Abstract
Background:
We compared the diagnostic accuracy of procalcitonin, C-reactive protein (CRP), and ferritin in differentiating an infectious aetiology versus disease flare in children with systemic juvenile idiopathic arthritis (sJIA).
Methods:
This prospective diagnostic accuracy study included 52 consecutive hospitalisations of 25 children diagnosed with sJIA (International League of Associations for Rheumatology 2001) presenting with a fever to the rheumatology department of a quaternary care centre. We categorised children post-discharge as infectious versus sJIA flare, depending on the clinical course. A subset of sJIA flare developed macrophage activation syndrome (MAS). We compared the area-under-curve (AUC) with its 95% confidence interval (95%CI) of the three biomarkers.
Results:
The median age of the children was 7 ± 7 years. We categorised 12 hospitalisations (23.1%) as infectious and 40 hospitalisations (76.9%) as sJIA flare. Thirteen hospitalisations (32.5%) with sJIA flare developed MAS. Neither procalcitonin (AUC = 0.584 ± 0.101, 95% CI: 0.387–0.782) nor CRP (AUC = 0.363 ± 0.096, 95% CI: 0.174–0.551) nor ferritin (AUC = 0.419 ± 0.093, 95% CI: 0.236–0.602) discriminated infectious aetiology from sJIA flare adequately. Both serum procalcitonin (AUC = 0.828, 95% CI: 0.694–0.961) and ferritin (AUC = 0.937, 95% CI: 0.862–1) demonstrated higher diagnostic accuracy in ascertaining MAS in sJIA flares, compared to CRP (AUC = 0.709, 95% CI: 0.543–0.876).
Conclusion:
Procalcitonin can be elevated in children in sJIA flare with MAS, even without an infectious aetiology. Clinicians should continue to rely on clinical history and examinations to discern infectious versus non-infectious fevers in children with sJIA.
Keywords
Introduction
Systemic juvenile idiopathic arthritis (sJIA) is a chronic inflammatory disease that affects children under 16 years of age and is characterised by persistent fever, arthritis, rash, and lymphadenopathy. 1 sJIA can also be complicated by macrophage activation syndrome (MAS), a life-threatening condition that results from excessive activation and proliferation of macrophages and T cells, leading to cytokine storm, organ failure, and coagulopathy. 2
One of the challenges in managing sJIA is to differentiate between a disease flare and an infection, as both can present with similar clinical features. The treatment strategies for these two scenarios are different and may have opposite effects. Immunosuppressive drugs used to treat sJIA flares may worsen infections, while antibiotics for a presumed infection may delay diagnosis and treatment for a developing MAS. Currently, clinicians rely on a combination of clinical and laboratory parameters to distinguish between a sJIA flare and an infection. 3 Novel biomarkers suggested as potential markers for sJIA disease activity have not been adopted widely, possibly due to tedious laboratory requirements.4,5 Among the laboratory parameters, C-reactive protein (CRP), ferritin, and serum procalcitonin are widely available as markers of inflammation and infection. Serum procalcitonin is a peptide precursor of calcitonin produced by various tissues in response to bacterial infections and is considered a more sensitive marker for sepsis than CRP and ferritin.6,7
However, the diagnostic accuracy of serum procalcitonin in differentiating between an sJIA flare and an infection is yet to be evaluated in a homogenous sJIA study population. 8 Moreover, the role of serum procalcitonin in detecting MAS in children with sJIA flares is unclear. Therefore, we conducted a comparative diagnostic test accuracy analysis of 52 hospitalisations of 25 children with sJIA who initially presented with high-grade fever to our rheumatology department. We compared the diagnostic accuracy of serum procalcitonin, CRP, and ferritin in differentiating between an infectious aetiology and a sJIA flare and detecting MAS in children with sJIA flares.
