Abstract
Background
Hypothermia is a significant concern in most neonatal units in both term and preterm, which increases the risk of mortality and morbidity. We aimed to reduce the incidence of hypothermia among at-risk preterm neonates upon admission to the neonatal intensive care unit (NICU) from a baseline of 75%-30% over 15 months.
Methods
This quality improvement study in a tertiary NICU implemented multiple plan-do-study-act (PDSA) cycles for all inborn neonates with a gestational age of less than 34 weeks or birth weight under 1500 g. The key outcome measure was the incidence of hypothermia, expressed as a percentage. Potential barriers to normothermia were evaluated using fishbone analysis and a key-driver diagram. Baseline data were collected over 3 months, and two PDSA cycles were performed over 18 weeks using a bundled approach and a sustenance phase over 9 months. Key interventions involved the optimization of environmental temperature at various locations, the use of polyethylene wraps for resuscitation, the proper use of transport incubators, and through raising awareness among healthcare professionals.
Results
A total of 319 neonates were part of the study. The baseline incidence of hypothermia (outcome measure) decreased from a median of 83% to 20.5% after two PDSA cycles and was sustained for over 9 months with reinforcement measures. Only two neonates had hyperthermia during the study period (balancing measure), and 96% compliance with documentation was achieved.
Conclusions
Quality improvement measures with simple policy reinforcement and a bundled approach can help reduce the incidence of hypothermia in at-risk preterm neonates.
Keywords
Introduction
Neonates are highly susceptible to respiratory complications, hypothermia, sepsis, hypoglycemia, and feeding difficulties, with hypothermia being a critical factor. Preterm neonates are at greater risk because of a larger body surface area, immature vasomotor control, permeable skin, and insufficient glycogen and subcutaneous fat stores, making them more vulnerable than term neonates. 1 Neonatal transport significantly contributes to hypothermia. 2 A study by the National Institute of Child Health and Human Development Neonatal Research Network (NRN) found that increased prematurity and lower birth weight correlated with higher incidence and severity of hypothermia. 3 Hypothermia is closely linked to severe neonatal morbidities and mortality.4–7 Various studies have explored interventions to reduce hypothermia in neonates requiring neonatal unit admission.8, 9 Team involvement, education, and staff awareness are essential for reducing hypothermia in neonates. 10
Despite the widespread implementation of the “World Health Organization Warm Chain,” neonatal hypothermia remains prevalent among term and preterm neonates, even in warm climates, with estimates ranging from 32% to 85%. 11 Poor implementation of the standard proven interventions to prevent hypothermia is a common fallacy in many neonatal units, leading to neonatal hypothermia, especially among preterm neonates born at <34 weeks of gestation. Hence, using quality improvement (QI) measures, we aimed to reduce the incidence of hypothermia in at-risk preterm neonates at admission to the neonatal intensive care unit (NICU) from 75% to 30% over 15 months (July 2021-September 2022).
Methods
This study was conducted in a 35-bed tertiary care neonatal unit serving multiple primary and secondary health centers in the area, with annual admission rates of 1200 to 1500. An annual audit revealed that many preterm infants were hypothermic upon NICU admission, prompting this study’s design. The study was conducted from July 2021 to September 2022 by implementing multiple plan-do-study-act (PDSA) cycles as per the World Health Organization Point of Care Quality Improvement (POCQI) model. 12 A multidisciplinary team, including neonatologists, pediatric residents, pediatric surgeons, obstetricians, anesthesiologists, radiologists, and registered nurses, was formed to address this issue, propose solutions, and execute the PDSA cycles. The Institute Ethics Committee clearance was obtained. Written informed consent was obtained from the parents of the eligible neonates. All inborn neonates with gestational age less than 34 weeks or birth weight less than 1500 g admitted to the NICU were included in the study. Neonates less than 24 weeks of gestation, birth weight less than 500 g, major congenital malformations, and those undergoing cardiac surgeries were excluded.
