Abstract
GMCSF, discovered in 1977, was one of the first growth factors for myelopoiesis. However, over time, it has been found to be redundant for basal myelopoiesis. Rather, its important function seems to be its effect on mature myeloid cells and the immune system. It is produced by a variety of cells, including macrophages, endothelial cells, T-cells etc. and works on the GMCSF receptor, which is mainly expressed by mature myeloid cells such as neutrophils, monocytes and macrophages. It has been found to be an important mediator of at least two autoimmune diseases: rheumatoid arthritis and multiple sclerosis. Animal models prove that GMCSF produced from T-cells play a crucial role in their pathogenesis of both these autoimmune diseases. This has led to the development of therapeutics which target GMCSF or its receptor. Clinical trials have been carried out in rheumatoid arthritis, which have shown modest efficacy. In addition, anti-GMCSF therapy has shown promise in giant cell arteritis.
Introduction
Granulocyte-macrophage colony stimulating factor (GMCSF) is a small glycoprotein which was among the first of the hematopoietic growth factors discovered in the late 1970s. 1 However, over time, it has been found to be relatively redundant for the production of granulocytes and other cells in that lineage, and mice with its knockout do not develop any deficiency in these cells. Rather, these mice die from alveolar proteinosis, due to a dysfunction of alveolar macrophages in clearing surfactants. 2 Although GMCSF was cloned nearly four decades ago, and has become commercially available since the last three decades, its utility has been surpassed many times over by recombinant G-CSF, which is favoured in conditions such as bone marrow transplant, neutropenia etc. 3
Over time, there has been increasing appreciation of its role as an important component of the immune response. It is produced by a variety of immune cells such as T-cells, macrophages and monocytes and has a central role in driving inflammation by the activation of macrophages and neutrophils, differentiation of monocytes to monocyte-derived dendritic cells (MoDC) and macrophages to M1 type.4,5 It has been found to be an important component of pathogenesis of various autoimmune diseases, mainly multiple sclerosis (MS) and rheumatoid arthritis (RA). GMCSF knockout renders mice resistant to developing experimental autoimmune encephalomyelitis (EAE), the animal model for MS, and resistant to CIA. 6 Even in humans with a disease, there has been appreciation of the high concentration of GMCSF in the synovial fluid (in RA) as well as in brain tissue (in MS) and increased frequency of T lymphocytes secreting GMCSF in both of these diseases.
Thus, in the last decade or so, therapeutics in the form of targeting the GMCSF receptor or GMCSF per se have been developed and have undergone clinical trials in RA (up to phase 3) as well as some other autoimmune diseases.
This review focuses on recapitulating the interesting journey from growth factor to inflammatory cytokine for GMCSF and evidence of its important role in autoimmune diseases like RA. Finally, we examine the status of clinical trials with this molecule and examine what the future holds for targeted therapeutics against this molecule.
Biology of GMCSF
GMCSF is a small glycoprotein of 23 KDa consisting of 127 amino acids (human) with two N-linked glycosylation sites. It is found in both humans and mice, and it is encoded by highly homologous genes.7,8 GMCSF can be produced by both hemopoietic and non-hemopoietic cell populations depending on the inducing stimulus and model. 7 It is produced by various cell types such as activated T-cells, macrophages, monocytes, vascular endothelial cells, mast cells and fibroblasts.
GMCSF receptor (GMCSFR) is a type I cytokine receptor, of the β-chain family (sharing β-chain with IL-3 and IL-5). GMCSFα chain binds directly with GMCSF with low affinity, while GMCSFß does not bind alone but increases affinity when binding along with GMCSFα.9,10 It is expressed in myeloid cells and some non-hematopoietic cells but not expressed by T-cells. 11 Four distinct signalling pathways, namely PI3K-Akt, ERK1/2, JAK2/STAT5 and NF-kB, are known to be involved in GMCSF. JAK-2 is closely related to the β-chain and gets phosphorylated on GMCSF binding to its receptor and leads to STAT5 activation. 12 In addition, GMCSF receptor activation promotes cell survival via PI3K pathway and JAK/STAT5-Bcl-2 and proliferation by Erk and NF-kB signalling. GMCSF promotes immune responses by signalling through ERK1/2 and NFκB activation. 13
Hematopoietic Role of GMCSF
In the 1960-1970s there was a huge effort to find ‘growth factors’ which were important for driving the generation of mature cells from bone marrow precursors. This was made possible due to the development of mouse bone marrow culture in vitro, which served as indicators for the presence of these factors by showing colony formation. 14 The first such growth factor isolated was erythropoietin (for RBCs). Its discovery accelerated research to find a similar ‘poietic’ factor for granulocytic–macrophage lineage. The first member to be isolated for these cells was murine MCSF followed closely by GMCSF (both in 1977) from a mouse-lung-conditioned culture media. 1 It was so named due to its ability to generate ‘colonies’ of granulocytes and macrophages from hematopoietic precursors.
