Abstract
Background:
Patients with connective tissue disease-associated interstitial lung disease (CTD-ILD) frequently experience sleep-related disorders like insomnia, obstructive sleep apnoea (OSA), and restless leg syndrome (RLS). Poor sleep quality in ILD significantly impacts health-related quality of life, leading to increased morbidity and mortality.
Methodology:
A cross-sectional, questionnaire-based study was conducted at a tertiary care centre in Eastern India. Consecutive cases of CTD-ILD and age-matched controls were recruited and were administered a Sleep Questionnaire that included questions related to insomnia, RLS, parasomnias, Epworth Sleepiness Scale (ESS), Berlin, STOP-BANG questionnaires.
Results:
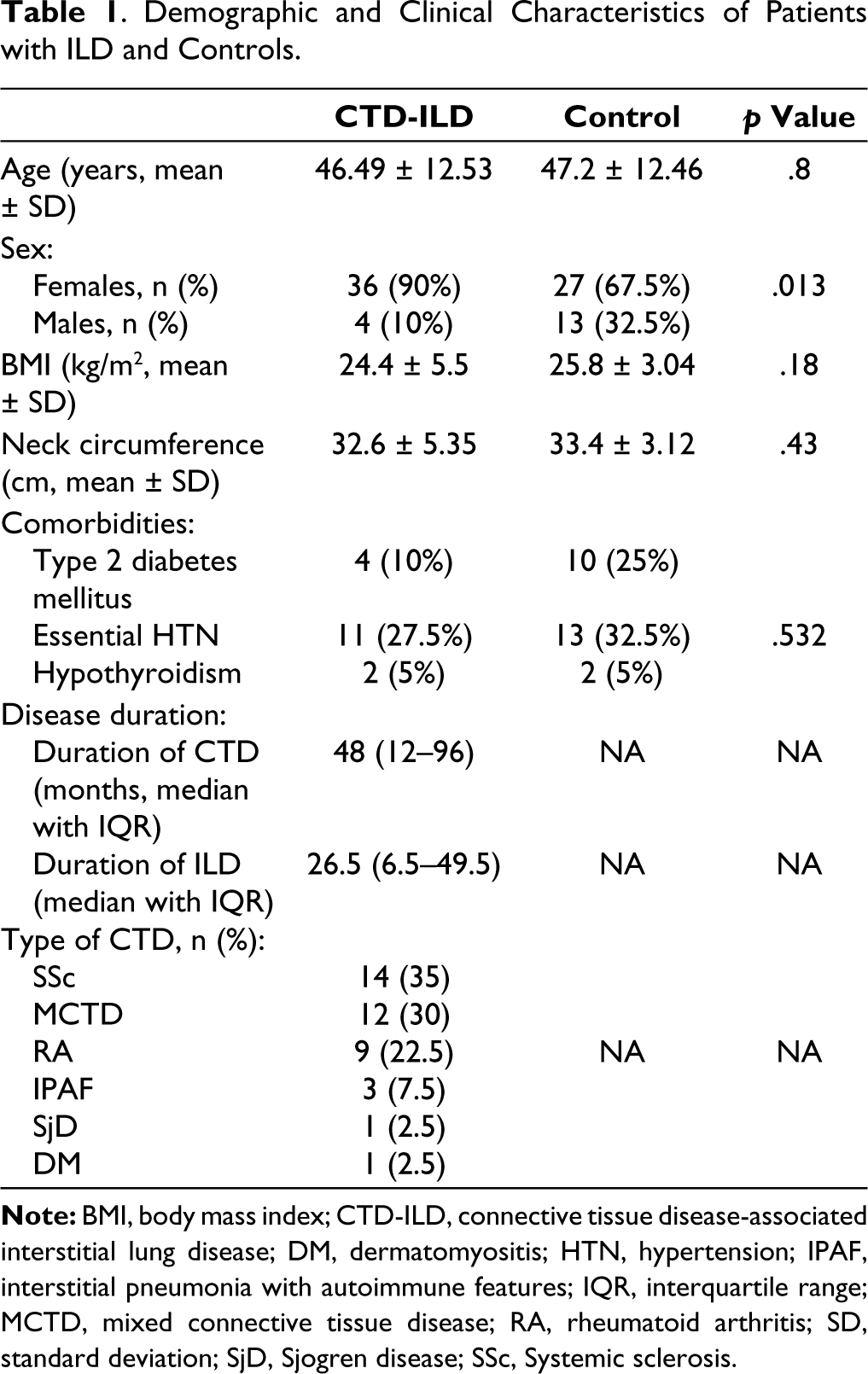
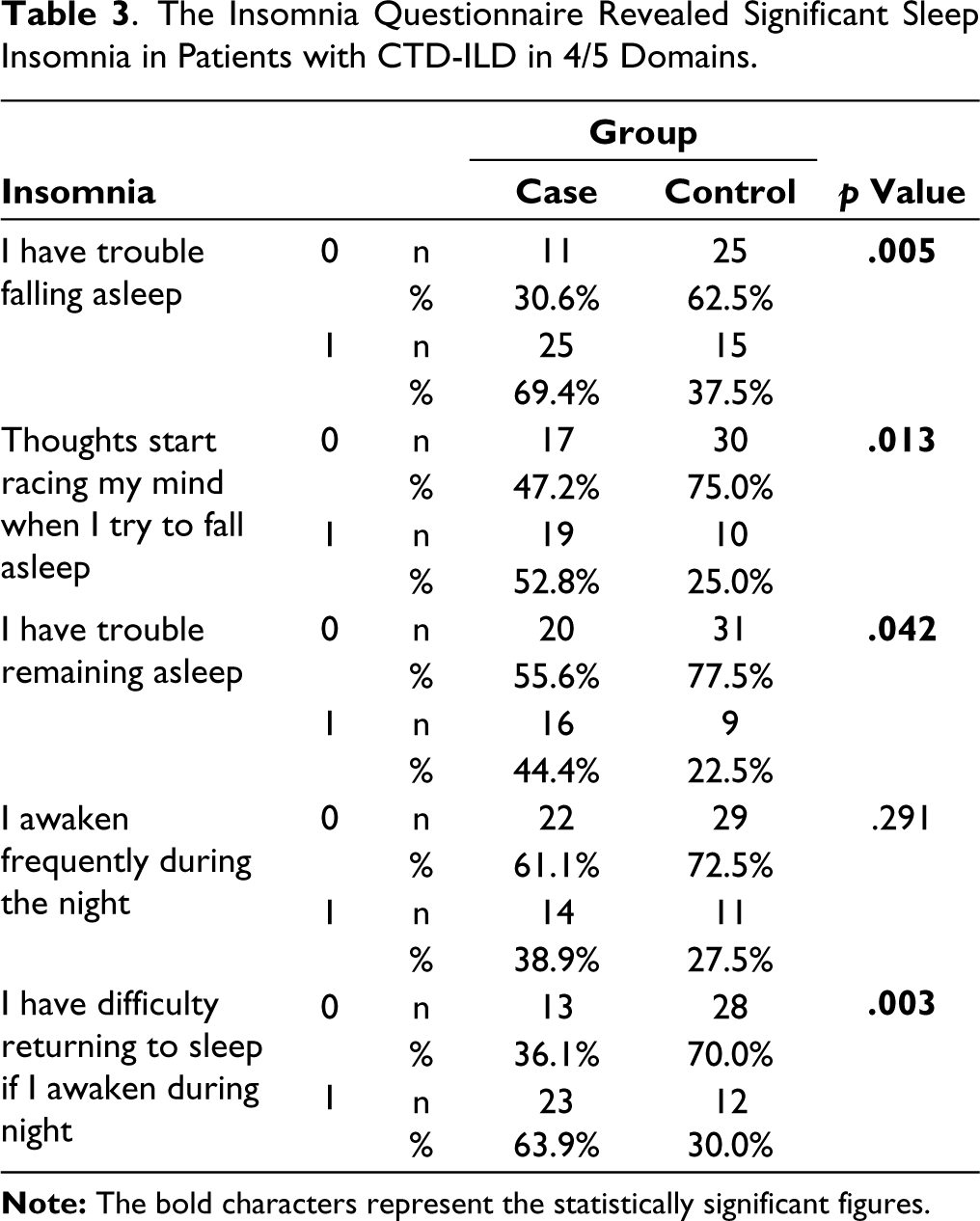
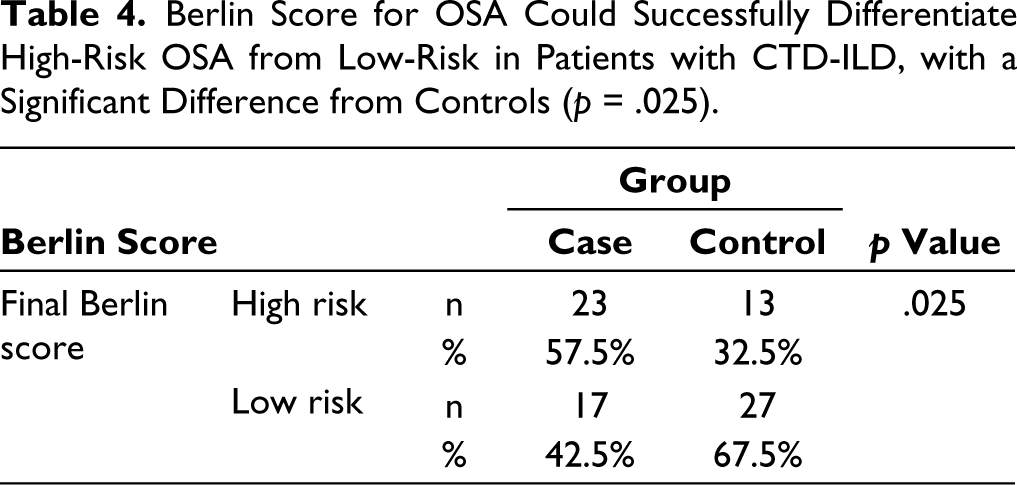
Forty cases of CTD-ILD and age-matched controls were enrolled. The mean age of patients was 46.5 ± 12.53 years and 90% were female. On the ESS, where a score of >10 is significant, the patients with CTD-ILD had significantly higher daytime sleepiness as compared to controls (p = .014). Per the Berlin questionnaire, 57.5% of the patients were at high risk (p = .025). RLS was significantly higher in patients compared with controls (Table 5, p < .05).
Conclusion:
Sleep disorders and sleep-disordered breathing (SDB) are highly prevalent in CTD-ILD. In resource-limited settings, sleep-related questionnaires can serve as a screening tool to identify patients who require further comprehensive evaluation.
