Abstract
Introduction:
There is a pressing need for non-invasive biomarkers to monitor lupus nephritis (LN) disease activity, as it is a critical complication. This study aimed to evaluate the role of both interleukin-18 (IL-18) and anti-α-enolase as biomarkers for predicting lupus renal disease activity.
Methods:
This case-control study enrolled 90 participants: 30 with active LN, 30 with inactive LN, and 30 healthy matching age and sex controls. Serum concentrations of IL-18 and anti-α-enolase were estimated. The activity of disease and organ damage were determined utilising the systemic lupus erythematosus (SLE) Disease Activity Index (SLEDAI-2K), the Systemic Lupus International Collaborating Clinics/American College of Rheumatology (SLICC/ACR) Damage Index (SDI), and the activity of renal disease was evaluated utilising the renal SLEDAI score.
Results:
Serum levels of both IL-18 and anti-α-enolase were significantly higher among patients with active LN (89.97 ± 14.71 ng/mL and 51.67 ± 8.84 ng/mL) compared to patients with inactive LN (24.03 ± 18.21 ng/mL and 18.53 ± 11.36 ng/mL) and controls (1.217 ± 0.611 ng/mL and 1.153 ± 0.532 ng/mL) (P < .001). Both biomarkers positively correlated with SLEDAI-2K score (r = 0.89, r = 0.845), ESR (r = 0.689, r = 0.69), 24-hour urinary proteins (r = 0.601, r = 0.559) (P < .001). While they both negatively correlated with e-GFR (r = −0.58, r = −0.593), complement C3 and C4 levels (r = −0.727, r = −0.665), (r = −0.567, r = −0.56), (P < .001). ROC curve analysis had high diagnostic accuracy (AUC = 0.95), with IL-18 >25 ng/mL and anti-α-enolase >24 ng/mL yielding 96.7% sensitivity and >90% specificity for active LN detection (P < .01).
Conclusions:
Anti-α-enolase antibodies and IL-18 can be useful biomarkers for detecting and monitoring renal disease activity in SLE and may serve as therapeutic targets in the future.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a multifaceted autoimmune condition defined by diverse immunological irregularities. These comprise the autoantibodies (auto-Abs) production, the self-reactive B and T lymphocytes, and a dysregulated cytokine network that collectively trigger widespread inflammatory processes and cause multisystem organ damage. 1 The underlying mechanisms of SLE remain incompletely elucidated, with significant variability in the factors precipitating disease onset. The clinical presentation of SLE is remarkably diverse, with inflammatory processes potentially affecting numerous tissue systems and organs throughout the body. Among the various clinical manifestations, kidney involvement, known as lupus nephritis (LN), represents a particularly significant aspect of the disease. 2
An estimated 60%-80% of people with SLE experience LN, a serious and common consequence that greatly increases morbidity and death from the illness. 3 Renal inflammation is the condition’s hallmark, which is caused by complex immunological interactions between kidney tissue and innate and adaptive immune responses. 4 In spite of substantial advances in medical interventions over recent decades that have improved survival rates for patients with severe LN, clinical complete remission following immunosuppressive treatments remains achievable in only nearly 50% of cases. Consequently, early and precise identification of LN is crucial for implementing timely therapeutic strategies and optimising patient prognosis. 5
There are now biomarkers that are promising diagnostic and monitoring tools for LN, although their precise role in the disease’s pathogenetic mechanisms continues to be complex and not fully comprehended. 6 Whereas renal biopsy maintains its status as the definitive diagnostic method for identifying LN’s histological subtypes and guiding clinical management, the molecular intricacies underlying specific renal lesions remain substantially unexplored and represent a significant area for continued research and investigation. 7
As critical biomarkers, auto-Abs shed light on both the activity and progression of the disease. Anti-α-enolase Ab may be a sign of active autoimmunity and fibrinolysis suppression and is associated with active kidney disease in SLE. The complex neutrophil extracellular trappings (NETs), which are released by neutrophils through an active process known as NETosis, include α-enolase. One of the main causes of autoimmunity in lupus is NETosis, which externalises α-enolase during flare-ups and causes circulating α-enolase antibody (Ab) to develop. 8
Several serum proteins have demonstrated promise as LN markers in addition to auto-Abs. One of these, interleukin-18 (IL-18), may be a key player in the pathophysiology of SLE; however, research comparing the levels of circulating IL-18 in SLE patients to that of healthy individuals, as well as research looking at the connection between IL-18 levels and the activity of SLE renal disease, has produced conflicting findings. In mouse spleen cells, IL-18 was first identified as a factor that increased interferon-gamma (IFN-γ). Because it alters either the T helper 2 or T helper 1 immune responses, it is essential in autoimmune diseases. 9
Kupffer cells, activated macrophages, keratinocytes, intestinal epithelial cells, osteoblasts and adrenal cortex cells are among the cell types that produce IL-18. Its expression boosts the cytotoxic effects of NK and T cells in SLE and triggers the production of IFN-γ, granulocyte/macrophage colony-stimulating factor, and tumour necrosis factor-α. We might conclude that IL-18 plays a complex role in autoimmune lupus, appearing to be involved in both the early pathogenic events in specific organs and the effector phases of the late organ destruction. 10 To our knowledge, IL-18-targeting treatment approaches for autoimmunity are being developed, so in this study we selected these two biomarkers to evaluate their diagnostic and prognostic utility in detecting renal disease activity in SLE instead of invasive techniques such as renal biopsies and may be they can serve as a potential therapeutic targets in the future.
