Abstract
Objective:
The study aimed to evaluate serum biomarkers (Galectin-9, TNFRII, APRIL, CXCL10, and BAFF) for their utility in identifying disease activity in idiopathic inflammatory myopathies (IIM)
Methods:
We enrolled adult patients who satisfied the 2017 EULAR/ACR criteria for IIM and categorised them into active (n = 16) and inactive (n = 24) subgroups. Inactive disease was defined as the absence of global and extramuscular activity, stable manual muscle testing 8 (MMT-8), and normal muscle enzymes over at least 6 months. We measured serum biomarkers by enzyme-linked immunosorbent assay (ELISA) at baseline, 3 and 6 months. We employed Spearman’s correlation, ROC curves, and multiple linear regression for evaluation.
Results:
We included 40 patients (mean age 38 ± 10 years; median disease duration 27.5 months). The majority had dermatomyositis (DM) (62.5%), followed by anti-synthetase syndrome (ASSD) (27.5%) and polymyositis (10%). Galectin-9, APRIL, and TNFRII levels were significantly higher in active disease (P < .05). Galectin-9 demonstrated a modest correlation with MMT-8 changes (r = 0.534).
A combination of Galectin-9, TNFRII, APRIL and creatine kinase (CK) levels showed the highest ROC AUC (0.953) to detect active disease. During the study, four patients initially categorised as ‘inactive’ patients exhibited improvement of 10% in MMT-8 over 6 months. To enhance sensitivity for detecting these clinically active diseases, we assessed Galectin-9 at lower cut-offs. Levels of 1146 pg/mL and 1109 pg/mL improved sensitivity to identify active disease to 81% and 87%, respectively, without compromising specificity.
Conclusion:
Serum biomarkers, Galectin-9, TNFRII, and APRIL, may help in differentiating between active and inactive disease in adult IIM. Notably, persistently elevated Galectin-9 levels, even in clinically quiescent disease, may signify ongoing activity.
Introduction
Traditional biomarkers in idiopathic inflammatory myopathies (IIM) including serum creatine kinase (CK), serum alanine transaminase (ALT), serum aspartate transaminase (AST) and lactate dehydrogenase (LDH) are important in diagnosis, treatment and prognostication of IIM. The International Myositis Assessment and Clinical Studies Group (IMACS) and The Paediatric Rheumatology INternational Trials Organisation (PRINTO) core set criteria for disease improvement includ changes in these biomarkers.1,2 The problem with these biomarkers is variability in their levels with gender, age, exercise intensity, and muscle mass. 3 There is also an inconsistent correlation of disease activity with CK across studies in IIM.4–6 In addition, these traditional biomarkers are not specific to IIM and can also be elevated in other forms of myopathies like metabolic, toxic or endocrine causes. 7
Serum troponin and ferritin which are easily available have also been used as biomarkers in certain situations. Ferritin was shown to be a reliable biomarker in assessing disease severity and response to treatment in anti-MDA5 positive dermatomyositis (DM). 8 In a few studies serum troponin, and CK to CK-MB ratio were shown to be a reliable biomarker for cardiac involvement.9,10 However, none of these have been proven to be reliable in assessing overall disease activity in IIM. Hence, there is need for biomarkers which have good sensitivity and specificity in accurately reflecting overall changes in diseases status.
In the past decade, it has been known that type 1 interferon mRNA expression has increased in peripheral blood and muscle tissue of IIM.11–13 It is also shown that interferons trigger the production of chemokine interferon-γ inducible protein 10 kDa (CXCL10) and Galectin9 by both immune and non-immune cell types.14,15 Further in muscle tissue from a patient with DM, TNF alpha and its receptors, TNFRI and TNFRII, were expressed.16,17 Several studies have thus implicated TNF alpha 308A polymorphism as a risk factor for juvenile DM (JDM). 18 Much work on these new biomarkers has been carried out in JDM. Wienke et al. have demonstrated that in JDM Galectin‐9, and CXCL10 can reliably differentiate active from inactive disease.19,20 This finding was validated in a multicentre JDM cohort, 21 where it was demonstrated that Galectin-9, CXCL10, and TNFRII distinguished between active disease and remission in a cross sectional cohort of JDM patients. 21
Amongst other new biomarkers, B cell activating factor (BAFF) is elevated in IIM, with modest correlations with CK levels, particularly in certain myositis-specific antibodies (MSA) subgroups. It has been reported that BAFF and proliferation-inducing ligand (APRIL) levels in the serum are associated with severe interstitial lung diseases in JDM.22,23 Of all the new biomarkers, the most common discriminative were Galectin-9 and CXCL10, and both were identified as promising prognostic biomarkers for adult patients with DM.24,25 However, studies looking at the role of these biomarkers longitudinally in adult IIM other than DM are sparse. Hence, we sought to assess the performance of Galectin‐9, CXCL10, TNFRII, BAFF and APRIL in determining clinical disease activity and disease flare in a cohort of adult IIM.
Methods
This observational cohort study was carried out from August 2019 to August 2021 at a tertiary care teaching hospital in India. It involved consecutive adult patients who met the definite IIM score of 2017 EULAR/ACR criteria for IIM, 26 and were part of the hospital’s myositis registry. The study excluded patients diagnosed with inclusion body myositis (IBM), overlap myositis, those with chronic kidney disease or a coexisting malignancy, and pregnant individuals. The collected data encompassed demographic details such as age, gender, age at onset of symptoms, and age at diagnosis. Additional data on disease subtypes, clinical features, serological markers, and treatment approaches were also systematically recorded. All the patients received treatment as per current standard of care. At baseline (time of enrolment) patients had either active or inactive disease status. Inactive disease subgroup was defined as a lack of evidence of activity as assessed by global and extramuscular assessments, stable muscle strength defined as <15% change in manual muscle testing 8 (MMT-8), and normal muscle enzyme levels over at least a 6-month period. 27
Antinuclear antibody (ANA) was tested by indirect immunofluorescence using HEp-2 2010 cell line (Euroimmune, Germany) and MSA and myositis-associated antibodies (MAA) were tested using Euroline myositis profile 4 (Euroimmune Germany). Serum AST, ALT, CK and LDH were done at baseline for all patients, and at 3 and 6 months for those in the active disease group. Similarly, for estimation of biomarkers (Galectin‐9, CXCL10, TNFRII, BAFF and APRIL) serum samples were collected at enrolment for all patients, and at 3 and 6 months in those with active disease. Biomarkers were also assessed at the time of disease flare for inactive disease groups. Serum samples (5 mL each at enrolment, 3 and 6 months) were stored at –40°C immediately after collection and were not thawed till the analyses at the end of the study. Galectin-9, CXCL10, TNFRII, BAFF and APRIL, were measured by enzyme-linked immunosorbent assay (ELISA) using commercially available kits (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s protocol (catalogue numbers of kits mentioned in Supplementary Table S1). All patients were followed up to assess clinical disease activity every 3 months for at least 1 year.
