Abstract
This review aims to provide a comprehensive analysis of filgotinib, a selective Janus kinase 1 (JAK1) inhibitor, and its potential applications in the treatment of rheumatoid arthritis (RA). It assesses filgotinib’s efficacy, safety, and role compared to traditional disease-modifying antirheumatic drugs (DMARDs) and other JAK inhibitors. Authors conducted a systematic search of PubMed, Scopus and Google Scholar to identify relevant studies, including phase I, II, and III clinical trials. We extracted and synthesised data on efficacy end points (ACR20, ACR50, and ACR70), safety profiles, and long-term outcomes. We performed comparative analyses with traditional DMARDs and other JAK inhibitors. Filgotinib demonstrates high efficacy in RA management, with ACR20 response rates ranging from 70% to 80%, ACR50 from 40% to 50%, and ACR70 from 20% to 30%. It significantly reduces disease activity (as measured by DAS28-CRP) and improves physical function (as indicated by Health Assessment Questionnaire Disability Index [HAQ-DI] scores). Compared to traditional DMARDs, filgotinib offers superior efficacy and a favourable safety profile. In comparison to other JAK inhibitors, filgotinib’s selective JAK1 inhibition contributes to a similar or improved safety profile while maintaining high efficacy. Long-term data confirm its sustained benefits and manageable safety profile. Filgotinib is a promising treatment option for RA, offering significant benefits. Clinical trials have observed improvements in disease activity and quality of life. Its selective JAK1 inhibition and favourable safety profile make it a competitive alternative to both traditional DMARDs and other JAK inhibitors. Ongoing research into personalised medicine and emerging therapies will further define filgotinib’s role in RA management.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease-causing chronic inflammation of synovial joints, leading to painful swelling, stiffness and joint deformity. 1 It is a progressive condition that can cause severe physical disability if not managed. The disease’s pathogenesis involves genetic, environmental and immunological factors. The immune system attacks healthy joint tissues, causing inflammation and the release of pro-inflammatory cytokines. 2 RA affects individuals of any age, with a higher prevalence in women. It is associated with cardiovascular disease, lung involvement and increased infection risk, which complicate its management. 3
Importance of Effective Management to Prevent Joint Damage and Disability
