Abstract
Introduction:
Due to notable distinctions in infection patterns, vaccine accessibility, and approaches to treatment among patients with autoimmune inflammatory rheumatic diseases (AIRD) in India compared to other regions globally, the Indian Rheumatology Association Vaccine (IRAVAC) group was formed to develop expert panel consensus statements on vaccination for adults with AIRDs in India.
Methods:
The IRAVAC group comprised seven rheumatologists, one epidemiologist, two infectious disease specialists, one clinical virologist, one specialist nurse, and a patient representative. The group determined the scope of the consensus statements, framed the research questions, and assigned a team of few members to review the evidence who designed the search strategy and conducted the systematic literature search and data extraction. Another group of members subsequently reviewed the articles for each vaccine, prepared a summary, generated draft statements, and assigned them an appropriate level of evidence. Eighty-eight such draft statements were circulated to the members of IRAVAC for review and deliberation. The draft statements were revised accordingly, and the final set of 48 statements on vaccinations for eight diseases was voted upon by the IRAVAC group, with a predefined 75% agreement set as a threshold for acceptance. It is envisaged that these consensus statements related to vaccination in AIRD would be helpful not only to rheumatologists in India but also to those in other countries.
Introduction
Infectious illnesses and their consequences are a major cause of sickness and death in people with autoimmune inflammatory rheumatic disorders (AIRD). The increase in susceptibility to infections among these people can be linked mostly to the immunomodulatory effects of the disease and/or the use of immunosuppressive drugs. 1
Vaccination is widely recognised as a safe and cost-effective method of preventing particular illnesses. Vaccination, however, may be less successful in some subsets of people with AIRD due to compromised immune systems. Furthermore, immunisation has the potential to aggravate the underlying AIRD illness.2,3
Evidence-based vaccination guidelines for patients with AIRD were developed by the European Alliance of Associations for Rheumatology (EULAR) task force to assist healthcare professionals dealing with vaccine-related questions in AIRD patients in clinical practice, with the goal of lowering infection-related morbidity and mortality in these patients. 4
In India, the need for guidelines in this area has been felt for a long time due to notable distinctions in infection patterns, vaccine accessibility, and treatment approaches among patients with AIRD compared to other regions globally. With this in mind, the Indian Rheumatology Association Vaccine (IRAVAC) group was formed to develop expert panel consensus statements on vaccination for adults with AIRDs in India. The present report describes the methodology, summary of evidence, and consensus statements on the effectiveness, immune response, and safety of vaccination in adults with AIRD, including individuals on immunomodulatory medications.
Methods
Expert Panel
The IRAVAC group consisted of seven rheumatologists (BGD, VR, SK, RV, KM, VKJ, CG), one epidemiologist (RS), two infectious disease specialists (SS, NM), one clinical virologist (BV), one specialist nurse (SB), and one patient representative (DL). All panel members were required to disclose any potential intellectual or commercial conflicts of interest before taking up their positions. Funding for this academic exercise was through an unrestricted educational grant, with the sponsor having no role in the development of this consensus.
The IRAVAC group defined this initiative’s scope and formulated research questions which were as follows:
What is the efficacy of providing vaccination? What is the safety of providing vaccination?
The IRAVAC group assigned a team of members the task of reviewing the evidence (VR, SK, RS, RV). Research questions were further elaborated upon following the population, intervention, control, and outcomes (PICO) method, which involves combining and adjusting the definitions of population, intervention, comparison, and outcome to serve as search terms (Tables 1 and 2). Accordingly, the search strategy (Supplementary data A-search strategy) was finalised, and a systematic literature search and data extraction were carried out (vide infra).
Vaccines and Diseases Included.

Vaccines and Diseases Not Included.
Another group of members was tasked with reviewing the data extracted and preparing a summary (Supplementary Data- B, Tables 1–14), generating draft statements, and grading the quality of evidence (VKJ, SK, KM, RV, CG, RS, NM). The quality of evidence in support of each statement was evaluated using the evidence‑assessment frameworks prescribed by the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) system. 5
Subsequently, 88 such draft statements generated were circulated to the members of IRAVAC for review and deliberation. Statements were then accordingly kept unchanged, modified, or removed after deliberation among all members that were held virtually. In case of disagreements, the evidence was reviewed, and a consensus was reached. The draft statements were revised accordingly, and the final set of 48 statements (Table 3A–C) were voted upon by the IRAVAC group using Google Forms with a predefined 75% agreement set as a threshold for acceptance. Eventually, all the statements had >75% level of agreement. These meetings were held online.
Search Strategy
The literature search covered MEDLINE (PubMed), EMBASE, the Cochrane Library, ClinicalTrials.gov, the World Health Organization (WHO) International Clinical Trials Registry Platform, conference proceedings from EULAR, American College of Rheumatology (ACR), and IRA, along with the manual examination of reference lists in the selected articles. Details of the search strategy used are available as Supplementary data A-search strategy. Data extraction was carried out utilising Rayyan software.