We conducted ROC analysis to assess the performance of biomarkers to predict active disease. One of the criteria in the definition of inactive disease (at the start of the study) was <15% change in MMT-8 over the preceding 6 months. However, we found that MMT-8 in a few patients categorised as inactive disease improved during the study. We assessed the improvement status of the subjects by considering specific cut-off values of Galectin-9 derived from the ROC analysis to redefine active disease. Galectin-9 was chosen for this purpose based on the observation that it was elevated at the time of flare in a patient of inactive disease and also because it had the highest coefficient of regression (B) with respect to MMT-8 in the regression analyses.
The study was approved by the Institutional Ethics Committee (hidden for anonymity). The study was conducted as per the Indian Council for Medical Research (ICMR) Guidelines for Biomedical Research on Human Participants and in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Statistical Analysis
The normality of data was assessed using the Kolmogorov–Smirnov test. An unpaired t-test and Mann Whitney U test were used to compare independent continuous variables at two-time points based on their normality, while the Kruskal Wallis ANOVA with post hoc Dunn’s multiple comparisons test was used to compare samples at three-time points. Correlation of change in biomarkers in the active disease group with the change in muscle strength and function parameters was assessed using Spearman’s rank correlation. The performance of the muscle enzymes (CK, AST, ALT, LDH) and biomarkers to predict active disease was assessed by plotting an ROC curve. Sensitivity and specificity for the muscle enzymes were selected based on the value of the cut-off specified by the laboratory (CK-145 IU/L, AST – 35 IU/L, ALT – 35 IU/L, LDH – 378 IU/L). For the newer biomarkers, cut-off values were selected based on the Youden index. 28 The performance of the combination of biomarkers and CK to predict active disease was assessed by plotting a ROC curve of the predicted probabilities following binary logistic regression. Multiple linear regression models were built to identify the disease factors [MMT-8 and Extramuscular Global disease activity (Extramuscular global assessment)] that predict biomarker levels at baseline, 3 and 6 months. Another multiple linear regression model was built to control for the IIM subtype along with disease factors to predict biomarker levels. Data were analysed using SPSS version 19.0; the significance level was set at <5%. Graphs to depict biomarker levels were generated using GraphPad Prism 10.