Patients and Methods
Design and Population
This prospective one-year case-control investigation was executed within the Rheumatology Department of the hospitals, comprising a comprehensive cohort of 90 research participants. The study population was strategically stratified into three distinct groups: a control group of 30 healthy individuals, a cohort of 30 sufferers of SLE who presented with active LN, and a third group of 30 SLE patients characterised by inactive LN. The research protocol was rigorously reviewed and received formal institutional approval from the Research Ethics Committee of the Faculty of Medicine. Every participant signed a written informed consent form.
Patient Selection
The study encompassed adult participants who were over 18 and fit the SLICC categorisation criteria for SLE. 11 The SLEDAI-2K was employed to gauge disease activity, 12 whereas the SLICC/ACR Damage Index (SDI) was utilised to estimate disease damage. 13 Exclusion criteria encompassed: cancer history, primary glomerulonephritis diseases or other infectious, chronic or autoimmune diseases.
Clinical Evaluation
All patients underwent a thorough medical history review and comprehensive musculoskeletal examination. Baseline laboratory results were obtained from the medical reports of the patients: erythrocyte sedimentation rate (ESR), blood urea nitrogen (BUN), serum creatinine, C-reactive protein (CRP), a complete blood count (CBC) and urinalysis.
Further tests included antinuclear antibody (ANA) and anti-dsDNA (using indirect immunofluorescence), serum complement levels (C3, C4), 24-hour protein in urine, serum creatinine, estimated glomerular filtration rate (eGFR) calculated with the Modification of Diet in Renal Disease (MDRD) equation, 14 antiphospholipid antibodies, and renal biopsy class (if available), according to the most recent classification provided by the Renal Pathology Society and the International Society of Nephrology. 15
The SLEDAI-2K
The SLEDAI-2K, a validated index assessing 24 laboratory and clinical parameters, measures SLE disease activity. Higher scores indicate increased disease activity, with specific cutoffs for flare-ups and remission. 12
The SLICC/ACR Damage Score
The SLICC/ACR Damage Score measures permanent organ damage to 12 organ systems, including cardiovascular, renal, musculoskeletal and neurological, offering a method to track long-term outcomes in SLE patients. 16
Biomarker Analysis
Each participant had six millilitres of venous blood drawn under strict aseptic conditions. The blood was then put in a sterile container with a clot activator and allowed to coagulate for half an hour. The samples were centrifuged for 20 minutes at 2000-3000 rpm. The separated serum was kept at −20°C for use in the α-enolase and IL-18 assays. Analysis of both markers was done using quantitative enzyme-linked immunosorbent assay (ELISA) kits that were both supplied by Bioassay Technology Laboratory, Nanhu dis, Zhejiang, China, according to the manufacturer’s instructions. 10% of the study sample participated in a pilot study to evaluate the method’s viability, applicability and clarity.
In principle, to deduce the concentration of both markers in the serum samples, standard curves using linear scales were constructed. Each standard’s mean absorbance was displayed on the y-axis, while the concentration was plotted on the x-axis. The graph’s points were used to build the best-fit curve. All recorded absorbances had zero standard absorbance deducted from them. The concentrations of the samples were read directly from this standard curve by using their optical density. The standard curve range for IL-18 was (0.5 ng/L-100 ng/L) and Sensitivity (0.2 ng/L). While the standard curve range for α-enolase was (0.2 ng/L-60ng/mL) and sensitivity was 0.102 ng/mL.
