Abstract
Introduction:
Systemic sclerosis (SSc) is a chronic disorder, characterised by endothelial dysfunction and fibrosis of skin and internal organs. Endothelium dysfunction leads to a decrease in nitric oxide levels, consequent to reduced Nitric Oxide synthase 3 (NOS3) activity due to decreased NOS3 gene function. Besides endothelium, macrophages too play an important role in the pathogenesis of SSc; classically activated M1 macrophages (express NOS2) and alternatively activated M2 macrophages (express Arginase1) are differentially altered in favour of M2 macrophages. Micro-RNAs (miRNAs) regulate expression of various genes and may favour disease phenotype.
Aim:
Our aim was to identify differentially expressed micro-RNAs in SSc, which target NOS2, NOS3 and Arginase1 genes by in-silico approach.
Methods:
Data on miRNAs upregulation reported in various studies was collected and their sequence ID from miRBase was identified in FASTA format from NCBI. Binding strength of these miRNAs against NOS2, NOS3 and Arginase1 genes was evaluated by RNA22.
Results:
After scanning 39 publications reporting miRNA expression in SSc, a total of 13 miRNAs were found to be up-regulated for NOS2 (Gibbs free energy ≤18.0Kj/mol) and 15 miRNAs up-regulated for NOS3 (Gibbs free energy ≤18.0Kj/mol) whereas for Arginase1 only 2 miRNAs were up-regulated.
Conclusion:
There is differential upregulation of miRNAs in SSc. Micro RNAs targeting NOS2 and NOS3 genes are highly up-regulated in comparison to Arginase 1. Role of epigenetic regulation of genes by miRNAs may play a key role in pathogenesis of SSc.
Keywords
Introduction
Systemic sclerosis (SSc) is a chronic autoimmune disease, which is characterised by fibrosis of the skin and internal organs as well as vasculopathy. Its pathogenesis starts in genetically predisposed individuals through encounters with environmental factors such as cold, stress, chemical exposure, certain viruses, silica dust, medications or drugs, resulting in endothelial injury. 1 Normally, Nitric Oxide synthase 3 (NOS3) expressed by healthy endothelial cells helps control vascular tone, leucocyte migration, and blood clotting and helps in repair and homeostasis. 2 In SSc patients, NOS3 expression is highly reduced, which results in endothelial cell dysfunction. 3 The mechanism behind this discrepancy is still unknown. Macrophages are one of the major pathogenic cells involved in the pathogenesis of SSc. 4 They mount an immune response against foreign antigens, phagocytose foreign bodies and help in immune clearance of cellular debris and plays role in tissue repair and remodelling. 5 There are two types of macrophage subpopulations in circulation: classically activated or M1 macrophages and alternatively activated or M2 macrophages. 6 M1 macrophages express inducible nitric oxide synthase (iNOS), a product of NOS2 gene, and secrete cytokines like interleukin-1β, IL-6, IL-12, and tumour necrosis factor-α (TNF-α) causing tissue injury while M2 macrophages express Arginase 1, product of Arginase 1 gene, and help in the resolution of inflammation by secreting excessive amounts of IL-10 and TGF-β1 6 and regulates Th2 cell differentiation and excessive production of IL-4, IL-13, and TGF-β1. These are the key cytokines involved in the pathogenesis of SSc.7–9 Various studies have reported M1/M2 macrophage alteration in fibrotic diseases. 10 The population of M2 macrophages is highly up regulated in SSc.
Micro RNAs (miRNAs) are ~22 nucleotides sequence long RNAs, which are generated from pre-miRNAs. miRNAs interact with the 3′ UTR of target mRNAs to suppress their target gene expression. 11 Previous studies have reported that the expression of NOS2, 12 NOS3, 13 and Arginase 14 is regulated by human miRNAs. Therefore, we evaluated miRNAs up regulated in SSc that may target NOS2, NOS3 and Arginase 1 genes by in silico approach.