Results
Demographic, Clinical and Serological Characteristics in Patients With IIM
Of 40 patients included in the study, 16 were categorised as active and 24 as inactive disease at enrolment. The mean age of the total study population was 38 ± 10 years and the median disease duration was 27.5 months (IQR 12–68). The majority [n = 25 (62.5%)] were DM, 11 (27.5%) were anti-synthetase syndrome (ASSD), and four (10%) patients were polymyositis. Baseline MMT-8 (IQR) among active and inactive disease subgroups was 56 (47.5–63) and 74 (69–78), respectively. Other baseline characteristics are shown in Table 1.
Baseline Characteristics Among the Study Population.
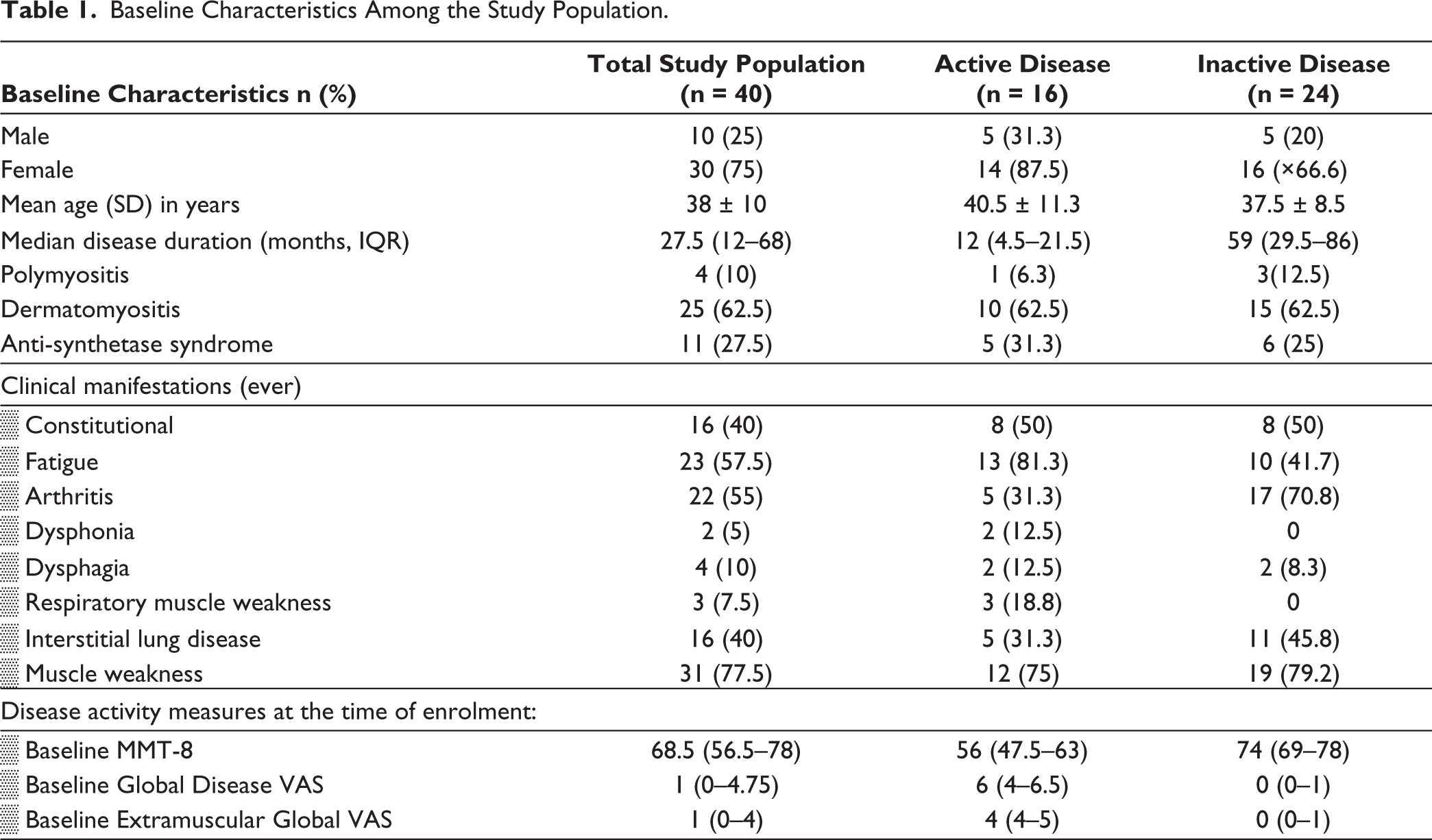
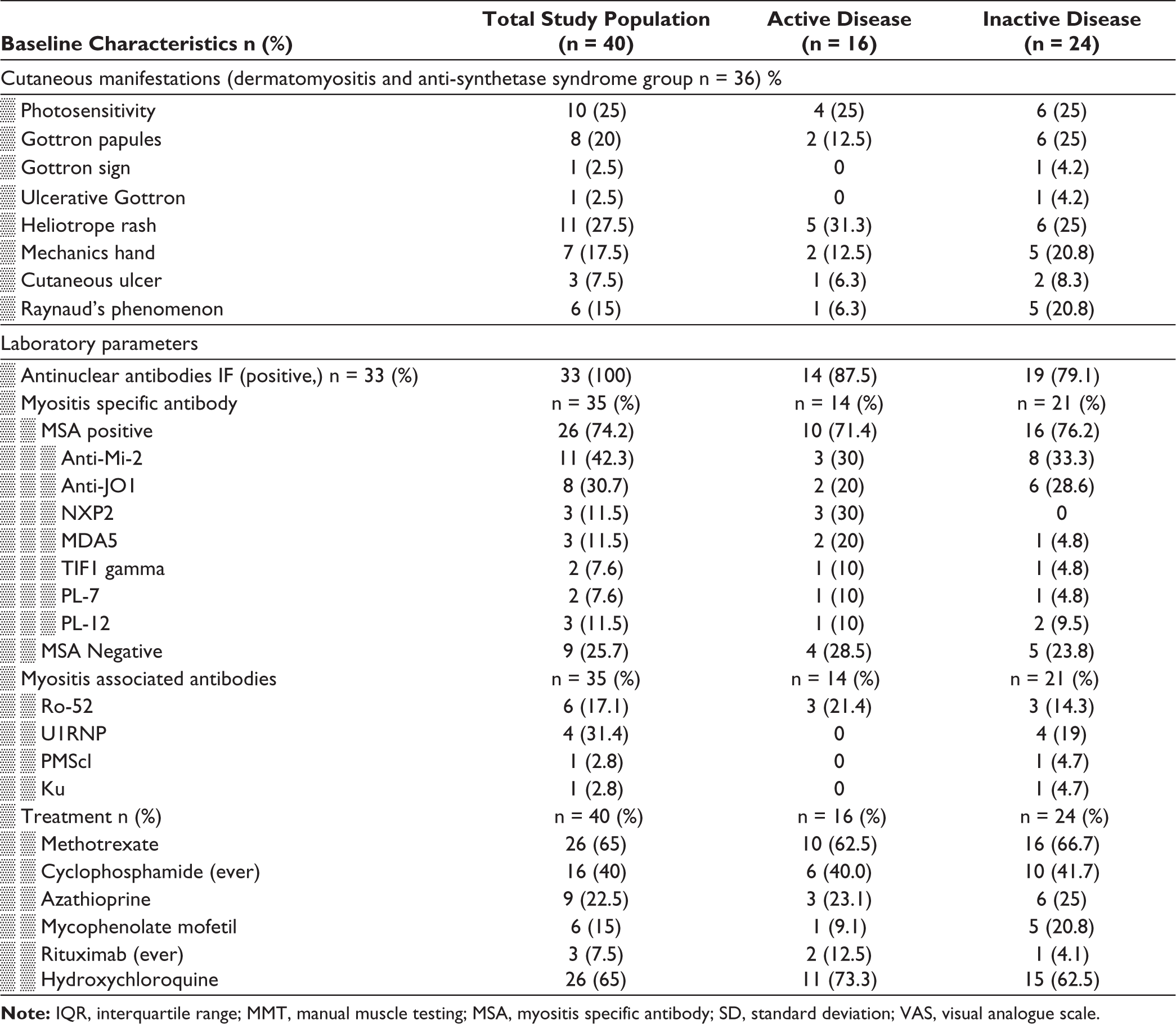
Anti-Mi-2 antibody (27.5%) was the most common MSA, followed by anti-Jo-1 antibody (22.9%). MSAs were negative in 25.7% (n = 9) of patients. Among the MAA, the anti-Ro-52 antibody was the most prevalent, seen in 17.1% (n = 6). Most of the patients were currently on methotrexate, followed by azathioprine. Seven patients (17%) had received pulse methylprednisolone, while five patients (12%) had received intravenous immunoglobulin, and 16 (40%) patients had received cyclophosphamide at least once.
Comparison of Biomarker Levels Among the Active and Inactive Disease Subgroups
Median baseline levels of Galectin-9 (2235.19 pg/mL vs. 1455.47 pg/mL), APRIL (0.806 ng/mL vs. 0.381ng/mL) and TNFRII (15.60 ng/mL vs. 8.95 ng/mL) were significantly higher in the active disease subgroup than in the inactive disease, P < .05 (Figure 1). Among those with active disease, levels of these biomarkers decreased over time with treatment at 3 and 6 months (P < .05). Levels of CXCL10 and BAFF were not different between active and inactive disease at baseline, and levels did not differ at 3 or 6 months in those with active disease (P > .05) (Figure 1).
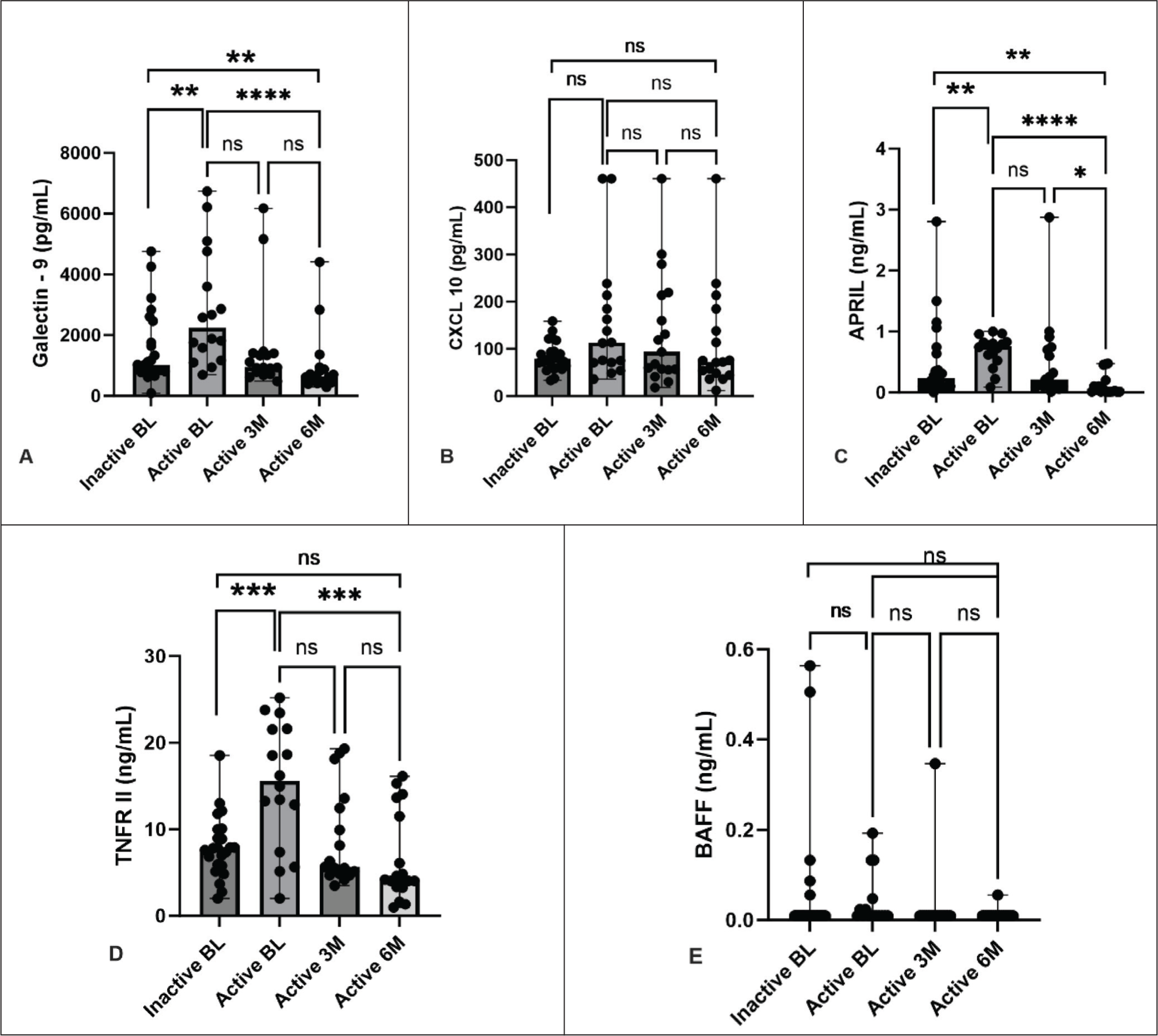
Notes: BL, Baseline; 3M, 3 months; 6M, 6 months; ns, non-significant; *P < .05; **P < .005; ***P < .001; ****P < .0001; APRIL, A proliferation inducing ligand; BAFF, B cell activating factor; CXCL, (C-X-C motif) ligand; TNFRII, Tumour necrosis factor receptor II. Unpaired t-test and Mann Whitney U tests were used to compare independent continuous variables (mean and median respectively) at two-time points based on their normality, while Kruskal Wallis ANOVA with post hoc Dunn’s multiple comparisons test was used to compare samples (median) at three-time points.