Effective management of RA is crucial to prevent irreversible joint damage and disability. The goal is to achieve and maintain clinical remission or low disease activity, improving physical function and patient quality of life. Early diagnosis and prompt initiation of disease-modifying antirheumatic drugs (DMARDs) are essential. 4 Conventional DMARDs like methotrexate (MTX) are essential, but some patients may not achieve adequate disease control or experience adverse effects. Janus kinase (JAK) inhibitors, like filgotinib, offer an alternative to traditional DMARDs providing additional options for disease control and preventing long-term joint damage.
Introduction to Janus Kinase Inhibitors
Janus kinase (JAK) inhibitors represent a novel class of targeted synthetic disease-modifying antirheumatic drugs (tsDMARDs). These agents interfere with the JAK-STAT signaling pathway, playing a critical role in modulating immune and inflammatory responses in rheumatoid arthritis.
Mechanism of Action of Janus Kinase Inhibitors
Janus kinase (JAK) inhibitors are synthetic pharmaceuticals that suppress the JAK-STAT signalling system. This is a crucial component of the immune response in RA. The JAK-STAT pathway is a crucial modulator of cytokine signalling, regulating immunological responses, haematopoiesis and inflammation. 5 . Four JAK enzymes phosphorylate specific STAT proteins, affecting gene expression in immune and inflammatory responses. Overactive JAK-STAT signalling in RA leads to pro-inflammatory cytokines, causing chronic inflammation. JAK inhibitors block this pathway, reducing inflammation and controlling the disease’s progression. These drugs are effective across multiple cytokines in RA, making them a potent option for patients (Figure 1). 6
Mechanism of Action of Janus Kinase (JAK) Inhibitors in Rheumatoid Arthritis.
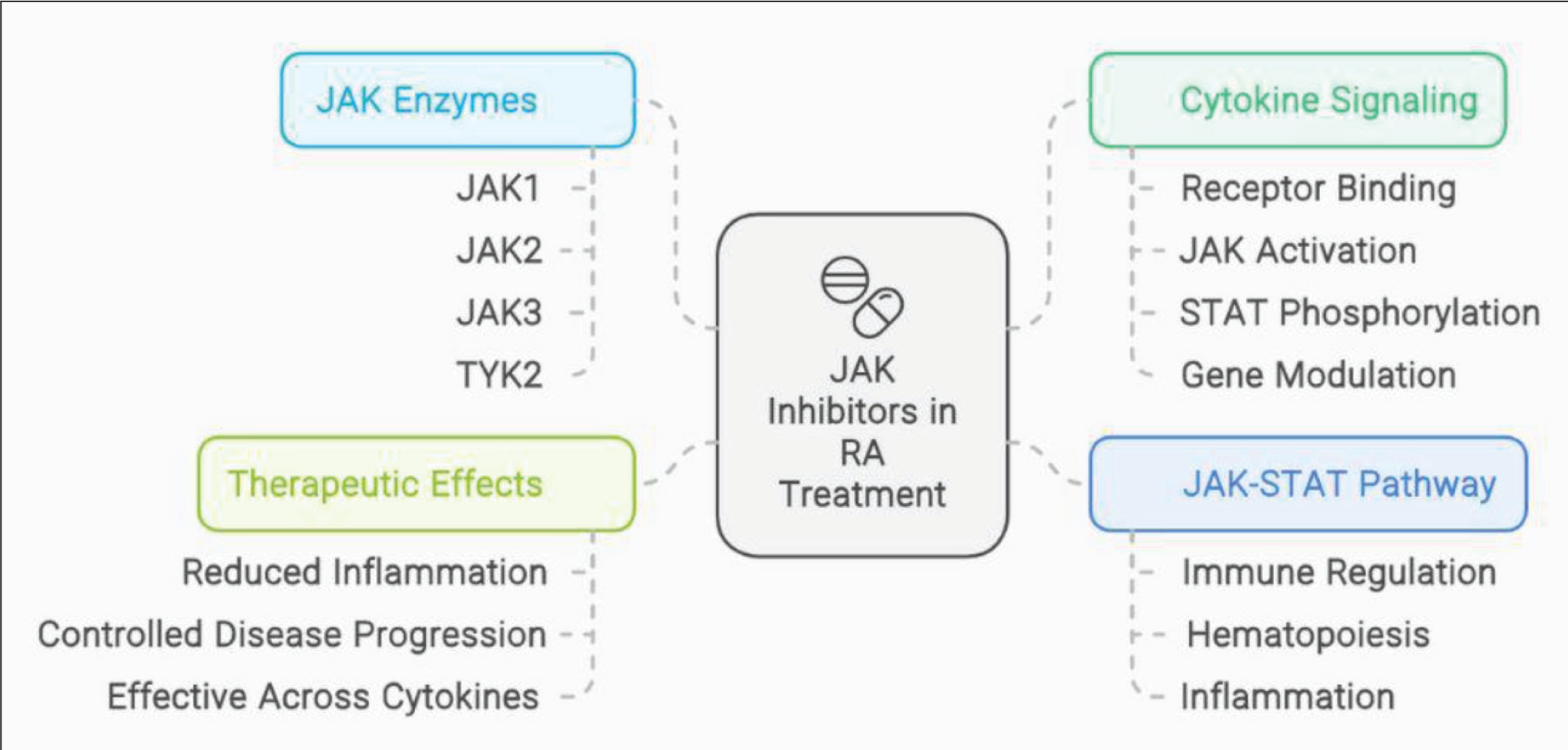
Overview of JAK Inhibitors Used in Rheumatoid Arthritis Treatment
JAK inhibitors represent a significant advancement in the treatment of RA, especially for patients who have an inadequate response to conventional DMARDs or biologic therapies. The following are the available treatment options or in development JAK inhibitors for the treatment of RA.
Objectives of the Review
The primary objective of this systematic review is to thoroughly evaluate and synthesise the current evidence on the efficacy and safety of filgotinib in the treatment of RA. The review aims to:
Assess the Efficacy of Filgotinib
Analyse the effectiveness of filgotinib in achieving key clinical outcomes in RA, such as:
Improvement in disease activity scores Response rates according to ACR20/50/70 criteria. Reduction in radiographic progression of joint damage. Enhancement of physical function and quality of life.
Evaluate the Safety Profile of Filgotinib
Investigate the incidence and severity of adverse events (AEs) associated with filgotinib, including:
Commonly reported side effects Serious adverse events (SAEs) Laboratory abnormalities
Compare Filgotinib with Other RA Treatments
Compare the efficacy and safety of filgotinib with other JAK inhibitors and biologic DMARDs.
