relevant data is presented in the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Permission to Reproduce Material from Other Sources
The authors have noted that no third-party content was reproduced in the article.