The serial change in biomarker levels in the active disease subgroup is depicted in Supplementary Figure S1. One patient in the inactive subgroup had a disease flare between 3 and 6 months. Levels of Galectin-9 (3412.31 pg/mL), CXCL10 (86.53 pg/mL), and APRIL (0.54 ng/mL) increased at the time of flare from baseline, while TNFRII (4.21 ng/mL) and BAFF (0.01 ng/mL) did not have any change.
Correlation Between Changes in Levels of Biomarkers with Change in Muscle Strength (MMT-8) and Various Diseases Activity Measures
Of all the biomarkers, changes in Galectin-9 (r = 0.534; P < .05), CXCL10 (r = 0.409; P < .05) and BAFF (r = 0.534; P < .05) showed moderate correlation with the change in MMT-8 at 6 months. In relation to change in CPK, a standard biomarker in myositis, only the change in Galectin-9 had moderate correlation (r = 0.465; P < .05), while it had mild correlation with BAFF (r = 0.348; P < .05). The rest of the measured biomarkers did not show any correlation (Table 2).
Correlation of the Change in Biomarkers With Change in MMT-8 and Traditional Serum Biomarkers at 6 Months (n = 16).
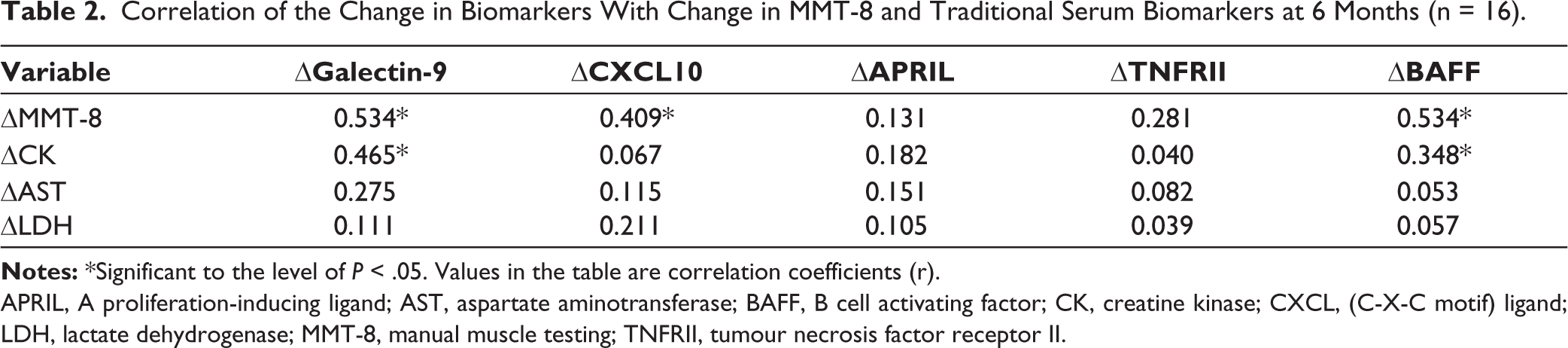
APRIL, A proliferation-inducing ligand; AST, aspartate aminotransferase; BAFF, B cell activating factor; CK, creatine kinase; CXCL, (C-X-C motif) ligand; LDH, lactate dehydrogenase; MMT-8, manual muscle testing; TNFRII, tumour necrosis factor receptor II.
In the correlation analysis between changes in biomarker levels at 3 and 6 months from baseline with changes in MDAAT score, cutaneous VAS and Muscle VAS, Galectin-9 showed mild to moderate correlation with MDAAT score (r = 0.652, 0.661), cutaneous VAS (r = 0.461, 0.427) and Muscle VAS (r = 0.614, 0.707) at 3 and 6 months (Table 3). CXCL10 showed mild to moderate correlation at 3 and 6 months with a change in MDAAT score (r = 0.533, 0.512) and Cutaneous VAS (r = 0.442, 0.421). At 6 months, the change in BAFF had mild correlation with both changes in MDAAT and Cutaneous VAS (r = 0.417, 0.494).
Correlation of the Biomarkers With Measures of Myositis Disease Activity at 3 and 6 Months (n = 16).
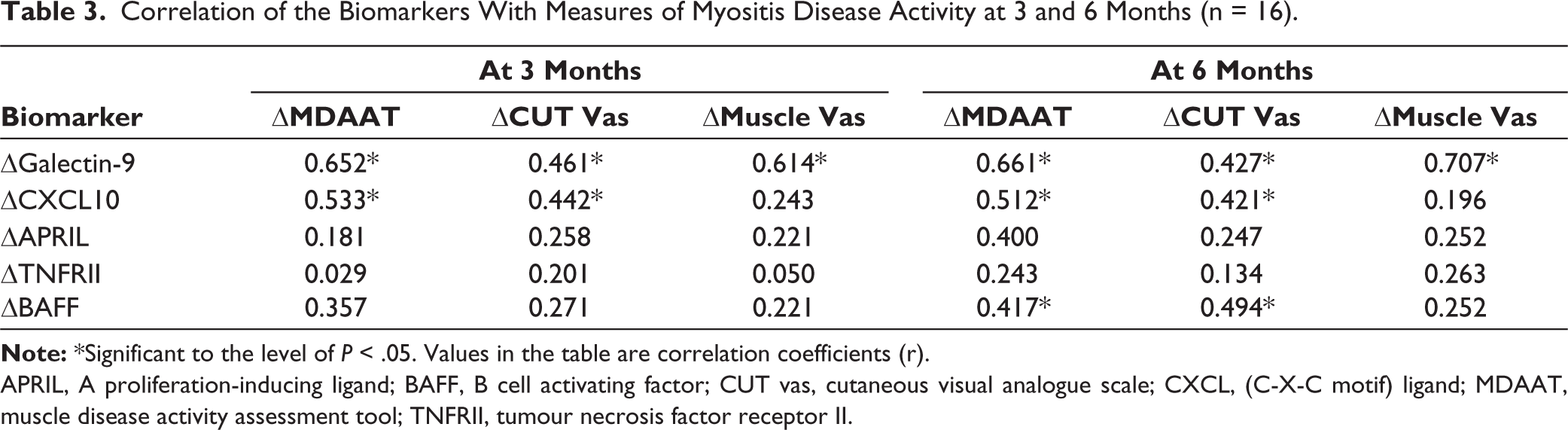
APRIL, A proliferation-inducing ligand; BAFF, B cell activating factor; CUT vas, cutaneous visual analogue scale; CXCL, (C-X-C motif) ligand; MDAAT, muscle disease activity assessment tool; TNFRII, tumour necrosis factor receptor II.