Materials and Methods
Study Design, Settings, Population, and Ethical Consideration
We conducted a prospective, diagnostic accuracy, comparative study amongst children (<16 years) with sJIA diagnosed earlier as per the International League of Associations for Rheumatology (ILAR) classification criteria, 2001. 9 We considered consecutive children with sJIA for inclusion, who presented with a fever (≥38oC) for ≥48 hours, to our rheumatology department of a quaternary care centre between March 2018 and October 2019. Multiple visits of children with recurrent fever episodes were also included consecutively. We sought informed consent from the parents or legal guardians of the children before their inclusion into our analysis. We obtained ethical clearance from the institutional ethics committee. As a pilot study, we empirically aimed to recruit at least 30 hospitalisations for the analysis because there was no previous published research to calculate a minimum sample size. We reported the current study as per the Standards for Reporting Diagnostic Accuracy (STARD) 2015 guidelines. 10
Definitions
At discharge from the hospital, we ascertained that the included children with sJIA had fever due to an infectious aetiology as diagnosed by a senior paediatric rheumatologist (>20 years clinical experience) with a clinical history and physical examination, confirmed with positive signs of infection in imaging or bacteria isolated from blood/urine/tissue cultures. We disregarded commensal organisms isolated without correlated clinical signs. They must also have responded promptly to antibiotic therapy (within 48–72 hours) without titrating up the immunosuppressive therapy (intravenous pulse steroids, biologic disease-modifying anti-rheumatic drugs). 3 We categorised other children with fever ≥38oC) for ≥48 hours, who did not fit the above clinical profile, into the ‘sJIA flare’ group when their clinical status promptly improved with a hike in immunosuppressive therapy.
Among the group of children experiencing a sJIA flare, we identified a ‘MAS’ subgroup who were diagnosed with MAS, as per the 2016 Classification Criteria for Macrophage Activation Syndrome Complicating Systemic Juvenile Idiopathic Arthritis: A European League Against Rheumatism (EULAR)/American College of Rheumatology (ACR)/Paediatric Rheumatology InterNational Trials Organisation (PRINTO) Collaborative Initiative. 11 Namely, serum ferritin >684 ng/ml, with any two of the following—platelet count ≤ 181 × 10⁹/L, AST > 48 units/L, triglycerides > 156 mg/dl or fibrinogen ≤ 360 mg/dl.
Sample Collection and Measurement
We collected the blood samples for procalcitonin from the included children at the time of admission before initiation of any therapy. The blood workup included a complete hemogram, erythrocyte sedimentation rate (ESR), CRP, liver function tests, renal function tests, ferritin, fibrinogen, triglycerides, and blood ± urine cultures performed in the in-house laboratory. We also obtained the necessary imaging as part of their disease management.
We stored the blood samples at -20°C in our in-house laboratory and analysed serum procalcitonin after the child’s discharge. To measure serum procalcitonin, we used Elecsys BRAHMS PCT assay (Roche, USA) on the Cobas 8000 e602 module, which employs the Electrochemiluminescence immunoassay ‘ECLIA’ method, for the in vitro quantitative determination. We assessed CRP and ferritin levels in the Cobas 8000 602/801 modules (Roche, USA)—using the turbidometry and ECLIA methods, respectively. We blinded the treating physician and the laboratory staff to the procalcitonin values and disease status, respectively.
Data Collection and Analysis
We collected data on demographic information, clinical history, physical examination, and medication information. We planned to analyse any missing data patterns and use imputation if appropriate. We considered data from each hospitalisation independently for analysis, assuming that procalcitonin levels do not remain elevated in the index hospitalisation after a previous infectious/flare episode.
For descriptive analysis, we reported estimates as frequencies (percentages) for categorical data and median ± interquartile range (IQR) for continuous data (due to the non-normal distribution of data)—and compared between groups using Pearson’s χ 2 and Mann-Whitney U tests, respectively. In a receiver operator characteristic (ROC) curve analysis, we compared the area-under-the-curve estimates of procalcitonin, CRP, and ferritin with their 95% confidence interval (CI) and P value. We further determined the best cutoff value for the above three biomarkers from the ROC analysis in determining an infectious aetiology. We reported the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), diagnostic accuracy as percentages, and P value. We repeated the above analyses amongst the subset of the sJIA flare group to determine the diagnostic accuracy of the three biomarkers for differentiating MAS in an sJIA flare. We performed all statistical analyses on SPSS Statistics Version 26 (IBM Corp, Armonk, NY, USA).
Results
We included 52 consecutive hospitalisations of 25 children with sJIA for our analysis and excluded none. There were no missing data. A majority of the included children were male (n = 14/25, 56.5%), with a median age of 7 ± 7 years. In 12 hospitalisations, we ascertained an infectious aetiology; in the other 40 hospitalisations, we ascertained an sJIA flare. In 13 hospitalisations (of the 40 hospitalisations with sJIA flare), we confirmed a diagnosis of MAS—three of whom were preceded by a mild viral upper respiratory infection. There were no deaths among these hospitalisations.