Introduction
Sleep is vital in maintaining health and homeostasis, and its complex connection with the immune system is well-recognised. 1 Poor sleep quality, including sleep disorders or dysregulation, is associated with an increased risk of autoimmunity and inflammation. 2 Studies have consistently shown that disorders of sleep result in poor outcomes in patients with connective tissue diseases (CTD) like rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), Sjogren’s disease (SjD), systemic sclerosis (SSc). The prevalence of insomnia reaches nearly 60% in RA. 3 Similarly, data from SSc also points to a high prevalence of lower sleep quality in 65%–70% of the patients, which correlates with the severity of interstitial lung disease (ILD). 4 CTD-associated ILD (CTD-ILD) is one of the major causes of morbidity and mortality across the spectrum of autoimmune diseases. Patients with ILD with autoimmune features, who do not meet the defined criteria for any CTD, are referred to as interstitial pneumonia with autoimmune features (IPAF) and this warrants immunosuppression and definitive treatment just as any CTD-ILD. 5 During the course of management of CTD-ILD, while addressing the disease activity with optimal immunosuppression forms one arm of treatment, close attention to the quality of life, including sleep and mood disturbances, is a major treatment domain often sidelined.
Patients with CTD-ILD tend to have a variety sleep disturbances, including but not limited to obstructive sleep apnoea (OSA), sleep-disordered breathing (SDB), anxiety and depression-associated insomnia, restless leg syndrome (RLS), and parasomnias. OSA is one of the most well-studied sleep-related breathing disorders with a reported prevalence of up to 60% in CTD, while the others are scarcely represented in research. 6 Poor sleep in these patients can be due to the disease itself, for example, gastro-oesophageal reflux disease (GERD), pruritus that is commonly associated with SSc, MCTD, fatigue, myalgia or depression. 7 The broader term of SDB encompasses OSA, central sleep apnoea (CSA), sleep-related hypoxemia or hypoventilation disorders. 8 The prevalence of OSA in ILD ranges from 20% to 88%, while it is around 5% in the general population.9–12 The symptoms of poor sleep hygiene include chronic fatigue, pain, depression, early morning headaches, cognitive decline and an increased inflammatory milieu at the cellular level. 13
Though the literature suggests an association between OSA and other sleep disorders in CTD, the data on CTD-ILD is scarce. Hence, we sought to study and screen the patients with CTD-ILD with a primary objective to estimate the prevalence of sleep disorders and also secondarily, to categorise and describe the prevalence of sleep-associated disorders like RLS, OSA and parasomnias.