Performance of the Biomarkers in Differentiating Active and Inactive Disease
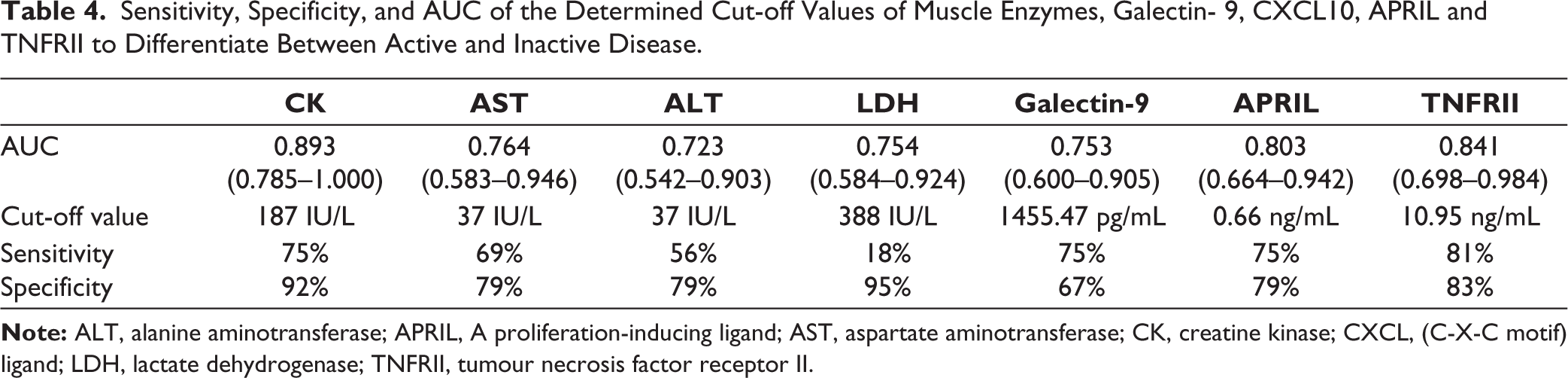
To assess the performance of the significantly different biomarkers that could differentiate between active and inactive disease (namely Galectin-9, APRIL, and TNFRII), we performed ROC curve analysis. Based on the coordinates of the ROC curve and Youden index, we obtained a cut-off value of 1455.47 pg/mL for Galectin-9 (with sensitivity of 75% and specificity of 67%), 0.66 ng/mL for APRIL (with sensitivity of 75% and specificity of 79%), and 10.95 ng/mL for TNFRII (with sensitivity of 81% and specificity of 83%) (Table 4).
Sensitivity, Specificity, and AUC of the Determined Cut-off Values of Muscle Enzymes, Galectin- 9, CXCL10, APRIL and TNFRII to Differentiate Between Active and Inactive Disease.
Performance of Combination of Biomarkers to Increase the Accuracy
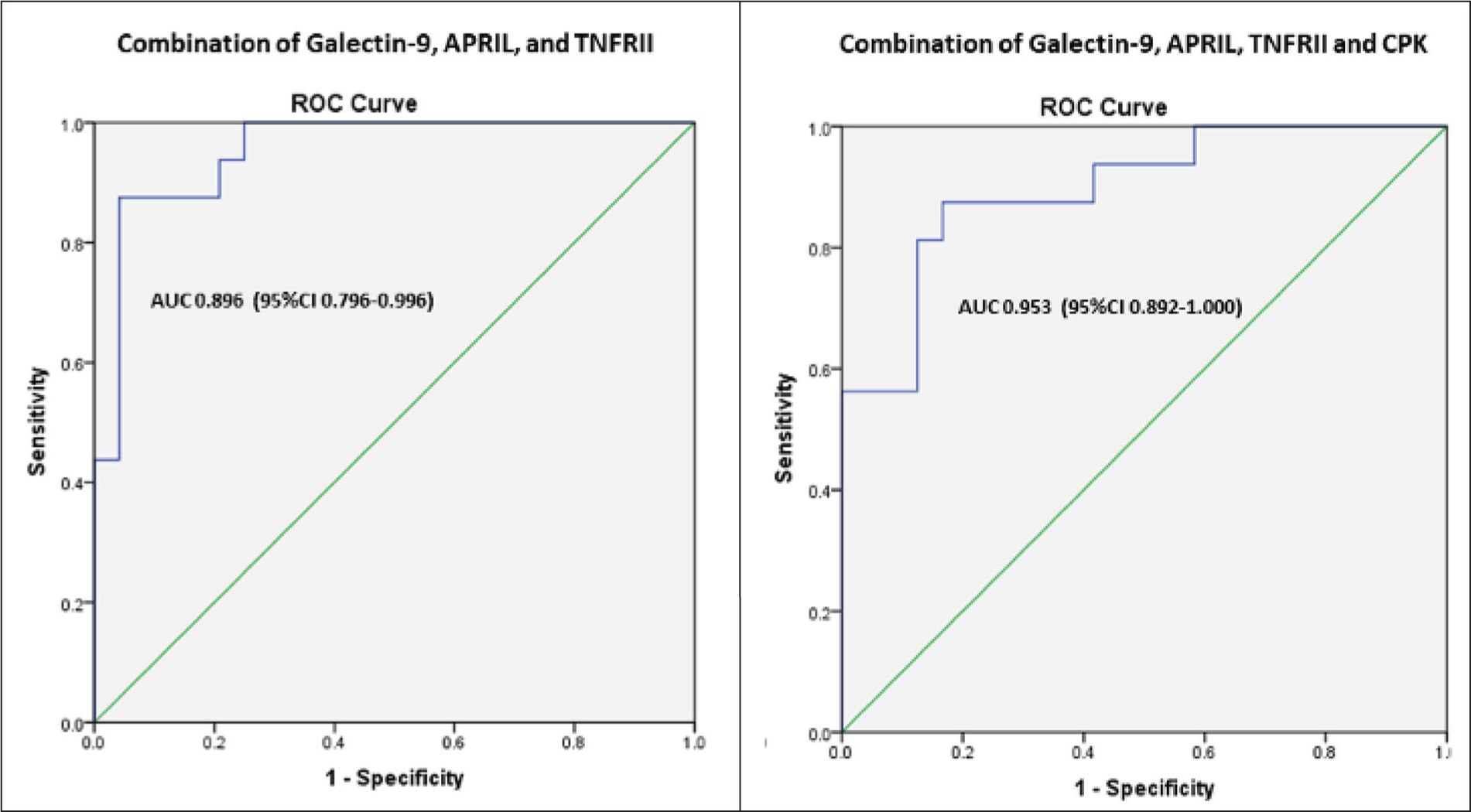
We then analysed if various combinations of these significantly different biomarkers could increase diagnostic accuracy. Using a binary logistic regression model with these different combinations, we calculated predicted probabilities. ROC curve analysis for these predicted probabilities of the combination of CK, Galectin-9, APRIL, and TNFRII yielded an AUC of 0.953 (95% CI = 0.892–1.000; P < .001). Similarly, we obtained an AUC of 0.896 (95% CI = 0.796–0.996) for the combination of Galectin-9, APRIL, TNFRII (Figure 2); AUC 0.930 (95%CI = 0.856–1.000), 0.896 (95%CI = 0.799–0.993), 0.951 (95%CI = 0.880–1.000) for combination of CK&Galectin-9, CK&APRIL, CK&TNFRII, respectively.