Discuss filgotinib’s place in the treatment algorithm for RA, particularly in patients who have inadequate responses to conventional or biologic DMARDs.
Identify Patient Subgroups Benefiting from Filgotinib
Explore the efficacy and safety of filgotinib in specific patient populations, such as:
MTX-naive patients. Patients with prior exposure to biologics or other JAK inhibitors. Subgroups based on disease duration, severity, and comorbid conditions.
Highlight Gaps in Current Knowledge
Identify areas where further research is needed, including long-term safety studies and real-world effectiveness data.
Discuss the potential for filgotinib in combination therapy and its implications for personalised medicine in RA.
The ultimate goal of this review is to provide clinicians, researchers, and healthcare professionals with a detailed understanding of filgotinib’s role in RA treatment, guiding informed decision-making and optimising patient care.
Methodology
This review was conducted through a comprehensive literature search using databases such as PubMed, Scopus, and Google Scholar. Relevant clinical trials, review articles, and regulatory documents related to Filgotinib in rheumatoid arthritis were identified and critically analyzed (Figure 2).
Methodology Overview of the Systematic Review Process.
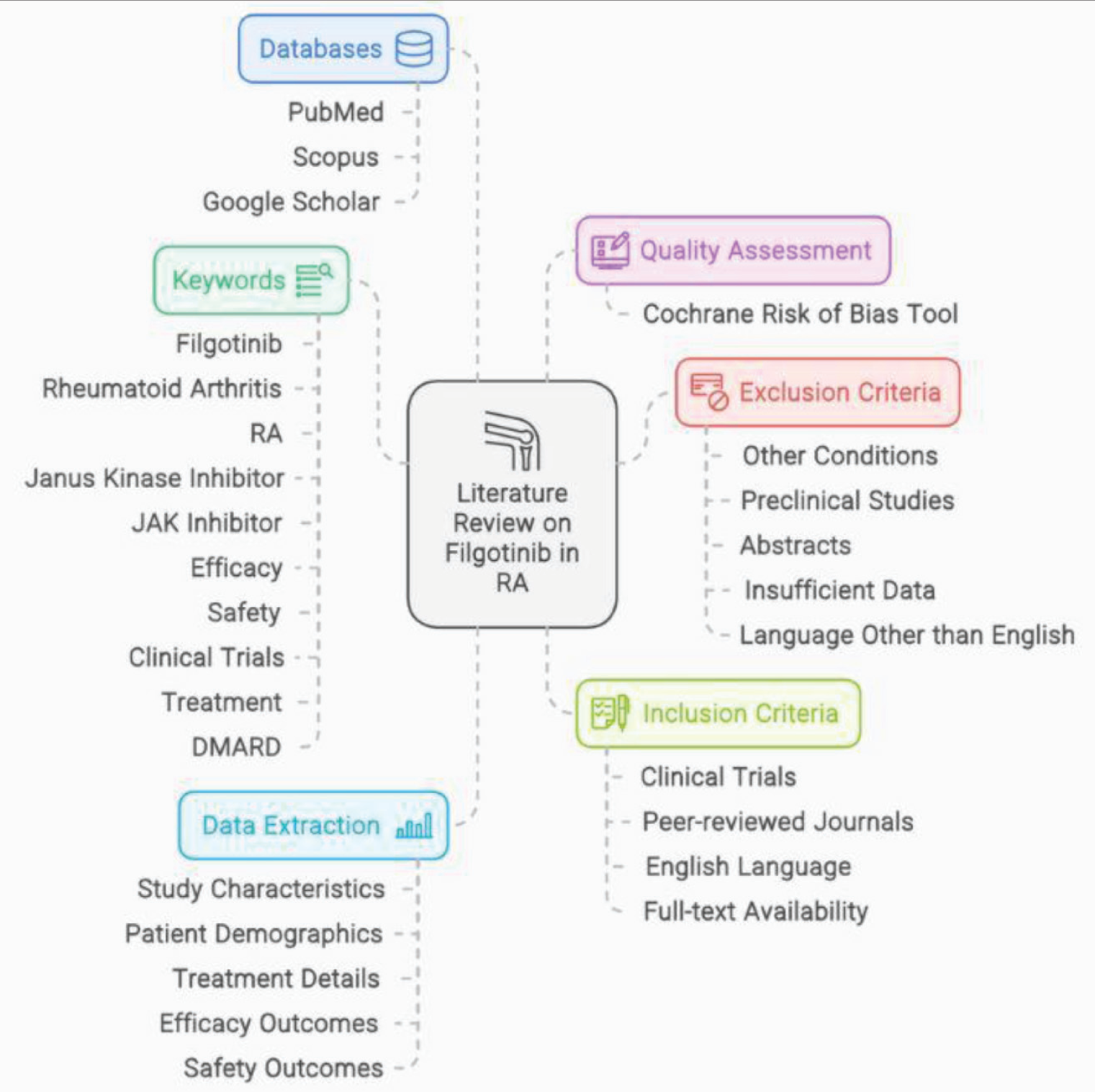
Search Strategy
We conducted the literature review using three databases: PubMed, Scopus, and Google Scholar. PubMed is a primary biomedical database for clinical trials and reviews relating to RA and pharmacological treatments. Scopus is a multidisciplinary database for peer-reviewed health sciences literature.
The search strategy used a combination of Medical Subject Headings (MeSH) terms and free-text keywords to capture relevant studies on filgotinib and its use in RA. It is intended that the following keywords and search phrases:
‘Filgotinib’, ‘Rheumatoid Arthritis’, ‘RA’, ‘Janus Kinase Inhibitor’, ‘JAK Inhibitor’, ‘Efficacy’, ‘Safety’, ‘Clinical Trials’, ‘Treatment’, ‘DMARD’
Inclusion and Exclusion Criteria
Studies were included if they focused on the efficacy, safety, or mechanism of action of Filgotinib in patients with rheumatoid arthritis. Articles not in English, case reports, and studies unrelated to Filgotinib or RA were excluded from the review.
The study selection process for this systematic review is outlined in Figure 3, which presents the PRISMA flow diagram illustrating the identification, screening, eligibility, and inclusion of studies evaluating Filgotinib in rheumatoid arthritis.
PRISMA Flow Diagram for the Selection of Studies in the Systematic Review of Filgotinib in Rheumatoid Arthritis.
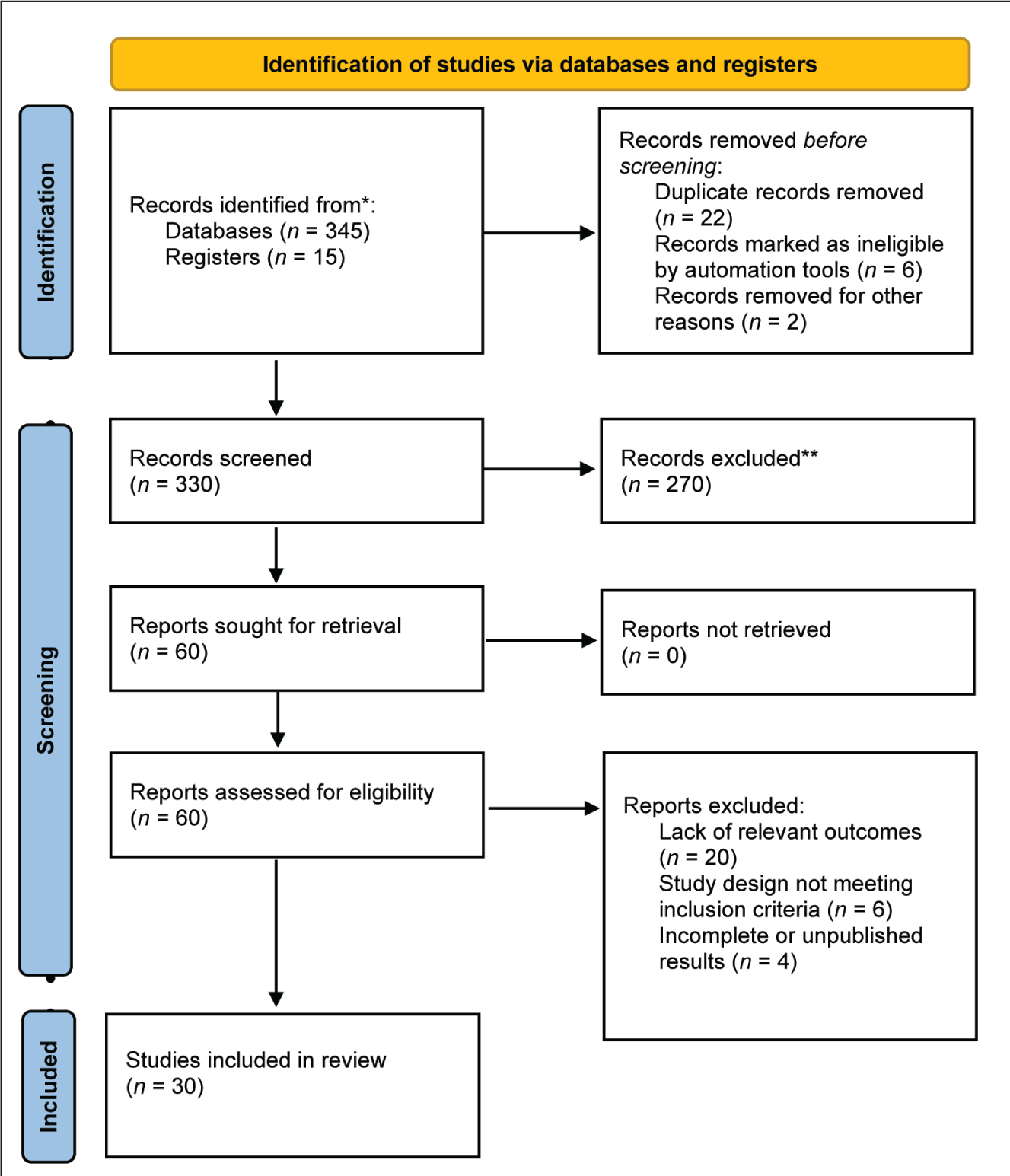
Types of studies included
Inclusion Criteria
Studies that assess the efficacy and/or safety of filgotinib in patients diagnosed with RA.