It was soon realised that in contrast to erythropoietin, which is the sole regulator for haematopoiesis, there were many such factors which existed for the granulocyte–macrophage lineage. The other members of what is now known as the ‘colony stimulating factor family’ comprise (apart from GMCSF) macrophage colony-stimulating factor (M-CSF) and granulocyte (G) colony-stimulating factor (G-CSF).15,16
Paradoxically, despite it being one of the first growth factors which promoted myelopoiesis, GMCSF was found to be redundant for steady state myelopoiesis (unlike G-CSF). GMCSF knockout (GMCSF−/−) mice developed normally (with normal cell counts), whereas G-CSF−/− mice died due to hematopoietic failure. However, GMCSF−/− mice were found to be prone to the development of pulmonary alveolar proteinosis (PAP) due to the dysfunction of alveolar macrophages in clearance of surfactants.2,17-19 An interesting fact is that some humans also develop autoantibodies to GMCSF and, similarly, have development of pulmonary alveolar proteinosis and some increased susceptibility to infections. 20 Unlike knockouts with only slight defect in immunity, overexpression of GMCSF is pathological, and in mice in whom bone marrow cells are transfected with GMCSF genes via virus, there is a high GMCSF level, causing a fatal myeloproliferative syndrome characterised by infiltration of macrophages and neutrophils in the liver, spleen and various other organs. 21
Subsequent to cloning of its genes in 1985, Sargramostim, a recombinant GMCSF, was developed in yeast and underwent clinical trials from 1987 to 1991. In 1991 it was first approved by US FDA for treatment after bone marrow transplant.22-24 However, in subsequent years, most of the times, the recombinant G-CSF Filgrastim, which was approved in the same year as GMCSF, was preferred as the agent to combat neutropenia whether after transplant or chemotherapy or to mobilise stem cells rather than GMCSF. The reason for the same is that G-CSF works better than GMCSF for the same role of mobilising neutrophils (in the case of neutropenia) or mobilising stem cells (in the case of hematopoietic stem cell transplant) with lesser adverse effects. 3
Role of GMCSF in Immune Response
As mentioned above, GMCSF is redundant in steady state production of granulocyte–macrophage cells, apart from some important roles like alveolar macrophage cell maturation. So, the question arose whether there was another role of GMCSF?
It was soon realised that apart from myelopoiesis, GMCSF (and G-CSF) also had an effect on mature cells of the granulocytic–macrophage series. They had an effect on all mature myeloid cells including neutrophils, monocytes and macrophages. They played a role in activation, differentiation, mobilisation and function of myeloid lineage cells. GMCSF (along with TNFα) had an indirect effect on neutrophils, which, when given before, made them hyperresponsive to a usual triggering stimulus. This was called ‘priming’ of the neutrophil and is thought to occur partly due to the upregulation of receptors like the f-Met-Leu-Phe (N-formylmethionine-leucyl-phenylalanine) on the surface of neutrophils.25-27
Its effect on antigen-presenting cells such as monocytes and macrophages was to increase the expression of key molecules involved in the class II MHC pathway along with increased expression of CD86 and CD40. 28 These cells are thought to be important mediators of inflammation at local sites as they migrate to areas of inflammation like joints and are thought to perpetuate inflammation. GMCSF also converts tissue macrophages to proinflammatory M1-macrophages and stimulates them to secrete a variety of pro-inflammatory cytokines like IL-1β, TNFα, IL-6 or IL-23. Also, it leads to the upregulation of various other receptors such as scavenger receptors, Fcγ receptors and toll-like receptors on the surface of macrophages. This is mediated by a transcription factor PU, 1 which is also involved in an alveolar macrophage–clearing surfactant in the lungs.29,30
GMCSF possesses the capacity to differentiate monocytes into monocyte-derived ‘dendritic cells’. 31 Otherwise its direct effect on dendritic cells is variable, promoting the development of one type but inhibiting another type of DC. 32 It has been suggested that the GMCSF functions in expanded myeloid cells in vivo are influenced by their tissue microenvironment. Those cells responsive to GMCSF are expected candidates that are able to release mediators such as cytokines, chemokines, reactive oxygen species and proteases. 33
Interestingly, GMCSF at low concentrations supports cell survival without proliferation, while at higher concentrations, GMCSF promotes both survival and proliferation, showing a dose-dependent effect in a concentration-dependent manner.