ROC Analysis for the Combination of Biomarkers.
Disease Improvement at Different Cut-off Values of Galectin-9
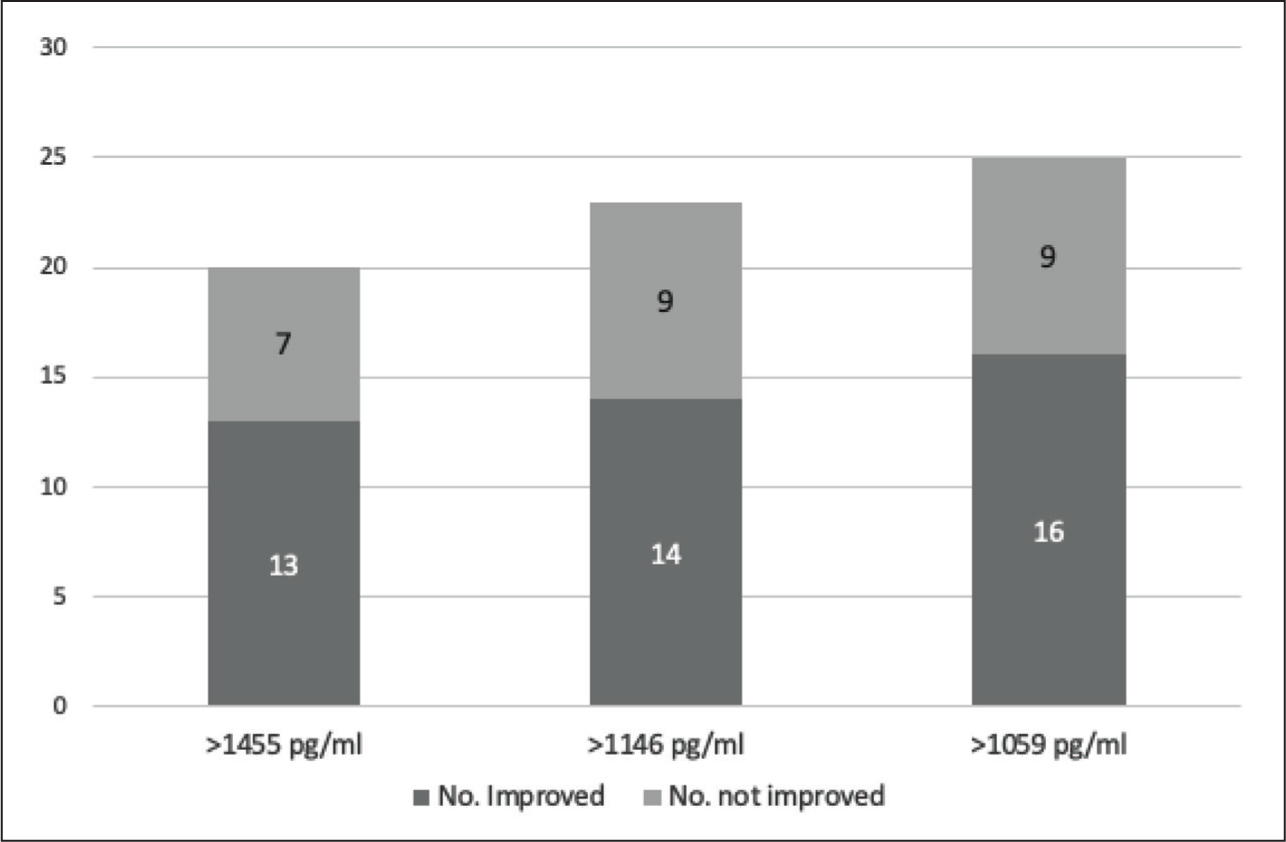
One of the criteria we used to define inactive disease was <15% change in MMT-8 over the preceding 6 months. However, during the course of the study, a few patients categorised as ‘inactive’ showed improvement in MMT-8. Thus, among the inactive disease group, four patients achieved 10% improvement in MMT-8 at the end of 6 months. The sensitivity of Galectin-9 to predict active disease at lower cut-off values than 1455 pg/mL (sensitivity of 75%) was assessed. New cutoffs at 1146 pg/mL and 1109 pg/mL were considered, and their sensitivity to identify clinically active disease improved to 81% and 87%, respectively. At each of these cut-off values, the number of subjects who achieved 10% improvement at 6 months after recruitment improved from 13 at 1455 pg/mL to 14 and 16 at 1146 pg/mL and 1109 pg/mL, respectively (Figure 3).
Bars Represent the Number of Patients Improved Versus not Improved Beyond Specific Cut-off Values of Galectin-9.