Methods
This single-centre, cross-sectional questionnaire-based study was carried out at a tertiary care hospital in Eastern India over four months, to determine the prevalence of sleep disorders in patients with CTD-ILD, after obtaining ethics approval (Ref no: KIIT/KIMS/IEC/1043/2022) and a written informed consent. Patients aged 18-80 years, diagnosed with ILD secondary to an autoimmune rheumatic disease (i.e., CTD-ILD) like SSc, mixed connective tissue disease (MCTD), RA, Sjogren’s disease (SjD) or having interstitial pneumonia with pulmonary fibrosis (IPAF) were included in the study. Patients with primary idiopathic ILD or with aetiology other than CTD were excluded. Patients who had underlying respiratory comorbidities of chronic obstructive pulmonary disease (COPD), bronchial asthma and a history of pulmonary tuberculosis, lung malignancy were also excluded. The diagnoses of SSc, MCTD, RA, SjD, dermatomyositis were made by the standard classification criteria as per ACR/EULAR.14–17
Consecutive patients attending the Pulmonology or Rheumatology clinics, who consented and met the inclusion criteria during the data collection period, were enrolled in the study. After enrolment, patients were administered the questionnaire (Supplementary file), which they reviewed, and it was then duly filled up by one of the investigators.
A focus on assessing multiple domains of sleep disturbance was sought, and the questionnaire was curated accordingly. First was the Epworth Sleepiness Scale (ESS) which is a generic scale to assess daytime sleepiness and has found clinical use in OSA and other SDB syndromes. It used eight questions and a total score of >10 denotes excessive daytime sleepiness. 19 OSA was assessed by two independent questionnaires: (a) Berlin questionnaire—which includes 10 items and can be divided into three domains 20 ; (b) the STOP-Bang questionnaire—which includes questions on four symptoms and demographic data. 21 Other questionnaires were included to assess RLS and parasomnias. The protocol also included medical, treatment and social histories. The questionnaire for RLS was derived from the diagnostic criteria. 22
Statistical analysis was conducted using IBM SPSS 23. All the continuous variables were expressed by mean ± standard deviation (SD) and categorical variables were expressed by frequency or percentage. For mean comparison, the student t-test or ANOVA was used for the data following normal distribution, and the Mann-Whitney U test or Kruskal Wallis test was used for the data not following normal distribution. p value < .05 was considered statistically significant.
Results
Sixty-two patients were screened, and 40 patients and age-matched controls were enrolled during the study duration of four months. Demographic and baseline characteristics are presented in Table 1. The study population was female predominant (90%) with SSc-ILD constituting the majority in the study group (14, 35%), closely followed by MCTD-ILD (12, 30%). The remaining was constituted by RA-ILD (9, 22.5%), IPAF (3, 7.5%) and one patient each with SjD-ILD, and dermatomyositis-ILD. The median age of the study participants was 40 years (IQR 30.5-56). All the patients were on some form of immunosuppressive therapy, the details and duration of which are listed in Table 1.
Demographic and Clinical Characteristics of Patients with ILD and Controls.
On assessing the sleep onset, latency and quality of sleep, we found that 56.5% had a sleep latency of longer than 30 minutes. The number of awakenings in the night can also be one of the indirect indicators of apnoea and in our cohort, 72.5% (29/40) of patients had experienced at least 2 or more awakenings per night, irrespective of the cause and they reported longer latency to fall asleep once awoken in the night.
For assessing sleep quality, we discovered that 57.5% (23/40) of the patients experienced excessive daytime sleepiness, with 47.82% of them sleeping for less than six hours per night. Another indicator of poor sleep quality, early morning headaches, were seen in over half of the study population (52.5%).
Prevalence of Excessive Daytime Sleepiness
42.5% of the patients scored >10 on the ESS, deemed significant. Further, the patients of CTD-ILD had significant daytime sleepiness as per the ESS, with a mean score of 10.41 ± 5.02 versus 7.88 ± 3.86 in the controls (p = .014) (Table 2). These findings were supported by the evidence from the insomnia questionnaire (Table 3).