Overall, GMCSF is thought to be important particularly for defence in the lung against infections. GMCSF−/− mice are found to have some increased susceptibility to pulmonary infections (apart from PAP). 34
GMCSF in Autoimmunity
One of the major fields where GMCSF research has progressed rapidly has been in autoimmunity. GMCSF-producing T-cells may delineate into a subset that might be involved in inflammation and pathogenesis of various autoimmune diseases (Figure 2).13,33
The first autoimmune disease where GMCSF was found to have an important role in pathogenesis was EAE, the animal model for MS. Initially, Th1 cells produced under the influence of IL-12 were thought to be important mediators of EAE. But it was soon realised that it was actually IL-23 (which shares a common p40 subunit with IL-12) which was the important polarising cytokine (and not IL-12), and the major mediators were actually Th17 cells (and not Th1 cells). A series of elegant experiments showed that IL-23-deficient mice were resistant to the development of EAE, and the critical pathway for EAE was IL-23-induced proliferation of Th17 cells.35,36
It is in the context of the function of Th17 cells that GMCSF was found to be important. Surprisingly, IL-17A, after which the Th17 cells were named, was not the major mediator of the damage in this model, as the knockout of IL-17 did not prevent brain lesions. However, the knockout of GMCSF completely prevented the development of any brain pathology, suggesting that GMCSF (rather than IL-17) produced from these Th17 cells was critical to encephalitogenicity. 37
GMCSF induces proliferation and activation of microglial cells essential for the onset of EAE. 25 These activated microglial cells are known to produce highly neurotoxic molecules such as reactive nitrogen and oxygen species, TNF-α and glutamate.38,39 The current concept is that Th17 cells that migrate to CNS in the EAE model convert to the Th1 subtype with more pathogenicity (now called the exTh17 cells or Th1/Th17 cells), and these secrete a variety of pro-inflammatory cytokines including GMCSF, IL-1β and IL-17A. The transcription factors involved are both RORc and Tbet.40,41
In humans with MS, increased frequencies of T-cells producing GMCSF, particularly the CD8+ T-cell subset, have been found in the cerebrospinal fluid and brain lesions.42,43 Interferon-β, which is used as a treatment for MS, has been shown to reduce the frequencies of these cells.44,45 Some studies have also found higher CD8+ T-cells producing GMCSF in peripheral blood, although this has not been consistently shown by all studies. 46
In addition to T-cell-mediated immunity, humoral immunity, that is, B-cells, are also known to produce cytokines that potentially modulate the inflammatory microenvironment.47,48 Even these have been shown to produce GMCSF and other pro-inflammatory cytokines. In this context, treatment with the anti-CD20 antibody has shown decline in Th1 and Th17 cells and also depletes B-cells. 48 Another study explored dimethyl fumarate (DMF) for the depletion of B-cells producing GMCSF.49,50
The second autoimmune disease, which is of course of much more interest to rheumatologists, in which much research has linked GMCSF with pathogenesis, is RA. As GMCSF was cloned in 1985 and came to the clinic few years later, there were reports of arthritis flare upon GMCSF administration in RA patients. 51 In addition, animal models of arthritis too suggested it to have an important role. GMCSF−/− knockout mice were found to be resistant to the induction of collagen-induced arthritis (CIA). 6 Administration with antibodies against GMCSF resulted in the blocking of disease progression and reduction in the pro-inflammatory cytokines. 52 In the SKG mice model for arthritis, treatment with GMCSF amplified the production of IL-6 and IL-1β in macrophages that induced the differentiation of CD4+ T-cells and promoted the production of IL-17 and GMCSF.