Results
A total of 48 statements covering eight diseases (Table 3A–C) were approved. The relevant rationale and pertinent literature have been discussed below. The details of the studies can be found in the supplementary material (Supplementary Data- B, Tables 1–14).
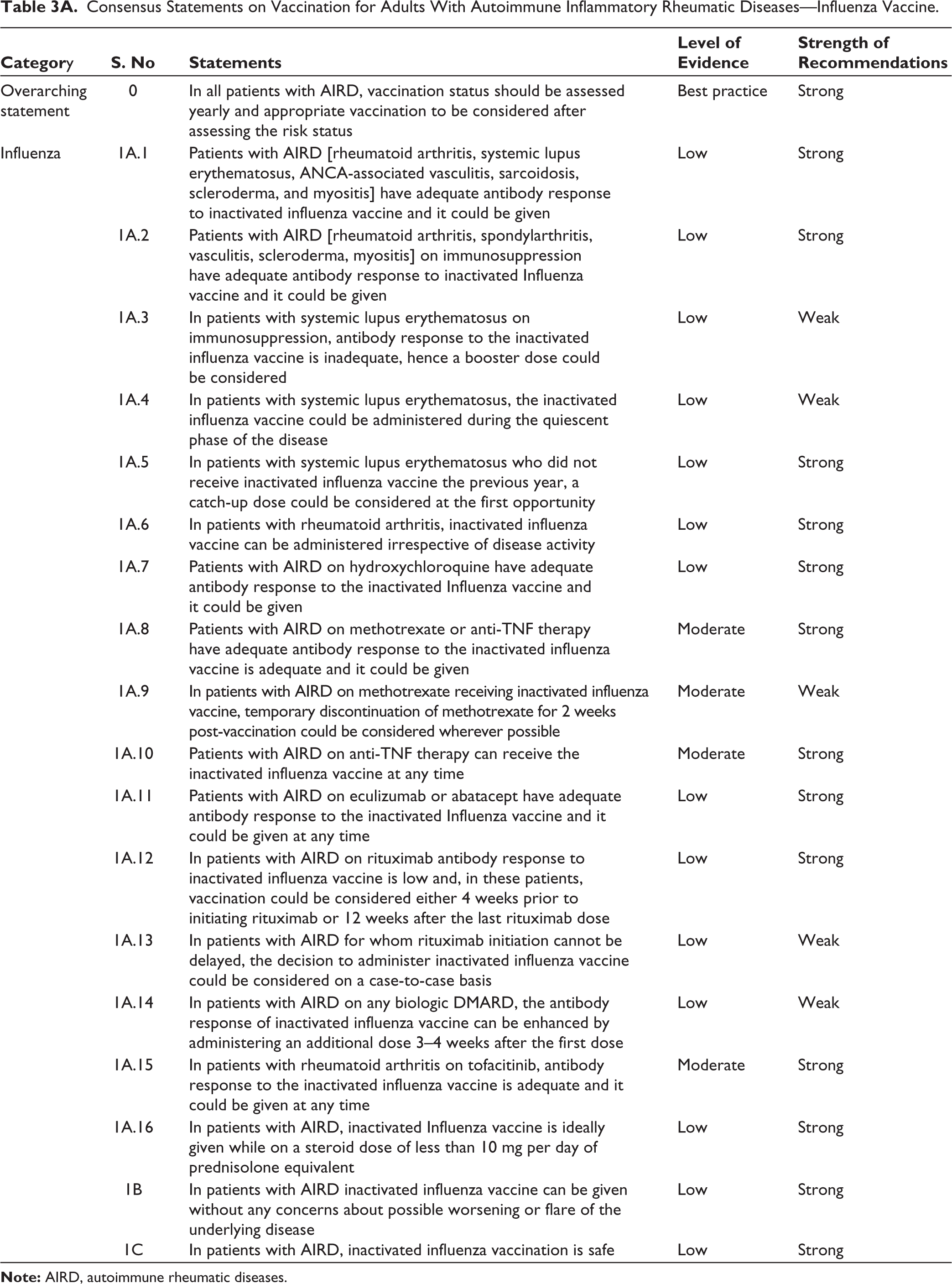
Consensus Statements on Vaccination for Adults With Autoimmune Inflammatory Rheumatic Diseases—Influenza Vaccine.
Consensus Statements on Vaccination for Adults With Autoimmune Inflammatory Rheumatic Diseases—Pneumococcal Vaccine.

Consensus Statements on Vaccination for Adults with Autoimmune Inflammatory Rheumatic Diseases—Other Vaccines.