The primary outcome measure was the percentage of eligible preterm neonates with hypothermia (axillary temperature <36.5 °C) upon NICU admission. The numerator is the number of eligible preterm neonates with hypothermia at admission, and the denominator is the total number of eligible preterm neonates admitted to the NICU during the study period. The balancing measure was the incidence of hyperthermia with an axillary temperature of greater than 37.5 °C at admission to monitor any unintended consequences of the study interventions. The nursing staff measured the axillary temperature of newborns at admission before transferring them from the transport incubator to the warmer. They documented the temperature in degrees Celsius in the admission register and neonatal files. Investigators collected these data from the admission register, recorded them manually in a Microsoft Excel spreadsheet, and reviewed them biweekly. Hypothermia was classified using WHO guidelines as mild hypothermia (cold stress) at 36.0 °C to 36.4 °C, moderate at 32.1 °C to 35.9 °C, and severe at <32 °C. 12
Over 3 months, baseline demographic data, including admission temperatures, were collected. Using fishbone analysis (Figure 1), the team identified fallacies and barriers to low admission temperatures and developed a key-driver diagram (Figure 2) to analyze the main problems, propose change concepts, and prioritize them.
Fishbone Analysis.
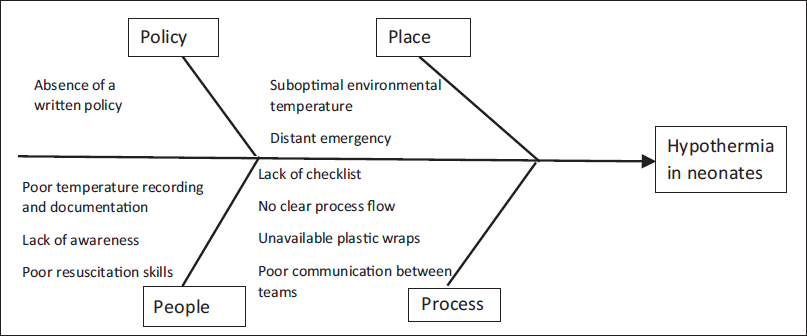
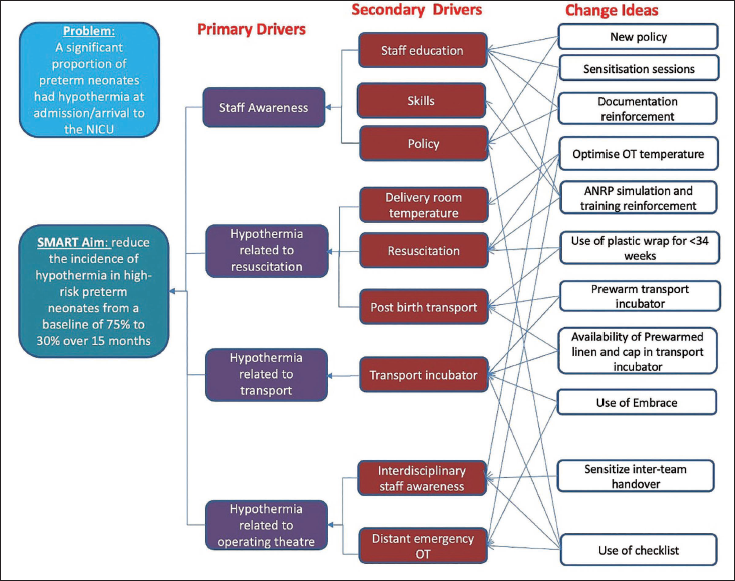
Table 1 outlines the process improvement initiatives through 2 PDSA cycles. In PDSA 1, a written policy was introduced, mandating nursing staff to record admission axillary temperatures. A bundled approach (change package) with evidence-based interventions was implemented to prevent hypothermia, addressing multiple drivers simultaneously. Educational sessions were conducted for various healthcare professionals to highlight the new policy. A checklist enforced the use of prewarmed transport incubators before expected deliveries, and a biomedical engineer optimized air temperatures daily. The use of caps was reinforced for all preterm deliveries and neonate transport. Temperature of delivery room and that of the operating theater (OT) were optimized to 25 °C to 28 °C, adhering to WHO guidelines. Changes in outcome measures and barriers were analyzed after PDSA1.
Summary of Process Improvement Initiatives Through PDSA Cycles 1 and 2.
