Abstract
Thrombocytopenia is a common manifestation of systemic lupus erythematosus (SLE) and is often associated with other haematological manifestations such as leukopenia and haemolytic anaemia. It is a poor prognostic marker and is an independent marker of mortality and morbidity. Several autoantibodies, such as anti-glycoprotein IIb/IIIa (GpIIb/IIIa), anti-GpIa/IIa, anti-GpIb/IX, anti-thrombopoietin receptor (TPO-R) and antiphospholipid antibodies, play a role in the pathogenesis of thrombocytopenia in SLE. Glucocorticoids are considered the first line in the management. However, when prescribed alone, it results in relapses and poor outcomes; therefore, guidelines recommend concomitant immunosuppressants along with glucocorticoids. The choice of concomitant immunosuppression may depend on other SLE-related manifestations. Rituximab and immunoglobulin have been used in relapsed and refractory thrombocytopenia. TPO-R agonists such as eltrombopag and romiplostim approved for chronic immune thrombocytopenia are found to be efficacious in SLE thrombocytopenia. However, TPO-R agonists are associated with a risk of thrombosis and rebound thrombocytopenia after stopping them. Therefore, an individualised approach is required when treating thrombocytopenia in SLE.
Introduction
Thrombocytopenia is a prevalent clinical symptom of systemic lupus erythematosus (SLE), reported in roughly one-third to half of all patients.1–4 Thrombocytopenia, which is characterised by a platelet count (PC) <100 × 109 l−1, is a part of the Systemic Lupus Erythematosus International Collaborating Clinics 2012 and the 2019 European Alliance of Associations for Rheumatology/American College of Rheumatology (EULAR/ACR) classification criteria.5,6 The overall prevalence of thrombocytopenia as per this cut-off was recorded in one-fourth of patients in the Indian SLE inception cohort (INSPIRE) and at enrolment in 10.4%.7,8 PCs less than 20,000 μl−1 are termed severe thrombocytopenia in SLE, those between 20 and 50,000 μl−1 are considered moderate and those over 50,000 μl−1 are called mild thrombocytopenia. 9 Severe thrombocytopenia is relatively uncommon in SLE and is observed in about 5% of patients. 3 In the INSPIRE cohort, it was noted in only about 1% of patients. 8
Thrombocytopenia typically clusters with other haematological manifestations such as autoimmune haemolytic anaemia and leukopenia, which was also observed in the INSPIRE cohort. Thrombocytopenia in SLE is reported to be an independent predictor of mortality and contributes to morbidity and increased damage.8,10–14 Rightly, severe thrombocytopenia (<25,000 μl−1 is considered as the British Isles Lupus Assessment Group A category and has been assigned a score of 1 in Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) at a PC of <100,000 μl−1.15,16