The Need for Vaccination in Patients with AIRDs
Every year, it is critical to check the immunisation status of patients with autoimmune rheumatic diseases (AIRD). Given the dynamic nature of diseases and vaccinations, this routine examination assists healthcare practitioners in making educated decisions tailored to each patient’s risk profile. Following a thorough evaluation, relevant immunisations might be prescribed to provide the best possible protection against preventable diseases. This proactive strategy not only meets the health requirements of AIRD patients but also adds to overall public health efforts by lowering the risk of vaccine-preventable illnesses among this at-risk population. Regular and tailored vaccine assessments are thus an essential component of comprehensive therapy and preventive actions for people suffering from AIRD.
Influenza Vaccination
A large case-control study 6 revealed that individuals with AIRD exhibited a suboptimal immune response compared to healthy controls, while many smaller case-control studies reported a sufficient response. The immunogenicity was found to be satisfactory in RA patients, regardless of their use of DMARDs or steroids, based on a case-control study. 7 In three smaller case-control studies, the immune response in systemic lupus erythematosus (SLE) appeared to be diminished compared to that of healthy controls but adequate.8–10 A small case-control study of patients with sarcoidosis found a comparable serological response when compared to healthy controls 11 (Supplementary Table 1).
In single-centre prospective controlled research from India, methotrexate at a dose of 15 mg/week did not affect vaccination response to the trivalent influenza vaccine in RA. 12 In another prospective cohort utilising adjuvanted pandemic influenza vaccination, antibody titres were dramatically lowered by treatment with methotrexate (MTX), leflunomide (LEF), or immunosuppressive medications [azathioprine (AZA), mycophenolate mofetil (MMF), and cyclophosphamide (CYC)]. In the same investigation, sulfasalazine (SSZ), and hydroxychloroquine (HCQ) showed no deleterious effect on anti-H1N1 antibody levels. 13 In a prospective cohort study also employing adjuvanted H1N1 influenza vaccination, MTX [subcutaneous] at a mean dose of 15.8 mg/week resulted in 15% lower antibody titres, whereas individuals not on immunomodulators had greater and longer antibody response. 14
There was no difference in antibody response between individuals with and without immunosuppressive medicines in an RCT on patients with Wegener’s granulomatosis who had quiescent illness. 15 Similarly, no effect of immunosuppressive treatment on antibody response was observed in a single prospective study of patients with scleroderma and myositis.16,17 The vaccine committee noted the varied results on the effect of csDMARDs but still decided to recommend vaccination given safety and efficacy (Supplementary Table 2).
Patients with SLE who are receiving immunosuppressive medication have insufficient antibody responses to the inactivated influenza vaccine. In two prospective cohorts and one observational study, immunogenicity was reduced in SLE patients receiving immunosuppressive medications such as azathioprine/Methotrexate/Mycophenolatemofetil/cyclophosphamide/prednisolone. Hydroxychloroquine appears to restore immunogenicity.18,19,20
Low-quality studies showed that the booster [additional dose after 3–4 weeks] was of value in SLE patients not vaccinated the previous year and those on immunosuppressives.19,21
SLE patients with haematologic criteria [especially haemolytic anaemia] or higher ACR criteria were more likely to respond poorly to immunisation. 22 Patients with serologic abnormalities consistent with active lupus (low complement or elevated anti-dsDNA antibodies) had lower antibody titres than controls and other patients, although this was not statistically significant. 8 An observational study found patients with quiescent illness had a trend toward higher efficacy. 23
The panel of experts felt that though the evidence was of low quality but was unanimous in suggesting vaccination during the quiescent phase of the disease.
Given the lower immunogenicity of influenza vaccination in SLE on immunosuppression and during active disease and keeping in mind the benefits of vaccination, the expert panel suggested that a catch-up dose be considered at the first opportunity for those who did not receive inactivated influenza vaccine in the previous year (Supplementary Table 3).
Disease activity in RA had no effect on the immune response to inactivated influenza vaccine, as evidenced by similar seroconversion and seroprotection rates across different disease activity levels. 10 This study, however, investigated a monovalent A/H1N1 influenza vaccine. Further research is needed to confirm these findings with currently available seasonal influenza vaccines. Healthcare providers should encourage and facilitate influenza vaccination for all RA patients, regardless of disease activity.
The effect of hydroxychloroquine on immunogenicity was noted in a prospective cohort study, where no significant reduction in immunogenicity was noted. 13 This is consistent with the immunomodulatory rather than immunosuppressive action of hydroxychloroquine (Supplementary Table 4).
The current evidence suggests that the effects of methotrexate on the immunogenicity of influenza vaccine are complex and conflicting. While a meta-analysis 24 and cohort study 25 show no significant impact; others including a meta-analysis demonstrate a decrease in immunogenicity. 26
Anti-TNF medications do not decrease the effectiveness of influenza vaccination. 27 While some studies have suggested a slight decrease in vaccine response,28-31 these findings have not been consistently observed in meta-analyses. 26 Additionally, research indicates that etanercept may have even less impact on vaccine immunogenicity 13 (Supplementary Table 4).