Studies published in peer-reviewed journals.
Articles published in English.
Studies with full-text availability.
Exclusion Criteria
Studies not focused on RA or that involve other conditions.
Preclinical studies (e.g., in vitro or animal studies).
Abstracts, letters, and editorials without substantial data.
Studies published in languages other than English.
Studies with insufficient data or those lacking proper methodological rigor.
Timeframe and Language Restrictions
The review focused on studies released between 2010 and present, which spans the development and study of JAK inhibitors, including filgotinib.
Data Extraction and Synthesis
Relevant data were systematically extracted from selected studies, including trial design, patient population, dosage, treatment duration, efficacy outcomes, and safety profiles. The findings were then synthesized to provide a comprehensive overview of Filgotinib's role in rheumatoid arthritis management.
Data Extraction
We collected relevant information from each included study using a standardised data extraction form. Key data points included:
Study characteristics (e.g., authors, publication year, study design, sample size). Patient demographics (e.g., age, gender, disease duration). Treatment details (e.g., dosage, duration of treatment, concomitant therapies). Efficacy outcomes (e.g., ACR response rates, DAS28 scores, radiographic progression). Safety outcomes (e.g., incidence of adverse events, SAEs, laboratory abnormalities).
Quality Assessment
We evaluated the quality of included studies using appropriate tools, such as the Cochrane Risk of Bias tool for RCTs. This assessment helps ensure the reliability of the findings.
Clinical Efficacy of Filgotinib
Clinical trials evaluating Filgotinib have demonstrated its significant efficacy in improving symptoms of rheumatoid arthritis. Key outcomes such as ACR response rates, disease activity scores, and patient-reported improvements support its therapeutic potential in diverse patient populations.
Table 1 summarizes the clinical efficacy outcomes of Filgotinib reported across Phase I, II, and III trials, highlighting key parameters such as study design, dosing regimen and key findings.
Summary of Clinical Efficacy of Filgotinib Across Phase I, II and III Trials.

Efficacy End Points
As shown in Table 2, Filgotinib demonstrated consistent efficacy across various clinical trials, with notable improvements in ACR20, ACR50, and ACR70 response rates, reflecting its potential in reducing disease activity in patients with rheumatoid arthritis.
Efficacy End Points of Filgotinib in Rheumatoid Arthritis (ACR20, ACR50, ACR70 Response Rates).
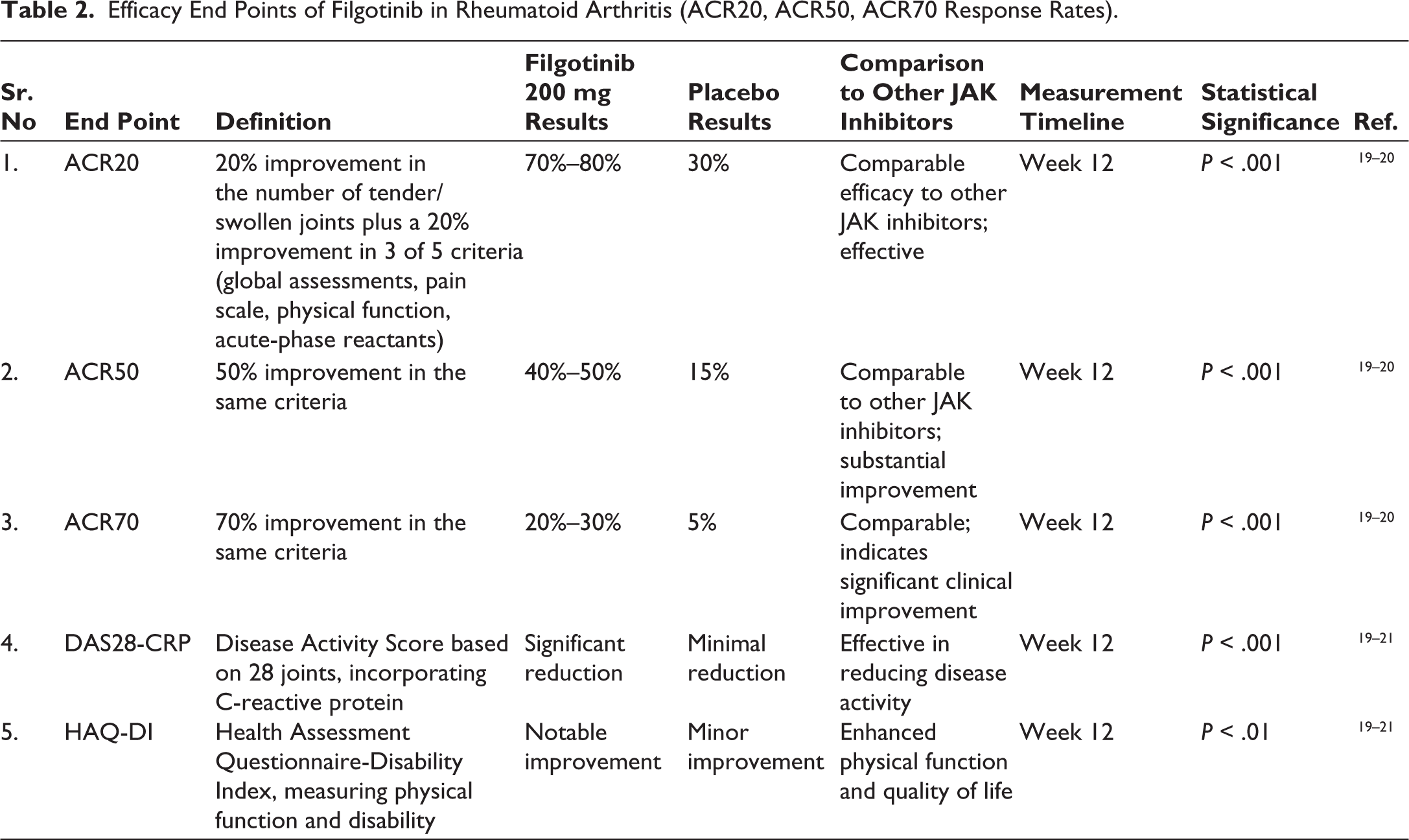
Comparative Efficacy
Comparative studies have assessed Filgotinib against other JAK inhibitors and conventional DMARDs to determine its relative effectiveness. These evaluations help position Filgotinib within the broader RA treatment landscape, highlighting its strengths and limitations.