Statistical Methods
The Statistical Package of Social Sciences (SPSS) software was used for data analysis, with version 28 being used for thorough statistical analysis. Categorical variables were analysed through frequency and percentage reporting, with inter-group comparisons executed via Chi-square testing. Continuous numerical data were represented through mean values and standard deviation, with statistical comparisons across groups performed utilising the non-parametric Mann-Whitney test. Statistical significance was constituted at a predetermined threshold of P value < .05. To enhance diagnostic precision, Receiver Operating Characteristic (ROC) curve analysis was implemented to figure out optimal cut-off points for interleukin 18 and Anti-α-enolase as predictive markers for LN, facilitating more nuanced diagnostic potential and discriminatory capabilities in identifying disease states.
Results
This study comprised sixty patients with LN (30 having inactive LN and 30 having active LN) and 30 healthy controls. The gender distribution was predominantly female, with 66.7% females (Male: Female ratio = 1:2) in the active LN group and 93.3% females (Male: Female ratio = 1:14) in the inactive LN group. Individuals with active LN had an average disease duration of 4.533 ± 2.874 years, compared to 4.633 ± 2.953 years in those with inactive LN. Nearly all patients underwent renal biopsy. Detailed results of the renal biopsy, SLEDAI-2K score, renal SLEDAI score and SLICC damage score are provided in Table 1.
Clinical and Histopathological Features Between Studied Active and Inactive Lupus Nephritis Patients.
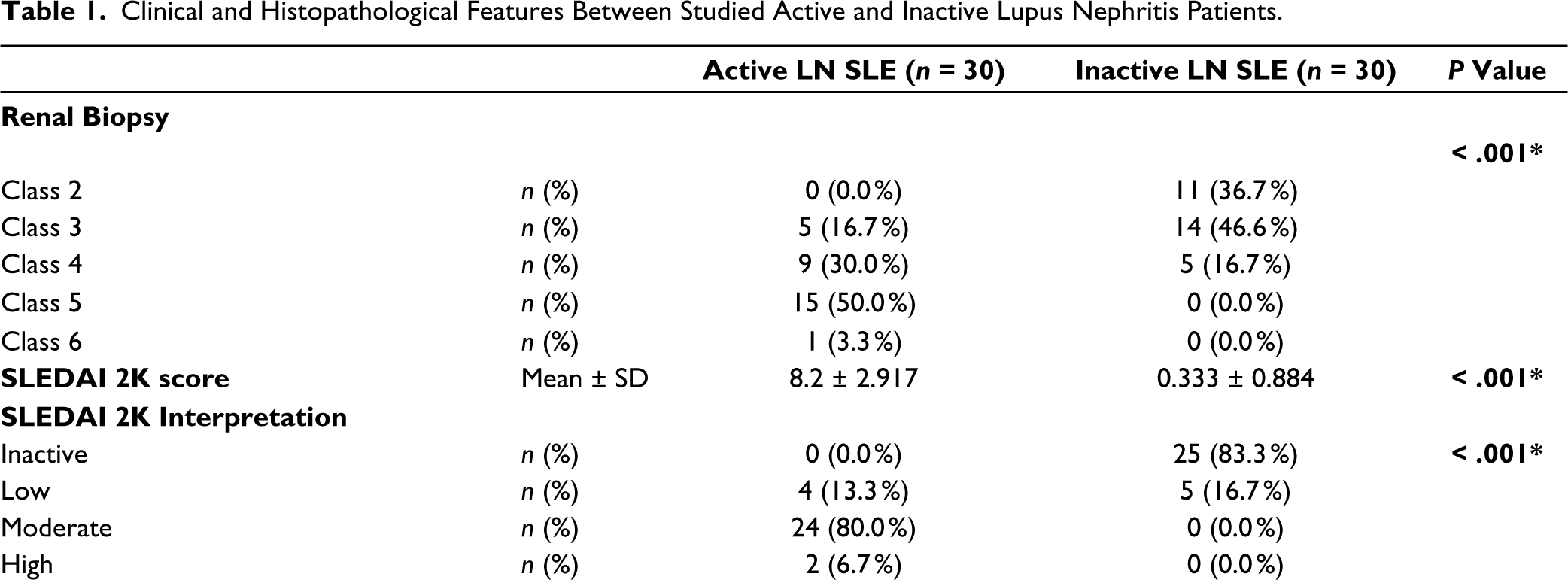
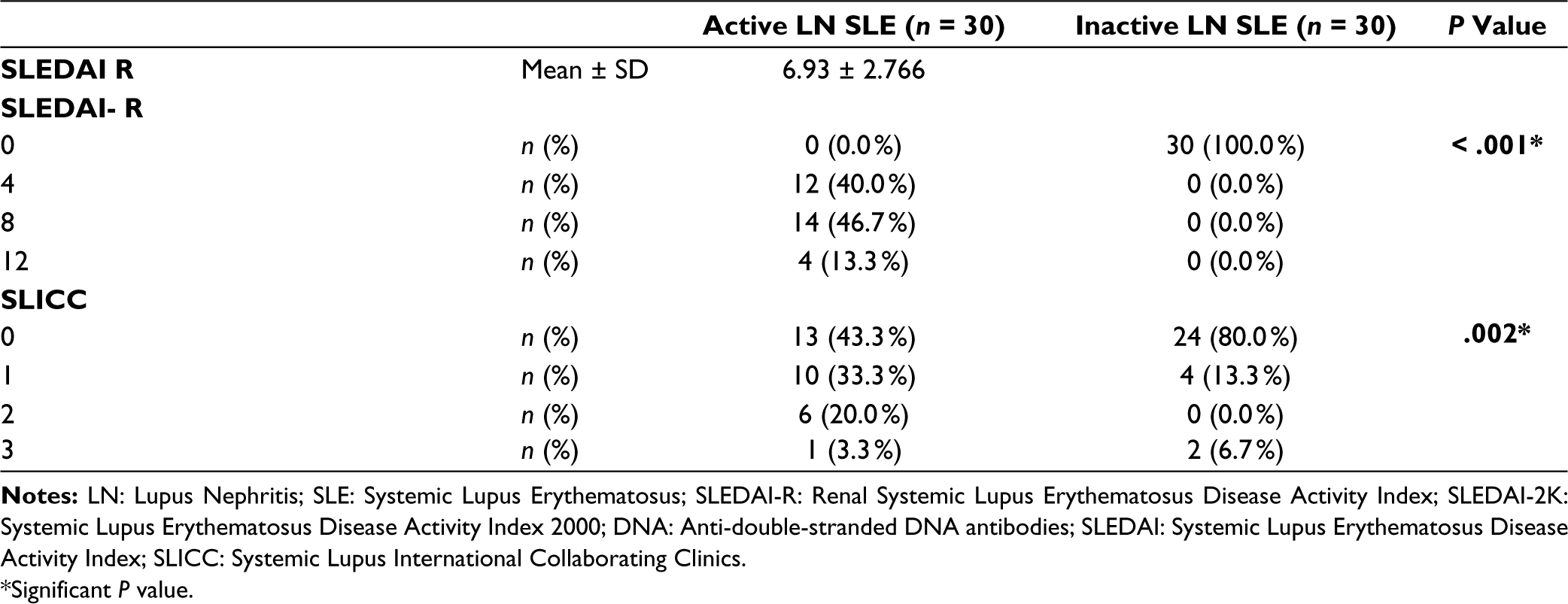
*Significant P value.