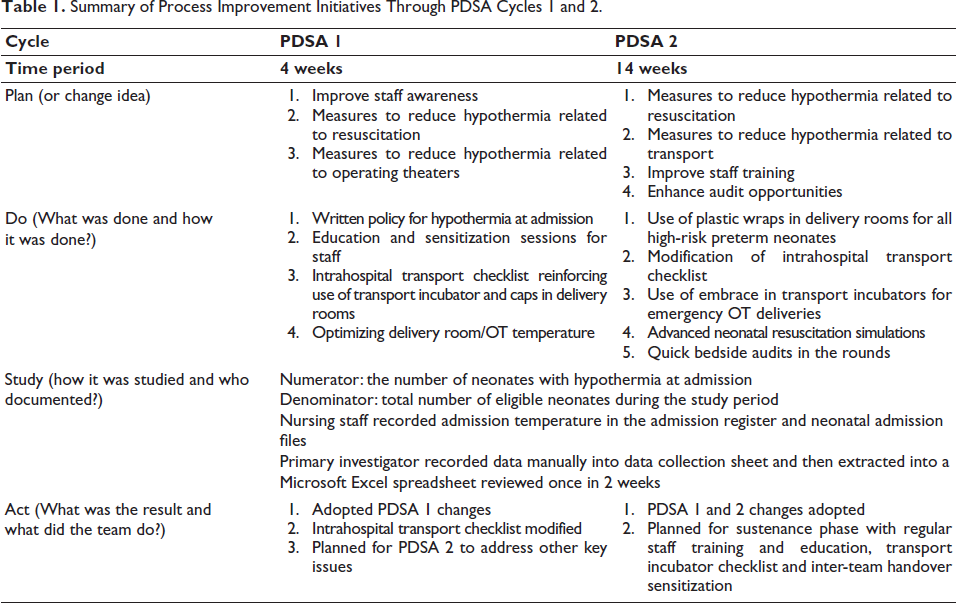
In PDSA 2, we introduced “Neohelp,” a sterile polyethylene plastic wrap with a cap in which the baby is placed immediately after birth without drying, to reduce heat loss in neonates of less than 34 weeks of gestation and weight of less than 1500 g. The family bore the cost as it is a standard practice. The checklist was updated to include two prewarmed linens in the transport incubator and the use of embrace to transport neonates from the distant emergency OT to the neonatal unit. Monthly, we reinforced the advanced neonatal resuscitation program (NRP) with simulations for postgraduates and respiratory therapists and basic NRP for nurses. Quick audits during clinical rounds ensured proper documentation of admission temperature. Sustenance phase data were collected for 9 months to maintain the implemented changes and monitor outcomes. Staff received bimonthly sensitization on the importance of the thermoregulation policy and documentation. Ongoing basic and advanced NRP training for all staff and daily transport incubator checks using a checklist by nurses and biomedical personnel were maintained, along with the change ideas from PDSA 1 and 2.
Statistical Analysis
SPSS 2021 version 1.0.0.1406 was used to analyze the dataset. Descriptive statistics summarized baseline demographics. Real-time Statistical Process Control (SPC) or run charts were used to track outcome trends, with standard rules distinguishing special from common-cause variations. The median of baseline data was calculated, and a shift was identified when 8 consecutive points were on the same side. Control charts incorporated study intervention timelines, analyzing outcome changes from baseline to post-intervention. We followed the Standards for Quality Improvement Reporting Excellence 2.0 guidelines (SQUIRE) for reporting. 13
Results
Of 3485 neonates born during the study period, 319 (9.2%) eligible preterm neonates were included. Table 2 presents baseline neonatal characteristics. The fishbone analysis identified the lack of a written policy, suboptimal delivery room and OT temperatures, insufficient staff awareness, and resuscitation and transport issues (Figure 1). Key drivers and interventions are summarized in Figure 2.
Baseline Demographic Characteristics and Admission Temperatures of Eligible Neonates.