GMCSF is thought to play an important role by activating or promoting differentiation and survival of macrophages and neutrophils in the synovium in RA.53-55 GMCSF in the synovium has been found to be produced from many cells. Previous studies have also implicated innate lymphoid cells 2 (ILC-2) as the possible source of GMCSF. 56 Hirota et al have elucidated that ILC-2s that are CD25+IL-33Ra+GATA3+ are the predominant ILCs that secrete GMCSF steadily at the site of the disease. 57 Also, fibroblast-like synoviocytes (FLS) in response to stimulation by IL-1 and TNFα have shown to trigger the production of GMCSF.58,59
It has been found that CD4+ T-cells in RA synovial fluid promote the differentiation of CD14+ monocytes to CD1c+ inflammatory DC population in an indispensable GMCSF-dependent manner. Another study showed that the ex vivo culture in the presence of GMCSF of differentiated human monocyte–derived dendritic cells (MoDCs) (CD14+CD33+) is capable of class II–mediated expression of two very important and prominent immune epitopes of auto-antigens [type II collagen (CII) and cartilage gp39 (HCgp39)] in the inflamed synovial joints of RA. 60 These GMCSF-stimulated MoDCs in the presence of TLR 2 and TLR 4 ligands led to a higher production of proinflammatory cytokines such as TNFα and IL6 in RA patients. 61 MoDCs in the synovial fluid derived from GMCSF stimulation have a more proinflammatory phenotype. 62
Makris et al have reported increased frequencies of GMCSF-producing B- and T-cells in the peripheral blood of RA patients and that the GMCSF+ B-cells were more of plasmablast cells or in a transitional phase and likely contributors to the auto-antibody production in RA pathogenesis. They also found the increased expression of GMCSF in B- and T-cells to be correlated to the disease activity. 63 Studies in RA have also reported increased frequencies of GMCSF-producing CD4+ T-cells in the synovial fluid (SF), with the capability to generate inflammatory dendritic cells (inf DCs) from monocytes in vitro, suggesting an important pathogenic link.42,63-65 These cells reduced in frequency after anti-TNF treatment.42,63,66 Recently, a study from our group has reported an increased frequency of CD8+ T-cells producing GMCSF in the SF of RA patients that is polyfunctional for the production of TNFα and IFNɤ. 67