As per the Epworth Sleepiness Scale, the Patients of CTD-ILD had Significantly Higher Daytime Sleepiness, Compared to Healthy Controls (p = .014); (An ESS Score of >10 Suggests Significant Daytime Sleepiness).
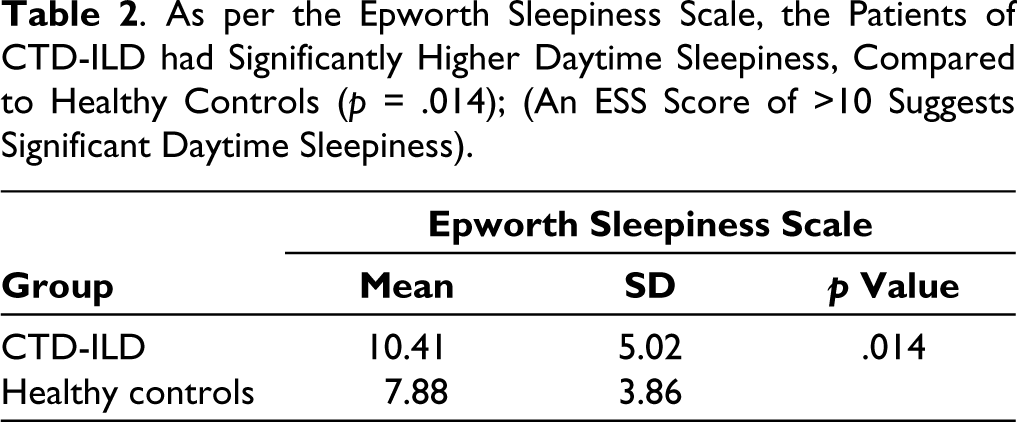
The Insomnia Questionnaire Revealed Significant Sleep Insomnia in Patients with CTD-ILD in 4/5 Domains.
Prevalence of OSA
Per the Berlin questionnaire, there was a significantly higher prevalence of OSA among patients with CTD-ILD compared to controls (57.5% vs. 32.5%, p = .025) (Table 4). However, as per the STOP-Bang questionnaire, we found a numerically higher number of patients at intermediate risk of OSA compared to controls (40% vs. 27.5%), but the difference did not reach statistical significance (data not shown).
Berlin Score for OSA Could Successfully Differentiate High-Risk OSA from Low-Risk in Patients with CTD-ILD, with a Significant Difference from Controls (p = .025).
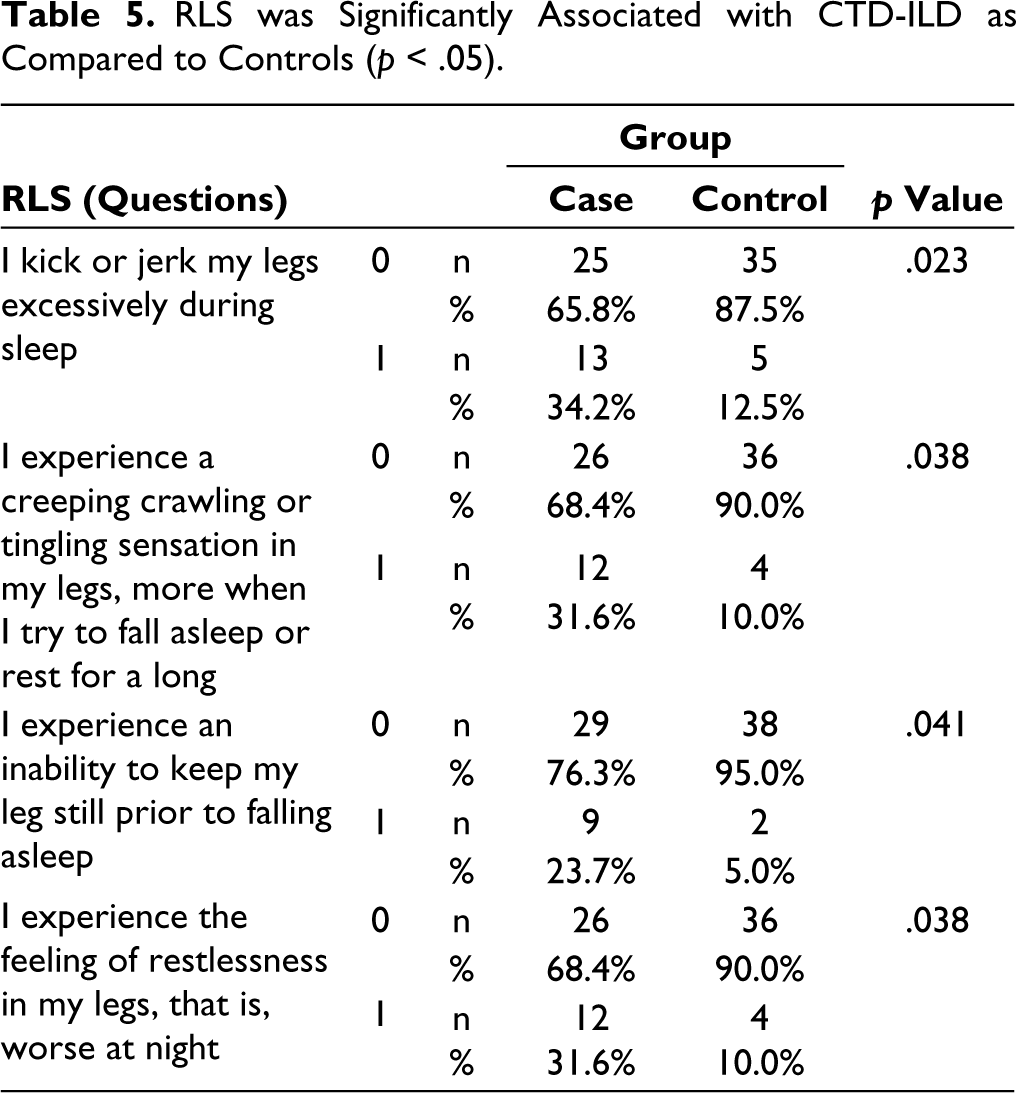
Restless Leg Syndrome
Four questions capturing the common symptoms of RLS were used as a part of a questionnaire. There was an increased prevalence of RLS in CTD-ILD (Table 5).
RLS was Significantly Associated with CTD-ILD as Compared to Controls (p < .05).
Parasomnias
There were no significant differences in the prevalence of parasomnias (Supplement Table S1).
Discussion
Disorders of sleep, including sleep-related breathing disorders contribute to the morbidity in CTDs. Beyond affecting the quality of life, poor sleep is associated with an array of other conditions like higher inflammation and inflamm-ageing, metabolic syndrome, higher risks of cardiovascular diseases, and insulin resistance. 2 CTDs per se are associated with accelerated atherosclerosis and this associated with sleep disorders can potentially lead to worsening risk.