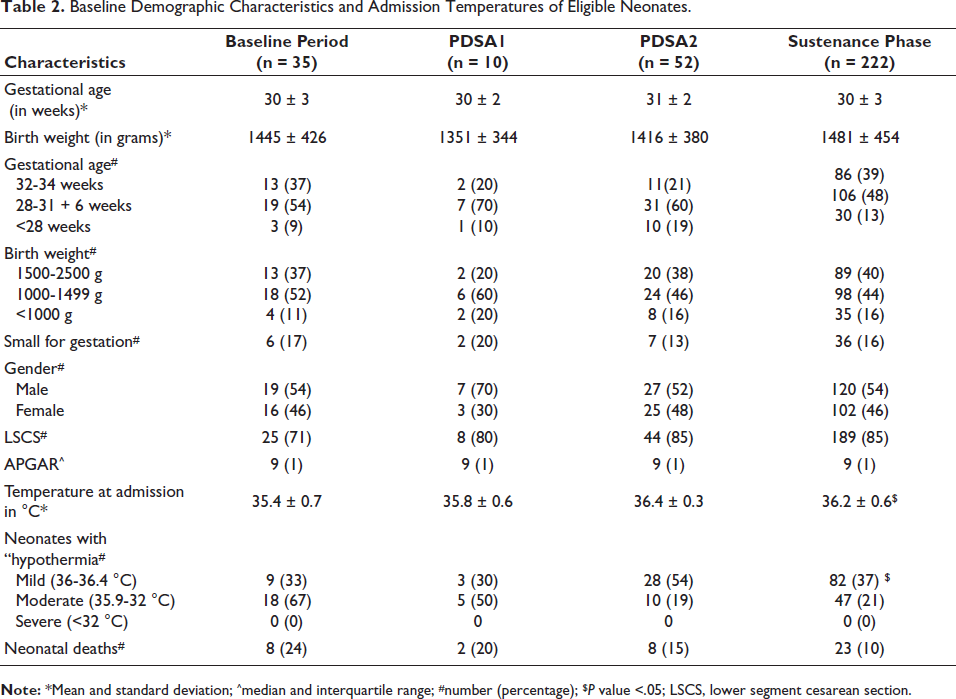
Figure 3 displays a run chart illustrating the trend in hypothermia incidence among at-risk preterm neonates. The baseline hypothermia incidence ranged from 80% to 100%, with a median of 83%. Following the PDSA 1 bundled approach, the median shifted to 50% by week 14 and declined to 20.5% by 24 to 26 weeks, with adjustments in the control limits, as shown in Figure 3. A shift of 8 data points was noted after 14 weeks and another shift after 50 weeks in the sustenance phase. There were four data points with zero hypothermia incidence during the sustenance phase. Less than 1% of neonates (n = 2) had temperatures above 37.5 °C during the study period, with 96% having a documented admission temperature.
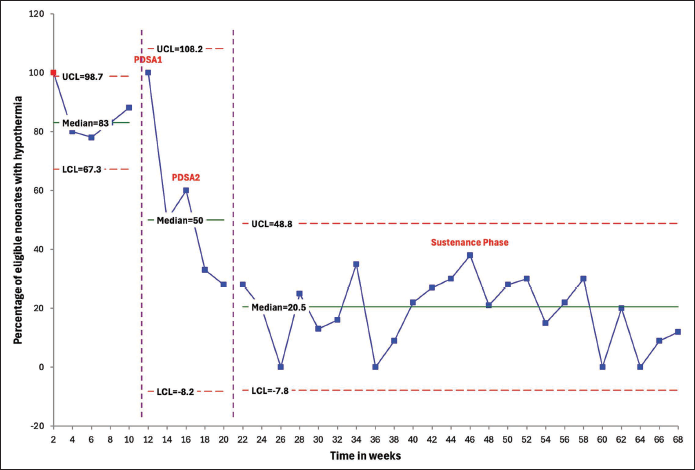
Mean admission temperature and the percentage of neonates with normothermia were notably higher during the PDSA cycles and sustenance phase than the baseline period (Table 2). The incidence of moderate hypothermia was significantly lower during these phases. There were no cases of severe hypothermia (<32 °C) during the study. Although neonatal deaths declined, this decrease was not statistically significant. Mean NICU admission temperatures for neonates born in the labor room and OT near the NICU were 35.5 °C ± 0.9 °C, 36.1 °C ± 0.5 °C, and 36.3 °C ± 0.3 °C during the baseline, PDSA cycles, and sustenance phases, respectively, showing statistical significance. During the baseline period, 50% of neonates were born during off-duty hours (5
Discussion
This QI initiative in a tertiary care NICU was conducted over 15 months. During the initial 12-week baseline data collection, a temporary decline in hypothermia incidence occurred as healthcare professionals became more conscious of temperature maintenance (Hawthorne effect). PDSA cycle 2 lasted 14 weeks, unlike the 4-week PDSA 1, owing to logistical delays in procuring medical-grade plastic wraps (Neohelp) through the hospital’s purchase department. The primary outcome was maintained for over 9 months, validating the effectiveness of the PDSA cycle changes.