Methods
PubMed database (
From various published studies, we prepared details of miRNAs whose expression was altered (up-regulated or down-regulated) in SSc patients and prepared a library including
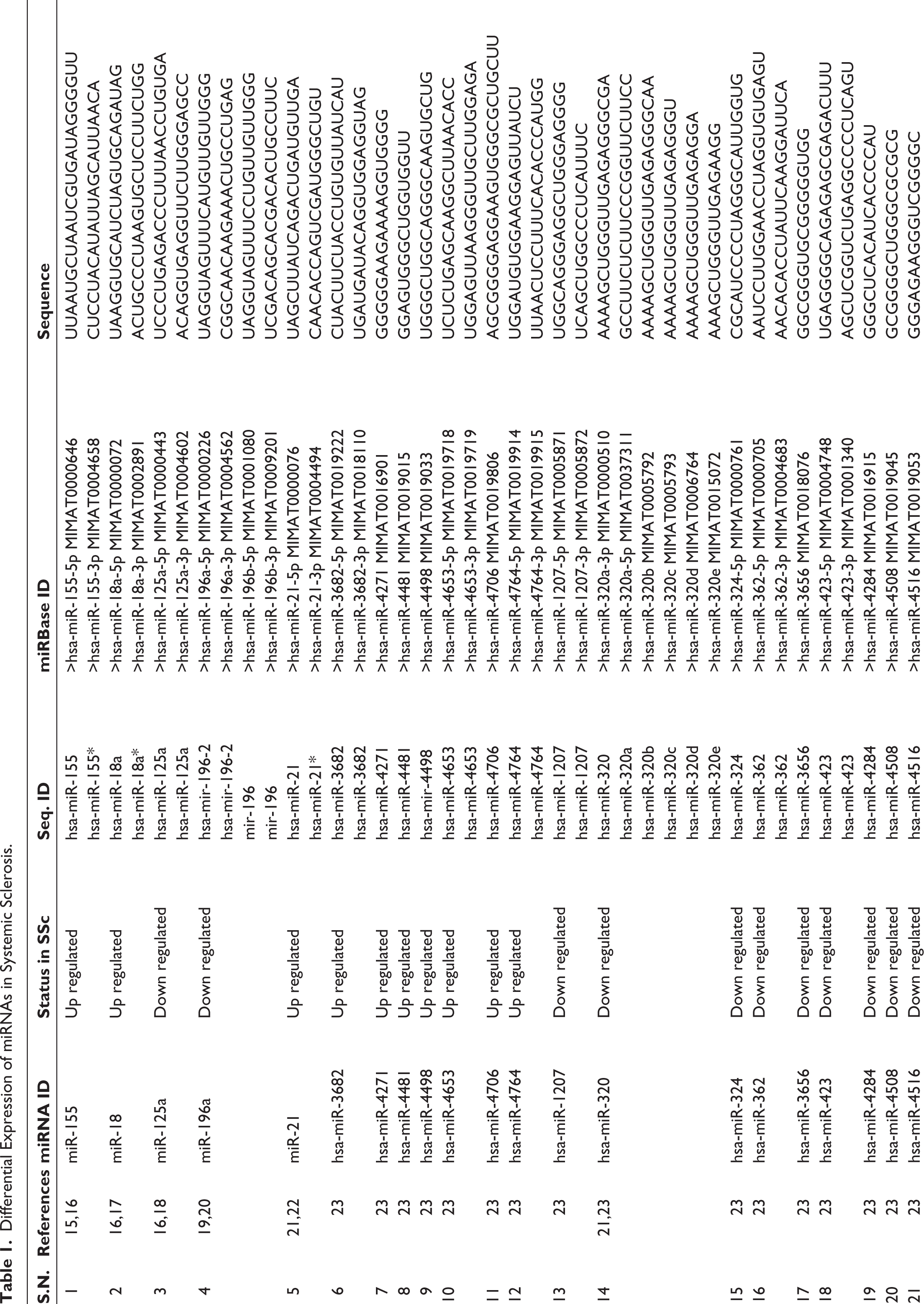
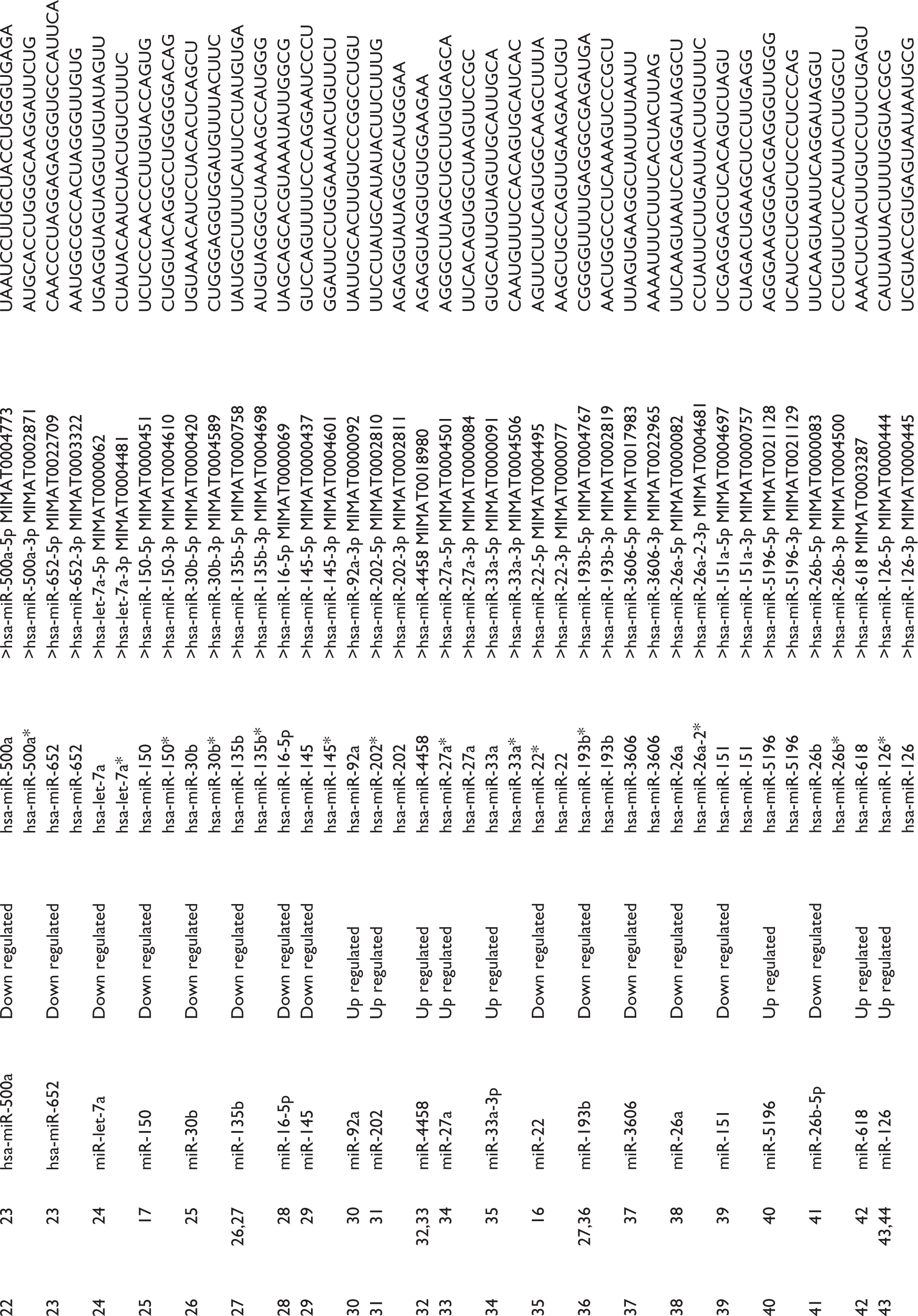
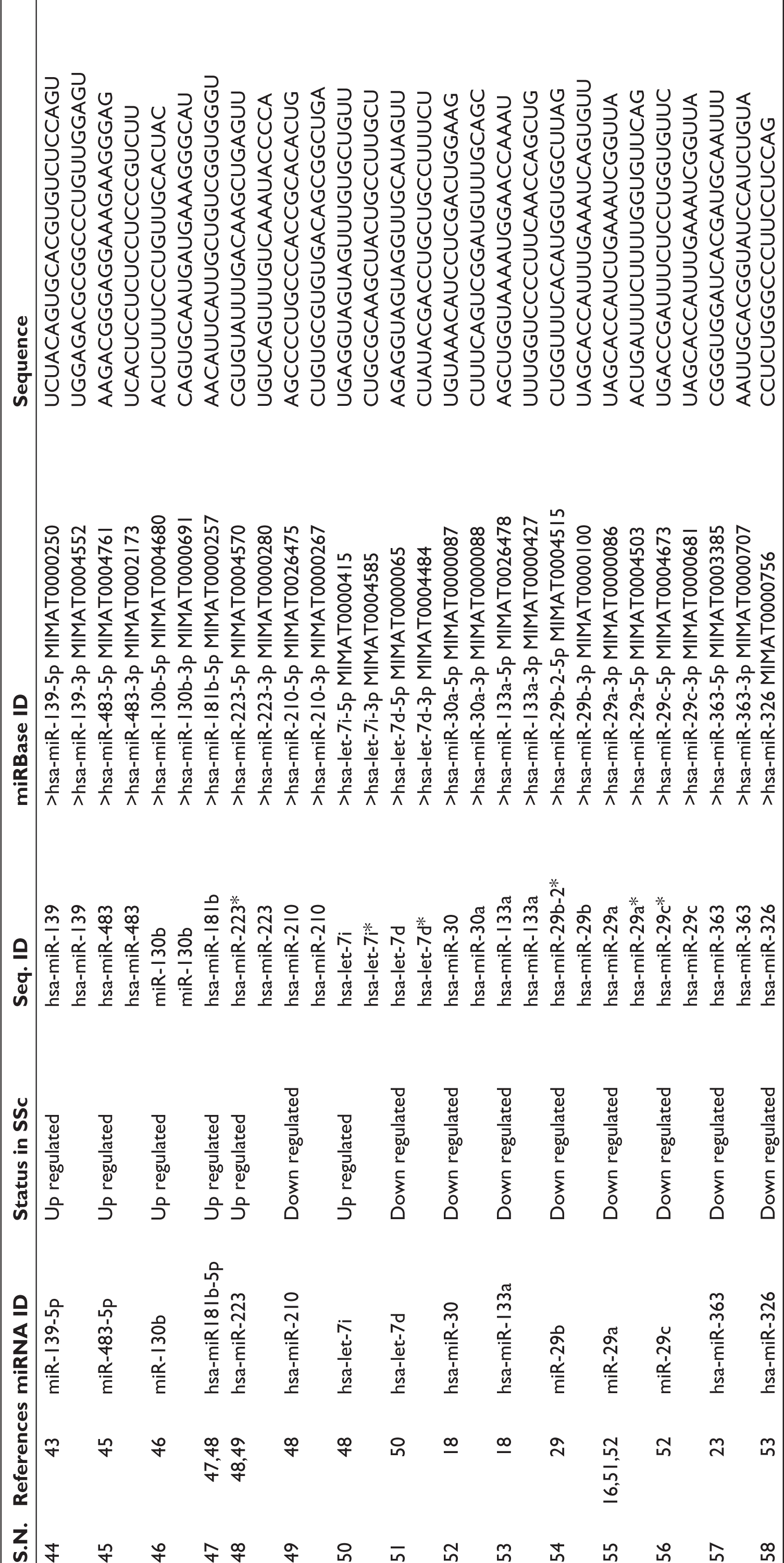
Differential Expression of miRNAs in Systemic Sclerosis.
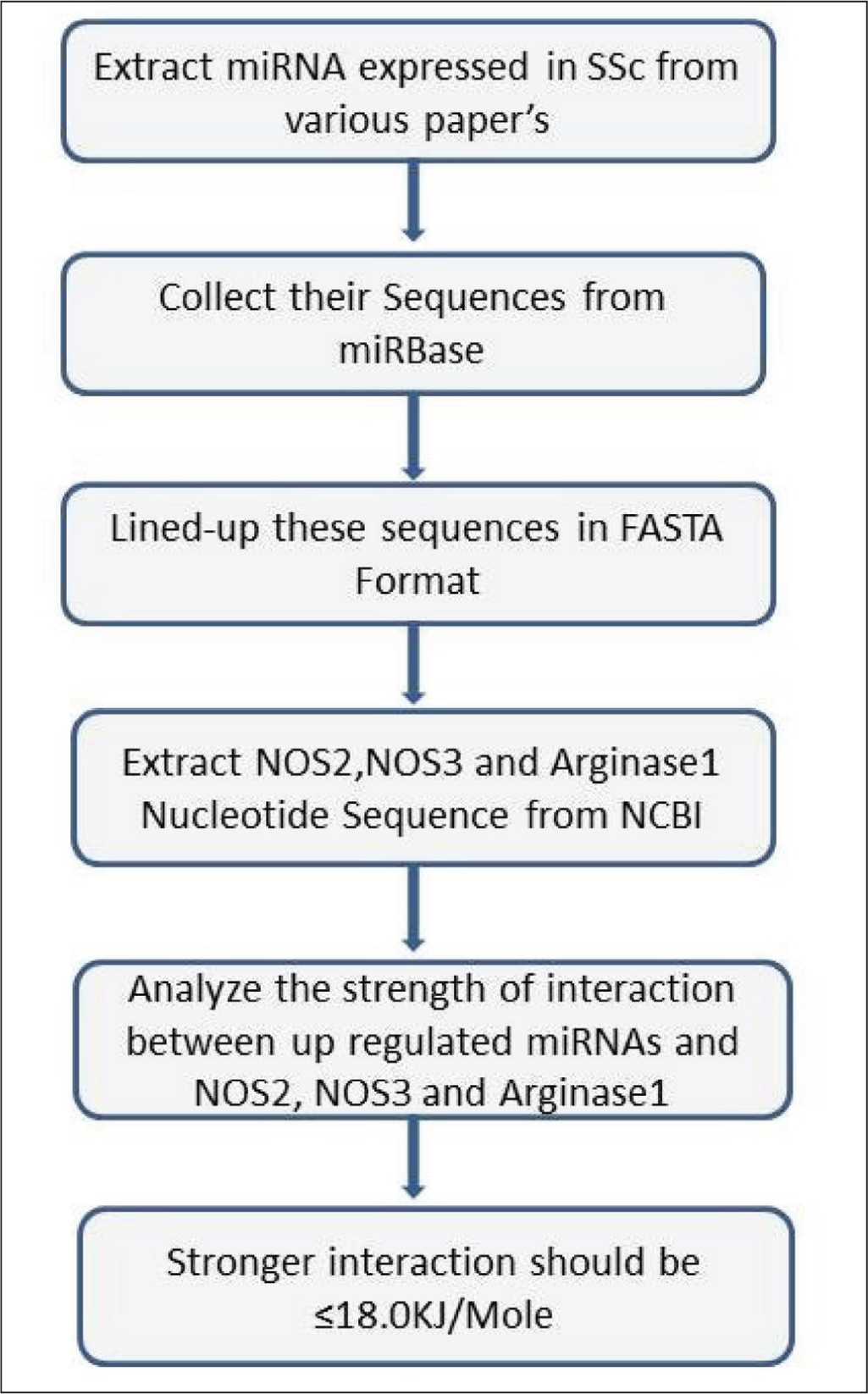
Flow Diagrammatic Representation of Identification of miRNAs up Regulated in Systemic Sclerosis, Extraction of Sequences, and Analysis of their Interaction with NOS2, NOS3 and Arginase1.
We extracted the miRNA sequences by using their sequence ID’s from miRBase (