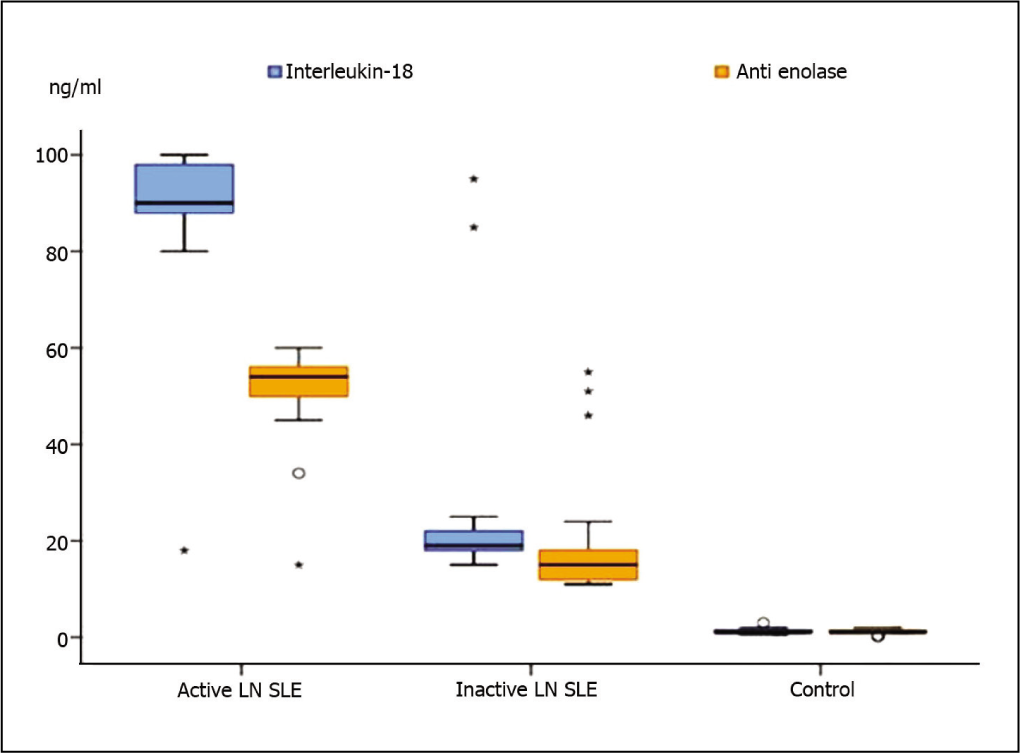
The mean IL-18 levels were significantly higher in the active LN group (89.97 ± 14.71 ng/mL) compared to the inactive LN group (24.03 ± 18.21 ng/mL) and the control group (1.217 ± 0.611 ng/mL), with a P value of < .001 (Figure 1). Likewise, anti-α-enolase levels were significantly elevated in the active LN group (51.67 ± 8.84 ng/mL) relative to the inactive LN group (18.53 ± 11.36 ng/mL) and the control group (1.153 ± 0.532 ng/mL), with a P value of < .01 (Figure 1).
Serum Levels of Both IL-18, Anti-α-enolase Levels Among the Studied Groups.
Notably, both IL-18 and anti-α-enolase show strong positive correlations with ESR (P < .001), anti-DNA titre (P < .001), SLEDAI 2K score (P < .001) and dose of corticosteroids (P < .001) (Table 2). Additionally, significant negative correlations are observed with complement levels, particularly C3 (P < .001) and C4 (P < .001). Furthermore, IL-18 and anti-α-enolase are significantly negatively correlated with eGFR (P < .001). Significant but moderate positive correlations are also seen with 24-h urinary proteins. Anti-α-enolase and CRP exhibited a significant positive correlation (P = .002), where there were no significant correlations observed with the other parameters (Table 2).
Performance Characteristics of Markers (IL-18, Anti-α-enolase) Between Patients with Active Lupus Nephritis Versus Inactive Lupus Nephritis.
Correlations Between Different Laboratory Markers, Disease Activity Scores, Disease Damage Scores and the Serum Levels of Both IL-18 and Anti-α-enolase Among the Two Studied Groups.
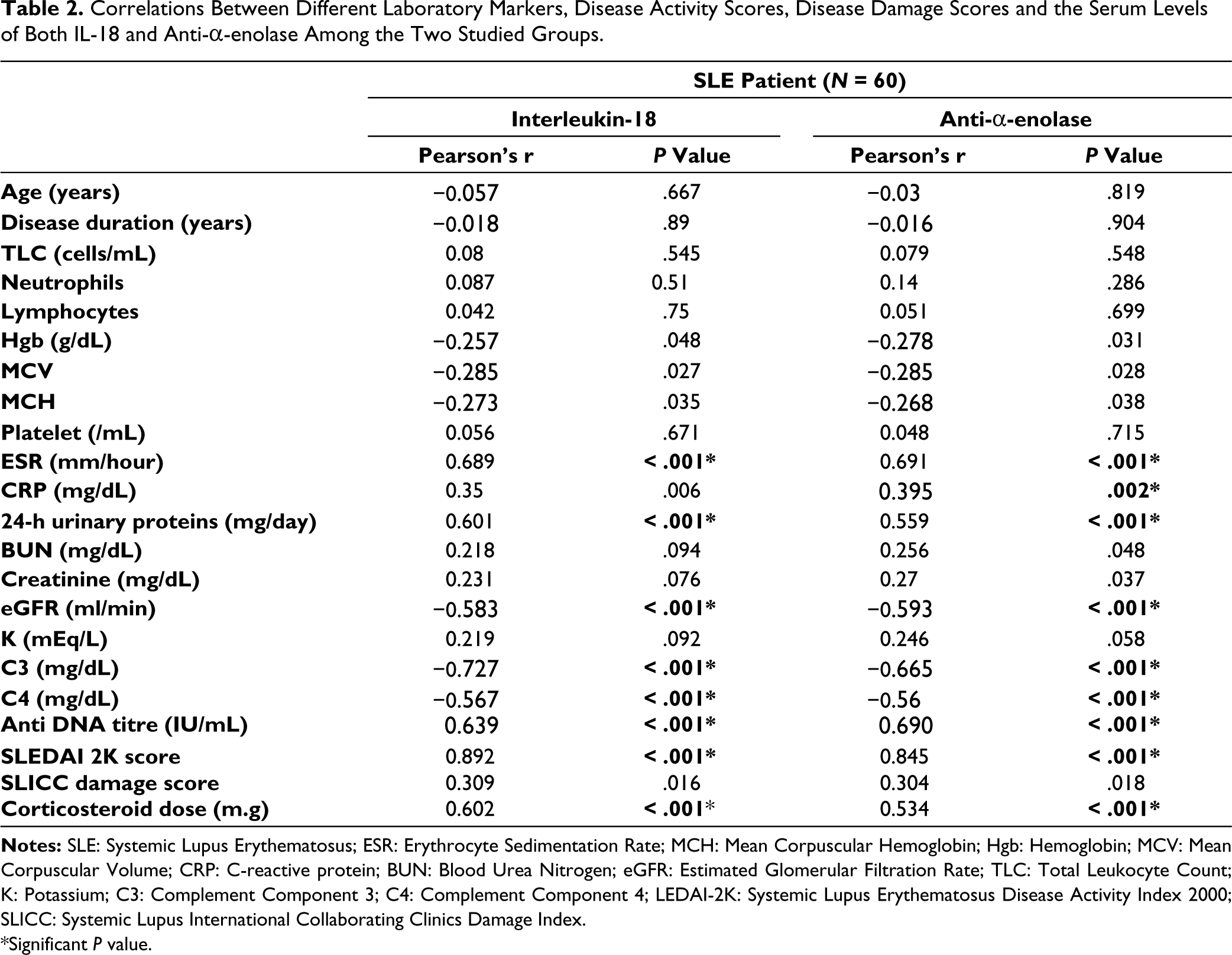
*Significant P value.
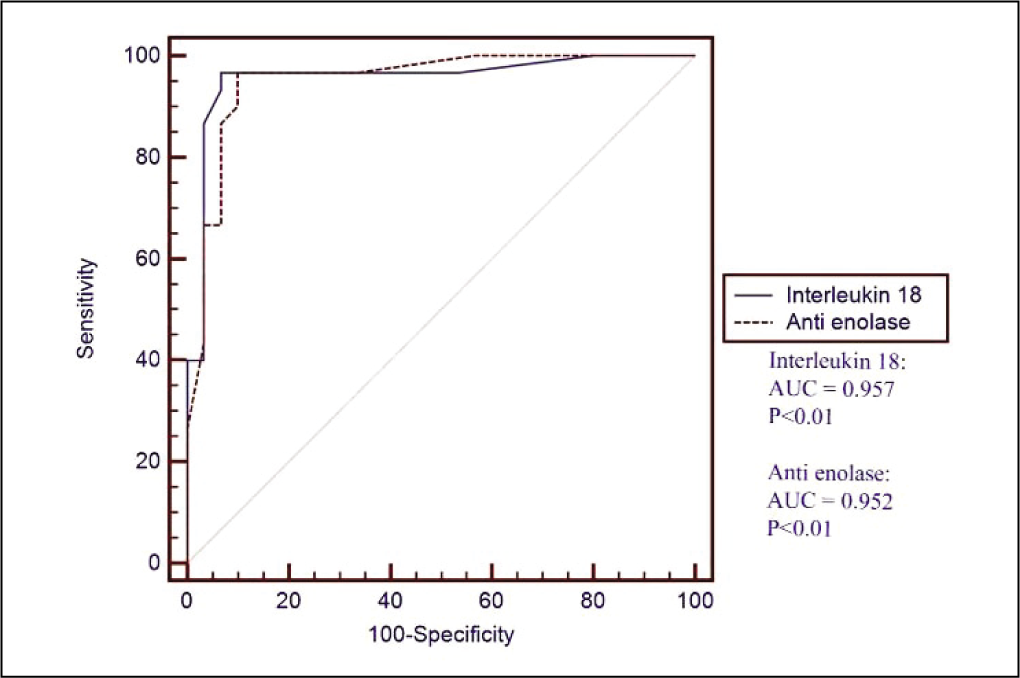
The performance characteristics for IL-18 and anti-α-enolase demonstrate high diagnostic accuracy in distinguishing active from inactive LN, with both markers achieving an AUC of 0.95, at their respective cutoff values (IL-18 > 25 ng/mL, Anti-α-enolase > 24 ng/mL) with a highly significant P value (< .01) (Table 3, Figure 2).