Two RCTs demonstrated that temporarily stopping MTX for 2 weeks after receiving the quadrivalent influenza vaccine significantly improves the immune response in patients with RA, without worsening RA symptoms. 32 This finding was further supported by another RCT, which showed that temporary MTX discontinuation also enhances the immune response to the trivalent seasonal influenza vaccine in RA patients. 33 Individuals with AIRD who are receiving methotrexate and the inactivated influenza vaccine may hence consider temporarily discontinuing methotrexate for 2 weeks after vaccination.
Anti-TNFs do not show a reduction in immunogenicity of influenza vaccination. 27 A modest reduction in immunogenicity of influenza vaccine was reported in a few studies28-31 though the results were not consistent in meta-analysis. 26 Studies also show less effect on vaccine immunogenicity when on etanercept. 13
A small case-control study showed that secukinumab does not significantly affect the immune response to TIV in patients with psoriatic arthritis (PsA) or AS compared to healthy controls. 34 Abatacept had similar seroconversion and geometric mean titre (GMT) of antibodies to those treated with MTX and more than those treated with RTX. 14 There is a need for more data; however, with the available evidence, it was suggested that inactivated influenza vaccine could be given at any time during treatment with non-TNF biologics such as secukinumab and abatacept (Supplementary Table 4).
In a prospective controlled cohort study comparing vaccine response to influenza vaccine was compared in RA treated with rituximab, RA treated with DMARDs alone, and healthy controls. Rituximab-treated patients had a significantly lower response rate to the California antigen (H1N1) (21%) compared to non-rituximab-treated RA patients (67%) and healthy controls (57%). Response rates for the B and H3N2 antigens were similar across all groups. 35 Another prospective cohort study found that humoral responses to trivalent influenza vaccination were significantly lower in RA patients treated with rituximab compared to those receiving MTX and healthy controls. In the rituximab group, humoral responses were partially restored 6–10 months after rituximab treatment. Previously vaccinated patients in the rituximab group had higher pre- and post-vaccination antibody titres for the A/H1N1 strain. 36 Administration of the vaccine after 12 weeks of RTX had a higher likelihood of having seroconversion [80% vs. 25% for <12 weeks, P = .054]. 37
Yearly influenza vaccination does elicit some response in patients treated with rituximab, even though the response may be lower than in healthy individuals. Given this, the panel recommended that influenza vaccination can be considered on a case-to-case basis in which initiation of rituximab cannot be delayed.
A prospective cohort study showed that after receiving two doses of the vaccine, patients with AIRD on biological DMARDs achieved an equivalent immunological response compared to healthy controls receiving a single dose of the vaccine. Based on this, the panel suggested an additional dose to enhance antibody response can be considered. 13
The immunogenicity of influenza vaccine in patients with RA on tofacitinib was evaluated in one RCT and long-term extension study of the same. Tofacitinib-treated patients taking background methotrexate had a decrease in response, although the proportion of patients achieving satisfactory immune responses was generally similar in all exposure subgroups (Supplementary Table 5).
Among current tofacitinib users, temporary discontinuation of tofacitinib for vaccination had little impact on immunogenicity. Hence, tofacitinib has a limited effect on influenza vaccine responses and vaccination could be undertaken yearly (as recommended), regardless of tofacitinib therapy38,39 (Supplementary Table 6).
The effect of steroids on the immunogenicity of the influenza vaccine was deduced from three prospective studies. None of the studies evaluated this as a primary objective. One study showed steroids at 0.15 mg/kg/day impaired immune response, 19 while three other studies showed that low-dose steroids did not impair antibody response.
A multivariate analysis from a prospective cohort study showed that after controlling for confounding factors, steroids at doses below 10 mg/day did not show a significant impact on vaccine response. 13 Similarly, glucocorticoids (GCs) at a mean dose of 7.4 mg/day did not significantly impair antibody response even when separating for doses <10 and ≥10 mg/day (P = .11) for H1N1 vaccination. 14 More research is, however, needed to clarify the relationship between different types and doses of steroids and vaccine effectiveness in various populations.
Disease flare was assessed in terms of worsening of disease activity measures, erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP); increase in the titre of autoantibodies, and increase in the dosage of GCs. Meta-analysis in SLE and RA showed no increase in disease activity measures post-vaccination.28,40 Two studies on RA patients showed a statistically significant reduction in disease activity after vaccination.10,35 Some studies also reported flares after vaccination, but most lacked comparators. Change in the presence or level of autoantibodies was reported in many studies. Most found no significant changes, but two studies reported statistically significant increases in some autoantibodies after vaccination.23,41 Four studies reported no change in steroid dose or steroid usage after vaccination.14,17,42,43 Overall, these findings suggest that influenza vaccination is safe for patients with AIIRD and does not appear to increase the risk of disease flares (Supplementary Table 7).