Pathogenesis of Thrombocytopenia in SLE
Megakaryopoiesis takes approximately 4–7 days, and several cytokines support several aspects of platelet production. Among these cytokines, genetic elimination or low levels of steel factor, interleukin-11 or thrombopoietin (TPO) affect megakaryocyte and platelet production. Thrombocytopenia is often complex in SLE. Platelet degradation by antibodies is the most prevalent pathogenic mechanism. Antigen specificity of antiplatelet antibodies is mainly directed to glycoprotein IIb/IIIa (GpIIb/IIIa), as well as to GpIa/IIa and GpIb/IX, similar to that in immune thrombocytopenia (ITP). Additionally, in 23–39% of patients with SLE, autoantibodies against TPO and the TPO receptor (TPO-R) Human myeloproliferative leukemia virus oncogene (c-mpl) have been identified.10,17 These antibodies disappear in treated patients with thrombocytopenia and reappear in relapses, thus confirming their role in the pathogenesis. Nevertheless, individuals with SLE who do not have thrombocytopenia have also been found to have antiplatelet antibodies.17,18 In a study in which the role of anti-GPIIb/IIIa and anti-TPO-R (c-mpl) antibodies was assessed in patients with SLE with and without thrombocytopenia, healthy controls and patients with chronic ITP, 91% of patients with SLE with thrombocytopenia had either anti-GPIIb/IIIa or anti-TPO-R antibodies, and 18.7% of patients had both antibodies. 18 Patients with SLE who have positive anti-TPO-R antibodies had much greater megakaryocyte hypoplasia and a poorer response to immunoglobulin (IVIg) and corticosteroids. 18
The association between Antiphospholipid antibody (APLA) and thrombocytopenia has been well documented in medical literature and is considered an antiphospholipid syndrome (APS) non-criteria manifestation. In the recent EULAR/ACR classification criteria for APS, thrombocytopenia has been ascribed a weight of 2. 19 It is unclear how antiphospholipid antibodies cause thrombocytopenia; however, it is thought to be related to platelet lysis, reduced platelet synthesis and increased platelet pooling. Destruction of platelets can be either immune-mediated or thrombotic microangiopathy, namely catastrophic antiphospholipid syndrome (CAPS). About 40–70% of thrombocytopenic patients with APS exhibit antibodies against glycoproteins on the platelet cell wall (GPIIb/IIIa, GPIb/IX, GPIa/IIa and GPIV). 