Out of the 12 hospitalisations categorised as having infectious aetiology, we diagnosed six hospitalisations (50%) as lower respiratory tract infections (LRTI), three hospitalisations (25%) as upper respiratory tract infections (URTI), two hospitalisations (16.7%) as urinary tract infections (UTI) and one hospitalisation (8.3%) as a case of acute gastroenteritis. Of the three URTI hospitalisations, two were for severe oral mucositis and lip infection with pus discharge and cultures that grew Staphylococcus aureus. The third hospitalisation was for granular pharyngitis, and the throat swab culture grew Streptococcus spp. The urine cultures from two hospitalisations for UTIs grew Escherichia coli and Morganella spp, respectively. One hospitalisation was for acute gastroenteritis with septic shock. Even though we could not get any growth on the blood or stool cultures, the bacterial aetiology of the illness was confirmed based on the typical symptoms, blood parameters and disease progression, including hypotensive shock with an optimal response to parenteral antibiotics.
As per exploratory comparisons of clinical features and laboratory parameters (Table 1), patient hospitalisations with sJIA flare were overwhelmingly more likely to present with quotidian fever, evanescent maculopapular rash, arthralgia and joint swelling than hospitalisation for infectious aetiology. Patients during sJIA flare hospitalisation, on average, had higher total white blood cell counts, platelet counts, ESR, aspartate aminotransferase, alanine aminotransferase, while haemoglobin, fibrinogen, and triglyceride levels were similar, with none of the above comparisons showing any evidence of an association. Of the biomarkers of interest, median procalcitonin was higher in the infectious group, while median CRP and ferritin were higher in the sJIA flare group, with considerable overlap in the variability (IQR) between groups and none of the comparisons showing any evidence of a true difference.
Demographic and Clinical Characteristics of 52 Included Hospitalisations of 25 Children at Admission.
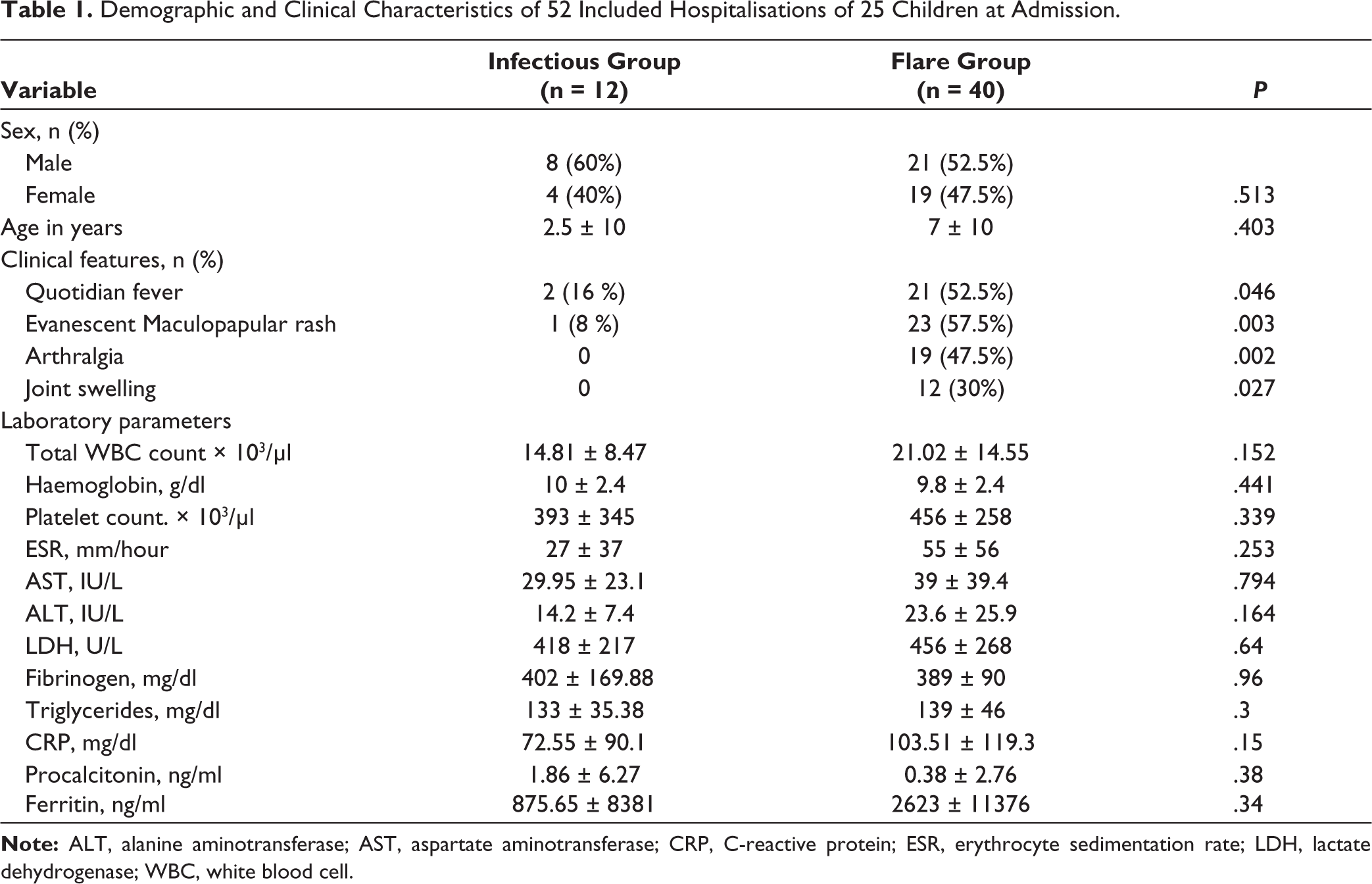
In a ROC analysis to diagnose infectious aetiology (Figure 1), procalcitonin demonstrated an area-under-curve (AUC) = 0.584 ± 0.101 (95% CI: 0.387–0.782), while CRP and ferritin demonstrated