AIIRD patients experience slightly higher rates of mild adverse events (AEs) compared to healthy controls,10,28 particularly systemic reactions such as myalgia and arthralgia. Local reactions such as pain/swelling/itching are more variable, with some studies showing higher rates44,45 in AIIRD and others showing lower rates.20,29 No serious AEs or deaths attributable to vaccination were reported in any study. Overall, inactivated influenza vaccination is safe for AIIRD patients (Supplementary Table 7).
Pneumococcal Vaccination (Supplementary Table 8)
There are two types of pneumococcal vaccines approved for use in India to prevent pneumococcal disease: the conjugated 13-valent pneumococcal conjugate vaccine (PCV13) and the 23-valent pneumococcal polysaccharide vaccine (PPSV23). It is recommended that immunocompromised individuals receive one dose of PCV13 followed by PPSV23 at least 8 weeks later [prime-boost strategy].
The pneumococcal vaccine demonstrated immunogenicity in individuals with SLE. 46 Patients with early disease onset, older age, high disease activity, and immunosuppressive therapy had poor immunogenicity. 46 In terms of immunogenicity related to vaccine types, SLE patients who received PPSV23 exhibited a robust antibody response similar to that of healthy controls, whereas SLE patients who received PCV12 displayed a reduced antibody response, although it still provided protection, in comparison to healthy controls.46–48
Among SLE patients with stable disease, and even in active disease, pneumococcal vaccines were found to be adequately immunogenic, either PPSV23 or PCV13-PPSV23 can be considered. 49 Pneumococcal vaccination is recommended for all SLE patients, particularly before initiating immunosuppressive therapy, regardless of their disease activity level. 46
Considering the increased susceptibility of SLE patients to pneumococcal infection, it is crucial to adopt preventive measures, including immunisation, across all age groups. In a meta-analysis study, high ESR, old age, high disease activity, and immunosuppressive therapy were reported to be predictors of poor immunogenicity. This is particularly vital for individuals undergoing immunosuppressive therapy. The recommendation is to administer immunisation before initiating treatment to enhance overall safety. 46
Pneumococcal vaccine (PPSV23) was found to be immunogenic in RA patients.47,49 Patients with RA, whether receiving immunosuppressive therapy or not, and receiving pneumococcal vaccination (PCV13), exhibited comparable safety and immunogenicity to those of healthy controls.50,51 Therefore, regardless of the immunosuppressive treatment, RA patients should be vaccinated.50,51
Utilising the prime-boost vaccine approach (PCV13 followed by PPSV23) in RA patients receiving immunomodulatory therapy shows both safety and the ability to generate an immune response, underscoring the importance of incorporating pneumococcal vaccination (either PCV or PPSV) as a routine procedure for all RA patients. 47
PCV7 has been established as both safe and immunogenic for individuals with vasculitis who are on conventional treatment therapy. The potential for impaired immune response when treated with DMARDs such as AZA, MTX, or CYC, has been noticed but not with prednisolone. Therefore, it is recommended that all vasculitis patients receive pneumococcal vaccination (PCV7). 52
The pneumococcal vaccine has been demonstrated to be safe and effective in individuals with primary Sjogren syndrome (SJS). Additionally, PCV13 shows immunogenicity in SJS patients who are not on DMARDs. Therefore, it is advisable to administer vaccination to SJS patients before commencing methotrexate therapy. 53
The pneumococcal vaccination (either PCV13 or PPSV23) produced a sufficient antibody response in systemic sclerosis (SSc) patients who were not using disease-modifying anti-rheumatic drugs (DMARDs), but a reduced response was observed in those receiving DMARDs. Moreover, the choice of vaccination, either PCV13 or PPSV23 did not impact the antibody response in SSc patients. 54
AIRD patients such as RA, AS, and PsA when treated with MTX exhibit a reduced immune response to pneumococcal vaccination, specifically the PCV7 vaccine.49,55,56 RA patients undergoing MTX therapy and receiving PCV13 experience a reduction in antibody response and functionality.50,56,57
The concurrent use of MTX alongside tacrolimus, Tocilizumab, TNF inhibitors, rituximab, and tofacitinib led to a reduced immune response to pneumococcal vaccination.50,51,58–61 Additionally, studies support the administration of the pneumococcal vaccine before starting MTX in RA.46,62
The optimal suggestion is to give all pneumococcal vaccines at least 2 weeks before starting any immunosuppressive therapies. This is especially important for methotrexate, as studies have demonstrated its potential to reduce the immune response to pneumococcal vaccination. 52 In a systematic review and meta-analysis, immunosuppressive medication, that is, methotrexate impaired the serologic response to pneumococcal vaccination.62,63