20 Furthermore, anti-β2GPI–β2GPI dimers bind to platelet membranes and induce platelet activation and aggregation.21,22 Increased production of von Willebrand factor (vWF) by endothelial cells and direct platelet activation, which causes an increase in vWF–platelet interaction, is yet another means antiphospholipid antibodies cause an increase in the consumption of platelets. 23 The decreased production of platelets in APS can be due to bone marrow infarction, especially in the setting of CAPS.24–26 Finally, platelet agglutination and pseudothrombocytopenia—an erroneously low PC—can result from antiphospholipid antibodies binding to antigens modified by Ethylenediamine tetraacetic acid (EDTA) on the platelet membrane. 27
Other mechanisms for thrombocytopenia in SLE include decreased production via antibody- or T-cell-mediated suppression of megakaryocytes, thrombotic microangiopathy, macrophage activation syndrome and hypersplenism.
Bleeding Risk
The true incidence of serious bleeding in lupus-associated thrombocytopenia remains unknown.
Currently, the severity of thrombocytopenia is determined based on the PC and the assumption that the severity of bleeding increases as the PC decreases. 13 In the context of thrombocytopenia, cutaneous and visible mucosal bleeds are considered minor bleeds, while all other bleeds are major bleeds. 28
In the INSPIRE cohort, major bleeding was observed in only one patient among 22 patients with severe thrombocytopenia, nine patients had minor bleeding, and the rest had no visible bleeding. The risk of bleeding is known to increase significantly in individuals aged >60 years with a history of haemorrhage. 12
Management
The treatment algorithms for thrombocytopenia in SLE stem either from the overall SLE disease management protocols or extrapolated from the ITP treatment recommendations. An algorithmic approach for the management of thrombocytopenia in SLE is represented in Figure 1.
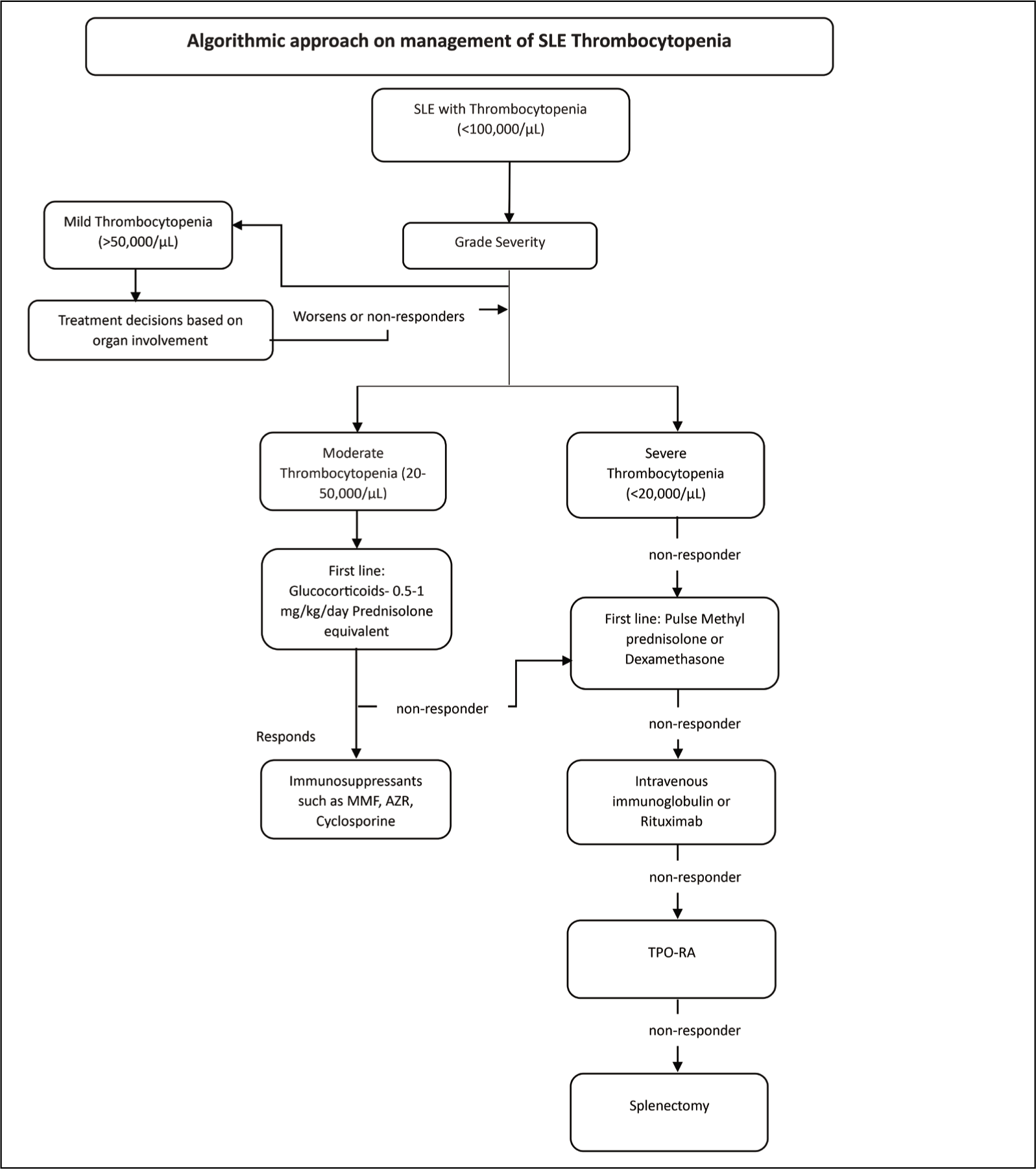
Immunosuppressive Therapy
PCs >50 × 109 l−1 generally do not require specific therapy unless bleeding manifestations and/or the involvement of other organs are present. Corticosteroids are considered the first-line treatment. 29 Among various corticosteroid formulations, dexamethasone and prednisolone are extensively used. Although equivalent doses of both formulations were effective, there were few differences between them. Table 1 highlights the differences between dexamethasone and prednisone.30–34 The American Society of Hematology recommends either prednisone (0.5–2.0 mg kg−1 day−1) or dexamethasone (40 mg day−1 for 4 days) as the initial therapy in newly diagnosed adult patients with ITP. 35 The EULAR guidelines recommend initial treatment with pulse methylprednisolone followed by 0.5–0.7 mg kg−1 day−1 prednisone equivalent. 36
Comparison of Different Steroid Formulations.