an AUC = 0.363 ± 0.096 (95% CI: 0.174–0.551) and AUC = 0.419 ± 0.093 (95% CI: 0.236–0.602), respectively. Based on the above ROC, a procalcitonin value of 1 ng/ml was chosen as the cutoff, demonstrating the highest possible combination of sensitivity (66.7%) and specificity (60%) with P = .186. At this cutoff, the overall diagnostic accuracy of procalcitonin at the 1 ng/ml cutoff was 61.54% with PPV = 33%, NPV = 85.7%, PLR = 1.67, and NLR = 0.56.
Receiver Operator Characteristic (ROC) Curve Analysis of Procalcitonin, Ferritin, and C-reactive Protein in Diagnosing an Infectious Aetiology in Hospitalisations for Fever, in Children Previously Diagnosed With sJIA (n = 52).
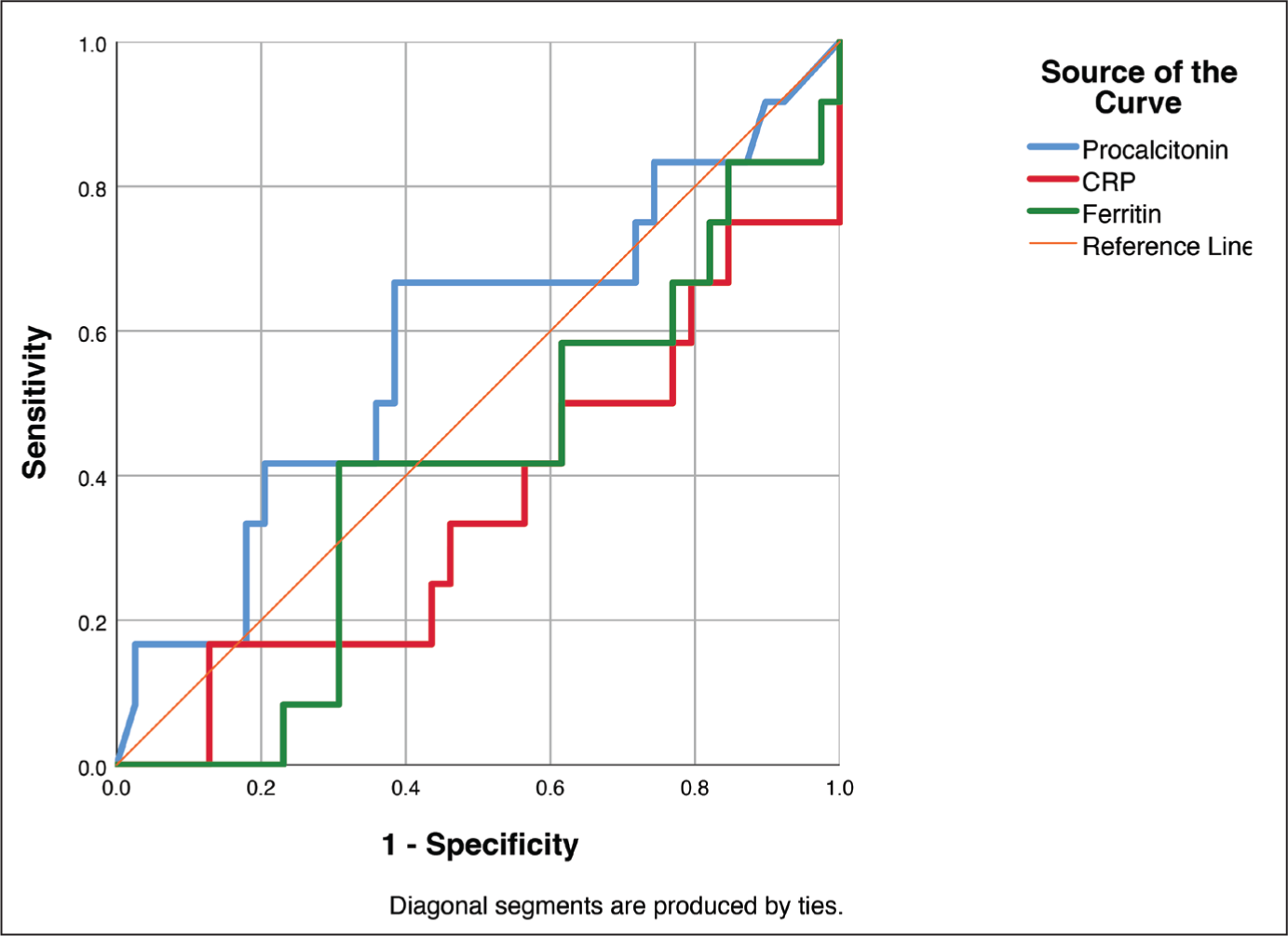
As depicted in Table 2, children during hospitalisation for MAS compared to sJIA flare hospitalisations, had lower median platelet count and fibrinogen levels but demonstrated much higher median ferritin, LDH, procalcitonin and CRP levels and triglyceride levels, with between-group comparisons showing strong evidence for a true difference.
Laboratory Parameters of a Subset of Patients With Confirmed sJIA Flare (n = 40), Categorised by Macrophage Activation Syndrome (MAS) Diagnosis.