The burden of OSA in CTDs and idiopathic ILD has been evaluated a priori.6,10,12,23–26 However, OSA in the context of CTD-ILD is under-represented. Data from studies that included all etiologies of ILD, demonstrated that among patients with concurrent ILD and OSA, idiopathic pulmonary fibrosis was the most common cause of ILD, while CTD-ILD constituted only 16% of the cases. 10 Sleep disorders are also influenced by other factors such as RLS, anxiety, and depression, which are frequently overlooked aspects of sleep health.
OSA in CTD-ILD
Patients with CTD-ILD, have multiple underlying risk factors that can contribute to the development of OSA. Since some of these patients fail to maintain remission after tapering steroids, they tend to be on a continued dose of at least low-dose steroids that can contribute to weight gain, metabolic syndrome, diabetes, hypertension, which are risk factors for the development of OSA. 27 In our study, we opted for a questionnaire-based cross-sectional assessment, which can be performed in a busy clinic, to screen and capture the group of patients who would benefit from a further detailed evaluation with polysomnography.
OSA in our patients was assessed by three questionnaires as described. One of the indirect evidence of OSA is excessive daytime sleepiness, which was assessed by the ESS. Independent of the entire Epworth scoring, nearly half of the patients (47.8%) reported that they experienced excessive daytime sleepiness, with 43% of them reporting a wake-up early morning headache, which can possibly be ascribed to hypoxia with or without hypercarbia during sleep. Berlin questionnaire, one of the more sensitive models for uncovering patients at risk of SDB, has a sensitivity of 86% in detecting those with a respiratory disturbance index (RDI) of >5. 20 Over half of our subjects (57.5%) fell in the high-risk category, and it is prudent to follow up on these patients with polysomnography. The other highly sensitive questionnaire we used was the STOP-Bang questionnaire, which reports 100% sensitivity with an apnoea-hypopnea index of greater than 30, when body mass index (BMI), neck circumference and gender are incorporated as in our study. 21 Accordingly, 40% of our subjects could be stratified as having an intermediate risk of OSA.