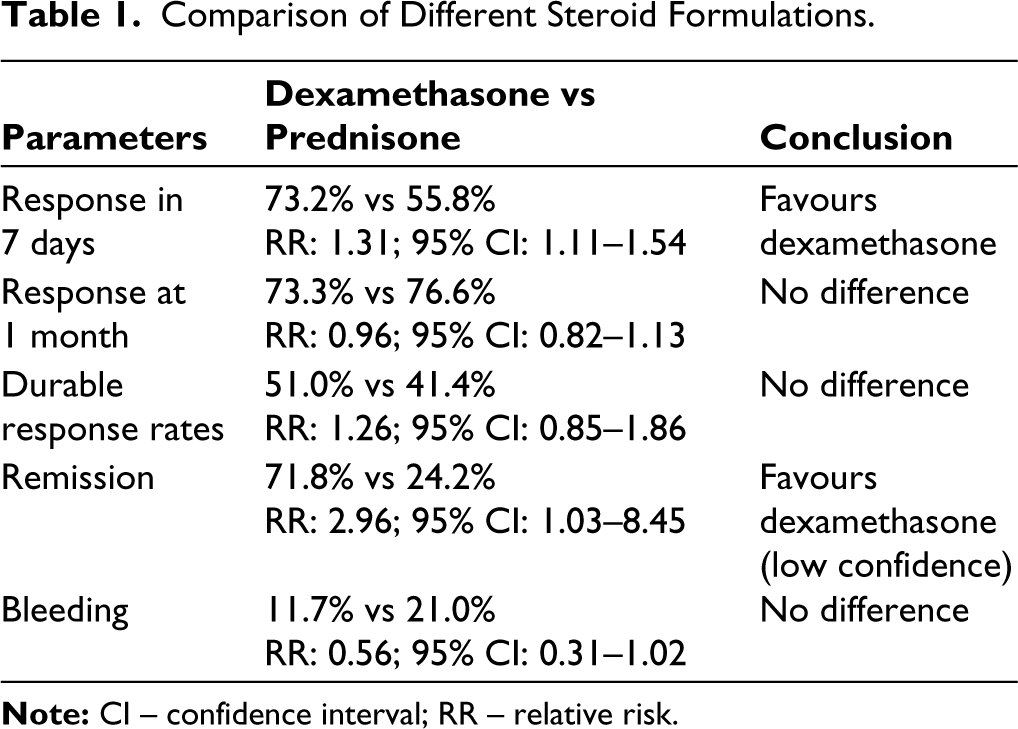
Thrombocytopenia responds well to corticosteroids; however, treatment is often lengthy and characterised by relapses of steroid tapering or stopping.30,31 However, there is a lack of well-designed randomised controlled trials (RCT) to determine an effective steroid-sparing immunosuppressive therapy for thrombocytopenia in SLE. The First line treatment pathways for newly diagnosed immune thrombocytopenia (FLIGHT) trial is an open-label RCT that evaluated the effects of glucocorticoids alone versus glucocorticoids in combination with mycophenolate mofetil (MMF) in adult patients with ITP. The patients were followed for a period of 2 years, during which the MMF group experienced a greater response (91.5% vs 63.9%; P < .001) and fewer treatment failures (22% vs 44% with a hazard ratio of 0.41). 37 Although this trial was conducted in patients with ITP, the results may be extrapolated to patients with SLE. The EULAR guidelines recommend the initiation of concomitant steroid-sparing immunosuppressive agents as per physician discretion. 29
Intravenous IVIg
Intravenous IVIg is the preferred alternative in situations where corticosteroids are contraindicated or steroid-sparing alternatives and in refractory patients. However, data on IVIg use in SLE were derived from case reports or observational studies. IVIg at a dose of 2 g kg−1 was administered for a variety of clinical symptoms, including subacute cutaneous lupus erythematosus, discoid lupus erythematosus, lupus nephritis, haematological (haemolytic anaemia, thrombocytopenia and pancytopenia) and neuropsychiatric lupus. In a large retrospective study involving 52 patients with SLE, about two-thirds of the cases demonstrated that IVIg was beneficial in practically all of these indications, and it is difficult to isolate the advantage in thrombocytopenia separately. 38 In another retrospective study that assessed the response of low-dose IVIg (0.5 g kg−1) in various disease manifestations, it was found that thrombocytopenia did not improve with low-dose IVIg. 39 In individuals with symptomatic ITP who had not received treatment before, an RCT examined the effects of prednisone alone, IVIg alone and a combination of the two. The results showed responses in 82%, 54% and 92% of patients, respectively. No difference was observed between the IVIg and prednisone groups, and IVIg offered no advantage over prednisolone as the primary therapy. Relapse was also not statistically significant between groups. 40
Rituximab
The use of rituximab in SLE is based on observational studies; however, it has been well-studied in chronic ITP. In two open-label RCTs including recently diagnosed patients with ITP, dexamethasone alone was compared with the combination of dexamethasone and rituximab (375 mg−1 m2) weekly for 4 weeks. The results showed that 6 months of combination therapy produced a higher sustained response (>50 × 10 9 l−1; 63% vs 36% and 58% vs 37%, respectively).41,42 Response to rituximab was observed within 1–8 weeks. The advantage of rituximab lies in maintaining a sustained response even after 5 years. According to a long-term follow-up study, 21–26% of adults and children with chronic ITP who received standard-dose rituximab were able to go at least 5 years without receiving any treatment; however, rituximab remains an off-label treatment for SLE. 43
Considering the pathophysiological mechanisms underlying SLE and the depletion of B cells is an attractive option. In a multicentre retrospective study in France, the efficacy of rituximab in 71 patients with SLE with cytopenia was studied. Among the 44 patients with thrombocytopenia, the overall response rate was 91%. 44 Rituximab is effective in treating a number of SLE symptoms, including thrombocytopenia, neurological symptoms, cutaneous lupus, arthritis and neurological manifestations (30.3%, 24.2% and 15.2%, respectively). 45 The use of low-dose rituximab (100 mg week−1 for 4 weeks) in SLE has been limited to a small case series. 46
Belimumab