Performance Characteristics of Markers (IL-18, Anti-α-enolase) Between Those with Active Lupus Nephritis Versus Inactive Lupus Nephritis.
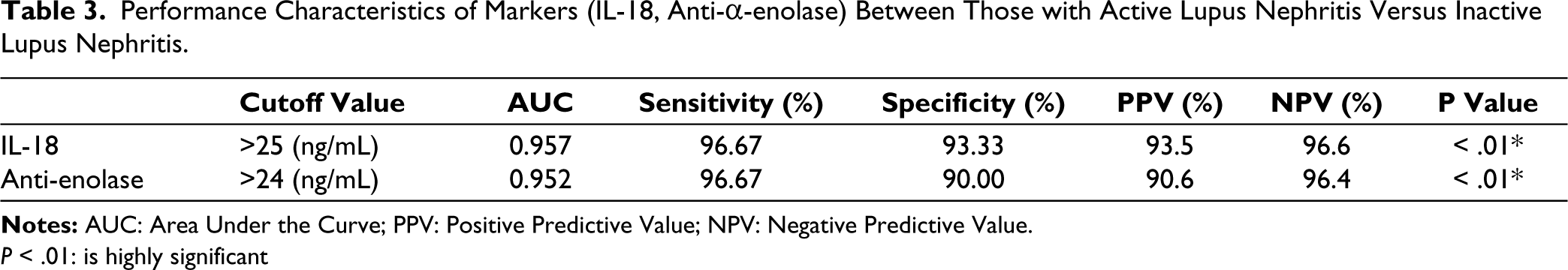
P < .01: is highly significant
Discussion
LN represents a significant complication in SLE, affecting a substantial portion of patients and contributing to increased morbidity and mortality. 17 There is a need for reliable biomarkers that can facilitate early detection, guide therapeutic decisions and monitor disease activity. 18 In this study, we evaluated the potential role of both IL-18 and anti-α-enolase antibodies as biomarkers for the activity of LN.
In the current work, the serum concentrations of both IL-18 and anti-α-enolase were found to be notably elevated in those experiencing active LN relative to those with inactive LN and healthy control subjects. These outcomes are in line with the prior work of Mende et al. 19 who substantiated that levels of serum IL-18 were elevated in SLE individuals, with particular prominence in cases involving active renal manifestations or irreversible organ impairment, thereby positioning IL-18 as a promising biomarker for active LN.
Complementing these observations, Faust et al. 20 provided additional corroborative evidence, documenting significantly enhanced levels of renal tubular epithelial cell-derived IL-18 among LN participants and importantly, establishing a correlative relationship between IL-18 elevation and the disease’s active state and severity.
As regard anti-α-enolase, Li et al. 21 in their study found significantly elevated serum levels of anti-α-enolase antibody in SLE patients compared to healthy controls, as in our study. Moreover, they demonstrated that patients with LN exhibited higher concentrations than those without renal manifestations. These observations were further confirmed by Huang et al. 22 who documented notably higher levels of anti-α-enolase among patients with LN compared to patients without nephritis.
In the present study, a strong positive correlation between serum levels of both IL-18, anti-α-enolase and ESR, SLEDAI-2K scores, anti-DNA titre was found, reflecting their potential as biomarkers for monitoring disease activity in LN. This finding aligns with the study by Mohsen et al. 23 who reported that in LN patients, IL-18 revealed a significant correlation with proteinuria, SLEDAI score, titre of anti-DNA, renal activity score and index of activity.
Also, in consistency with our results, when compared to normal control groups, Amerio et al. 24 discovered that among various cytokines, IL-18 was uniquely and strongly correlated with ‘active disease’, defined by a SLEDAI score exceeding eight points. Wu et al. 25 further investigated serum IL-18 as a potential biomarker for predicting long-term renal outcomes, revealing significant correlations between serum IL-18 and SLE global disease activity, along with the existence and severity of LN. Multivariate analysis suggested that serum IL-18 levels six months post-treatment represented the most unfavourable prognostic factor for clinical outcomes, irrespective of initial renal status.