Our target genes were NOS2, NOS3, and ARG1. We extracted their complete nucleotide sequences from NCBI database (National Center for Biotechnology Information) in FASTA format (
NOS2: (Accession No: NM_000625.4, BC144126.1 and BC130283.1)
NOS3: (Accession No: NM_000603.5, AF032908.1 and AF400594.1)
Arginase I (NM_000045.4, NM_001244438.2 and NM_001369020.1)
We evaluated the binding strength of miRNAs against nitric oxide synthases (NOS2, NOS3) Arginase (ARG1) by using an RNA22 webserver. 54 In RNA 22, input miRNA sequence (not more than 50) in FASTA format was followed by input single target sequence (NOS2, NOS3 and Arginase1) in FASTA format. Both the sequences aligned together and gave results in hetroduplex form.
Use of Gibbs Free Energy in Target Prediction
There are various criteria for target prediction of miRNA: strong, Watson Crick base-pairing of the miRNA to a complementary site in the mRNA, conservation of the miRNA binding site, favourable minimum free energy (MFE), also known as Gibbs free energy, for the local miRNA-mRNA interaction, and/or structural accessibility of the surrounding mRNA sequence. Gibbs free energy or MFE is used to measure the stability of miRNA-mRNA interaction. 55 Gibbs free energy less than –15 to –18 Kcal/mol indicates stable base pairing. Lower the Gibbs free energy more likely is the stable interaction between miRNA and mRNA. 56 We then identified the target sequences of various miRNAs, upregulated in SSc, having Gibbs free energy ≤ –18 Kcal/mol.
Results
First, we prepared the library of miRNAs whose expressions were altered in SSc patients (Table 1). We extracted the miRNAs sequence from miRbase and analysed the strength of targeting of NOS2 (Supplementary Table 1), NOS3 (Supplementary Table 2) and Arginase1 (Supplementary Table 3) by RNA22 webserver. A total of 15 miRNAs that strongly target NOS3, 13 miRNAs that target NOS2 and only two miRNAs that target Arginase-1 were identified. We interpreted the result according to Gibbs free energy (δG) of miRNA with target sequence which must be ≤18.0Kj/mole for the upregulated miRNA.