The immunogenicity of the pneumococcal vaccine in the presence of TNFi was investigated in one systematic review and meta-analysis, as well as in three RCT and four case-control studies. Three RCTs and several lower-quality studies indicate a satisfactory immune response to pneumococcal vaccines, including both PPSV23 and PCV7, in patients with AIRD, with a focus on those with RA, SPA, and PsA, who are undergoing TNFi treatment.50,51,55,58,59
Around four studies shown the impact of RTX on the immunogenicity of pneumococcal vaccine in patients with AIRD. Findings from an RCT demonstrated a modest antibody response of 25% among RTX-treated patients (only four out of 63 received RTX), compared to an 89% response in participants treated with bDMARDs with other modes of action. 64 As per a case-control study, only 10.3% of patients on RTX monotherapy and none of the patients on RTX combined with MTX demonstrated a positive antibody response for both serotypes, in contrast to 17.6% and 50% for abatacept and tocilizumab, respectively. 59 Interestingly a study cohort of AIRD patients on bDMARDS (including RTX) achieved functional antibody phagocytic response after prime-boost strategy (PCV13+PPSV23). However, most pneumococcal vaccines administered to patients using rituximab were given within the recommended timeframe of 4 weeks before and 6 months after the rituximab treatment. 62
In an RCT evaluating the effect of baricitinib on the immunogenicity of pneumococcal vaccination in AIRD patients, among the 106 RA patients who received PCV13 while being treated with baricitinib (with 90% of them also taking baricitinib in combination with methotrexate), nearly two-thirds of the participants attained favourable humoral and functional responses to PCV-13 vaccination. 65
The effect of (JAK) inhibitor on the immunogenicity of pneumococcal vaccination in AIRD patients was investigated in two RCTs involving a total of 589 RA patients. In one of these trials, a lower percentage of patients receiving tofacitinib (45.1%) achieved satisfactory pneumococcal responses to PPSV23 in comparison to those who received a placebo (68.4%), and the pneumococcal antibody titres were notably reduced with tofacitinib, particularly when used in combination with methotrexate. However, in another part of the same study, no significant difference in satisfactory response rate between continuous and withdrawn patients treated with tofacitinib and methotrexate (75.0% and 84.6%, respectively) was observed. 34
As per a case-control study conducted on RA patients, a similar antibody response was noticed between the tocilizumab and the control group. However, it is suggested that tocilizumab used in combination with MTX may result in reduced antibody response. 61 Similarly, it was also observed that the humoral immune response to PPSV23 remained largely unaffected by tocilizumab treatment in patients with AIRD. 32
The evaluation of the immunogenicity of the prime-boost strategy was done in five studies out of which two were RCT and three were cohort studies. One RCT found that the strategy was immunogenic in RA patients taking csDMARDs and bDMARDs (except rituximab), while another RCT involving patients with stable SLE did not find it superior to the polysaccharide vaccine alone.64,65 A cohort study on AIRD patients on bDMARDs showed a functional antibody phagocytic response after the prime-boost strategy. 62
The majority of patients undergo conventional therapeutic care, and research has explored the occurrence of disease flares in AIRD following vaccination. In a study involving 73 patients with SLE, after receiving the PPSV 23 vaccine, it was observed that while 8% of patients experienced a slight increase in their SLEDAI score, the change was not statistically significant. 66 Studies investigating the treatment of biological medications such as tocilizumab, anti-TNF (Adalimumab), and Abatacept in RA patients, did not document any instances of disease flares following the PPSV 23 vaccination.62,67 Although the primary focus of most studies on PCV13 vaccination in RA and SLE was its immunogenicity, none of them reported significant safety concerns or disease flares, and even with more than 50% of patients being on steroids, the presence of steroids did not adversely impact post-vaccination disease flares. Furthermore, when assessing serological changes via anti-dsDNA levels and complements C3 and C4 in SLE for pneumococcal vaccination (PPSV23), no significant alterations were observed following vaccination. 68 Thus, Pneumococcal vaccines PPSV 23 and PCV 13 appear to be safe for patients with AIRDs, regardless of their use of steroids, DMARDs, or biologics.
The primary vaccine-related AE encountered was localised injection site reactions, consistent with rates seen in the general population, while some studies documented transient diffuse musculoskeletal pain and muscle tenderness that resolved spontaneously. No serious adverse side effects were noted nor any death due to vaccination. Available pneumococcal vaccines PPSV 23 and PCV 13 appear to be safe for patients with AIRDs concerning vaccine-related side effects.32,33,52,53-67,70-82 While the quality of evidence concerning pneumococcal vaccination in AIRDs is robust, a significant constraint is that the majority of studies have focused solely on patients with RA and SLE, with findings being applied to other AIRDs.
