Abstract

Dear Editor,
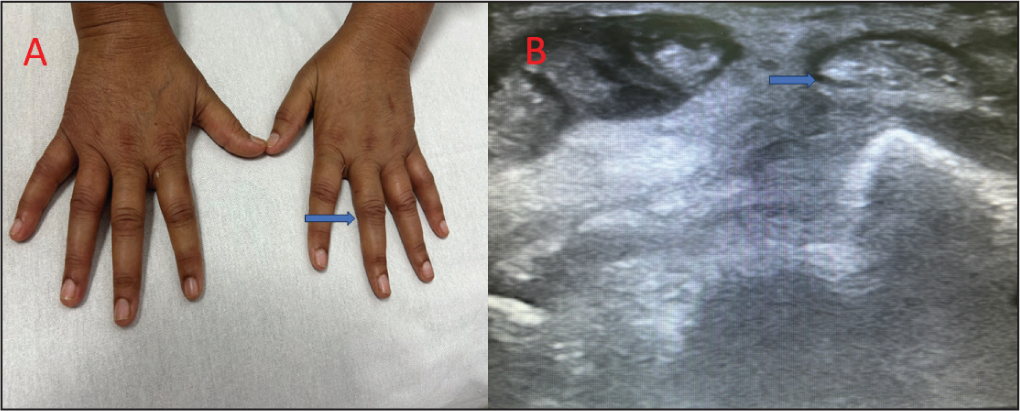
A 58-year-old female presented with a history of symmetrical, inflammatory arthritis of small joints of the hands involving metacarpophalangeal joints (MCP), proximal interphalangeal joints (PIP) and bilateral wrist of four weeks duration. She denied any history suggestive of connective tissue disorder or inflammatory low backache. Reviewing her medical documents revealed that she had advanced carcinoma endometrium and was undergoing chemotherapy, having started on nivolumab 200 mg biweekly four months ago. On examination, there was synovitis of small joints of hand and tenosynovitis of right wrist (Figure 1A). Her investigations revealed anaemia (haemoglobin (Hb): 10.2 g/dL) with markedly increased acute phase reactants (erythrocyte sedimentation rate (ESR): 64 mm/hr, C-reactive protein (CRP): 64 mg/L). Her rheumatoid factor, anti-cyclic citrullinated peptide antibody, anti-nuclear antibodies by Indirect Immunofluorescence, anti-Ro and La by enzyme-linked immunosorbent assay (ELISA) and Human Leukocyte antigen 27 by PCR were negative. Hand radiographs were normal. Musculoskeletal ultrasonography showed grade 3 power Doppler (PD) synovitis with tenosynovitis (Figure 1B). Given the background history of immune checkpoint inhibitor therapy and temporal course of arthritis, probable diagnosis of nivolumab-related arthritis was made. She was started on oral prednisolone 10 mg/day with minimal response after two weeks. After a shared decision with the oncology team and the patient, she was then advised to stop nivolumab and started on methotrexate 15 mg/week along with prednisolone 10 mg/day. She showed marked improvement in her symptoms on follow-up after a month.
Clinical Photography Showing Synovitis in Both Hands B Musculoskeletal Ultrasonography Showing Tenosynovitis of Right Wrist.
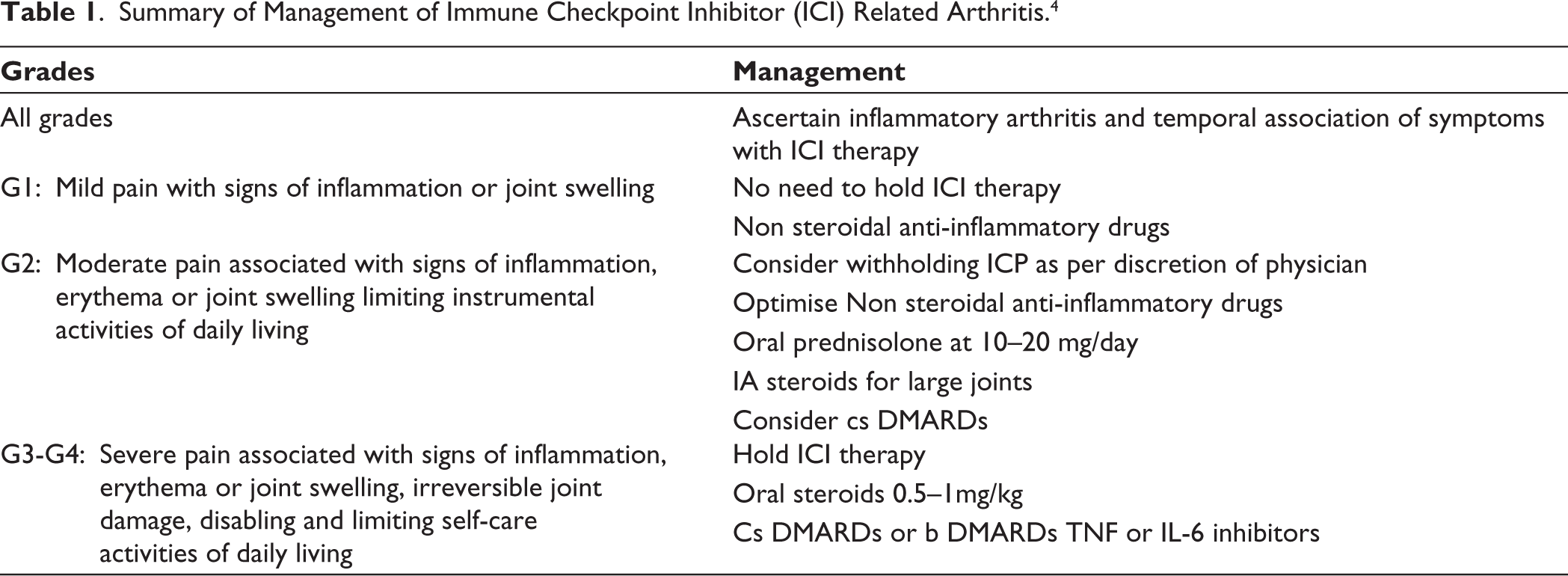
Inflammatory arthritis is the leading immune-related musculoskeletal adverse event amongst patients started on immune checkpoint inhibitor therapy, in approximately 5% of patients. 1 Studies have shown that pembrolizumab targeting PD1 is associated with highest risk of musculoskeletal adverse events, especially inflammatory arthritis. 2 One systematic review shown that half of reported immune checkpoint inhibitor-associated arthritis cases present with polyarthritis resembling seronegative RA, but only 9% are seropositive. 3 Low doses of corticosteroids or conventional synthetic disease modifying anti-rheumatic drugs (DMARDs) are usually the first line of therapy. Few patients may develop severe symptoms requiring biologic therapy. Most used biologic DMARDs are tumour necrosis factor (TNF) Inhibitors and interleukin-6 (IL-6) receptor inhibitors. In majority of the patients, arthritis resolves after stopping Immune checkpoint Inhibitor (ICI) therapy. Current guidelines on management of ICI therapy-related arthritis are summarised in Table 1. 4 Rheumatologists should be vigilant about this uncommon cause of arthritis while evaluating arthritis in the setting of malignancy.
Summary of Management of Immune Checkpoint Inhibitor (ICI) Related Arthritis. 4
Footnotes
Data Availability
All data relevant to the study are included in the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Clearance
Ethical clearance was not taken, as it was not required.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Written consent has been obtained from the patient for publication