Over the last decade, belimumab has been established as an effective and safe drug for the treatment of SLE in several clinical trials and real-world studies. Not only does it permit glucocorticoid tapering, but it also reduces disease activity and flares in several SLE domains, thereby limiting damage. 47 Belimumab’s efficacy in treating patients with SLE with refractory thrombocytopenia has been documented in case reports. 48 Additionally, it has demonstrated efficacy in APL-associated thrombocytopenia. 49
TPO Receptor Agonists (TPO-RA)
The US Food and Drug Administration (FDA) has approved two TPO agonists for use in refractory ITP, eltrombopag and romiplostim. Both attach to the TPO-R, changing its structure and initiating the Janus kinase 2/signal transducer and activator of transcription (JAK2/STAT5) pathway, which increases platelet production and megakaryocyte progenitor cell proliferation.
Romiplostim acts on surface receptors, thereby competing with endogenous TPO, whereas eltrombopag binds the transmembrane domain of the receptor. Romiplostim significantly enhanced the total platelet response in comparison to eltrombopag in a database review that indirectly compared the two drugs. 50 Additionally, both splenectomised and non-splenectomised individuals with ITP responded well to romiplostim. 51 with serious adverse effects, including cataracts, pneumonia and bone marrow fibrosis. Arterial and venous thromboembolisms were observed with both agents. The incidence of thromboembolism events includes 4.1–7.5 per 100 patient-years for romiplostim and 3.4 per 100 patient-years for eltrombopag. 52 These thromboembolic events were not associated with either the dose of TPO-RA or the degree of thrombocytosis, and at least one acquired and inherited thrombotic risk factor was present in most patients who experienced thrombosis.53,54 By this corollary, TPO-RA should be avoided in SLE with APS.
The effectiveness and safety of eltrombopag in the treatment of steroid-refractory thrombocytopenia in individuals with SLE were investigated in a single-centre trial. The median response time was 8 days. Most trial participants showed a sustained response without any major adverse effects. 55 In a retrospective cohort study, 93% of patients with SLE with thrombocytopenia receiving TPO-RA responded to eltrombopag or romiplostim. Patients on eltrombopag experienced two arterial thrombosis (both in the presence of APLA or APS), and one patient receiving romiplostim developed a dual thrombosis (no APLA or APS). 56 In a systematic review and meta-analysis of 15 RCTs, involving 3,026 adult thrombocytopenic patients, the relative risk for thromboembolism was 1.81 (95% confidence interval: 1.04–3.14). 57
Rebound thrombocytopenia after stopping TPO-RA was observed in most patients; however, in only 10% of patients, the PC dropped below the pretreatment level. Endogenous TPO activity is regulated by platelets, and megakaryocyte levels are suppressed and may not re-equilibrate after the abrupt withdrawal of TPO-RA. However, Repeated ExPosure to Eltrombopag in Adults with ITP, which evaluates the effect of three cycles of administration of eltrombopag on PCs in patients with chronic ITP, showed that eltrombopag is effective without any increase in adverse events. 54
Spleen Tyrosine Kinase (SYK) Inhibitor
In individuals with ITP, phagocytosis-based antibody-mediated platelet destruction is largely dependent on SYK activation. Fostamatinib inhibits platelet phagocytosis mediated by autoantibodies and suppresses SYK activation in macrophages. The US FDA has approved fostamatinib for the treatment of refractory chronic ITP. 58 Fostamatinib and lanraplenib, another SYK inhibitor, have been shown to ameliorate kidney and skin diseases in mouse models. 59 However, fostamatinib was withdrawn from the phase 2 study, while lanraplenib failed to achieve its primary endpoint in the proof-of-concept study. 60
Platelet Transfusion
Platelet infusion, either pooled or concentrated, is not recommended unless life-threatening bleeding occurs.