SSc miRNA Mediated Disrupted Endothelium Repair and Homeostasis
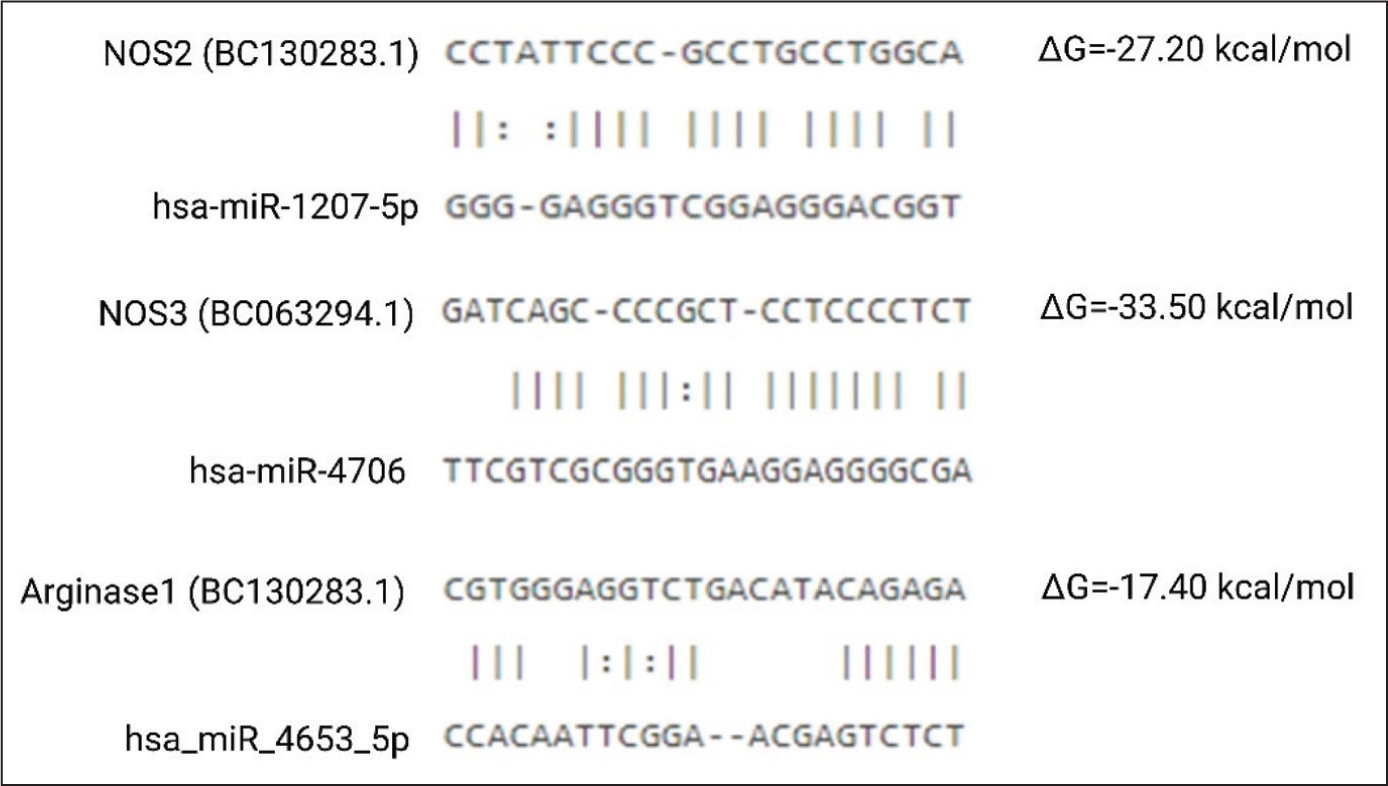
Fifteen unique miRNAs were upregulated in SSc that has potential to target NOS3 gene very strongly and may lead to impaired endothelial repair and homeostasis (Supplementary Table 2). Representative miRNA interaction with it is shown in Figure 2. This might be a cause of endothelial dysfunction. NOS3 dysfunction leads to excessive endothelium injury.
Interaction of miRNA up Regulated in Systemic Sclerosis Patients with NOS2, NOS3, and Arginase1 mRNA, Representative Image.