Another disease in which GMCSF may have a role to play is interstitial lung disease. In idiopathic pulmonary fibrosis (IPF), BAL fluid has been shown to have high GMCSF levels. 68 SKG mice, which are an animal model for RA, are known to develop interstitial lung disease. In this disease, lungs have been shown to be infiltrated by neutrophils, Th17 cells and T-cells producing GMCSF. Interestingly, blocking GMCSF prevented the development of lung fibrosis in this model. 69
Other autoimmune diseases where GMCSF has been thought to be important are inflammatory disease like Crohn’s disease (CD) and ulcerative colitis. 70 In earlier studies from a murine model for CD, GMCSF-deficient mice have been shown to be more vulnerable to developing acute dextran sodium sulphate (DSS)–induced colitis, which was ameliorated by GMCSF administration.71,72 This is contrary to the models for RA and MS and is thought to be mediated by GMCSF also influencing signalling pathways in myeloid-derived suppressor cells (MDSCs).These MDSCs expand vigorously during inflammation and infection and suppress T-cell responses, 73 although their exact role in models for experimental colitis and IBD pathogenesis is unclear. 74 An additional hypothesised function of GMCSF involves communication between mononuclear phagocytes and group 3 innate lymphoid cells (ILC3s) for intestinal homeostasis by upregulating the function of DCs and regulatory T-cells (T regs).75,76 Indeed, the administration of GMCSF in CD in clinical studies has shown good clinical response and remission in CD patients with moderate to severe disease activity.77,78 Furthermore, a phase 2 randomised clinical trial showed GMCSF to be more effective than placebo for remission and good response. 79
Anti-GMCSF Therapies
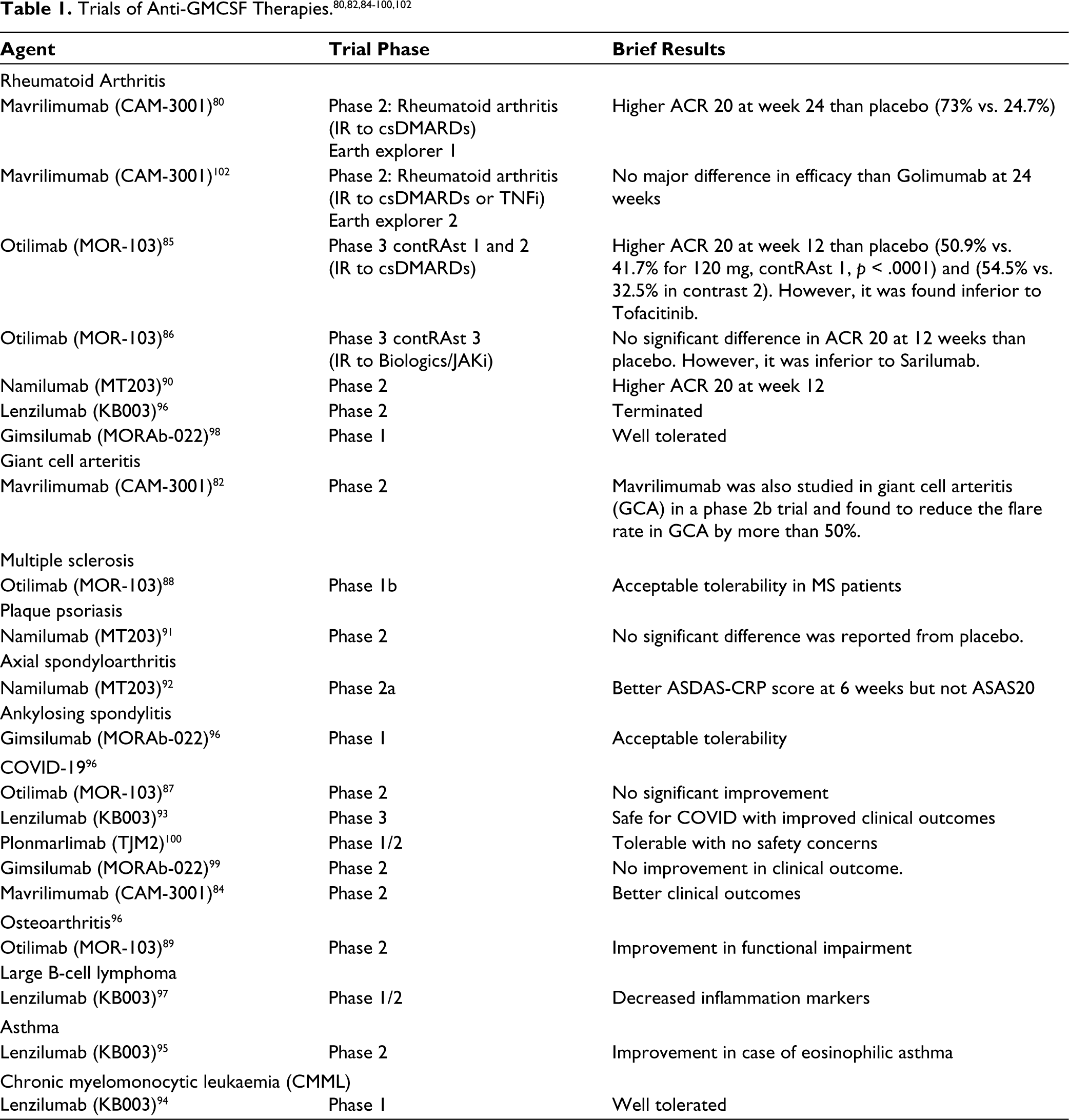
Given the role of GMCSF as an important player in autoimmune diseases, trials have examined its role as a therapeutic target (Table 1). The overall timeline of GMCSF and the development of anti-GMCSF therapies is shown in Figure 1.
Timeline of GMCSF Discovery and Anti-GMCSF Therapy Development.