Hepatitis A and Hepatitis B Vaccination
Among the AIRDs, immunogenicity of the Hepatitis A virus (HAV) vaccine has been studied only in rheumatoid arthritis (RA) with adequate antibody response. A single dose of the vaccine is insufficient while two doses of the vaccine administered 6 months apart showed an 83% seroconversion rate. 81 A three-dose regimen had superior and comparable seroconversion rates in both RA patients and healthy controls (99% vs. 100%). 82 Patients on tumour necrosis factor (TNF) inhibitors showed a better antibody response than other medications. There was no association between HAV vaccination and disease activity. Pain (6%) and haematoma (17%) were noted as adverse effects. Information about the safety, efficacy, and cost-effectiveness of HAV vaccination is lacking (Supplementary Table 9).
In individuals with AIRD, sufficient antibody responses to the standard Hepatitis B vaccine were observed. The immunogenicity and safety of the recombinant hepatitis B virus (HBV) vaccine in patients with AIRD was evaluated in seven studies.83–89 Among these studies, three reported high levels of seroconversion, ranging from 82% to 94%.83–85 However, the remaining studies reported lower levels of seroconversion in specific subgroups of RA patients, depending on the type of immunosuppressant used. Additionally, one study found zero seroconversion in patients with sarcoidosis 89 (Supplementary Table 10).
Richi P et al. 83 found significant seroconversion rates in both biological DMARDs (81.8%) and synthetic-DMARDs (93.8%) users. It is important to note that this study used additional vaccine cycles when patients did not achieve seroconversion with one cycle. In contrast, Intongkam S et al. 85 reported much lower seroconversion rates after 8 weeks of vaccination in biological (50%) and synthetic-DMARDs (69.7%) users. The variability in seroconversion rates was highly influenced by the specific immunosuppressants taken by the patients. The expert panel chose to incorporate higher doses and additional treatment cycles based on recommendations available in patients undergoing dialysis.
Extensive clinical trials in individuals with AIRD show that the routine Hepatitis B vaccine is safe, with no major side effects. Safety of administering the HBV vaccine was examined in two studies. Erkek E et al. 87 found no systemic side effects and identified pathergy in four out of 13 patients. Kuruma KA et al. 88 observed no significant changes in SLEDAI (Systemic Lupus Erythematosus Disease Activity Index) scores but did observe a significant increase in the prednisolone dose and reported flares in three out of 28 patients compared to the baseline. 87
Anti-rabies Vaccination (Supplementary Table 11)
The response to rabies vaccination using the purified vero cell culture vaccine was investigated in a case-control study that included patients with AIRD, such as RA and lupus, as well as other immunosuppressed individuals. In patients with definitive dog bites necessitating vaccination, the study evaluated vaccine responsiveness through the measurement of rabies-neutralising antibodies (NAb) and found that all subjects achieved NAb titres exceeding 0.5 IU/ml after the second vaccination dose on Day 14. All patients demonstrated a protective response and no adverse effects were observed in the immunosuppressed group, ultimately concluding that the rabies (PVCC) vaccine is both safe and elicits an adequate immune response. 90 The expert committee was of the opinion that rabies vaccination as per post-exposure prophylaxis in an immune-compromised patient protocol of the Government of India is to be followed after a dog bite. In immune-compromised people, GOI recommends antibody titre 14 days after completion of the standard 5-dose course to assess the need for revaccination.