Paralleling our results, Abou El-Fetouh et al. 26 also documented a highly significant positive correlation between elevated serum IL-18 levels and SLEDAI scores. A meta-analysis by Xiang et al. 27 further substantiated these findings, confirming that baseline serum IL-18 level variations related to SLEDAI-2K changes. In LN specifically, their research established significant correlations between serum IL-18 and SLEDAI, renal activity score and activity index. However, on the other hand the study done by El Bakry et al. 10 did not establish a correlation between Serum IL-18 and disease activity indices as SLEADI score this difference in the results could be attributed to that in their study non of their lupus patients had inactive disease and that most of the patients had moderate to severe disease activity.
With respect to disease damage, our study demonstrated no significant correlation between the SLICC damage scores and the serum level of IL-18 among the studied patients. This finding partially aligns with the study by Mende et al. 19 which reported a near-significant association between IL-18 and organ damage in a multivariable analysis.
The positive correlation of IL-18 and anti-α-enolase with 24-hour urinary proteins demonstrated in our study highlights their potential as non-invasive biomarkers for monitoring renal involvement, guiding treatment and predicting outcomes in LN. Consistent with our results, other studies such as Mohsen et al. 23 and Li et al. 21 found a positive correlation between serum anti-α-enolase antibody levels, serum IL-18 and serum total IgG, and 24-hour urine protein. On the other hand, Mosca et al. 28 reported that the levels of anti-α-enolase antibodies in their study did not correlate with any of the clinical or serological variables examined.
In the current work, significant negative correlations of both biomarkers and C3, C4 levels and eGFR were reported, indicating their potential links with renal function. This was validated by Mohsen et al. 23 as in their study only individuals with LN indicated a significant negative correlation between C3, C4 and serum IL-18; however, this finding partially agreed with another study by Abou El-Fetouh et al. 26 who reported a correlation between C3 and IL-18 however it does not reach a statistical significance.
Corticosteroids have the ability to reduce IL-18 production in healthy stimulated peripheral blood mononuclear cells. 29 In this study as regard the correlation between the daily dose of corticosteroids received by the patients and the levels of both IL-18 and anti-α-enolase, the current work revealed a statistically significant positive correlation. this finding goes with another study 30 who also found that the dose of corticosteroids correlated positively with serum levels of IL-18 and that patients with high disease activity had higher levels of IL-18 and had to receive higher doses of corticosteroids to control their disease activity, in the opposite to patients with low disease activity. However, the study done by Mohsen et al. 23 revealed no statistically significant correlation between serum level of IL-18 and daily dose of corticosteroids.
Finally, our study established a remarkable diagnostic accuracy for both IL-18 and anti-α-enolase, with 96.7% sensitivity and >90% specificity for active LN detection. However still few studies had been conducted on the role of anti α-enolase in LN in particular so we recommend further studies in this issue as there are ongoing studies on using anti-ENO antibodies as a potential novel approach in treatment of certain disorders as malignancies and may be autoimmune disorders in the future also In autoimmunity, therapeutic approaches that target IL-18 are being developed.
Despite the promising findings, several limitations exist in this investigation, one of the main ones being the small sample size, which could restrict the broader applicability of the findings, and the case-control design, while allowing for the identification of associations, it does not establish causality. Finally, to confirm the validity of these biomarkers and explore their potential therapeutic applications, more extensive, multi-centre cohorts and longitudinal follow-up are needed in future research.
Conclusions
Elevated serum concentrations of anti-α-enolase and IL-18 are significantly linked to active LN and demonstrate remarkable diagnostic accuracy in differentiating between active and inactive disease. Their significant correlations with scores of disease activity, renal function and complement levels highlight their potential as reliable biomarkers for detecting and monitoring renal disease activity in SLE.
Footnotes
Authors’ Contributions
There was equal participation from each author. The final manuscript was read and approved by all the authors.
Availability of Data and Materials
The corresponding author can provide the datasets utilised and/or analysed for this study upon request.
Consent for Publication
N/A
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approval
The Ethical Review Board of the Faculty of Medicine at Ain Shams University granted ethical approval for the study protocol, with the specific ethical clearance reference number FMASUR10/2023. All procedures for data collection were treated according to the Helsinki Declaration of Biomedical Ethics. All analyses were performed on de-identified and anonymised data.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Every participant signed a written informed consent form.