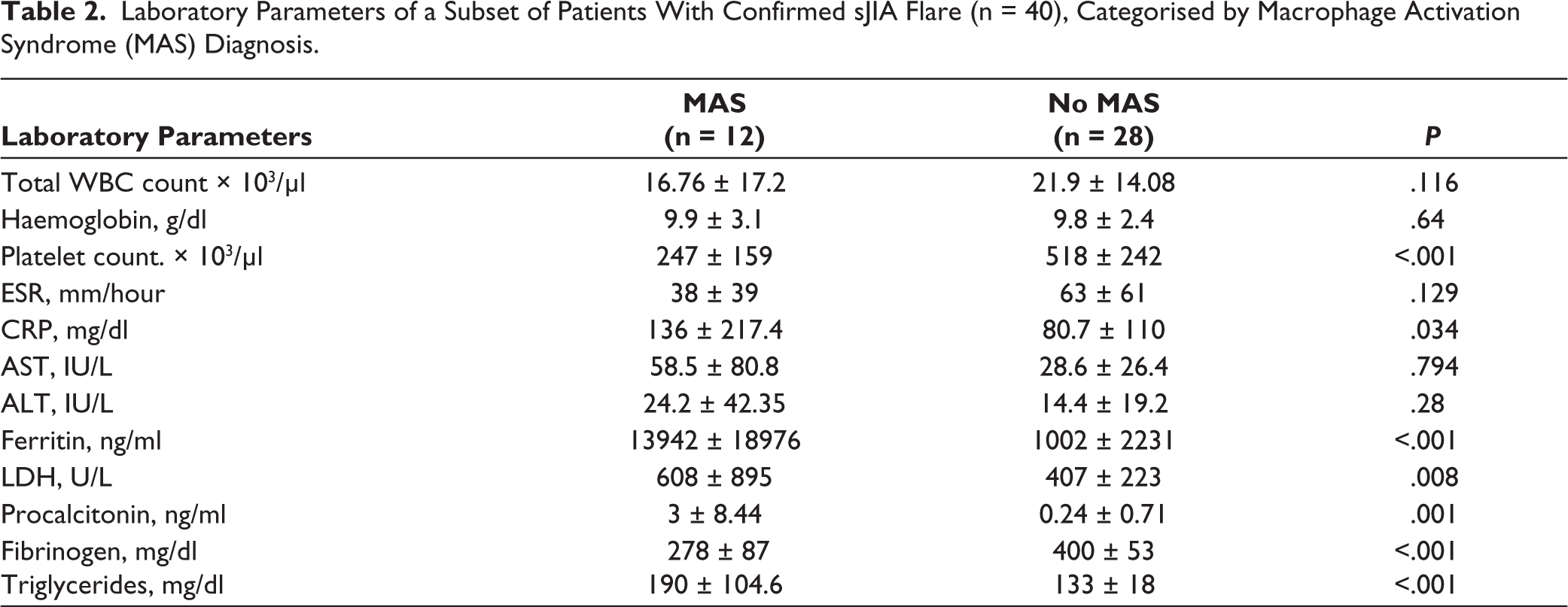
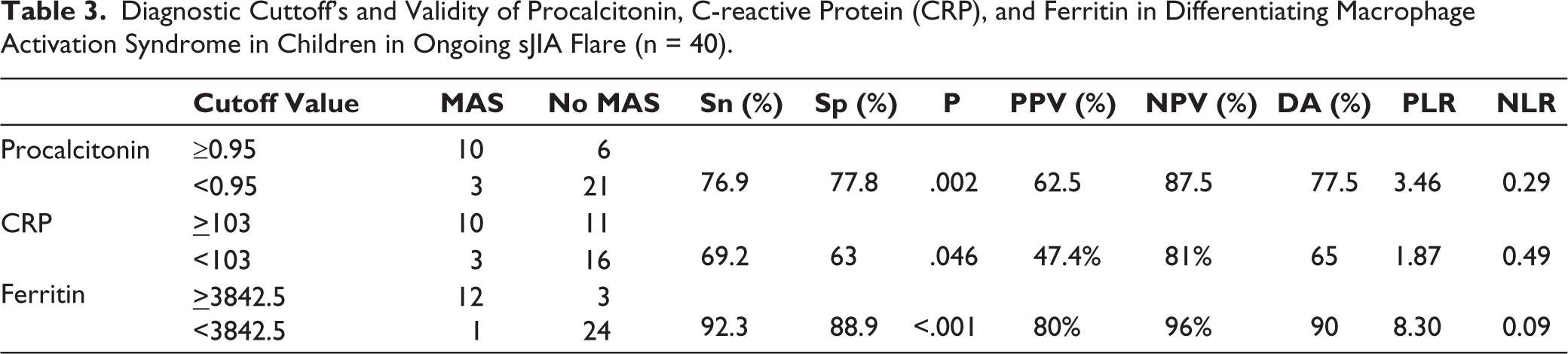
From a ROC analysis of the sJIA flare subset (n = 40) to diagnose MAS (Figure 2), procalcitonin demonstrated an AUC = 0.828 ± 0.068 (95% CI: 0.694–0.961), while CRP and ferritin demonstrated an AUC = 0.709 ± 0.085 (95% CI: 0.543–0.876) and AUC = 0.937 ± 0.039 (95% CI: 0.862–1), respectively. As per these ROC curves, we selected 0.95 ng/dl for procalcitonin, 103 mg/dl for CRP, and 3842.5 ng/ml for ferritin as the best cutoff values for diagnosing MAS in the background of an sJIA flare. At these cutoffs, procalcitonin (77.5%) and ferritin (90%) demonstrated high diagnostic accuracy for MAS, while CRP’s accuracy was considerably lower (65%) (Table 3).
Receiver Operator Characteristic (ROC) Curve Analysis of Procalcitonin, Ferritin and C-reactive Protein in Diagnosing Macrophage Activation Syndrome (MAS) in Hospitalisations for sJIA Flare (n = 40).
Diagnostic Cuttoff’s and Validity of Procalcitonin, C-reactive Protein (CRP), and Ferritin in Differentiating Macrophage Activation Syndrome in Children in Ongoing sJIA Flare (n = 40).