Herpes Zoster Vaccination (Supplementary Table 12)
The recombinant non-live zoster vaccine has been studied in two phase 3 trials in patients over 50/70 years old (ZOE50/70 studies). In patients with immune-mediated diseases not on immunosuppressive therapies at enrolment, a post hoc analysis demonstrated a 90.5% efficacy for the adjuvant recombinant zoster vaccine in patients with spondyloarthritis, RA, psoriasis, and celiac disease and showed a similar incidence of serious AEs between vaccine and placebo recipients, suggesting potential benefits of RZV vaccination for individuals with these conditions. However, in patients with pre-existing pIMDs, ZOE-50/70 studies were not designed to specifically measure the effectiveness and/or safety. 91
Tetanus Vaccination (Supplementary Table 13)
The tetanus vaccine’s efficacy was investigated in patients with sarcoidosis alongside other AIRDs, involving 48 patients and 31 controls, revealing that it was less effective in patients with sarcoidosis versus healthy controls (50% vs. 23%, P = .019), which may be due to their hypo-responsive humoral immune system, as indicated by lower lymphocyte levels (P = .013). Currently, there is a lack of data on its effectiveness, AEs, and cost-effectiveness in both sarcoidosis and other AIRDs. 92
Yellow Fever Vaccination (Supplementary Table 14)
Immunogenicity following the first dose of 17DD/17D-204 yellow fever vaccines in individuals with AIRDs has been investigated in several studies, with available data on immunogenicity assessed using the plaque reduction and neutralisation test (PRNT) for conditions such as RA, SLE, scleroderma, primary SJS, spondyloarthritis, and vasculitis. Certain studies with more extensive participant pools indicate a potential decrease in immunogenicity among patients with AIRD, ranging from 71% to 87% when contrasted with the 95% observed in healthy controls.93–95 Conversely, other specific studies indicated comparable seropositivity rates for the 17DD/17D-204 yellow fever vaccine in both populations with AIRD and healthy controls. 96
In a study conducted over a median duration of 21.1 years (with a range of 14.4–31.3 years) and had patients with AIRD and solid organ transplantation, the rate of seropositivity persistence was found to be 88% among individuals receiving immunosuppressive agents, which closely matched the 89% observed in the control group. 97 In another study, the persistence of seropositivity was documented in 71%–76% of both healthy controls and RA patients, even after more than a decade of getting vaccinated with the yellow fever vaccine.97,98
A single study is available that provides limited data on the effect of csDMARDs and bDMARDs on the immunogenicity of 17DD/17D-204 yellow fever vaccines in RA patients. 97 The data indicates that the vaccine’s immunogenicity is influenced by the duration of use of csDMARDs and/or bDMARDs. When used for <5 years, 100% of those using csDMARDs and 90% of those using bDMARDs demonstrated immunogenicity. However, the rate of seroconversion decreased to 76% after 5 or more years of corticosteroids plus bDMARD use but remained similar to the control group in individuals receiving only csDMARDs (89%). 98
The data on adverse effects of the 17D-204 yellow fever vaccine in AIRD is limited.94,97 Both mild and severe adverse effects, whether local or systemic, exhibited a similar incidence in individuals receiving corticosteroids and healthy controls. In a study investigating vaccine-induced viremia, it was observed that the peak yellow fever viral RNA levels in patients with AIRD occurred slightly later and at lower levels on Day 6 (47%) compared to Day 5 (78%) in healthy controls. 94 These studies neither included individuals with AIRD receiving high levels of immunosuppressive treatment with cyclophosphamide, tacrolimus, mycophenolate mofetil, cyclosporine, azathioprine (>2 mg/kg/day), sirolimus, methotrexate (>20 mg/week) and prednisone (≥20 mg/day) nor those concomitantly taking bDMARDs. 94 Moreover, revaccination with 17DD/17D-204 yellow fever vaccines exhibited comparable immunogenicity and safety in individuals receiving infliximab when compared to healthy controls. 95
Due to the limited availability of comprehensive safety data in AIRD, yellow fever vaccines have not received approval for use in these conditions from both EULAR and ACR. The authors, comprising rheumatologists, vaccinologists, and infectious disease experts, propose that the decision on yellow fever vaccination should be made collaboratively by the treating rheumatologist and the patient. This decision should consider the balance between potential risks and benefits, considering the patient’s current disease status, the level of immunosuppressive treatment, and the prevalence of yellow fever in the travel destination. If deemed necessary, revaccination might be recommended after 10 years of the initial dose, and it may be advisable to test for seropositivity a decade after the first yellow fever vaccine to determine the necessity for revaccination.
Other Vaccines
Data on vaccination for Japanese encephalitis virus, typhoid, human papillomavirus, diphtheria, pertussis, rubella, neisseria meningtiditis and polio was not included due to a paucity of relevant data.
Conclusion
This consensus document developed by the IRAVAC is a guide for optimal vaccination for adults with AIRD in India. In all patients with AIRD, vaccination status should be assessed yearly and appropriate vaccination should be considered after assessing the risk status. Vaccination with inactivated vaccines is safe and immunogenic in AIRD patients. Antibody response is preserved while on most immunosuppressive medications when vaccination is carried out preferably during a stable phase of the disease with no major risk of disease flares. In conclusion, this expert panel sincerely believes that the present consensus statements related to vaccination in AIRD would be helpful not only to rheumatologists in India but also to other countries.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
The expert panel wishes to acknowledge an unrestricted educational grant from Pfizer Vaccine Limited for the development of these consensus statements. We would like to acknowledge Scientimed Solutions Pvt. Ltd. for their assistance in developing this manuscript. The funding for this assistance was provided by the Indian Rheumatology Association. We would like to acknowledge Centre for Chronic Disease Control (CCDC) for assistance in the Systematic literature review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval was not obtained from the local Ethics Committee or Institutional Review Board as it is a consensus statement.
Funding
An unrestricted education grant from Pfizer Vaccine Limited supported the work. The funder had no input in the content of this manuscript.
Patient Consent
Consent was not needed/not applicable as this did not include any patient related data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