SSc miRNA Regulate M1 Versus M2 Macrophage Differentiation
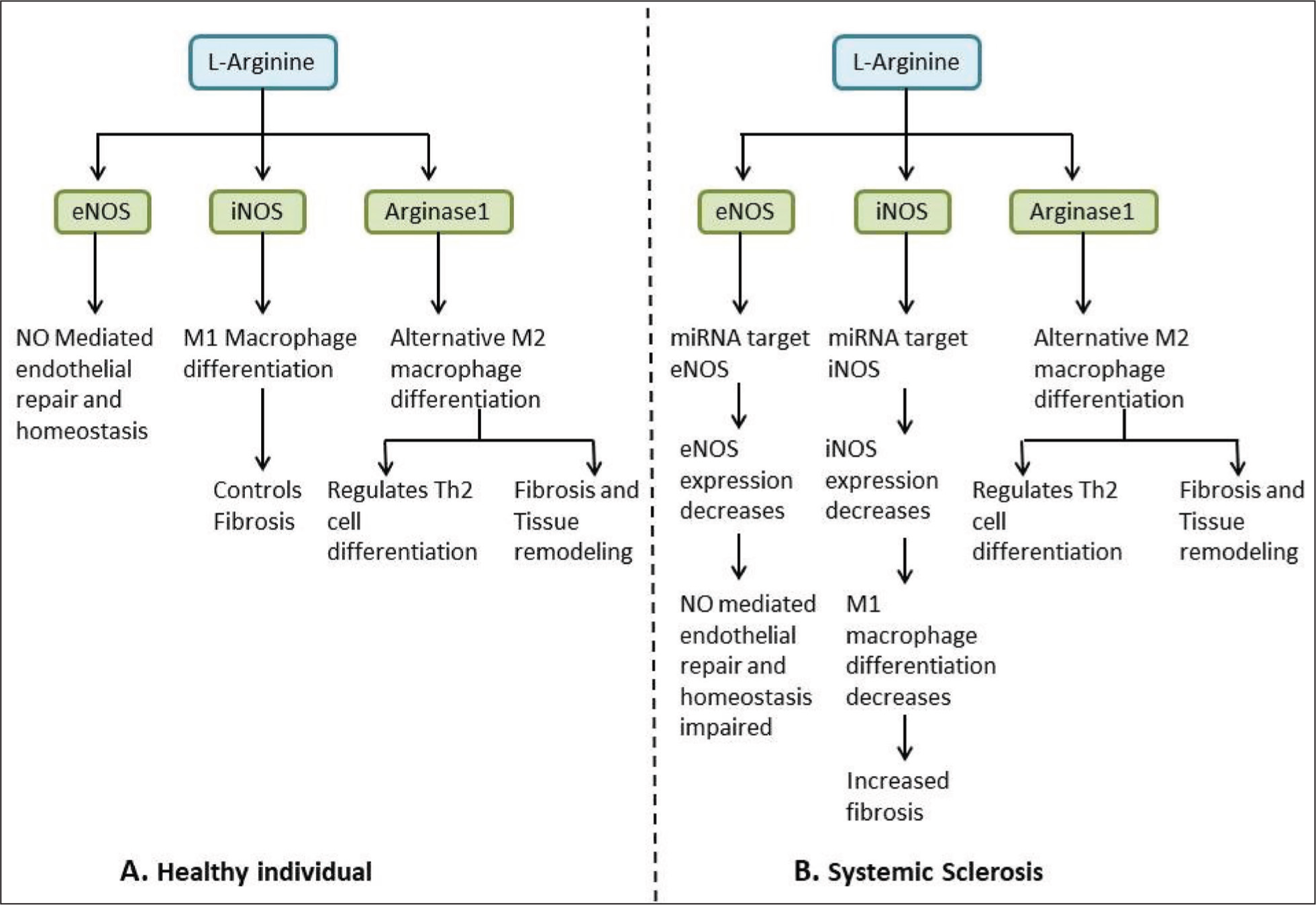
SSc begins with endothelial injury and M1/M2 macrophage dysregulation. In the initial phase, the endothelium gets disrupted and factors involved in repair are altered, resulting in enhanced damage to the endothelium and disruption of repair. M1 macrophages express NOS2 while M2 macrophages express Arginase1. In the present study, we observed that 13 microRNAs were strongly targeting NOS2, while only two miRNAs were targeting Arginase1. It suggested that miRNAs upregulated in SSc target NOS2 predominantly (Supplementary Table 2), as compared to Arginase1 (Supplementary Table 3), which may have resulted in reduced functionality of M1 macrophage and had no/minimal effect on the M2 macrophage population (Figure 3), resulting in M2 macrophage functional dominance. M2 macrophages express arginase1 and use arginine as a substrate. Arginase hydrolyses arginine to urea and ornithine, which acts as a precursor for polyamines and proline, which enhances collagen production and fibrosis. 57
L-Arginine Metabolism in Endothelial Cell and Macrophages in Healthy Control and Systemic Sclerosis Patients.