Discussion
In this diagnostic accuracy study, analysing 52 hospitalisations of 25 children with sJIA presenting with fever ≥48 hours, we found that procalcitonin was a poor diagnostic tool to differentiate a bacterial infection aetiology from a sJIA flare, albeit being slightly superior to CRP and ferritin. As evidenced in a subgroup analysis, this finding may be due to the high levels of these acute phase reactants detected in a large proportion of children with sJIA flare who subsequently developed MAS—a lethal hyper-inflammatory state. To our knowledge, we are the first to report on the diagnostic value of procalcitonin in differentiating infectious versus non-infectious in a homogenous cohort of sJIA children hospitalised with high-grade fever.
Our findings may inform the initial management of sJIA, which may vary from centre to centre, as currently, no international guidelines provide specific guidance for clinicians to choose between antibiotic therapy and immunosuppressive therapy.12,13
Our findings contradict those of studies with other autoimmune diseases, where serum procalcitonin levels were significantly higher in patients with infections versus patients with disease flare. A systematic review and meta-analysis of 15 studies, including patients with systemic lupus erythematosus (SLE) with bacterial infections versus disease flare, found that serum procalcitonin (AUC = 0.862) had higher diagnostic accuracy in detecting bacterial infections than CRP (AUC = 0.784)(14). Similarly, an earlier meta-analysis of 668 adult participants from eight assorted studies of autoimmune diseases (three SLE studies) reported serum procalcitonin had a superior diagnostic accuracy than CRP (AUC = 0.884 vs. 0.789) in diagnosing bacterial infections from disease flare. However, both meta-analyses only included adult participants, a majority with SLE and no sJIA participants, which may not be generalisable to our paediatric study population with sJIA. Compared to an SLE flare, a sJIA flare, involves a more widespread activation of proinflammatory cytokines and chemokines like IL-1, IL-6, IL-18, and TNFα, which in turn cause macrophages and CD8+ T cells to traffic to the bone marrow and liver, leading to MAS.15,16 Thus, there is little to no rise in acute phase reactants like CRP during SLE flares, compared to other autoimmune diseases. 17
Bearing some similarity to sJIA in pathophysiology is the adult-onset Still’s disease (AOSD). A cross-sectional study of 38 participants with active AOSD (>3 disease score)—12 with confirmed bacterial infections, reported that procalcitonin has very high diagnostic accuracy for diagnosing infections compared to CRP (AUC = 1 vs. 0.832). The authors suggested a cutoff of 1.4 ng/ml to diagnose bacterial infections, while values < 0.5 ng/ml maybe suggest nonspecific inflammation, with a 100% sensitivity as per their ROC analysis. However, none of the included AOSD participants had developed MAS in the above study, which would have demonstrated much higher serum procalcitonin levels as reported in other studies.18–20 Another study with similar limitations, which included 92 children with Juvenile Idiopathic Arthritis (JIA), reported that at a 0.15 μg/ml cutoff, procalcitonin reported excellent diagnostic accuracy for bacterial infections (Sensitivity = 94.1%, Specificity = 98.3%, PPV = 94.1% and NPV of 98.3%). 8 However, the investigators were unable to include children with sJIA in their analysis, as they were reportedly rare. As non-systemic variants for JIA are less likely to develop MAS than sJIA, the authors’ recommendation for serum procalcitonin to differentiate infectious versus non-infectious fevers cannot be extended to sJIA flares.
Our subgroup analysis of the biomarkers in children with sJIA flare validates our finding that children developing MAS will likely report high serum procalcitonin values that overlap those of children with infections. Even though three of the 12 children included in the infectious group reported only URTI, a localised infection—the group median ± IQR procalcitonin stood at 1.86 ± 6.27 ng/ml, which is representative of most systemic infections- and apt for comparisons with sJIA flare group. Procalcitonin itself is not a perfect predictor of all bacterial infections, as it remains low in localised infections affecting skin and skin structure, diabetic foot, septic arthritis, and osteomyelitis 21 —all likely diseases to present with a fever in children with sJIA.