Identifying Disease Factors Predicting Biomarker Levels
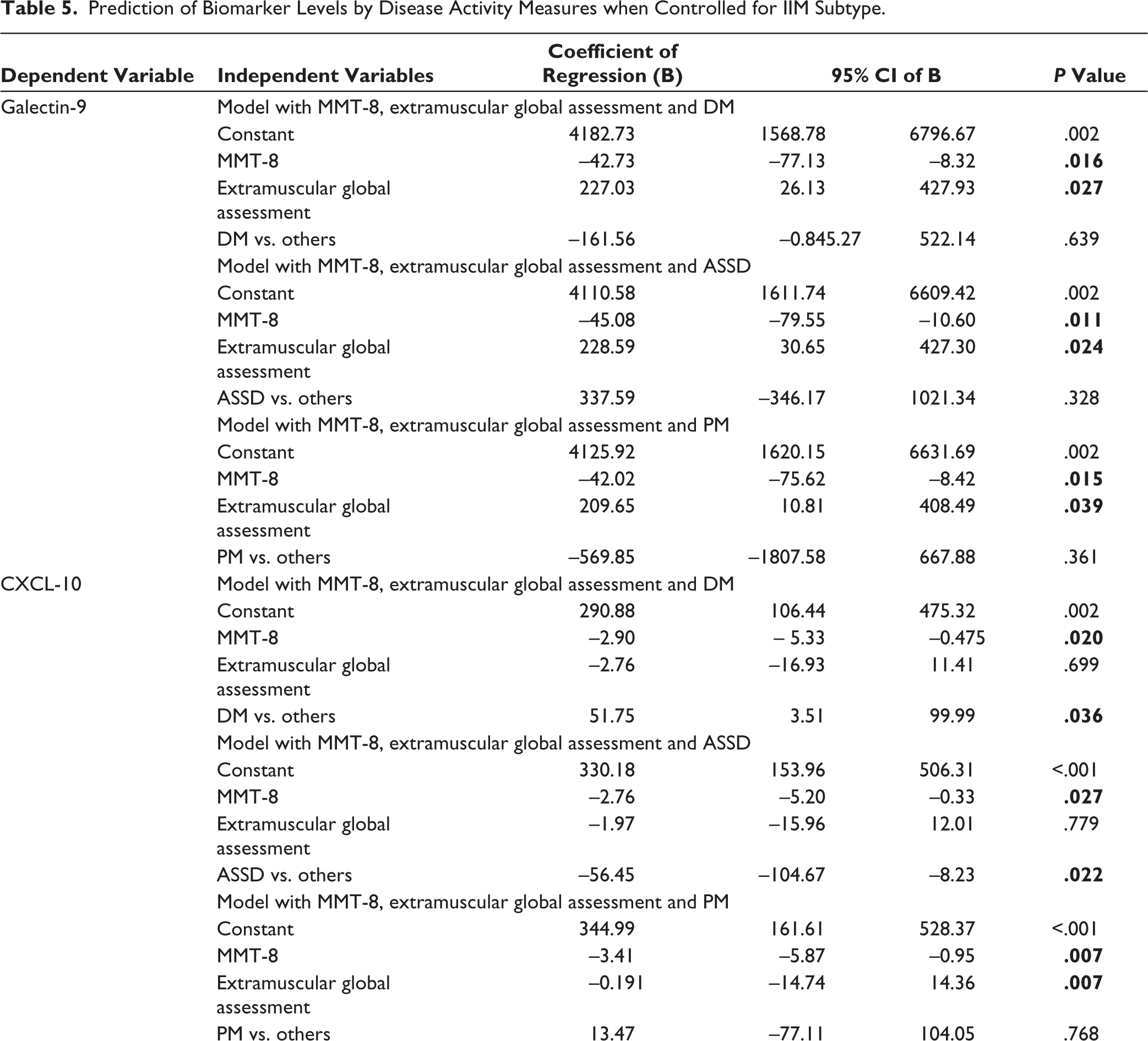
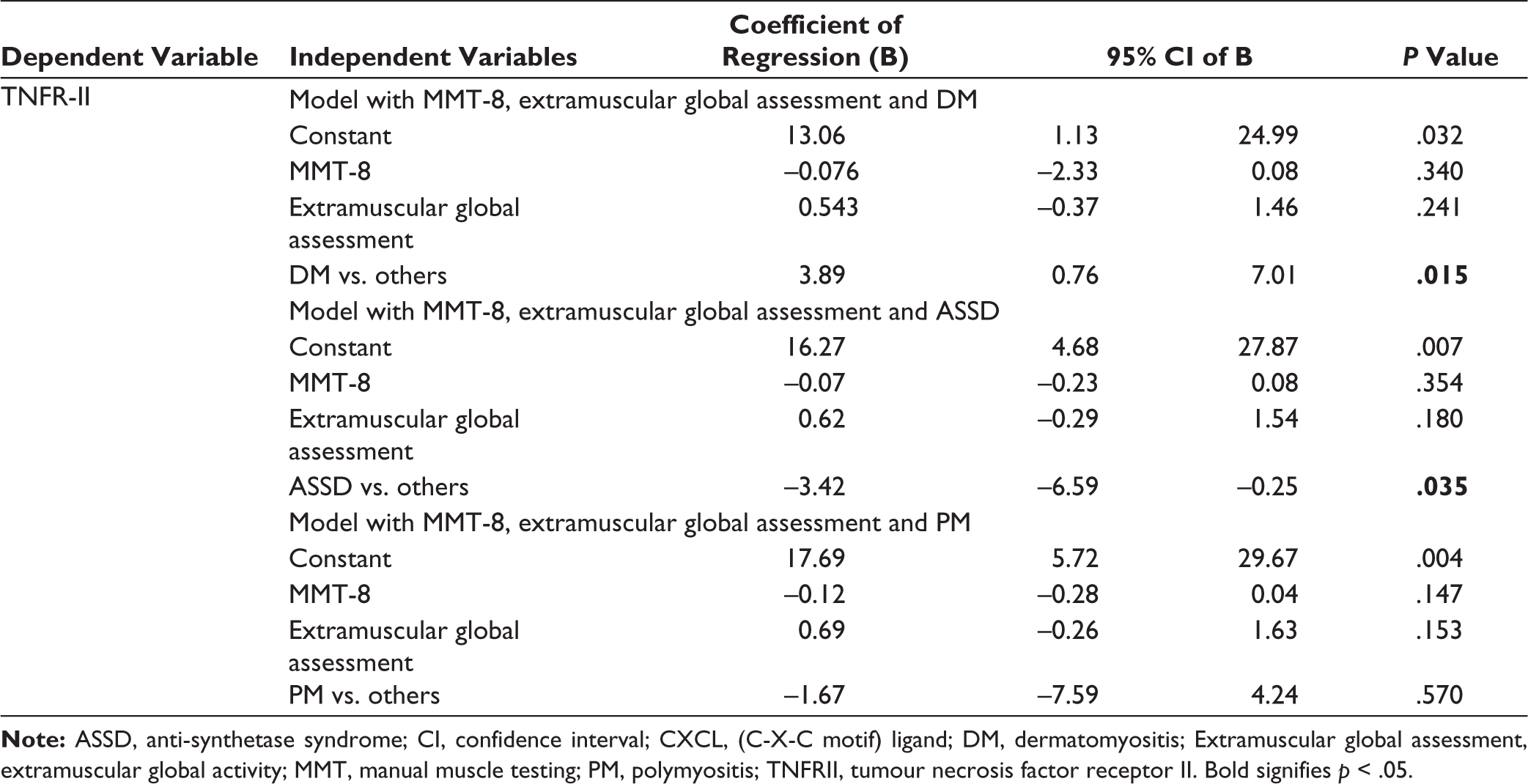
We performed multiple linear regression analyses to explore the relationships between specific biomarker levels and independent variables reflecting disease activity, including MMT-8, and extramuscular global assessment. We found that MMT-8 significantly predicted Galectin-9 (B = –41.082), CXCL10 (B = –74.575) and APRIL (B = –0.414) levels (P < .05) after controlling for other disease activity measures. Extramuscular global assessment predicted only Galectin-9 levels (B = 219.753, P < .05) (Supplementary Table S2).