Discussion
In our in-silico study, we observed that NOS2 and NOS3 mRNAs are strongly targeted by miRNAs upregulated in SSc patients. Previous studies have shown that SSc pathogenesis starts with endothelial dysfunction in genetically predisposed individuals after exposure to environmental factors; cold, stress, chemical exposure, and exposure to certain viruses, resulting in activation and aggregation of platelets to form thrombi. The imbalance between vasodilators and vasoconstrictors leads to ischemic reperfusion injury. 1 Endothelial NOS (NOS3) is produced by endothelial cells and acts in an autocrine manner to stimulate the growth of endothelial cells and migration of endothelial cell progenitors leading to angiogenesis. NO diffuses into vessel walls, causing arterial vessels to relax and increase blood flow. NO also acts in a paracrine manner to prevent thrombosis by inhibiting platelet adhesion and aggregation. NO produced by NOS3 binds to the prosthetic heme on soluble guanylate cyclase (sGC) to stimulate cyclic GMP synthesis, activating cGMP-dependent protein kinase (cGKI) and cGMP-gated channels to regulate downstream targets that increase endothelial cell proliferation, migration, survival, and permeability. 57 Therefore, targeting NOS3 by miRNA may lead to endothelial dysfunction in SSc patients.
Additionally, low expression of NOS3 leads to the activation of endothelial cells and overexpression of cell adhesion molecules like intercellular adhesion molecule-1 (ICAM1) and Vasopressin-activated Ca(2+)-mobilising (VACM-1) which leads to excessive recruitment of immune cells 1 and excessive platelet activation and thrombosis as well as reduced angiogenesis. 3 Activated platelets are a rich source of serotonin (5 hydroxy tryptamine, 5HT) which through 5HT2 receptors mediate widespread fibrosis.
Similarly, in our in-silico study, we found that NOS2 mRNA is strongly targeted by miRNA upregulated in SSc patients, but only two of the miRNAs targeted Arginase 1 and that too with very little Gibbs free energy. This alteration may also play an important role in SSc pathogenesis. Macrophages in SSc are the major source of fibrosis-inducing cytokines, such as transforming growth factor β1 (TGF-β1). 58 Macrophages, the M1 phenotype, produce nitrogen intermediates and inflammatory cytokines such as interleukin-1β (IL-1β), tumour necrosis factor-α (TNF-α) and IL-6 to promote active inflammation. 59 In contrast, M2 macrophages have high expression of haemoglobin scavenger receptor (CD163), class A scavenger receptor (CD204), and mannose receptor (CD206). 60 They are associated with a high degree of vascularisation and influence wound repair. 61 Ishikawa et al. also demonstrated that the ratio of infiltrating macrophages, CD68+ cells, to T cells were high, suggesting an important role of cutaneous macrophages in SSc. 62 In SSc patients, M2 Macrophages and Th2 cells are present predominantly and these cells secrete cytokines such as IL4, TGF-β1, and IL13 which are the major players of fibrosis. 63 During TGF-β1 canonical signalling, SMAD-2/3 gets phosphorylated and interacts with SMAD-4 and gets translocated inside the nucleus and binds to promoter sequences of various pro-fibrotic genes resulting in excessive expression of pro-fibrotic genes. 64 Our study has translational potential, whereby targeting miRNAs that target NOS2 and NOS3 genes by chemically modified antisense oligonucleotides 65 may help in restoring the balance between M1 and M2 macrophages and thus reduced endothelial dysfunction and fibrosis. Our study has certain limitations as well; first, we analysed miRNAs targeting three genes only and second, we did not validate the expression of either various miRNAs or the three genes that were the target of miRNAs in samples drawn from the patients with SSc.
To conclude, we observed that there are many miRNAs that are upregulated and strongly target NOS2 and NOS3 but scarcely Arginase-1 genes. This could explain endothelial dysfunction and excessive fibrosis in SSc. Validation of these miRNAs in endothelial dysfunction as well as differential alteration in macrophage populations is required.
Supplemental Material
The supplemental material for this article is available online.
Supplemental Material
The supplemental material for this article is available online.
Supplemental Material
The supplemental material for this article is available online.
Footnotes
Acknowledgements
Authors acknowledge voluntary contribution of Akshat Agarwal and Harshita Pandey towards data acquisition, English language editing, and bibliography annotations in preparation of this manuscript.
Authors Contribution
The conception and design of the study – NS, MKR VA; acquisition of data, analysis, and interpretation of data – NS, VA. Drafting the article – NS, MKR; Revising it critically for important intellectual content – MKR, VA, VA. Final approval of the version to be submitted – NS, MKR, VA, VA. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved – NS, MKR, VA, VA.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