As the PRINTO 2016 classification criteria 10 recommended, our subgroup analysis reports a superior diagnostic accuracy for ferritin to diagnose MAS than serum procalcitonin or CRP. However, the PRINTO criteria use a lower 684 ng/ml cutoff for ferritin, while we detected a much higher cutoff of 3842 ng/ml to have the best combination of excellent sensitivity and specificity. However, MAS may be considered a continuum of the sJIA flare pathology and can be occult or subclinical in 20%–30% of the cases and only clinically overt in about 10%. 22 Thus, the lower ferritin cutoff, dropping platelet counts, fibrinogen, rising AST, and triglycerides, as per PRINTO 2016, maybe more valuable in the early prediction of MAS. Nonetheless, to our knowledge, we are the first to evaluate the diagnostic accuracy of serum procalcitonin for MAS in an sJIA flare, and it may be a helpful biomarker after an infection has been definitively ruled out—which may prove difficult. Any planned larger, prospective validation study of the PRINTO 2016 criteria in sJIA patients may include procalcitonin to determine its diagnostic accuracy for MAS.
Strength and Limitations
The strengths of the current study include its relatively large sample size of children with sJIA—prospectively and consecutively recruited, with all common laboratory biomarkers available in a secondary or tertiary centre explored—lending external validity. However, we included multiple hospitalisations for fever, from the same participant, for analysis, assuming that after successful treatment of infection/sJIA, the procalcitonin levels drop dramatically by discharge and will not remain elevated at the time of the subsequent hospitalisation for fever. This assumption is likely accurate, given how serum procalcitonin values decline rapidly in other resolving inflammatory conditions. 23 However, it is unclear if the inclusion of the same children led to the clustering of unknown patient-related factors, which may have resulted in the null association in our analysis. A small sample size of 12 children in the bacterial infection group and the types of bacterial infections included are not generalisable to all settings, however, the included infections did demonstrate high procalcitonin levels, similar to most bacterial sepsis scenarios, thus likely avoiding a false null estimate. Since procalcitonin was collected at admission, stored, and analysed post-discharge to blind the treating paediatric rheumatologist, we avoided any inadvertent investigator bias. However, this also limits our analysis to diagnostic accuracy at admission and does not describe any trends during hospitalisation or how immunosuppressive or antibiotic therapies may have influenced the biomarker levels.
While we used validated international guidelines with objective laboratory parameters for diagnosing MAS, only five out of 12 infections were confirmed with microbiological evidence, while the rest were essentially clinical diagnoses supplemented with radiographs. This is also reflective of a real-world scenario in paediatrics, and we do not anticipate any misclassification of patients with infections as sJIA flare, and thus a false null association, as the classification was done at discharge, when we ensured that the child had improved with the initiation or a hike of antibiotic therapy, as opposed to immunosuppressive therapy. Three out of 12 children developed MAS—seemingly triggered by a viral upper respiratory tract infection. However, these few mild viral infections, in the ‘flare group’ are unlikely to cause a procalcitonin rise by themselves, thus not inducing a false null estimate. Instead, MAS, either triggered by a viral infection or developing as a continuum of a disease flare, is likely the cause of high procalcitonin levels in the flare group.
Conclusion
Serum procalcitonin may not be helpful as a diagnostic biomarker to differentiate between infectious versus non-infectious aetiology in children with known sJIA, hospitalised with a high-grade fever. A paediatric rheumatologist must continue to rely on a good clinical history with examination to select an appropriate treatment approach and must not solely rely on biomarkers to diagnose aetiology, which may be obfuscated in a hyper-inflammatory state like MAS.
Footnotes
Acknowledgements
We would like to thank our colleagues Dr. Harikrishnan, Dr. Arun Tiwari, Dr. Thomas, Dr. Salil, Mrs. Beenamol K.P, Mr. Sekhar V. Eswar, Ms. Kavya Panikar, Ms Ameesha from the Department of Rheumatology and Clinical Immunology, AIMS Kochi, India - in for their help in data collection. We thank Prof. KR Sundaram and Ms. Renjitha, Department of Biostatistics, for aiding in data analysis.
Author Contributions
RK, SB, VM, SK designed the study. RK and PC collected data for the study. RK and MP performed data analysis. RK, SB and MP wrote the first draft of this article, while others contributed significant revisions to the final draft.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This research was undertaken after ethical approval from the Institutional Review Board of Amrita Institute of Medical Sciences, Kochi, India (Approval no. IRB-AIMS-2019-033)
Funding
The first author undertook this study per the DM residency requirements at Amrita Institute of Medical Sciences, Amrita Vishwa Vidyapeetham, Kochi, India, and received monetary support for the conduct of the study from Amrita Vishwa Vidyapeetham University.
Patient Consent
Written informed consent form from the parents as well as assent from the children (>7 years) were taken before enrolment in the study.