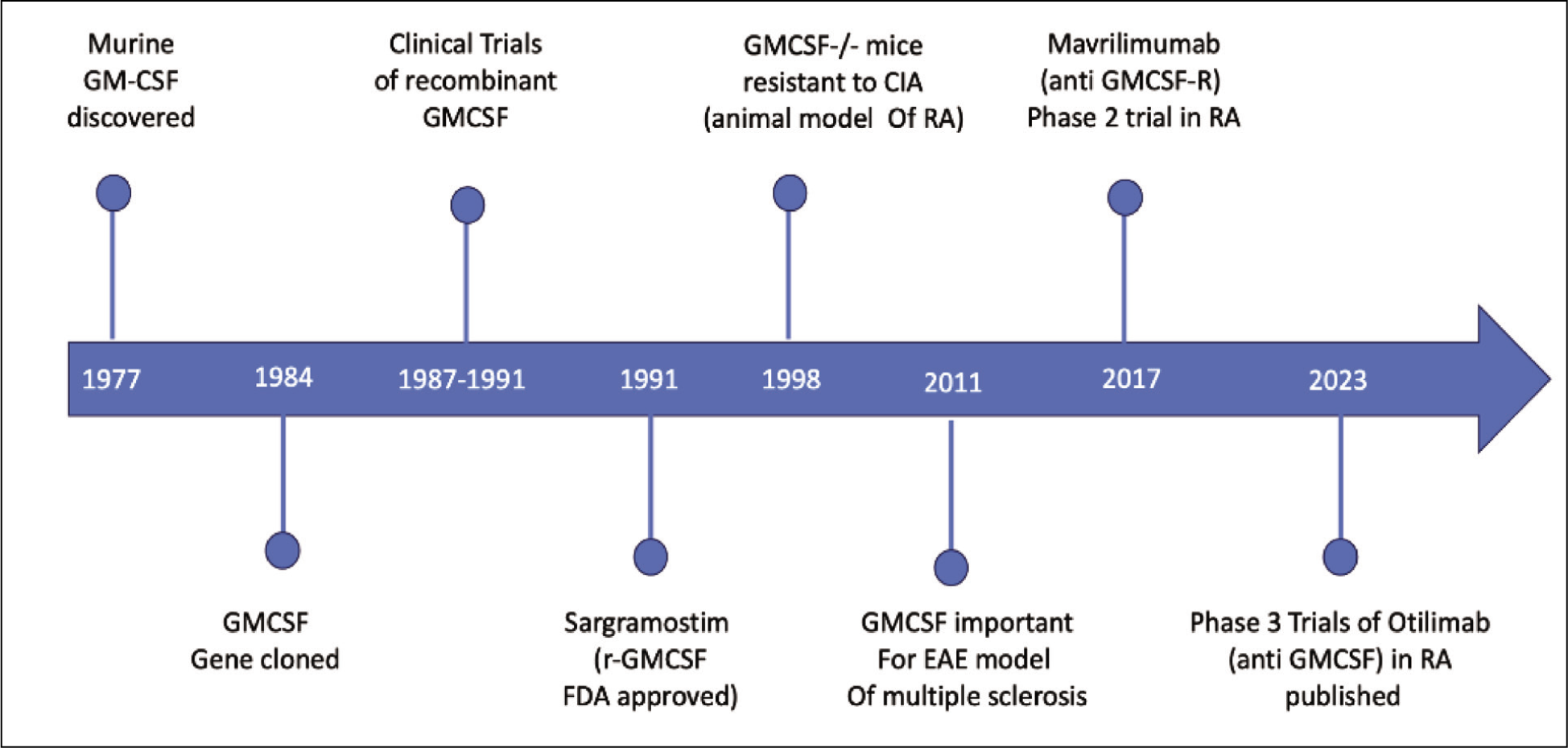
Schematic Diagram Highlighting the Sites and Role of GMCSF in Disease Pathogenesis.