When controlled for the disease subtype, MMT-8 (B = –42.73, P = .016; B = –45.08, P = .011; B = –42.02, P = .015) and extramuscular global assessment (B = 227.03, P = .027; B = 228.59, P = .024; B = 209.65, P = .039) independently predicted Galectin-9 levels in DM, ASSD and PM, respectively.
CXCL10 was mainly predicted by disease subtype DM (B = 51.75, P < .05) and ASSD (B = –56.45, P < .05). MMT-8 (B = –2.9, –2.76, –3.41, P < .05) had minimal prediction capability when controlled for in DM, ASSD and PM, respectively (Table 5). When controlled for disease subtype, MMT-8 and extramuscular global assessment could not independently predict APRIL and BAFF levels (P > .05) (Supplementary Table S3).
Prediction of Biomarker Levels by Disease Activity Measures when Controlled for IIM Subtype.
Discussion
In this study, we evaluated the performance of Galectin-9, CXCL10, APRIL, TNFRII and BAFF to determine disease activity in adult IIM. We also evaluated various disease factors that could predict these biomarker levels. We found that Galectin-9, TNFRII and APRIL were significantly higher in active disease compared to inactive disease.
We observed that baseline levels of Galectin-9, APRIL and TNFRII were significantly higher in the active when compared to the inactive disease group. Among those with active disease, the levels of these biomarkers decreased significantly at 3 and 6 months with treatment. Our observations were similar to those reported by Wienke et al. wherein they found that the levels of Galectin-9 and CXCL10 distinguished between active disease and remission in JDM, and subsequently confirmed in adult DM patients. It was also observed that Galectin-9 and CXCL10 were relatively specific for inflammatory myopathies as their levels were not highly increased or did not differentiate between active disease and remission in other autoimmune diseases. 21 Enders et al. also observed that plasma TNFRII is able to distinguish JDM with active and inactive disease. 29
In our study, one patient from the inactive disease group experienced a flare of disease between 3 and 6 months of recruitment. The levels of Galectin-9, APRIL and TNFRII increased at the time of flare and decreased in response to treatment. In a small study of 10 JDM patients who experienced a flare and were prospectively followed up, authors found that continuously elevated or rising levels of Galectin-9 and CXCL10 predicted flare several months before the onset of actual symptoms. These findings were true even in the absence of elevated CK levels. 21 Thus these biomarkers appear promising for use in monitoring disease activity and predicting flare.
When we correlated the changes(Δ) in levels of biomarkers with the change in clinical disease activity, we observed that changes in ΔGalectin-9 and ΔCXCL10 over 6 months correlated with ΔMDAAT and ΔCutaneous VAS. In addition, ΔGalectin-9 also correlated with ΔMuscle VAS, indicating that they could be reliable indicators of both muscular and extramuscular disease improvement. A study in JDM also showed that changes in CXCL10, Galectin-9 and TNFRII correlated with physician global assessment (PhGA) and childhood myositis activity score (CMAS). 29 A recent study using the SOMAscan assay also identified both Galectin-9 and CXCL10 among the top upregulated proteins in JDM, correlating with disease activity as assessed by the PhGA. 30
In a study by Kryštůfková et al. changes(Δ) in levels of both BAFF longitudinally correlated with ΔCK, Δcutaneous, Δmuscle, Δglobal, and Δskeletal disease activities. Though serum BAFF level has been reported to be significantly different in patients with Jo-1 positivity, with changes in BAFF level correlating well with disease activity measures and treatment, 31 this was not observed in our study when we did a subgroup comparison between patients with and without anti-Jo 1 antibody. This could likely be due to a limited sample size.
Although the performance of CK to predict active disease was better compared to any of the individual biomarkers, Galectin-9, APRIL, TNFRII together performed better than CK alone. Further, the ability to predict active disease increased when all three biomarkers were combined with CK. Wienke et al observed that both Galectin-9 and CXCL10 outperformed CK alone in two of their analysed cohorts. 21 It is known that CK levels vary with age, gender and race, 32 and the study by Wienke et al., demonstrated the outperformance of Galaectin-9 and CXCL10 in JDM. Our findings reconfirm CK as a good biomarker in adults but a combination of the novel biomarkers may outperform the performance of traditional muscle enzymes. Our cut-off value for Galectin-9 and CXCL10 as well as sensitivity and specificity are lower than previous studies, probably due to differences in the measuring technique and the ethnicity of the population studied.
We observed that 2/3rds of patients with higher Galectin-9 levels at baseline experienced disease improvement, including those four patients who were classified as inactive disease at the start of the study. This finding implies that Galectin-9 can be explored as a possible biomarker for disease activity and improvement in IIM, and there may be a scope for improvement in muscle strength even in seemingly inactive patients.
It is well documented that type 1 interferon mRNA expression is significantly increased in peripheral blood and muscle tissue in IIM. 33 Interferons stimulate a variety of immune and non-immune cells to produce CXCL10 and Galectin-9.14,15 Galectin-9 has also been implicated in the pathogenesis of extramuscular features like vasculopathy and interstitial lung disease in IIM.8,34 Thus, our observations that MMT-8 predicted Galectin-9, CXCL10 and APRIL levels, while extramuscular global VAS predicted Galectin-9 levels in addition to MMT-8, seem to confirm the positive relationship of interferons in IIM. It is well known that interferon signature is upregulated in DM and ASSD among the various IIM subtypes.35,36 In line with this, we too observed that DM and ASSD predicted CXCL10 and TNFRII levels.
Although there are many studies on the utility of these biomarkers in JDM, data on adult IIM is scarce. Thus, our study adds valuable information to the literature of IIM in this aspect. The strengths of our study include a homogeneous cohort of adult IIM patients with active and inactive disease subgroups and longitudinal measurement of serum biomarkers. Limitations include a small sample size, and a follow-up duration limited to 6 months. We also did not have enough patients who flared, which would have allowed us to test the robustness of these biomarkers in predicting flare. All our patients were from the south Indian Tamil population, which is both a strength and a weakness. It is a strength because the effect of varying ethnicity on IFN signature is negated, but it is also a weakness because generatability of the results to other populations will need validation.
We conclude that serum biomarkers like Galectin-9, TNFRII and APRIL in combination could be reliable biomarkers to differentiate between active and inactive disease in adult IIM. The levels of these biomarkers could be indicators of disease flare and improvement, especially when combined with CK levels. Tracking changes, especially in Galectin-9 and TNFRII, could help monitor disease activity and predict flare. Besides, elevated Galectin-9 among individuals who are classified as having inactive diseases based on clinical criteria may reflect an active disease that would show improvement. Further studies may be needed to understand the value of these biomarkers in guiding treatment decisions and as a potential outcome measure in adult IIM.
Supplemental Material
Supplementary material for this article is available online.
Footnotes
Acknowledgements
Saikumar Dunga and Aishwarya Gopal are to be acknowledged as equal contributions as first authors.
Data Availability Statement
All data are available with the corresponding author and may be made available upon reasonable requests.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Patient Consent
The institutional scientific and ethics committee of Jawaharlal institute of postgraduate medical education and research (JIPMER), Puducherry, India had approved the study protocol (JIP/IEC/2019/250), and all the study subjects signed the written informed consent.
Funding
The study was funded by grants from the Indian Rheumatology Association for the year 2020.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