We employed the change package concept (bundled approach) in the PDSA cycles instead of implementing one change at a time. Since most changes were evidence-based interventions to prevent neonatal hypothermia, this method saved considerable time. Establishing a unit policy requiring nursing staff to check the axillary temperature of neonates upon NICU admission altered staff behavior through increased awareness, achieving 96% compliance. A bundled approach, combined with staff awareness and the appropriate use of transport incubators and plastic wraps in delivery rooms, significantly improved admission temperatures, consistent with other studies. 14 This approach also significantly reduced the incidence of hypothermia in neonates, achieving a new median of 20.5% during the sustenance phase. Although mortality rates were lower in the PDSA and sustenance phases, the difference was not significant. This study cannot definitively link reduced mortality to improved admission temperatures because of the varied risk factors and confounders associated with high-risk neonates.
The WHO guidelines recommend a temperature of 25 °C to 28 °C in OTs and delivery rooms for preterm neonates, which was adhered to in this study. 11 Neonates born and transported from the distant emergency OT to the NICU experienced the greatest temperature drop because of long distances, suboptimal OT temperatures, and inadequate preparedness during off-duty hours. QI interventions, including checklists, plastic wraps for resuscitation, Embrace with a transport incubator, and enhanced team handover resulted in the highest increase in admission temperature for these preterm neonates.
In a study by Datta et al., 10 28% of neonates experienced cold stress, 39% moderate hypothermia, and 3% severe hypothermia. Contrarily, our study found no severe hypothermia, with only 2 cases of admission temperatures >37.5 °C, serving as a reassuring balance measure. The same study initially reported a 70% hypothermia rate, which improved to 25% over 9 months. Our study showed hypothermia incidence improved from 83% to a median of 20.5% over 15 months. While Datta’s study identified and addressed transport issues, it did not comment on room temperature optimization. Our research tackled various environmental issues in multiple birth locations accordingly.
Knobel et al. 15 demonstrated significant improvement in neonatal resuscitation using polyethylene wraps, consistent with our findings. We applied plastic wraps to neonates under 34 weeks’ gestation, unlike the previous study that targeted those under 28 weeks. Because of the high prevalence of small-for-gestational-age neonates, we opted for plastic wraps for all neonates born under 34 weeks of gestation or weighing less than 1500 g, compared to the NRP recommendation of under 32 weeks.16, 17 Most studies, including systematic reviews, indicate that plastic bags or wraps with a plastic cap enhance neonatal core body temperature. 18
Strengths and Limitations
This QI study, following the ePOCQI model, included all various departments involved in neonatal care. No significant funds were spent on new equipment or additional staff, nor were construction activities within the hospital. The study concentrated on simple process modifications to streamline tasks and enhance workflow efficiency, facilitating staff performance and potentially improving patient outcomes with existing resources and personnel. A major limitation is that this is a single-center study, which provides opportunities for QI collaboration. This can enhance quality and patient safety on a broader scale. Additionally, the study’s inability to measure process variables is another drawback.
Conclusion
QI measures significantly reduced hypothermia in at-risk preterm neonates in our NICU. The simplicity and ease of our changes suggest that they are sustainable and replicable in other settings. Regulating environmental temperature is crucial for maintaining euthermic conditions. Simple process changes can significantly impact at-risk neonates. A bundled approach facilitates implementing multiple evidence-based changes quickly. Polyethylene wraps are effective and economical for maintaining euthermia in neonates in low-resource settings.
Footnotes
Authors Contribution
SK, SB, JP: Data collection, implementation of intervention, working out of technical details and draft manuscript preparation. SB, JP, LL: Study conception and design, analytical calculations and numerical simulations verified the study methods, manuscript revision and final approval. SK, SB, JP: Analysis and interpretation of results. SK, SB, JP, AKP: Draft manuscript preparation. LL: Provided critical review. All the authors gave final approval of the version to be published.
Declaration of Conflict of Interests
The authors declare no potential conflicts of interest concerning this article’s research, authorship, or publication.
Ethical Approval
The Institute Ethics Committee clearance was obtained (clearance no: IEC 947/2020).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the parents of the eligible neonates.