Comparison with Traditional DMARDs
Filgotinib’s efficacy and safety profile have been compared with traditional disease-modifying antirheumatic drugs (DMARDs) like methotrexate. These comparisons offer valuable insights into its potential advantages in terms of symptom relief, disease progression control, and patient tolerability.
Table 3 presents a comparative analysis of Filgotinib versus traditional disease-modifying antirheumatic drugs (DMARDs), highlighting its superior or comparable efficacy in improving clinical outcomes among patients with rheumatoid arthritis.
Summary
Comparative Efficacy of Filgotinib Versus Traditional DMARDs in Rheumatoid Arthritis.
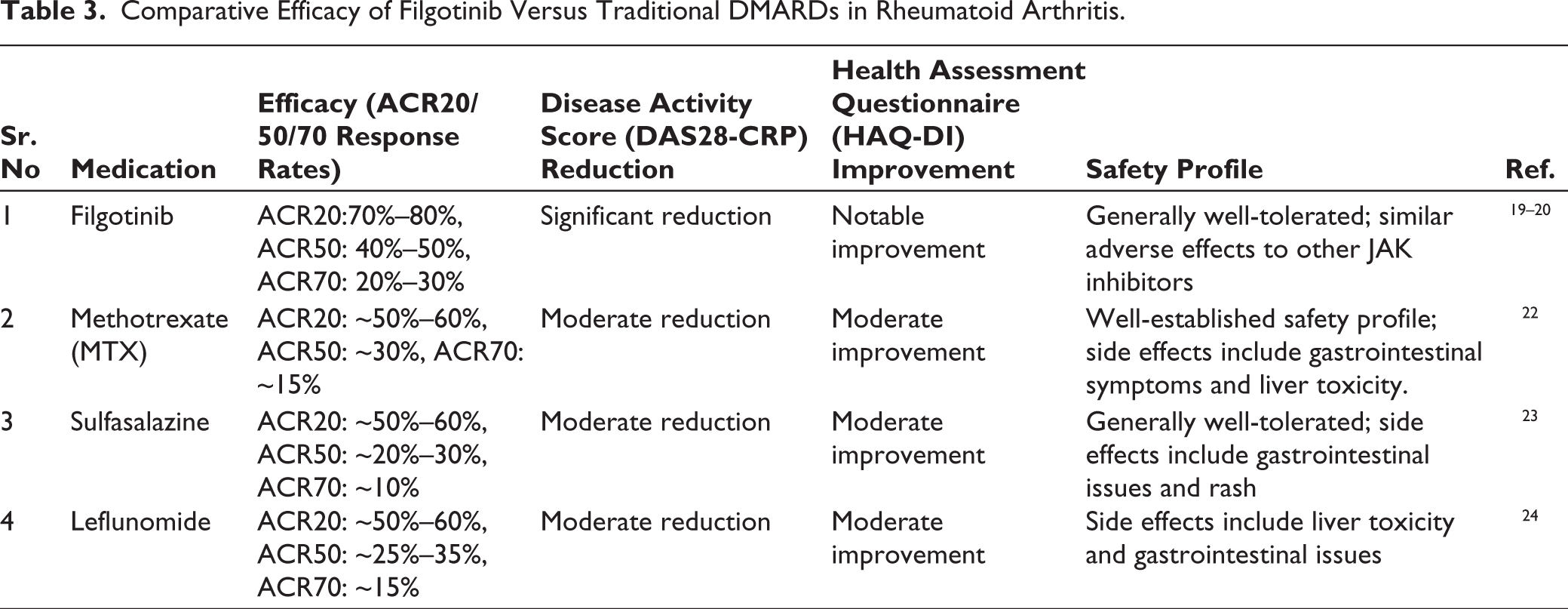
Filgotinib demonstrated ACR70 response rates of 20%-30%, resulting in a 70% improvement.19–20
Filgotinib monotherapy did not have a superior ACR20 response rate versus MTX. Filgotinib was well tolerated, with acceptable safety compared with MTX. 18
Comparison with Other JAK Inhibitors
Filgotinib has been evaluated alongside other JAK inhibitors such as tofacitinib, baricitinib, and upadacitinib. These comparisons help to highlight differences in selectivity, clinical efficacy, safety profiles, and tolerability, which are essential for guiding treatment decisions in rheumatoid arthritis.
Table 4 summarizes the comparative efficacy of Filgotinib against other Janus kinase (JAK) inhibitors, illustrating differences in clinical response rates, remission outcomes, and safety profiles across key phase II and III trials in rheumatoid arthritis.
Comparative Efficacy of Filgotinib Versus Other JAK Inhibitors in Rheumatoid Arthritis.