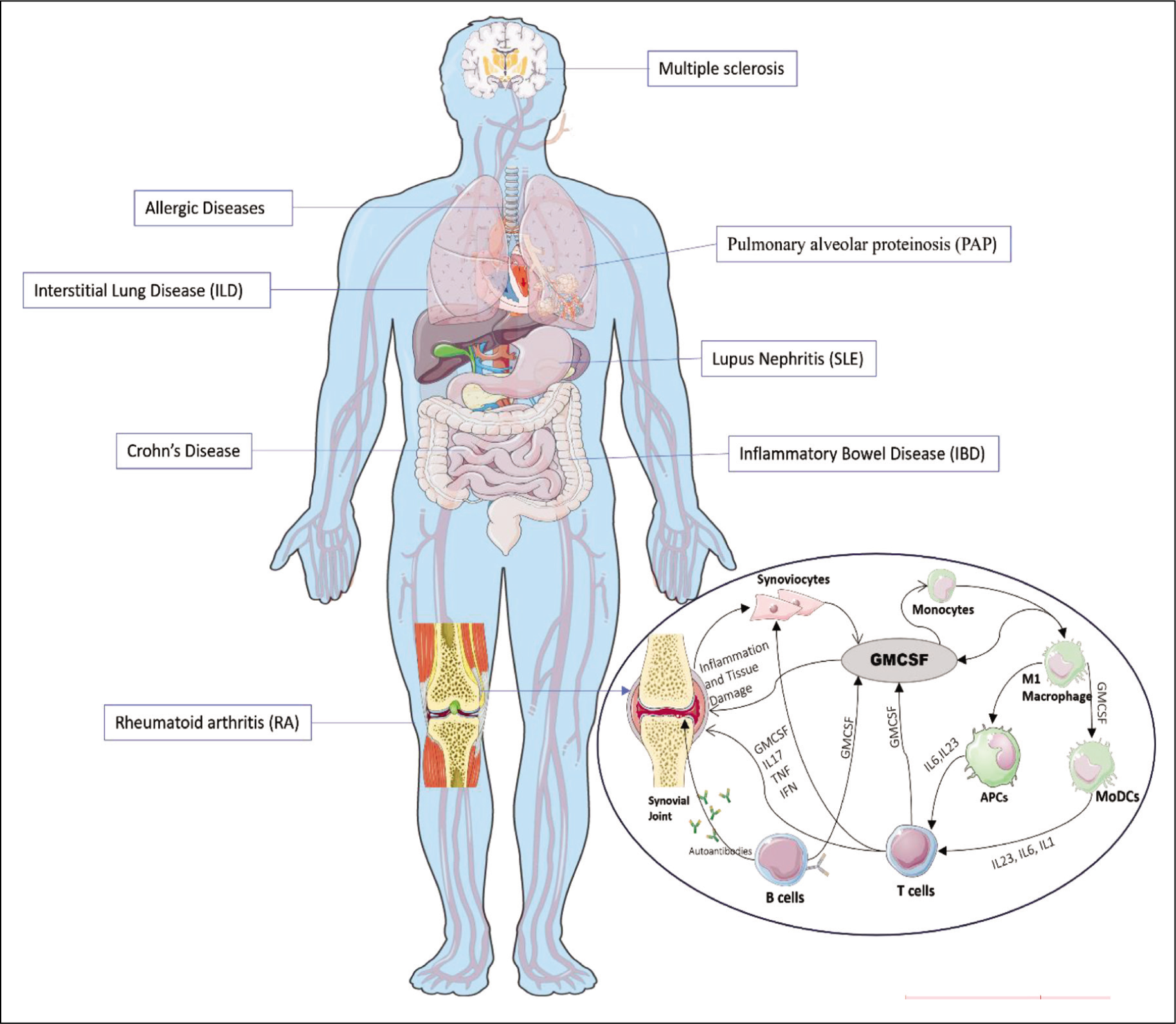
Mavrilimumab (CAM-3001)
Mavrilimumab is a monoclonal antibody targeting the GMCSF α receptor. It underwent a phase 2b trial in RA patients (IR to csDMARDs) and found significantly higher ACR 20 than placebo (73% vs. 24.7% at week 24). 80 In the long-term extension study, meaningful responses continued up to three years; however, there was a 20% reduction in forced vital capacity (FVC) in 2%-3% of the patients. However, in most cases, these were reported to be asymptomatic. 81 It was also studied in giant cell arteritis (GCA) in a phase 2b trial and reduced the flare rate in GCA by more than 50%. 82 This led to it Mavrilimumab being given an orphan drug designation for GCA in 2020 by the FDA. During the COVID pandemic, it was trialled as a treatment for COVID-19 but failed to show efficacy in the MASH COVID study, 83 whereas another study showed an improvement in the clinical outcome in severe COVID. 84
Otilimab (MOR-103)
Otilimab is a fully humanised monoclonal antibody against GMCSF. In two phase 3 randomised controlled trials (RCTs) in RA patients (with IR to csDMARDs), comprising more than 1,500 patients (contRAst 1 and 2), it showed only modestly higher ACR 20 responses at week 12 compared to placebo (gain of 10%-12%) (50.9% vs. 41.7% for 120 mg in contrast 1, p < .0001) and (54.5% vs. 32.5% in contRAst II). Tofacitinib, which was included as an active comparator in both studies, was superior to Otilimab at 24 weeks. 85
Another phase 3 RCT included patients of RA with an inadequate response to biologics or JAKis (contRAst 3) but could not demonstrate any difference in efficacy compared to placebo in ACR 20 at 12 weeks. This study also had an active comparator, which was similar in design to the previous two RCTs, but the agent in this study was Sarilumab. This was found to be superior to Otilimab. 86 No major unexpected severe adverse event (SAE) was found, no cases of PAP, with <1% MACE and <4% serious infections (half being COVID and no TB). It was trialled in COVID-19 but was not found to confer significant improvement; however, a satisfactory safety profile and decreased inflammation markers were seen. 87 It has also undergone safety and immunogenicity studies in MS and osteoarthritis (OA) patients and has been found to be safe.88,89
Namilumab (MT203)
Another monoclonal antibody against GMCSF, Namilumab, has been studied and has shown better DAS28 and higher ACR 50 along with acceptable tolerability in a phase 2 study in RA. 90 In plaque psoriasis, no significant difference was observed in comparison to placebo. 91 In axial spondyloarthritis, better ASDAS score was observed versus placebo (secondary end point), but not in ASAS20 versus placebo (NAMASTE study). 92