Splenectomy
Splenectomy has been declining because of physician and patient preferences for medical therapy over surgery and increased awareness of the possible risks associated with splenectomy. This may be considered only in patients with refractory diseases.
Pregnancy-Related Thrombocytopenia Management: Special Considerations
The intricate relationship between female sex steroids and SLE poses significant challenges to providing comprehensive care for women throughout their lifespans. Pregnancy can increase lupus activity, and flares can occur during any trimester. 61 SLE-associated thrombocytopenia accounts for only one score in the SLEDAI. Thrombocytopenia is not always associated with a disease flare during pregnancy. A retrospective study that evaluated SLE flares during pregnancy by organ involvement found that the haematological system is most commonly (15.6%) affected. 61 Moreover, baseline thrombocytopenia was found to be a predictor of unfavourable pregnancy outcomes with Odds ratio (OR) = 1.33 per 50K decrease. 62 Pregnant women with SLE with thrombocytopenia are at increased risk of miscarriage, intrauterine stillbirth and preterm delivery compared to those without thrombocytopenia. 63
In addition to the aforementioned causes of thrombocytopenia in SLE, pregnancy-related causes include gestational thrombocytopenia, pre-eclampsia/Hemolysis, Elevated Liver enzyme levels, and Low Platelet levels (HELLP) syndrome and acute fatty liver of pregnancy, to name a few. With up to 76% of all causes, gestational thrombocytopenia is the most frequent cause of thrombocytopenia in pregnancy.64,65 The PC will often stay between 130,000 and 149,000 μl−1, have minimal effect on the health of the mother and resolve on its own following delivery.
Both the management of disease activity and the potential harm to the feotus must be taken into account when selecting a treatment plan. There are no randomised studies comparing different first- and second-line treatments. The majority of treatment guidelines29,35 do not discuss the treatment of thrombocytopenia in pregnancy. First-line treatment included either oral corticosteroids or IVIg. A retrospective study that compared IVIg and corticosteroids did not find any difference in platelet response (38% vs 39%; P = .85) or maternal and neonatal outcomes. 66 Among the various formulations of corticosteroids, prednisolone is preferred as it does not cross the placenta. In refractory cases, among the myriad of available treatments, azathioprine and cyclosporin A have better safety profiles. However, these medications have a delayed response time with response rates of 40–60%. 67 Rituximab crosses the placenta and suppresses neonatal B-lymphocyte development, causing prolonged lymphopenia in newborns. Although the current literature indicates that the real incidence of neonatal infection following maternal rituximab administration is minimal, there is an elevated risk of infection in both the mother and the newborn. 68 Hence, rituximab may be considered in dire circumstances, as supported by numerous reports.69–71 Similarly, TPO-RA such as eltrombopag has been successfully tried in several reports. 72 Although platelet transfusion is normally not advised because of the short-term benefits of quick clearance, it can be taken into consideration in the event that an emergency caesarean section is necessary.
Prognosis
Mortality is considerably lower in individuals who have achieved complete remission from thrombocytopenia than in those who have not.9,11 According to the INSPIRE cohort, the death rate for individuals with severe thrombocytopenia is three times greater than that of those with moderate thrombocytopenia and controls. Interestingly, patients with moderate-to-severe thrombocytopenia had a higher hospitalisation rate. 8
Conclusion
Severe thrombocytopenia is uncommon in SLE and does not result in severe bleeding. It responds to steroid treatment briskly but may relapse despite concomitant steroid-sparing therapy. Rituximab and IVIG are useful in patients with refractory disease. Non-immunosuppressive agents such as TPO-RA are useful adjuncts in refractory thrombocytopenia; however, their use must be restricted to patients with non-APS and for a short duration until the PC is >30 × 10 9 l−1.
Footnotes
Ethical Approval and Patient Consent
Ethics committee, St. John’s Medical College Hospital IECStudyREF.NO.159/2017. The patient consent has been taken for the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