Summary
Studies have demonstrated that
Overall Comparison
Serious Infections and Outcomes with Filgotinib
The risk of serious infections is a critical consideration with JAK inhibitors, including Filgotinib. Clinical trials and long-term studies have evaluated the incidence, types, and outcomes of serious infections associated with Filgotinib use, providing important safety insights for clinical practice.
Comparison with Tofacitinib and Other JAK Inhibitors
Comparative studies between Filgotinib and other JAK inhibitors, such as Tofacitinib, have highlighted differences in efficacy, safety profiles, selectivity for JAK isoforms, and patient-reported outcomes. These comparisons are essential for informed clinical decision-making and personalized treatment approaches in rheumatoid arthritis management.
Long-term Efficacy
Table 5 presents the long-term treatment outcomes of Filgotinib in patients with rheumatoid arthritis, highlighting study design, population, key finding and patient-reported outcomes over extended follow-up periods in clinical trials.
Long-Term Treatment Outcomes of Filgotinib in Rheumatoid Arthritis.

Long-term efficacy is crucial in understanding the sustainability of a drug’s benefits over time, especially in chronic conditions like RA. Long-term extension studies have evaluated filgotinib, a selective JAK1 inhibitor, to assess its prolonged effectiveness and safety. 29
Safety and Tolerability
The safety and tolerability profile of Filgotinib has been extensively evaluated in clinical trials. This section summarizes common adverse events, serious risks, and the overall benefit-risk assessment associated with its use in rheumatoid arthritis.
Table 6 summarizes the safety and tolerability profile of Filgotinib in rheumatoid arthritis, including the incidence of adverse events, serious infections, and laboratory abnormalities observed across clinical studies.
Safety and Tolerability Profile of Filgotinib in Rheumatoid Arthritis.

Types of Malignancies Linked to Filgotinib
The overall malignancy risk with filgotinib appears to be relatively low. Long-term safety data from studies such as DARWIN-1–3 and FINCH-1 have reported a malignancy rate of approximately 1.5%-1.8%. Research indicates that people with RA exhibit an elevated incidence of malignancies, particularly among those on immunosuppressive treatment, in comparison to the general population. The trial recruited 739 of the 790 patients who completed the phase II parent investigations. Out of the 13 people who had cancer that got worse without treatment, four had blood cancer (3 non-Hodgkin lymphoma and 1 diffuse large B-cell lymphoma) and nine had solid tumours (2 lung cancers, 2 breast cancers, and 1 each of colon cancer, gallbladder adenocarcinoma, metastatic leiomyosarcoma, melanoma and kidney cancer). 31
Clinical Applications
Filgotinib has demonstrated significant therapeutic potential in the management of moderate-to-severe rheumatoid arthritis. This section discusses its approved indications, recommended usage, and its role in clinical practice based on current evidence.
Patient Populations
Filgotinib is effective in treating RA in both younger and older patients, with consistent efficacy across age groups. Its safety profile is similar to younger populations, making it a viable option for all age groups.
Clinical trials have evaluated filgotinib across a broad range of age groups, primarily focusing on adults with moderate to severe RA. Most of the studies have included patients aged 18–75 years, with older populations (≥65 years) being a key subgroup of interest given the age-related risks for RA and the potential for increased susceptibility to adverse events in older adults.
While the drug has shown efficacy in both younger and older adults, post-hoc analyses of clinical trials, including the FINCH 1, 2 and 3 studies, suggest that filgotinib is particularly well-tolerated in older patients, showing a favourable safety profile compared to other JAK inhibitors. Importantly, older patients did not exhibit a higher incidence of SAEs compared to younger patients, which is a significant consideration given the increased risks for infections or other complications in elderly populations. However, further real-world data on long-term safety and efficacy, particularly in patients over 75 years old, would be valuable. 29
Combination Therapy
Researchers have thoroughly investigated filgotinib in conjunction with MTX for individuals exhibiting insufficient response to MTX alone. The combined treatment has demonstrated increased effectiveness, enhancing ACR response rates, DAS28-CRP and physical function. It possesses a well-accepted safety profile. 18
Researchers have also studied the safe combination of filgotinib with other DMARDs like sulfasalazine and leflunomide for additional disease control benefits. This combination may offer an effective treatment strategy for patients. 38
Monotherapy
Filgotinib is a potent monotherapy for RA patients, particularly those with MTX insensitivity or contraindications. 18 Clinical trials show significant improvements in ACR20, ACR50 and ACR70 response rates, with the 200 mg dose being particularly effective.17–18
Comparative Analysis
A thorough comparative analysis of Filgotinib with other conventional and novel therapies provides valuable insights into its relative efficacy, safety, and clinical utility. This section highlights key differences and similarities with alternative treatment options.