Sleep Disturbances Beyond OSA in CTD-ILD
While OSA in the setting of CTDs and ILD has been studied extensively, there is an evident gap in addressing sleep disturbances unrelated to hypoxia or apnoea or OSA. This gains more inertia in patients living with AIRDs, especially those with concomitant ILD, as a baseline sub-optimal lung functioning, a state of chronic hypoxia may lead to anxiety, depression, insomnias and sometimes parasomnias. The association of RLS with CTDs has also been described in literature and this can contribute to the overall quality of life (QoL). Data from meta-analysis estimates the prevalence of RLS to be around 3% in the general population. 28 However, in the setting of autoimmune rheumatic diseases (AIRDs), its prevalence stands at around 15%–30% across cohorts, with iron deficiency anaemia and high disease activity being the common associations.29–33 In our study, nearly half of the subjects had some form of symptom associated with RLS, with creeping or crawling sensation of the legs while falling asleep and inability to keep the legs still before falling asleep and during the night being the most common symptoms.
Parasomnias associated with CTD-ILDs have hardly been described in the literature. We found that parasomnias were not uncommon, with 17.4% of patients reporting sleep-talking as noticed by their partners. An important observation is that all patients in our study had some form of sleep disturbance. The burden of anxiety and depression in the setting of CTDs has been studied and there is a reported higher prevalence in these patients.7,26,34 Similarly, ~43.5% of our subjects reported that thoughts raced through their minds when they tried falling asleep and 47.8% had difficulty in returning to sleep if they woke up during the night. This indirectly contributes to the possibly undetected burden of mental ill-health in these patients, and it is one of the neglected domains in the treatment of CTDs, as treat-to-target mainly includes chasing remission and maintenance.
Sleep disorders also contribute to the increased disease activity and researchers have also suggested that long-term sleep disturbances themselves can predispose to the development of AIRDs. Adequate sleep is closely related to maintaining a homeostatic balance between pro-inflammatory and anti-inflammatory cytokines. Animal models have shown that with total sleep deprivation or partial sleep deprivation, there was metabolic dysregulation, fraility and resulted in the death of the animal in the short run. Additionally, the inflammatory pathways including signal transducer and activator of transcription (STAT), nuclear factor kappa B (NF-κb) are upregulated with even partial sleep deprivation during the night, and this also results in higher mRNA transcription encoding pro-inflammatory cytokines.2,35–37 Higher levels of Toll-like receptor (TLR)-induced IL-6 and tumour necrosis factor (TNF) were also observed in patients who had disturbed sleep. 38 These homeostatic dysregulations in the setting of poor sleep quality efficiency or quantity are only augmented in the setting of an underlying CTD, as the sleep disturbances may add to the higher inflammatory burden of the disease.
Our study was limited by its small sample size and single-centre enrolment. Second, as CTDs are more prevalent in females, especially the diseases that are commonly associated with ILD, that is RA, MCTD and SSc, there is an inevitable gender bias in our study and the prevalence of OSA in CTD-ILD may be skewed. Correction was not feasible as our study included consecutive patients. So, sex as a risk factor for the development of OSA, could not be studied. The prevalence of ILD was highest in SSc and as this was a single-centre experience, there could have been a geographic bias. Further, other physical factors like microstomia, tightness of the skin on the chest resulting in decreased chest expansion and disordered breathing were not captured.
Conclusion
There is an increased prevalence of sleep disorders and SDB in patients with CTD-ILD, with OSA being one of the most common. In a resource-poor setting, and when financial constraints limit the use of polysomnography upfront for the evaluation of sleep disturbances, this questionnaire-based method is a plausible alternative where patients with a high risk of OSA can be uncovered and followed up with polysomnography.
While OSA is one domain, the other sleep-related disturbances with respect to sleep quality, and quantity were also assessed by these questionnaires, including RLS, parasomnias and the often-neglected mental well-being of these patients living with chronic diseases. Poor sleep translates to metabolic dysregulation and produces a pro-inflammatory milieu that can further augment the disease process in these patients.
Supplemental Material
Supplemental material for this article available online.
Footnotes
Data Availability Statement
Data will be available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