Lenzilumab (KB003)
Lenzilumab (KB003) was found to be safe and showed some improvement in oxygen requirement and clinical outcome in COVID-19 patients with severe pneumonia. 93 It was found to be tolerable and safe in chronic myelomonocytic leukaemia (CMML). 94 It was not efficacious in eosinophilic asthma.95,96 It has also been trialled in B-cell lymphoma 97 and RA (study terminated prematurely). 96
Gimsilumab (MORAb-022)
Gimsilumab (MORAb-022) was found to be tolerable and safe in RA and healthy people. 98 It was trialled in COVID-19 pneumonia but was not found to be beneficial. 99 A randomised trial in ankylosing spondylitis has been registered. 96
Plonmarlimab (TJM2)
Plonmarlimab was tried in severe COVID-19. 100
The major concern with targeting GMCSF has been the theoretical risk as to whether it would lead to respiratory problems like pulmonary alveolar proteinosis. 101 This is because knockout of GMCSF in mice, as well as autoantibodies to GMCSF in humans, has been shown to lead to this disease due to a deficiency in surfactant clearance by alveolar macrophages. However, this has not been found to occur in any of the clinical trials. The lower FVC in a long-term study was worrying although reported to be asymptomatic.
Conclusion
GMCSF is among the first ‘growth factors’ discovered which promoted myelopoiesis. However, in steady state it does not have an important role in myelopoiesis; rather, it seems to be much more important for its activating effect on mature myeloid cells—neutrophils, macrophages and monocytes. It is thought to play an important role in the pathogenesis of some autoimmune diseases such as MS and RA, as elucidated by their animal models as well as ex vivo studies of human samples. Therapeutics targeting GMCSF have been developed, and there have been phase 2 studies in RA, which have been promising, but phase 3 studies showed only modest results, which were lower than those of other approved therapies. Interestingly, it was also found to be effective in giant cell arteritis, for which the FDA approved an orphan drug label for one targeted therapy. In the near future we may find some therapy targeting GMCSF approval, if not for RA then in some other autoimmune disease.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval was not obtained from the local Ethics Committee or Institutional Review Board as it was a review article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: APLAR research grant 2022 was received by Varun Dhir and ICMR senior research fellowship was obtained by Aastha Khullar.
Patient Consent
Consent was not needed/not applicable as this did not include any patient related data.