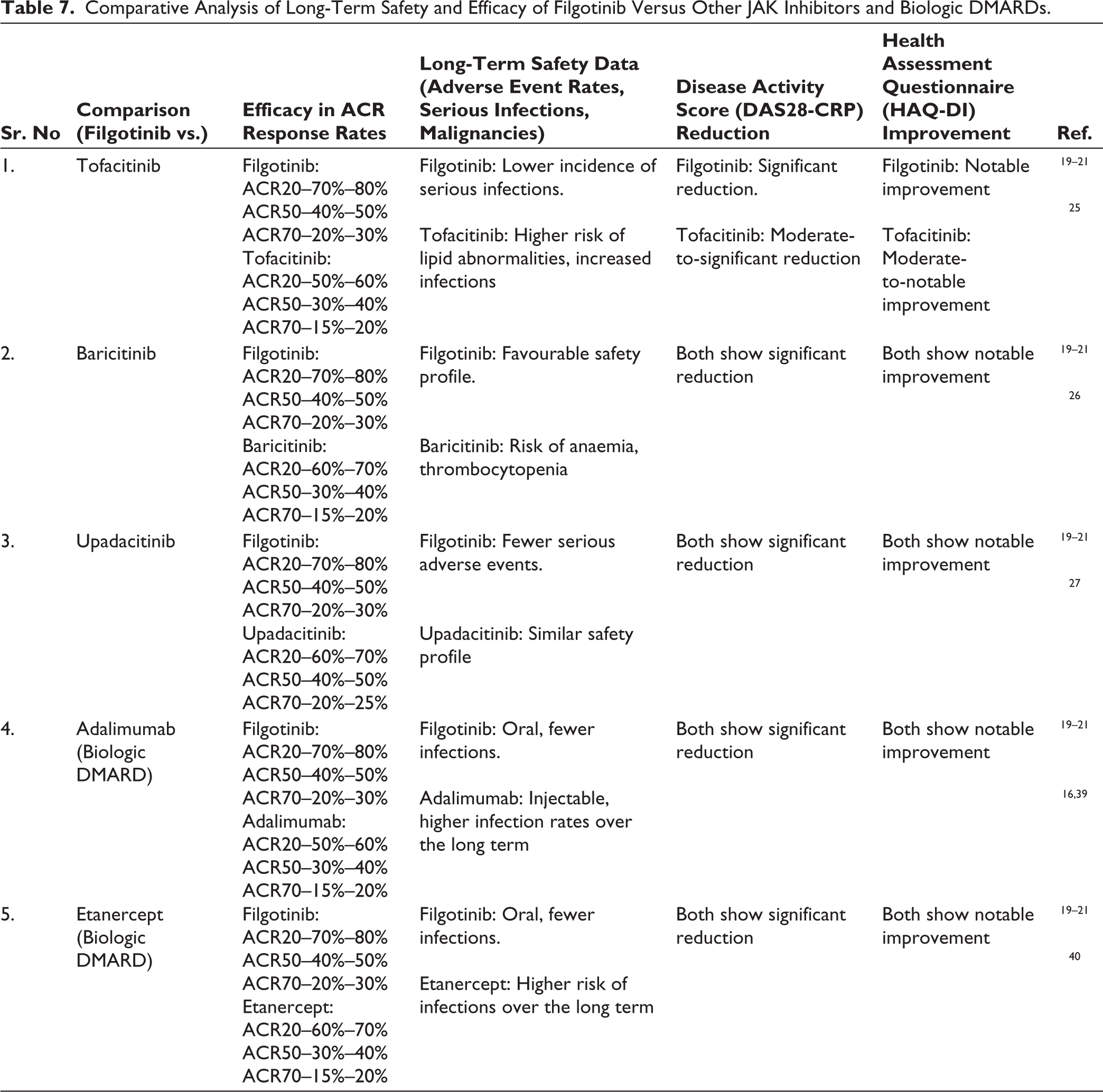
Table 7 presents a comparative analysis of the long-term safety and efficacy profiles of Filgotinib versus other JAK inhibitors and biologic DMARDs, highlighting key differences in clinical outcomes and adverse event rates reported in extended follow-up studies.
Comparative Analysis: Filgotinib versus Other JAK Inhibitors and Biologic DMARDs
Comparative Analysis of Long-Term Safety and Efficacy of Filgotinib Versus Other JAK Inhibitors and Biologic DMARDs.
Economic Considerations
This section evaluates the economic implications of Filgotinib therapy in the management of rheumatoid arthritis. It includes an analysis of cost-effectiveness, direct and indirect healthcare expenditures, and comparative pricing relative to other JAK inhibitors and biologic DMARDs, providing insights into its value proposition in clinical practice.
Cost-effectiveness
The cost-effectiveness of Filgotinib is assessed by examining its clinical benefits relative to its financial burden on healthcare systems. This includes evaluating quality-adjusted life years (QALYs), healthcare resource utilization, and treatment affordability compared to alternative JAK inhibitors and conventional DMARDs.
Direct Cost
Filgotinib’s procurement cost is comparable to other JAK inhibitors, but its oral administration method may reduce healthcare costs due to infusion procedures. Its dose schedule allows for more flexible patient care, potentially reducing hospital visits.
Indirect Cost
Filgotinib’s effectiveness in alleviating pain and inflammation can markedly enhance patients’ quality of life, perhaps resulting in heightened productivity and decreased job absenteeism. This enhancement can yield significant economic advantages for both patients and companies. Moreover, proficient management of RA symptoms might mitigate the likelihood of comorbidities, thereby reducing long-term healthcare expenditures.
Comparative Effectiveness
Economic evaluations comparing filgotinib with other JAK inhibitors have shown that it may provide superior health outcomes at a comparable or lower cost. Studies indicate that filgotinib can lead to greater improvements in quality-adjusted life years (QALYs) due to its favourable safety profile and effectiveness, making it a cost-effective choice in RA management. 41
Pharmacoeconomic models that incorporate data from clinical trials and real-world studies have generally concluded that filgotinib is a cost-effective treatment for RA. 41 These models often demonstrate that, despite its initial cost, the long-term benefits in terms of improved health outcomes and reduced healthcare utilisation justify its use.
Healthcare Utilisation
Filgotinib’s effectiveness in managing RA may lead to reduced healthcare utilisation compared to traditional DMARDs and other JAK inhibitors. Patients on filgotinib experience fewer disease flares and hospitalisations, resulting in lower healthcare costs. Studies suggest that filgotinib can contribute to significant cost savings by reducing the need for additional medications and interventions. It also improves patient quality of life, leading to enhanced physical function and reduced disease symptoms, potentially reducing indirect costs.
Future Directions
Future research on Filgotinib should focus on long-term safety outcomes, head-to-head comparative trials with other JAK inhibitors, and its effectiveness in diverse patient subpopulations. Additionally, studies exploring biomarkers for treatment response and real-world cost-benefit analyses will be crucial to optimizing its clinical utility.
Ongoing Research
Current studies include extended trials to investigate the long-term safety and effectiveness of filgotinib, its impact on comorbid conditions and real-world patient populations. Researchers are investigating combination therapies with other RA treatments to evaluate synergistic effects and optimise treatment regimens. To make filgotinib a better RA treatment, researchers are studying how it works by looking at how it affects certain immune pathways and how it interacts with other signalling molecules. 42
Research is exploring alternative formulations of filgotinib to improve patient adherence and ease of use, such as extended-release formulations or combination therapies. Additionally, studies are assessing its potential for treating other autoimmune diseases beyond RA.
Personalised Medicine
Progress in genetics and biomarker research is facilitating personalised therapy strategies for RA, enhancing results and reducing unwanted effects. 43 Researchers are formulating prediction models utilising genetic and clinical data to enhance therapy and dose optimisation. The clinical integration of personalised medicine necessitates guidelines and tools to assist physicians in making educated decisions based on genetic and biomarker data. 44 .
Innovative Therapies
Researchers are developing new JAK inhibitors with improved selectivity and safety profiles, which could potentially offer enhanced efficacy and reduced side effects compared to current options like filgotinib. Cell-based therapies, like CAR T-cell therapy and stem cell therapy, offer potential disease-modifying benefits for RA patients. 45
Potential Impact
Innovative therapies and personalised medicine approaches could improve disease management, leading to better long-term outcomes, reduced disability and improved quality of life for RA patients. These changes could shift treatment paradigms, offering more targeted and effective options.
Discussion
Filgotinib is a viable therapeutic option for individuals with RA, especially for those who have not sufficiently reacted to conventional DMARDs. As a second-line treatment after DMARDs, filgotinib offers unique benefits due to its specific inhibition of Janus kinase 1 (JAK1), setting it apart from conventional DMARDs and other JAK inhibitors.
Efficacy and response rates in clinical investigations have shown that filgotinib attains superior response rates and enhanced physical function relative to conventional DMARDs and non-selective JAK inhibitors. The medicine has demonstrated notable enhancements in ACR response rates and decreases in disease activity, rendering it a viable option for individuals necessitating more intensive treatment approaches.
Dosing guidelines recommend administering 200 mg of filgotinib once a day. Individual patient response and tolerability may modify this regimen, facilitating tailored treatment programmes that account for patient-specific characteristics and illness severity.
Filgotinib’s specific mode of action enhances its favourable safety profile. It is less likely for filgotinib to cause side effects like hepatotoxicity, dyslipidaemia, cytopenias, thromboembolic events and infections than other JAK inhibitors. This advantageous safety profile increases its appeal as a therapy alternative, especially for those susceptible to severe problems.
Particular advantages of filgotinib surpass its effectiveness and safety. Selective inhibition of JAK1 not only controls disease activity but also lowers the risk of serious side effects that are often linked to non-selective JAK inhibitors. Furthermore, filgotinib may offer potential steroid-sparing benefits, a crucial benefit for individuals requiring prolonged corticosteroid treatment.
Customised therapeutic approaches in the future for RA care may incorporate more individualised treatment strategies, including genetic indicators to forecast responses to filgotinib. Further research is necessary to identify specific indicators that could inform treatment decisions and improve outcomes for individual patients.
Despite the promising data on filgotinib, gaps remain in long-term efficacy and safety data. More research required for head-to-head trials to determine its comparative effectiveness against other JAK inhibitors. Ongoing research into next-generation JAK inhibitors and emerging therapies will further clarify filgotinib’s role in RA management and potentially expand its applications.
Conclusion
Filgotinib is a highly effective treatment for RA with high ACR response rates, substantial reductions in disease activity and improvements in physical function. It offers superior efficacy and a favourable safety profile compared to traditional DMARDs. Filgotinib’s selective JAK1 inhibition reduces side effects, making it a strong candidate for RA management. Its safety profile is comparable to or better than other JAK inhibitors, but ongoing monitoring is essential for long-term adverse effects. Filgotinib’s high efficacy and potential reduction in healthcare utilisation and costs support its cost-effectiveness, despite its higher cost.
It looks like filgotinib could be a big step forward in treating RA. It has a lot of advantages over older DMARDs and works just as well as other JAK inhibitors. As research continues, it will play a crucial role in future RA management strategies. Tailoring personalised medicine based on genetic and biomarker information could enhance filgotinib’s efficacy and optimise patient outcomes. Additionally, research into innovative therapies and next-generation treatments will complement filgotinib’s role in RA management, providing clinicians with a broader range of treatment options.
List of Abbreviations
RA: Rheumatoid arthritis
JAK: Janus kinase
DMARDs: Disease-modifying antirheumatic drugs
ACR: American College of Rheumatology
DAS28-CRP: Disease Activity Score in 28 Joints - C-Reactive Protein
HAQ-DI: Health Assessment Questionnaire-Disability Index
MTX:: Methotrexate
SAE: Serious adverse events
bDMARDs: Biologic disease-modifying antirheumatic drugs
csDMARDs: Conventional synthetic disease-modifying antirheumatic drugs
RCT: Randomised controlled trial
FDA: Food and Drug Administration
EMA: European Medicines Agency
PK: Pharmacokinetics
Footnotes
Acknowledgements
The author expresses sincere gratitude to Krescent Medical Research Pvt. Ltd. for their invaluable guidance and support throughout the preparation of this manuscript.
Authors’ Contribution
All authors contributed substantially to the conceptualization, drafting, and critical revision of this manuscript.
Dr. Krishnaa S. Upadhye: Conceptualized the study, defined the design and intellectual content, conducted comprehensive literature searches, supervised clinical and experimental aspects, acquired relevant data, and took the lead in manuscript preparation, editing, and review. Dr. Upadhye also served as the guarantor for the integrity and accuracy of the work.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
Not applicable as no new data were generated or analyzed for this review article.
Declaration of Conflicting Interests
Dr. Krishnaa S. Upadhye is the Director of Clinical Operations at Krescent Medical Research Pvt. Ltd. This affiliation has been disclosed to the journal and the publisher. Krescent Medical Research Pvt. Ltd. had no role in the conceptualization, writing, or approval of this manuscript, and no funding was received from the company for this work. The views expressed in this review article are solely those of the author and do not necessarily reflect the views of Krescent Medical Research Pvt. Ltd.
Ethical Considerations
Not applicable.
Funding Statement
The authors have not declared a specific grant for this research from any funding agency in the public, commercial, or not-for-profit sectors.
